Is Development of High-Grade Gliomas Sulfur-Dependent?
Abstract
:1. Introduction
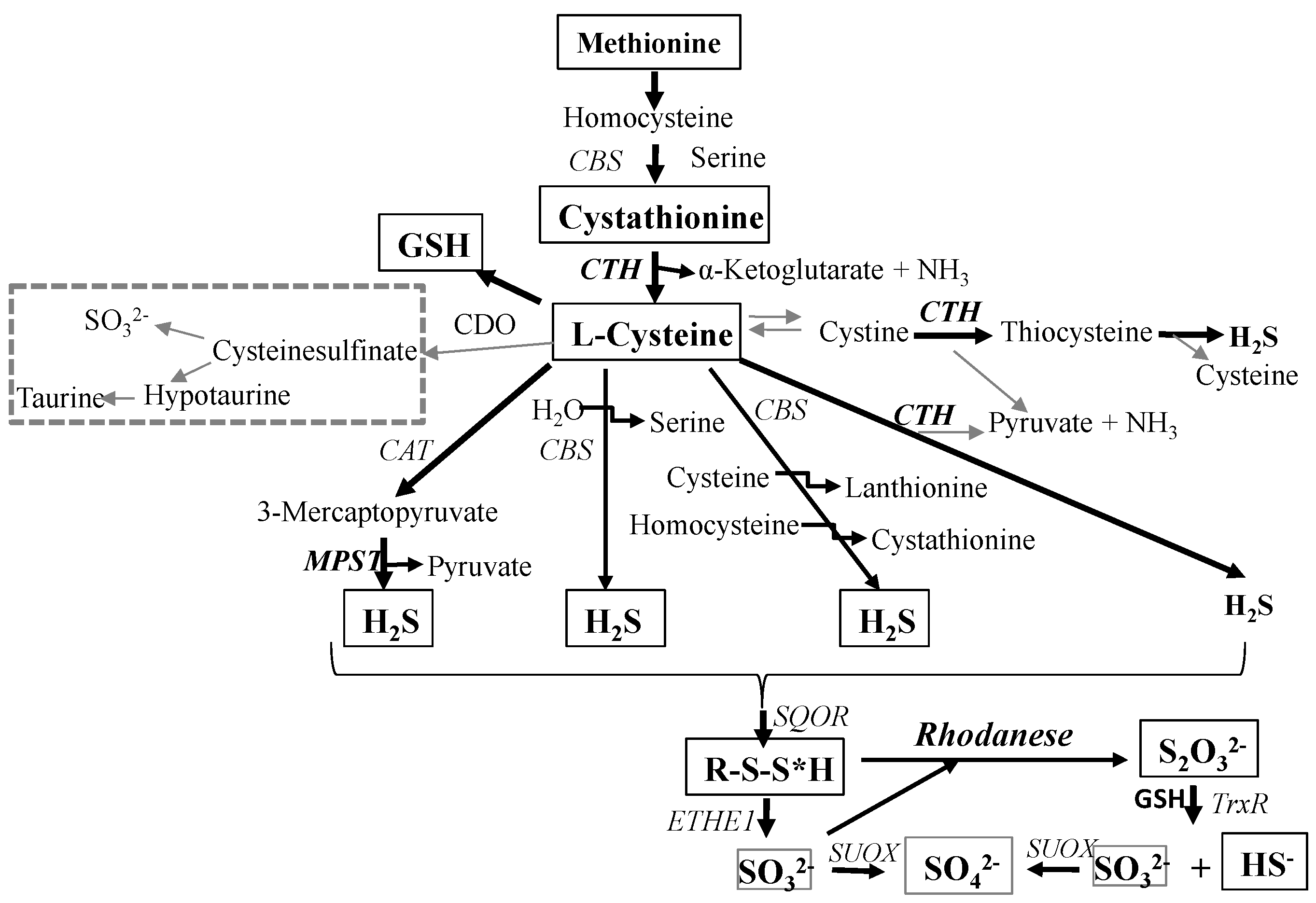
2. Results and Discussion
2.1. MPST, Rhodanese and γ-Cystathionase Activities and the Level of Sulfane Sulfur in Human Brain Regions
| Brain Regions | MPST | Rhodanese | CTH ** | Sulfane Sulfur |
|---|---|---|---|---|
| nmol·mg−1·min−1 | nmol·mg−1 | |||
| Cerebellum | 639 ± 103 | 212 ± 44 | 0.7 ± 0.2 * | 191 ± 41 |
| Hypothalamus | 745 ± 119 | 200 ± 54 | 0.7 ± 0.3 * | 221 ± 53 |
| Thalamus | 841 ± 142 * | 222 ± 63 | 0.4 ± 0.1 * | 209 ± 44 * |
| Nuclei subcortical | 732 ± 170 | 179 ± 68 | 0.6 ± 0.4 | 197 ± 40 |
| Hippocampus | 671 ± 205 | 177 ± 67 | 0.4 ± 0.3 | 225 ± 63 |
| Frontal cortex | 627 ± 181 | 144 ± 35 | 0.5 ± 0.4 | 231 ± 49 |
| Parietal cortex | 574 ± 142 | 139 ± 50 | 0.7 ± 0.3 * | 228 ± 70 |
| Liver | 7224 ± 2782 | 3031 ± 1128 | 1.2 ± 0.8 | 393 ± 133 |
2.2. MPST, Rhodanese and Cystathionase Activities and Sulfane Sulfur Level in Human Brain Gliomas
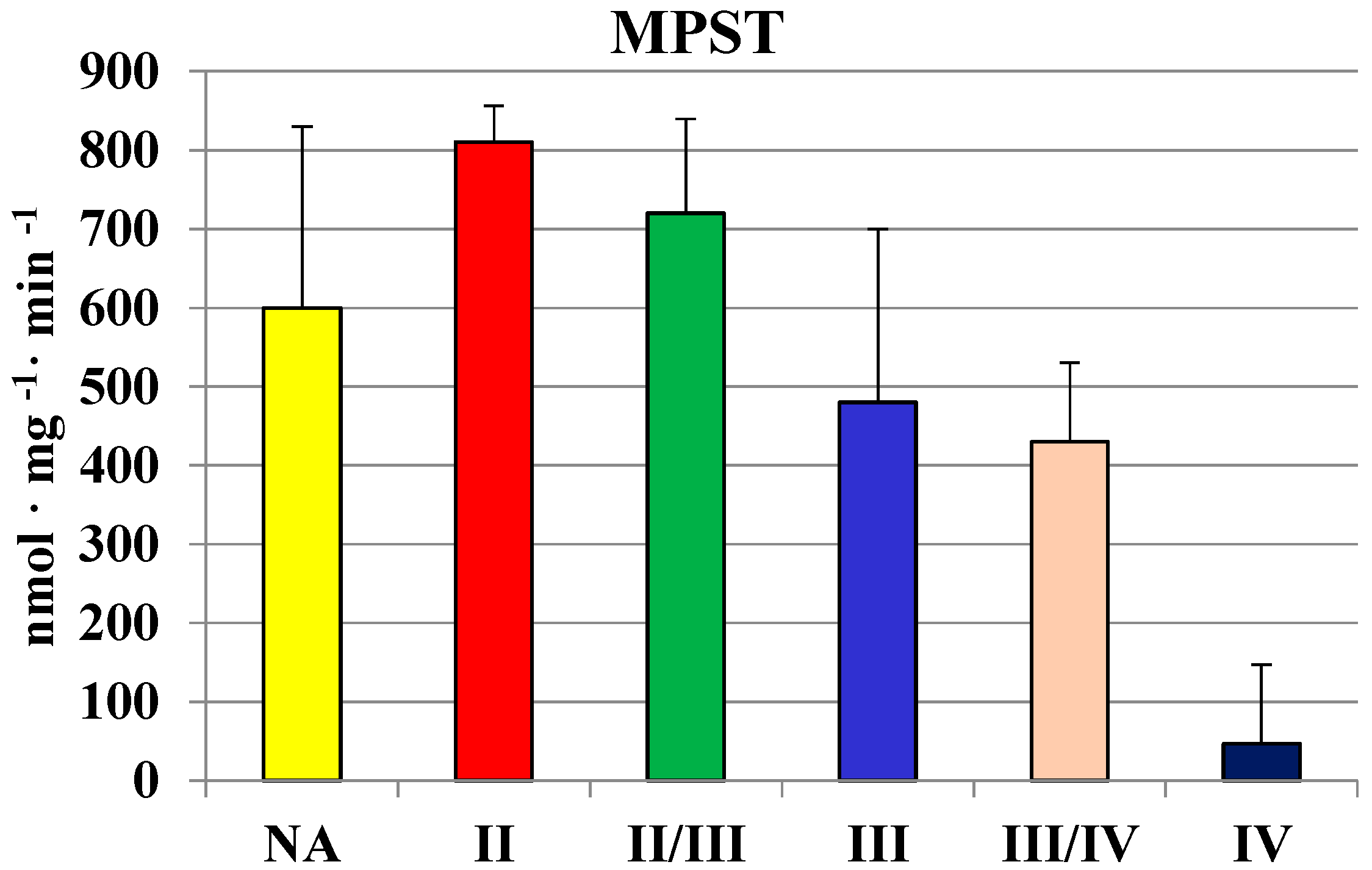

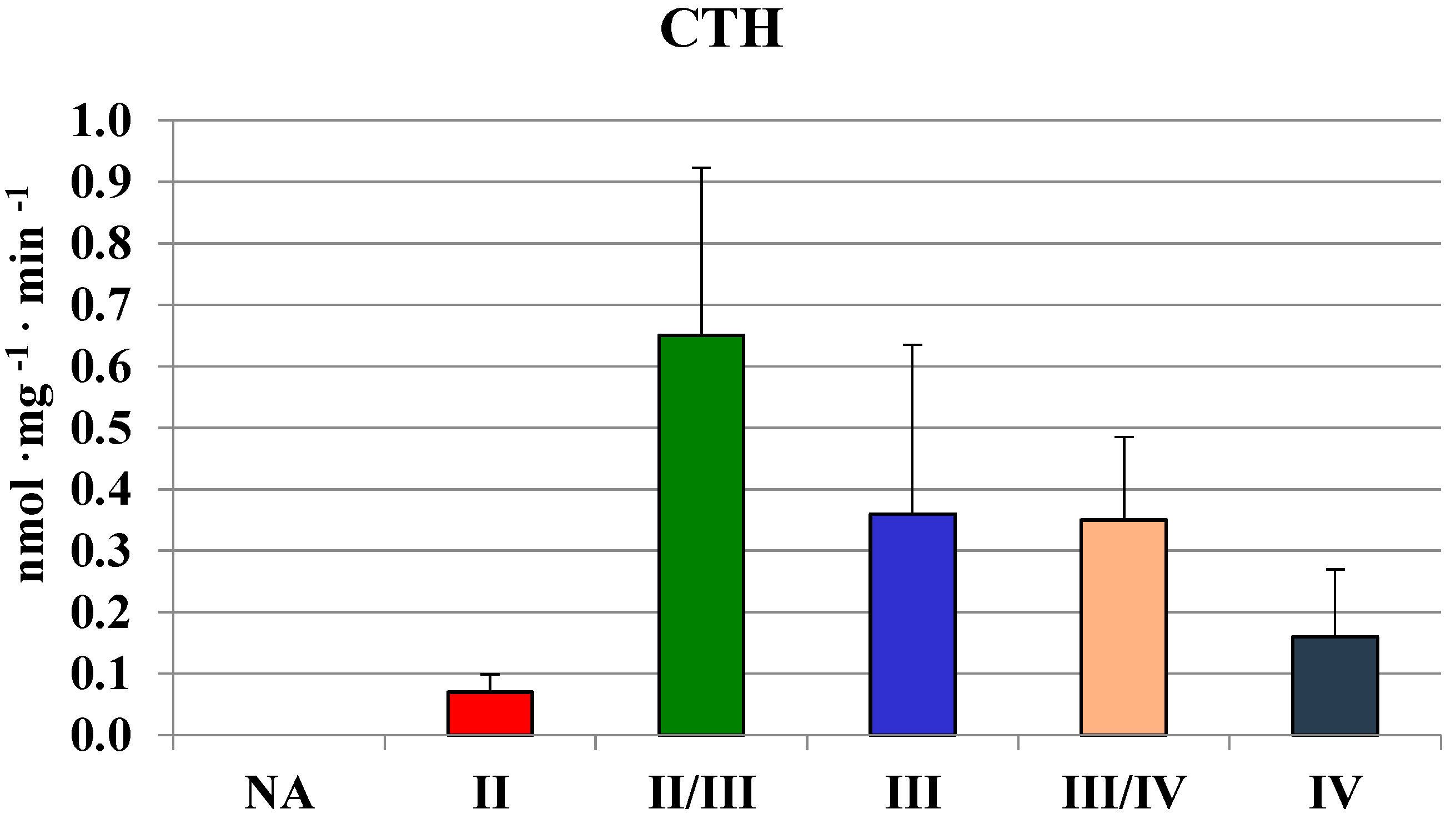
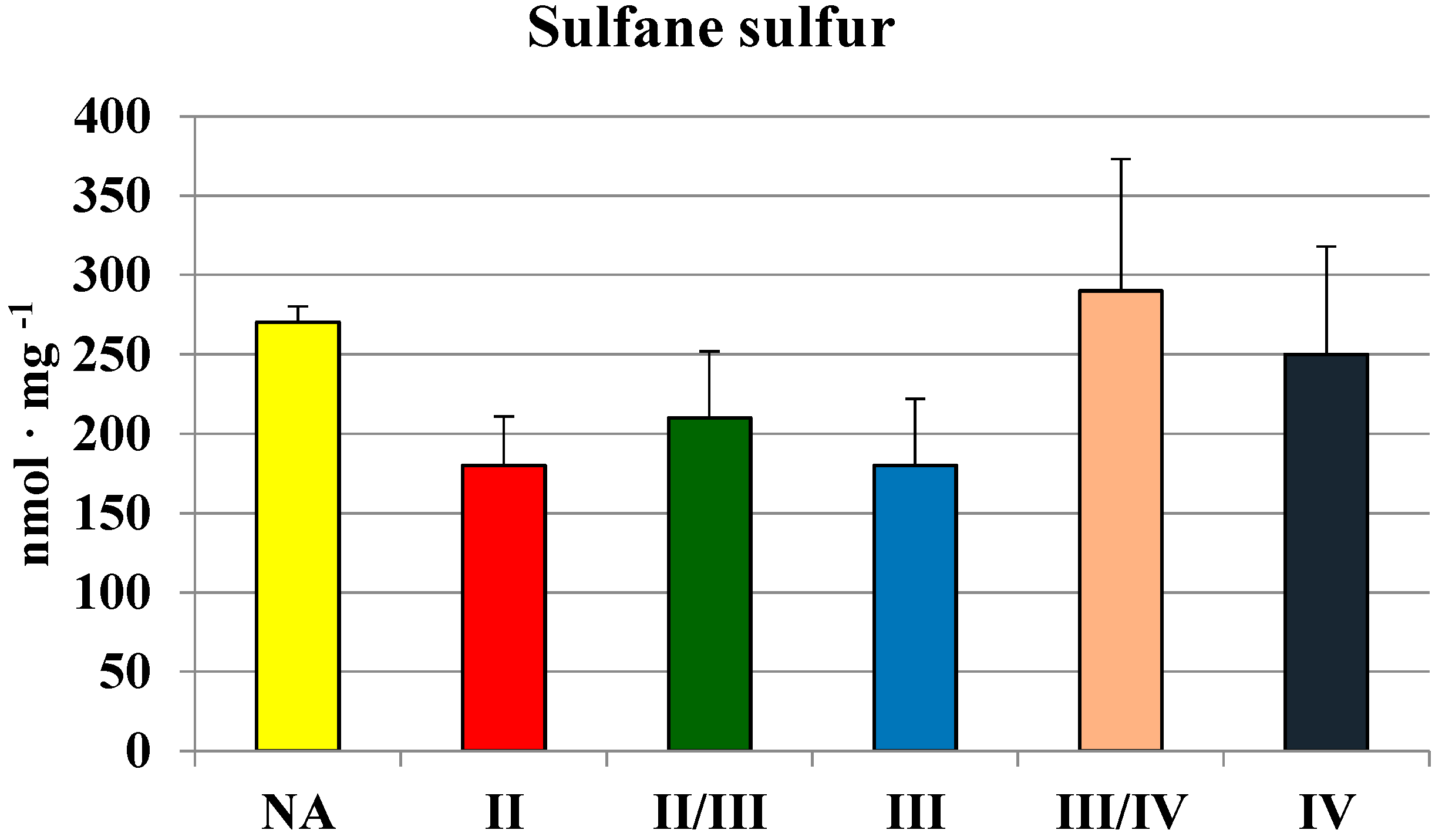
| Tissues | MPST | Rhodanese | CTH | Sulfane Sulfur |
|---|---|---|---|---|
| nmol·mg−1·min−1 | nmol·mg−1 | |||
| Brain | 689 ± 151 | 146 ± 54 | 559 ± 356 | 214 ± 51 |
| Glioma | 442 ± 118 | 63 ± 28 | 210 ± 200 | 250 ± 90 |
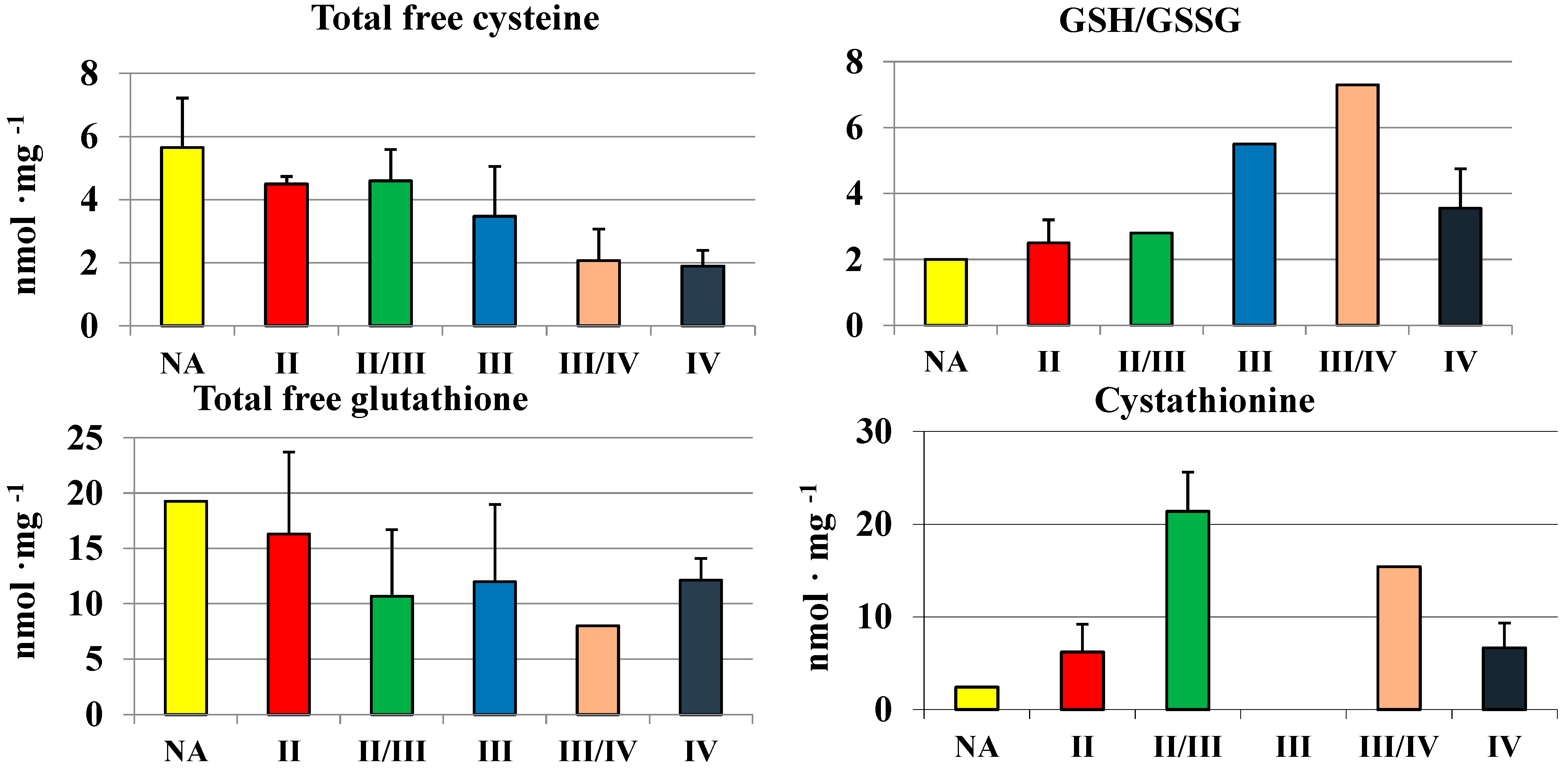
2.3. Cysteine, Glutathione and Cystathionine Levels in Human Brain Gliomas
3. Experimental Section
3.1. Chemicals
3.2. Human Tissues
3.2.1. Sections of Normal Human Brain Obtained Postmortem
3.2.2. Sections of Gliomas Obtained Intraoperatively
| No. | Tissue | WHO Grade | Number of Sections |
|---|---|---|---|
| 1 | Gliosis (around hemangioblastoma) | NA | 1 section |
| 2 | Diffuse astrocytoma (fibrillary) | WHO II | 1 section |
| 3 | Diffuse astrocytoma (gemistocytic) | WHO II | 1 section |
| 4 | Diffuse astrocytoma (fibrillary) | WHO II | 1 section |
| 5 | Diffuse astrocytoma | WHO II | 1 section |
| 6 | Oligodendroglioma | WHO II | 1 section |
| 7 | Oligoastrocytoma | WHO II/III | 1 section |
| 8 | Diffuse astrocytoma | WHO II/III | 1 section |
| 9 | Anaplastic oligodendroglioma | WHO III | 1 section |
| 10 | Anaplastic astrocytoma | WHO III | 2 section |
| 11 | Anaplastic astrocytoma/Glioblastoma | WHO III/IV | 4 section |
| 12 | Glioblastoma | WHO IV | 5 section |
3.3. Tissue Homogenates
3.4. Enzyme Assay
3.5. Sulfane Sulfur and Protein
3.6. RP-HPLC (Reverse Phase High Performance Liquid Chromatography)
3.6.1. Standard Curves
3.6.2. Sample Preparation
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Abbreviation
| CTH | γ-cystathionase |
| GSH | glutathione, reduced form |
| GSSG | glutathione, oxidized form |
| MPST | 3-mercaptopyruvate sulfurtransferase |
| TFG | total free glutathione |
| TFC | total free cysteine |
Conflicts of Interest
References
- Toohey, J.I.; Cooper, A.J.L. Thiosulfoxide (sulfane) sulfur: New chemistry and new regulatory roles in biology. Molecules 2014, 19, 12789–12813. [Google Scholar] [CrossRef] [PubMed]
- Nagahara, N.; Sawada, N. The mercaptopyruvate pathway in cysteine catabolism: A physiologic role and related disease of the multifunctional 3-mercaptopyruvate sulfurtransferase. Curr. Med. Chem. 2006, 13, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Wang, R. Two’s company, three’s a crowd: Can H2S be the third endogenous gaseous transmitter ? FASEB J. 2002, 16, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Shibuya, N.; Kimura, Y. Hydrogen sulfide is a signaling molecule and a cytoprotectant. Antioxid. Redox Signal. 2012, 17, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H. Physiological role of hydrogen sulfide and polysulfide in the central nervous system. Neurochem. Int. 2013, 63, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Jurkowska, H.; Roman, H.B.; Hirschberger, L.L.; Sasakura, K.; Nagano, T.; Hanaoka, K.; Krijt, J.; Stipanuk, M.H. Primary hepatocytes from mice lacking cysteine dioxygenase show increased cysteine concentrations and higher rates of metabolism of cysteine to hydrogen sulfide and thiosulfate. Amino Acids 2014, 46, 1353–1365. [Google Scholar] [CrossRef] [PubMed]
- Breillout, F.; Antoine, E.; Poupon, M.F. Methionine dependency of malignant tumors: A possible approach for therapy. J. Natl. Cancer Inst. 1990, 82, 1628–1632. [Google Scholar] [CrossRef] [PubMed]
- Cellarier, E.; Durando, X.; Vasson, M.P.; Farges, M.C.; Demiden, A.; Maurizis, J.C.; Madelmont, J.C.; Chollet, P. Methionine dependency and cancer treatment. Cancer Treat. Rev. 2003, 29, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Poirson-Bichat, F.; Gonçalves, R.A.; Miccoli, L.; Dutrillaux, B.; Poupon, M.F. Methionine Depletion Enhances the Antitumoral Efficacy of Cytotoxic Agents in Drug-resistant Human Tumor Xenografts Methionine Depletion Enhances the Antitumoral Efficacy of Cytotoxic Agents in Drug-resistant Human. Clin. Cancer Res. 2000, 6, 643–653. [Google Scholar] [PubMed]
- Jurkowska, H.; Wróbel, M. N-acetyl-l-cysteine as a source of sulfane sulfur in astrocytoma and astrocyte cultures: Correlations with cell proliferation. Amino Acids 2008, 34, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Jurkowska, H.; Uchacz, T.; Roberts, J.; Wróbel, M. Potential therapeutic advantage of ribose-cysteine in the inhibition of astrocytoma cell proliferation. Amino Acids 2011, 41, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Dulińska-Litewka, J.; Wróbel, M.; Bronowicka-Adamska, P. Comparative Effect of Diallyl Disulphide, Cystathionine, N-Acetylcysteine and Thiosulfate on PC-3 Cells Proliferation, Sulfurtransferases Expression and Sulfane Sulfur Levels. Int. J. Indig. Med. Plants 2014, 47, 1660–1668. [Google Scholar]
- Diwakar, L.; Ravindranath, V. Inhibition of cystathionine-gamma-lyase leads to loss of glutathione and aggravation of mitochondrial dysfunction mediated by excitatory amino acid in the CNS. Neurochem. Int. 2007, 50, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Toohey, J.I. Sulphane sulphur in biological systems: A possible regulatory role. Biochem. J. 1989, 264, 625–632. [Google Scholar] [PubMed]
- Bronowicka-Adamska, P.; Zagajewski, J.; Czubak, J.; Wróbel, M. RP-HPLC method for quantitative determination of cystathionine, cysteine and glutathione: An application for the study of the metabolism of cysteine in human brain. J. Chromatogr. B 2011, 879, 2005–2009. [Google Scholar] [CrossRef]
- Toohey, J.I. Persulfide sulfur is a growth factor for cells defective in sulfur metabolism. Biochem. Cell Biol. 1986, 64, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Jurkowska, H.; Placha, W.; Nagahara, N.; Wróbel, M. The expression and activity of cystathionine-γ-lyase and 3-mercaptopyruvate sulfurtransferase in human neoplastic cell lines. Amino Acids 2011, 41, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Herman-Antosiewicz, A.; Singh, S.V. Signal transduction pathways leading to cell cycle arrest and apoptosis induction in cancer cells by Allium vegetable-derived organosulfur compounds: A review. Mutat. Res. 2004, 555, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Banik, N.L.; Ray, S.K. Garlic compounds generate reactive oxygen species leading to activation of stress kinases and cysteine proteases for apoptosis in human glioblastoma T98G and U87MG cells. Cancer 2007, 110, 1083–1095. [Google Scholar] [CrossRef] [PubMed]
- Jhee, K.H.; Kruger, W.D. The role of cystathionine beta-synthase in homocysteine metabolism. Antioxid. Redox Signal. 2005, 7, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Borcsok, E.; Abeles, R.H. Mechanism of action of cystathionine synthase. Arch. Biochem. Biophys. 1982, 213, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Szabó, C. Hydrogen sulphide and its therapeutic potential. Nat. Rev. Drug Discov. 2007, 6, 917–935. [Google Scholar] [CrossRef] [PubMed]
- Whiteman, M.; Winyard, P.G. Hydrogen sulfide and inflammation: The good, the bad, the ugly and the promising. Expert Rev. Clin. Pharmacol. 2011, 4, 12–32. [Google Scholar] [CrossRef]
- Bergstrom, M.; Ericson, K.; Hagenfeldt, L. PET study of methionine accumulation in glioma and normal brain tissue: Competition with branched chain amino acids. J. Comput. Assist. Tomogr. 1987, 11, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Szczerbowska-Boruchowska, M.; Stegowski, Z.; Lankosz, M.; Szpak, M.; Adamek, D. A synchrotron radiation micro-X-ray absorption near edge structure study of sulfur speciation in human brain tumors—A methodological approach. J. Anal. At. Spectrom. 2012, 27, 239–247. [Google Scholar] [CrossRef]
- Goggins, M.; Scott, J.M.; Weir, D.G. Regional differences in protein carboxymethylation in postmortem human brain. Clin. Sci. 1998, 94, 677–685. [Google Scholar] [PubMed]
- Goggins, M.; Scott, J.M.; Weir, D.G. Methylation of cortical brain proteins from patients with HIV infection. Acta Neurol. Scand. 1999, 100, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Schmierer, K.; Wheeler-Kingshott, C.A.M.; Boulby, P.; Scaravilli, F.; Altmann, D.R.; Barker, G.J.; Tofts, P.S.; Miller, D.H. Diffusion tensor imaging of post mortem multiple sclerosis brain. Neuroimage 2007, 35, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Harish, G.; Venkateshappa, C.; Mahadevan, A.; Pruthi, N.; Srinivas Bharath, M.M.; Shankar, S.K. Glutathione metabolism is modulated by postmortem interval, gender difference and agonal state in postmortem human brains. Neurochem. Int. 2011, 59, 1029–1042. [Google Scholar] [CrossRef] [PubMed]
- Valentine, W.N.; Frankenfeld, J.K. 3-Mercaptopyuruvate sulfurtransferase (EC 2.8.1.2): A simple assay adapted to human blood cells. Clin. Chim. Acta 1974, 51, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Wróbel, M.; Jurkowska, H.; Sliwa, L.; Srebro, Z. Sulfurtransferases and cyanide detoxification in mouse liver, kidney, and brain. Toxicol. Mech. Methods 2004, 14, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Greenberg, D.M. A crystalline enzyme that cleaves homoserine and cystathionine: I. Isolation procedure and some physiochemical properties. J. Biol. Chem. 1958, 230, 545–560. [Google Scholar] [PubMed]
- Czubak, J.; Wróbel, M.; Jurkowska, H. Cystathionine γ-lyase (EC 4.4.1.1): An enzymatic assay of α-ketobutyrate using lactate dehydrogenase. Acta Biol. Crac. Ser. Zool. 2002, 44, 113–117. [Google Scholar]
- Wood, L. Sulfane sulfur. Methods Enzymol. 1987, 143, 25–29. [Google Scholar] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Dominick, P.K.; Cassidy, P.B.; Roberts, J.C. A new and versatile method for determination of thiolamines of biological importance. J. Chromatogr. B Biomed. Sci. Appl. 2001, 761, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wróbel, M.; Lewandowska, I.; Bronowicka-Adamska, P.; Paszewski, A. The level of sulfane sulfur in the fungus Aspergillus nidulans wild type and mutant strains. Amino Acids 2009, 37, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.E.; Roberts, B.; Holcenberg, J.; Glode, L.M. Cystathionine metabolism in neuroblastoma. Cancer 1988, 62, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wróbel, M.; Czubak, J.; Bronowicka-Adamska, P.; Jurkowska, H.; Adamek, D.; Papla, B. Is Development of High-Grade Gliomas Sulfur-Dependent? Molecules 2014, 19, 21350-21362. https://doi.org/10.3390/molecules191221350
Wróbel M, Czubak J, Bronowicka-Adamska P, Jurkowska H, Adamek D, Papla B. Is Development of High-Grade Gliomas Sulfur-Dependent? Molecules. 2014; 19(12):21350-21362. https://doi.org/10.3390/molecules191221350
Chicago/Turabian StyleWróbel, Maria, Jerzy Czubak, Patrycja Bronowicka-Adamska, Halina Jurkowska, Dariusz Adamek, and Bolesław Papla. 2014. "Is Development of High-Grade Gliomas Sulfur-Dependent?" Molecules 19, no. 12: 21350-21362. https://doi.org/10.3390/molecules191221350
APA StyleWróbel, M., Czubak, J., Bronowicka-Adamska, P., Jurkowska, H., Adamek, D., & Papla, B. (2014). Is Development of High-Grade Gliomas Sulfur-Dependent? Molecules, 19(12), 21350-21362. https://doi.org/10.3390/molecules191221350





