Investigation of Dendriplexes by Ion Mobility-Mass Spectrometry
Abstract
:1. Introduction
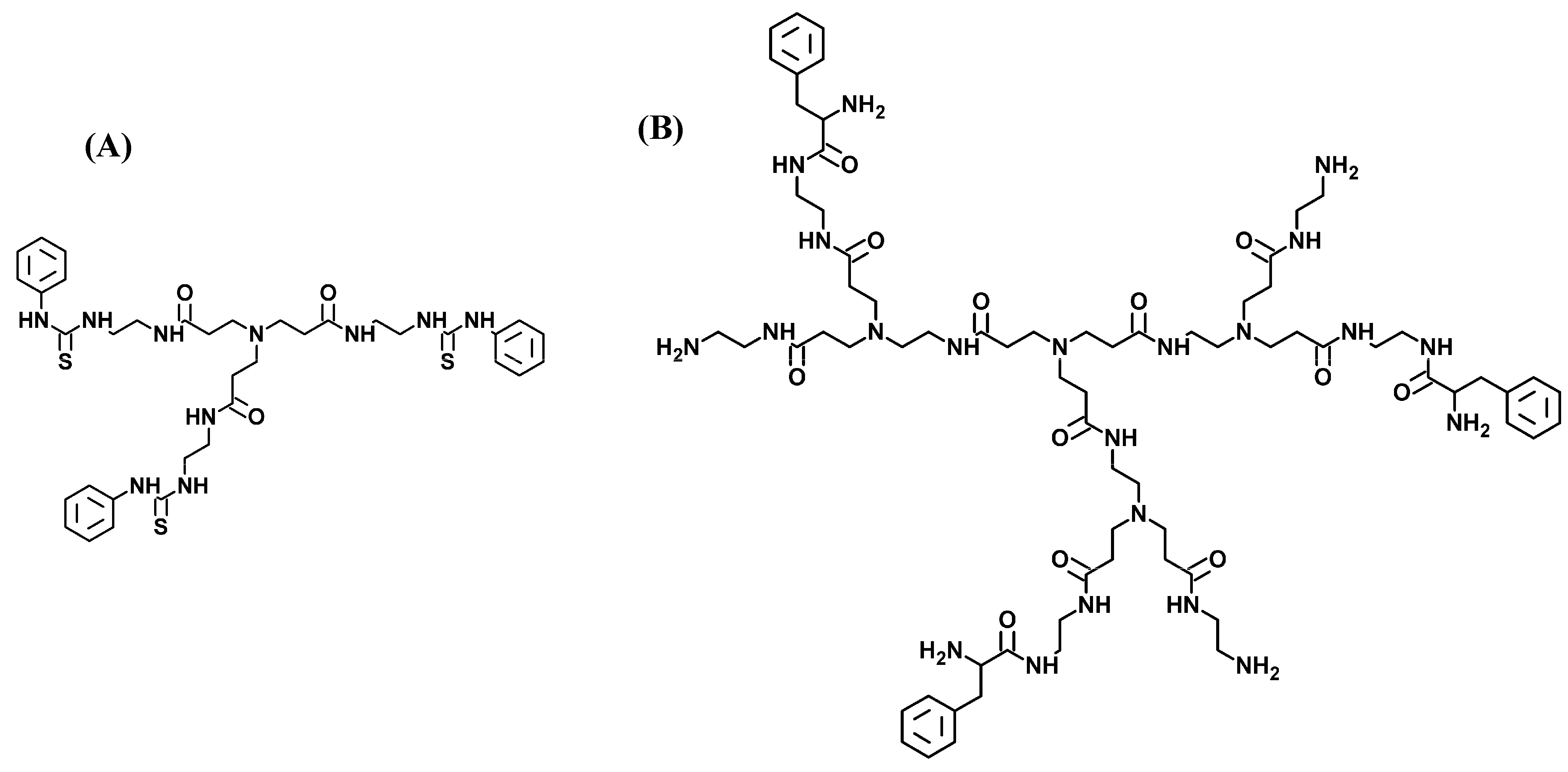
2. Results and Discussion
2.1. DNA Duplex
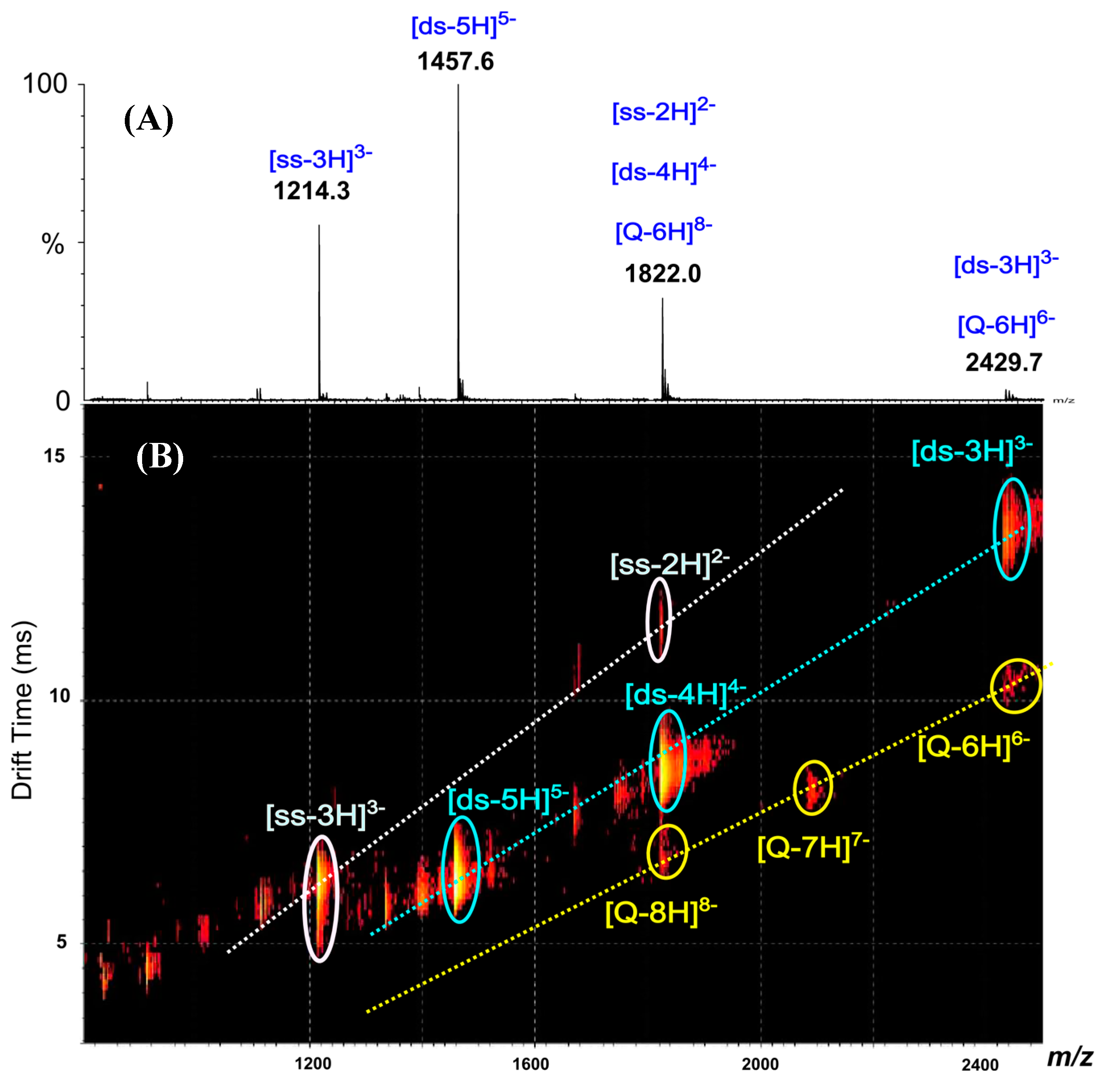
2.2. Dendriplexes
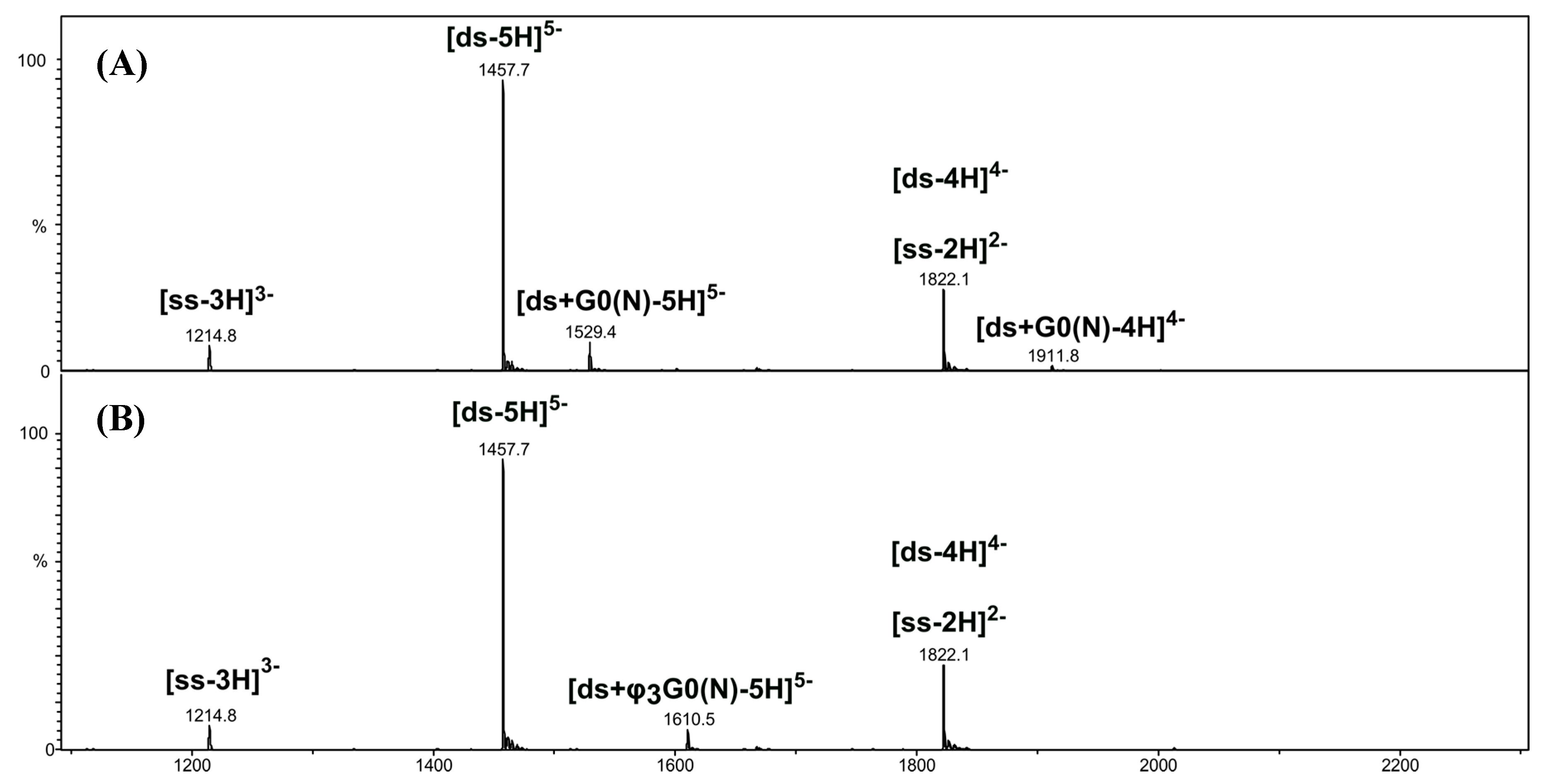
[ds/G0(N)] and [ds/φ3G0(N)] Dendriplexes

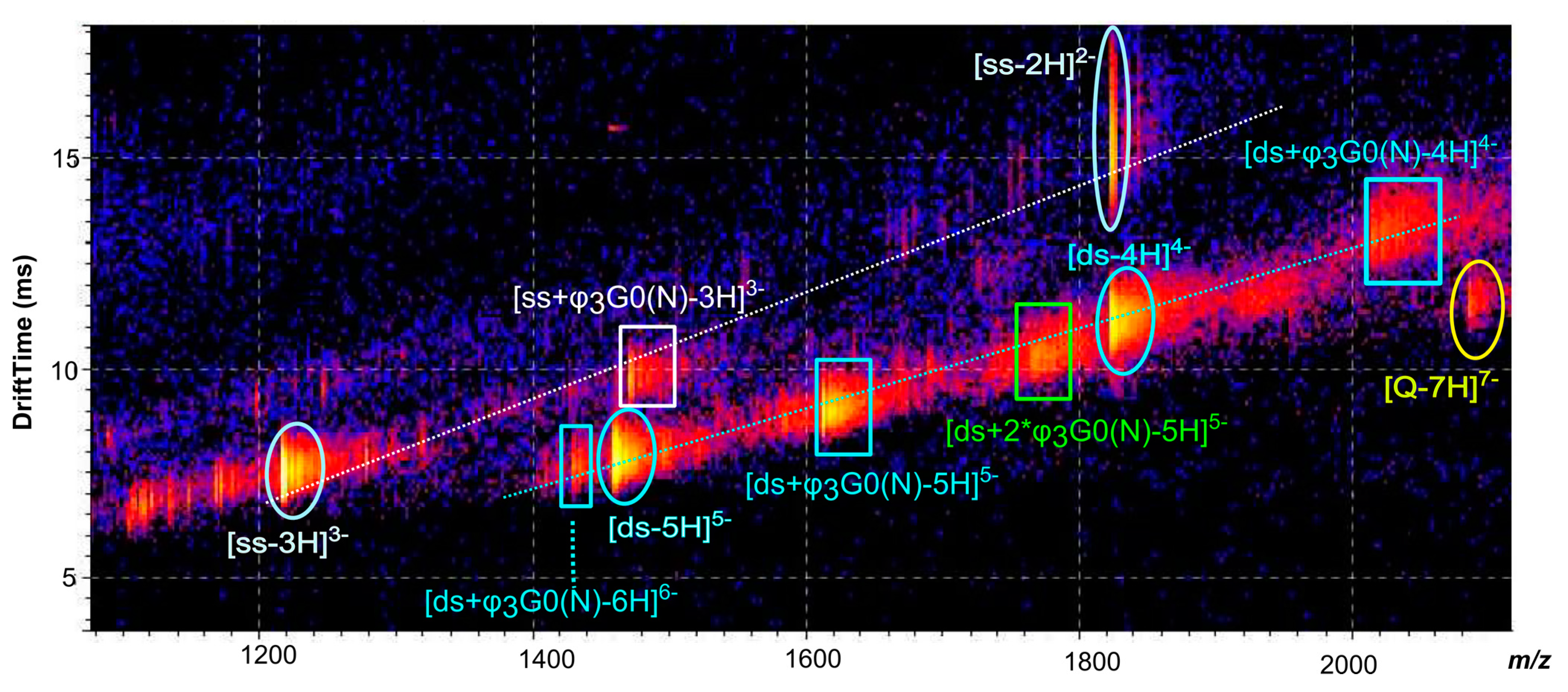
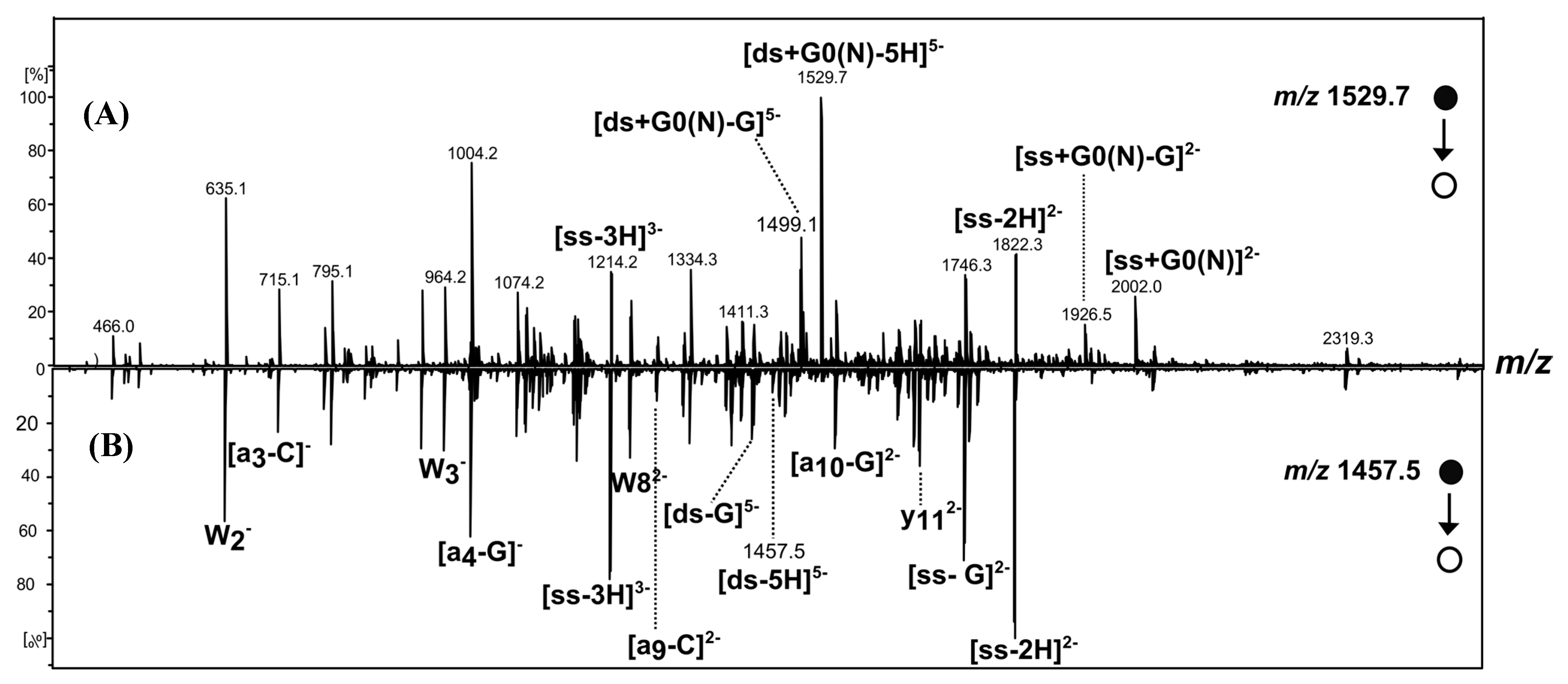
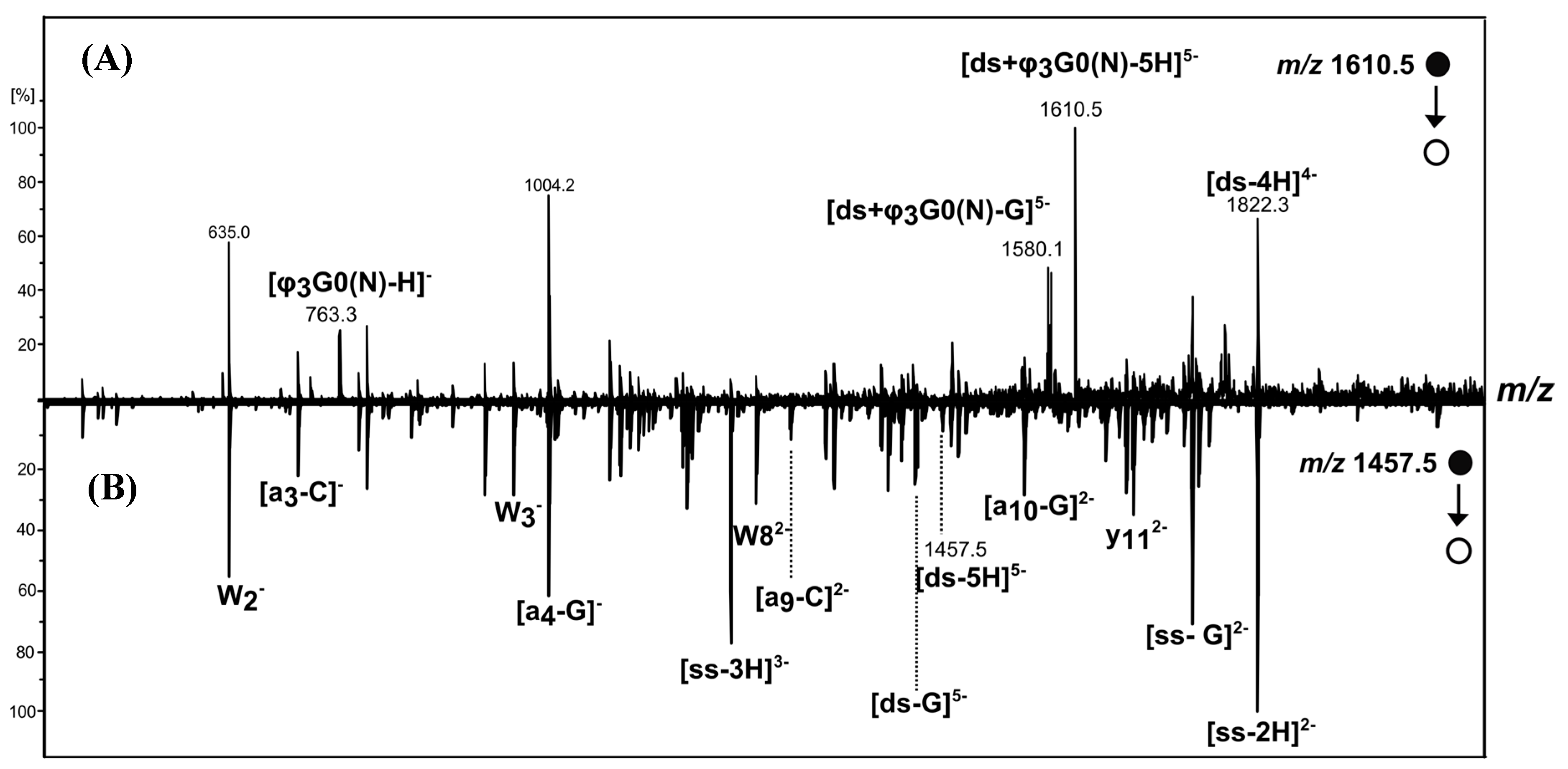
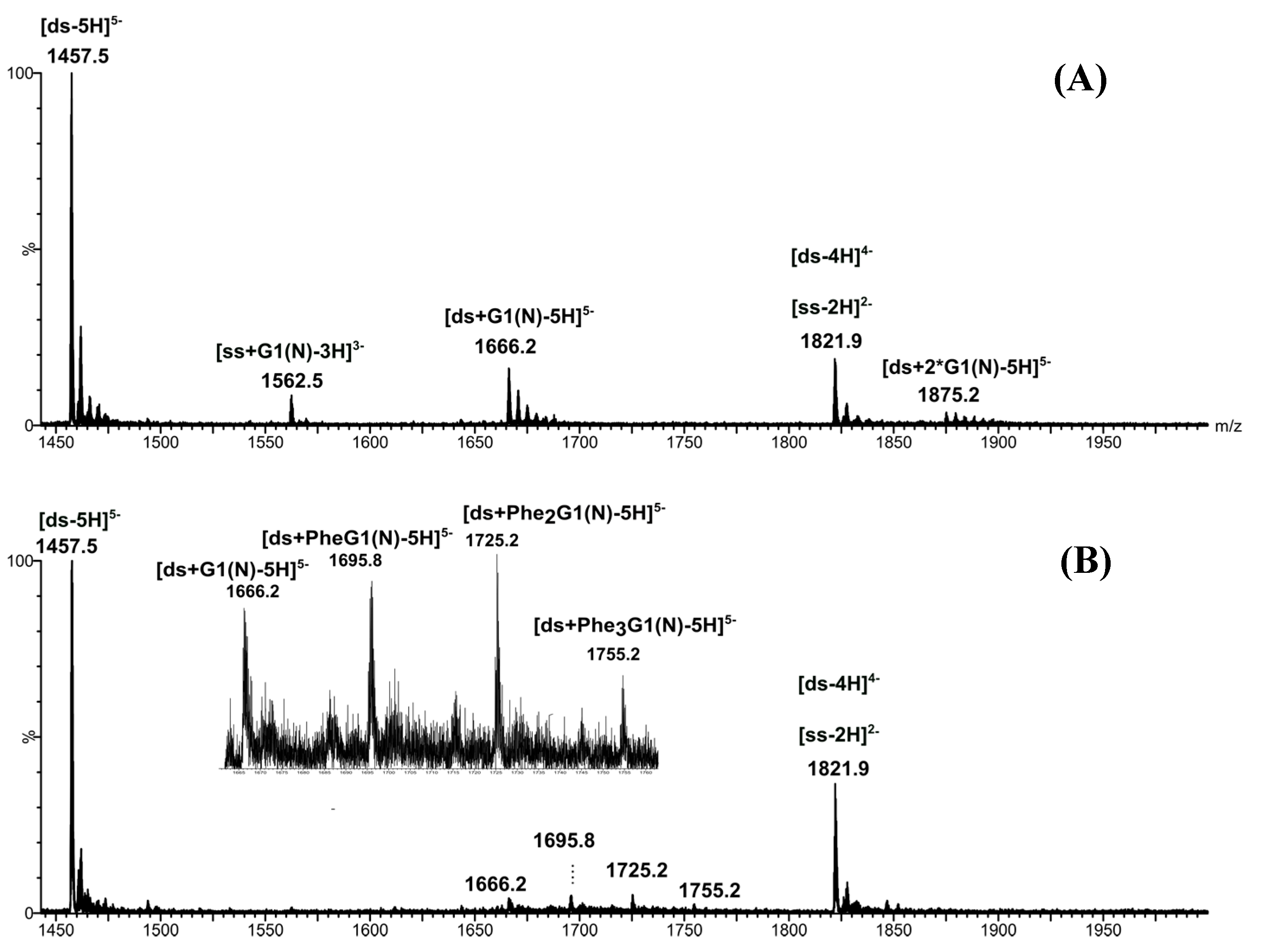
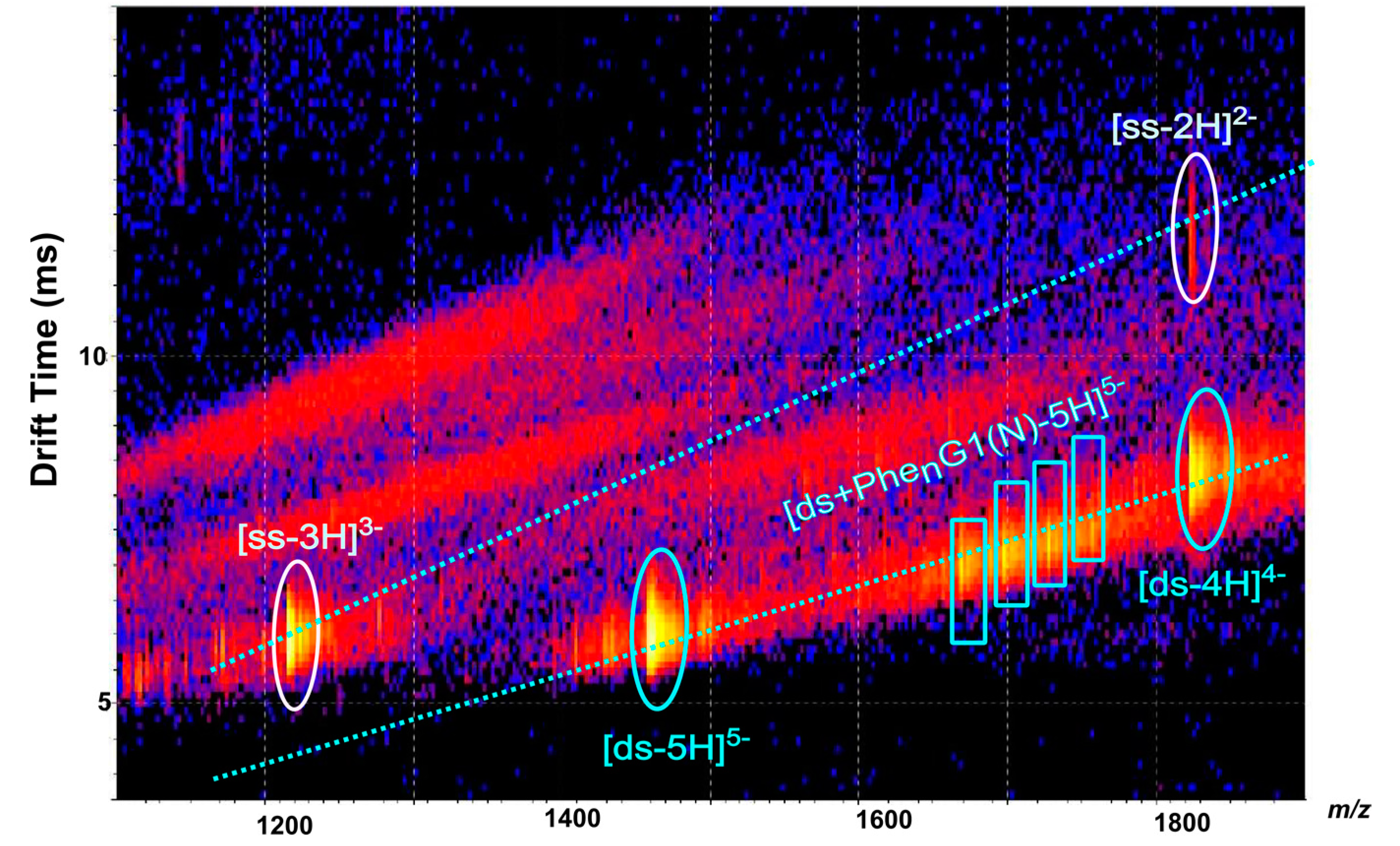
2.3. [ds/G1(N)/] and [ds/PhenG1(N)] Dendriplexes
3. Experimental Section
3.1. Reagents
3.2. Synthesis and Chemical Modification of PAMAM
| Compound | Ion | m/z Exp. a | m/z Calc. b | Error (ppm) | Elemental Compositions c |
|---|---|---|---|---|---|
| G1(N) | [M+2H]2+ | 522.8770 | 522.8780 | 1.90 | C45H93N19O9 |
| PheG1(N) | [M+2H]2+ | 596.4121 | 596.4122 | −0.08 | C54H102N20O10 |
| Phe2G1(N) | [M+2H]2+ | 669.9465 | 669.9464 | 0.10 | C63H111N21O11 |
| Phe3G1(N) | [M+2H]2+ | 743.4806 | 743.4807 | −0.06 | C72H120N22O12 |
3.3. Preparation of Samples
3.4. ESI-Q-TOF-MS(/MS) and ESI-IM-MS of Dendriplexes [ds/Native PAMAM] and [ds/Modified-PAMAM]
3.5. ESI-QIT-MS(/MS) of Dendriplexes [ds/Native PAMAM] and [ds/Modified- PAMAM]
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Esfand, R.; Tomalia, D.A. Poly(amidoamine) (PAMAM) dendrimers: from biomimicry to drug delivery and biomedical applications. Drug Discov. Today 2001, 6, 427–436. [Google Scholar]
- Eichman, J.; Bielinska, A.; Kukowska-Latallo, J.; Baker, J. The use of PAMAM dendrimers in the efficient transfer of genetic material into cells. Pharm. Sci. Technol. Today 2000, 3, 232–245. [Google Scholar]
- Fischer, D.; Li, Y.; Ahlemeyer, B.; Krieglstein, J.; Kissel, T. In vitro cytotoxicity testing of polycations: Influence of polymer structure on cell viability and hemolysis. Biomaterials 2003, 24, 1121–1131. [Google Scholar]
- Jevprasesphant, R.; Penny, J.; Jalal, R.; Attwood, D.; McKeown, N.B.; D’Emanuele, A. The influence of surface modification on the cytotoxicity of PAMAM dendrimers. Int. J. Pharm. 2003, 252, 263–266. [Google Scholar]
- Luo, D.; Haverstick, K.; Belcheva, N.; Han, E.; Saltzman, W.M. Poly(ethylene glycol )-Conjugated PAMAM Dendrimer for Biocompatible, High-Efficiency DNA Delivery. Macromolecules 2002, 35, 3456–3462. [Google Scholar]
- Kolhatkar, R.B.; Kitchens, K.M.; Swaan, P.W.; Ghandehari, H. Surface acetylation of polyamidoamine (PAMAM) dendrimers decreases cytotoxicity while maintaining membrane permeability. Bioconjug. Chem. 2007, 18, 2054–2060. [Google Scholar]
- Fant, K.; Esbjo, E.K.; Jenkins, A.; Grossel, M.C.; Lincoln, P.; Norde, B. Effects of PEGylation and Acetylation of PAMAM Dendrimers on DNA Binding, Cytotoxicity and in Vitro Transfection Efficiency. Mol. Pharm. 2010, 7, 1734–1746. [Google Scholar]
- Wang, X.; He, Y.; Wu, J.; Gao, C.; Xu, Y. Synthesis and evaluation of phenylalanine-modified hyperbranched poly(amido amine)s as promising gene carriers. Biomacromolecules 2010, 11, 245–251. [Google Scholar]
- Kono, K.; Akiyama, H.; Takahashi, T.; Takagishi, T.; Harada, A. Transfection activity of polyamidoamine dendrimers having hydrophobic amino acid residues in the periphery. Bioconjug. Chem. 2005, 16, 208–214. [Google Scholar]
- Yu, S.; Li, M.H.; Choi, S.K.; Baker, J.R.; Larson, R.G. DNA condensation by partially acetylated poly(amidoamine) dendrimers: Effects of dendrimer charge density on complex formation. Molecules 2013, 18, 10707–10720. [Google Scholar]
- Shcharbin, D.; Pedziwiatr, E.; Bryszewska, M. How to study dendriplexes I: Characterization. J. Control. Release 2009, 135, 186–197. [Google Scholar]
- Rosu, F.; de Pauw, E.; Gabelica, V. Electrospray mass spectrometry to study drug-nucleic acids interactions. Biochimie 2008, 90, 1074–1087. [Google Scholar]
- Mason, E.A.; McDaniel, E.W. Transport Properties of Ions in Gases; John Wiley&Sons Inc.: Hoboken, NJ, USA, 1988. [Google Scholar]
- Giles, K.; Pringle, S.D.; Worthington, K.R.; Little, D.; Wildgoose, J.L.; Bateman, R.H. Applications of a travelling wave-based radio-frequency-only stacked ring ion guide. Rapid Commun. Mass Spectrom. 2004, 18, 2401–2414. [Google Scholar]
- Pacholarz, K.J.; Garlish, R.A.; Taylor, R.J.; Barran, P.E. Mass spectrometry based tools to investigate protein-ligand interactions for drug discovery. Chem. Soc. Rev. 2012, 41, 4335–4355. [Google Scholar]
- Maire, F.; Coadou, G.; Cravello, L.; Lange, C.M. Traveling wave ion mobility mass spectrometry study of low generation polyamidoamine dendrimers. J. Am. Soc. Mass Spectrom. 2013, 24, 238–248. [Google Scholar]
- Gidden, J.; Baker, E.S.; Ferzoco, A.; Bowers, M.T. Structural motifs of DNA complexes in the gas phase. Int. J. Mass Spectrom. 2005, 240, 183–193. [Google Scholar]
- Ferreira, R.; Marchand, A.; Gabelica, V. Mass spectrometry and ion mobility spectrometry of G-quadruplexes. A study of solvent effects on dimer formation and structural transitions in the telomeric DNA sequence d(TAGGGTTAGGGT). Methods 2012, 57, 56–63. [Google Scholar]
- Smiljanic, D.; Wesdemiotis, C. Ternary complexes of poly(ethylene imine), single-stranded oligodeoxynucleotides and glutamic acid moieties. Int. J. Mass Spectrom. 2012, 316, 235–243. [Google Scholar]
- Smiljanic, D.; Wesdemiotis, C. Non-covalent complexes between single-stranded oligodeoxynucleotides and poly(ethylene imine). Int. J. Mass Spectrom. 2011, 304, 148–153. [Google Scholar]
- Schalley, C.A; Verhaelen, C.; Klärner, F.G.; Hahn, U.; Vögtle, F. Gas-phase host—guest chemistry of dendritic viologens and molecular tweezers: A remarkably strong effect on dication stability. Angew. Chem. Int. Ed. Engl. 2005, 44, 477–480. [Google Scholar]
- Loontiens, F.G.; Regenfuss, P.; Zechel, A.; Dumortier, L.; Clegg, R.M. Binding characteristics of Hoechst 33258 with calf thymus DNA, poly[d(A-T)], and d(CCGGAATTCCGG): Multiple stoichiometries and determination of tight binding with a wide spectrum of site affinities. Biochemistry 1990, 29, 9029–9039. [Google Scholar]
- Rosu, F.; Gabelica, V.; Houssier, C.; de Pauw, E. Determination of affinity, stoichiometry and sequence selectivity of minor groove binder complexes with double-stranded oligodeoxynucleotides by electrospray ionization mass spectrometry. Nucleic Acids Res. 2002, 30, 1–9. [Google Scholar]
- Dong, X.; Xu, Y.; Afonso, C.; Jiang, W.; Laronze, J.Y.; Wen, R.; Tabet, J.C. Non-covalent complexes between bis-beta-carbolines and double-stranded DNA: A study by electrospray ionization FT-ICR mass spectrometry (I). Bioorg. Med. Chem. Lett. 2007, 17, 2549–2553. [Google Scholar]
- Wan, K.X.; Shibue, T.; Gross, M.L. Non-Covalent Complexes between DNA-Binding Drugs and Double-Stranded Oligodeoxynucleotides: A Study by ESI Ion-Trap Mass Spectrometry. J. Am. Chem. Soc. 2000, 122, 300–307. [Google Scholar]
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar]
- Leriche, E.D.; Afonso, C.; Lange, C.M.; Grossel, M.C.; Truong, L.; Coadou, G.; Oulyadi, H.; Loutelier-Bourhis, C. Glycine-modified polyamidoamine dendrimers: Synthesis and structural characterization using nuclear magnetic resonance, ion-mobility mass spectrometry and capillary electrophoresis. RSC Adv. 2014, 4, 1744–1753. [Google Scholar]
- Ma, J.C.; Dougherty, D.A. The Cation−π Interaction. Chem. Rev. 1997, 97, 1303–1324. [Google Scholar]
- Rosu, F.; Pirotte, S.; de Pauw, E.; Gabelica, V. Positive and negative ion mode ESI-MS and MS/MS for studying drug–DNA complexes. Int. J. Mass Spectrom. 2006, 253, 156–171. [Google Scholar]
- Daniel, J.M.; McCombie, G.; Wendt, S.; Zenobi, R. Mass spectrometric determination of association constants of adenylate kinase with two noncovalent inhibitors. J. Am. Soc. Mass Spectrom. 2003, 14, 442–448. [Google Scholar]
- Sannes-Lowery, K.A; Griffey, R.H.; Hofstadler, S. a Measuring dissociation constants of RNA and aminoglycoside antibiotics by electrospray ionization mass spectrometry. Anal. Biochem. 2000, 280, 264–271. [Google Scholar]
- Mcluckey, S.A.; van Berkel, G.J.; Glish, G.L. Tandem mass spectrometry of small, multiply charged oligonucleotides. J. Am. Soc. Mass Spectrom. 1992, 3, 60–70. [Google Scholar]
- Vincent, T.J.; Dole, R.; Lange, C.M. Gas-phase fragmentation of half- and first-generation polyamidoamine dendrimers by electrospray mass spectrometry using a quadrupole ion trap. Rapid Commun. Mass Spectrom. 2008, 22, 363–372. [Google Scholar]
- Leriche, E.; Hubert-roux, M.; Grossel, M.C.; Lange, C.M.; Afonso, C.; Loutelier-Bourhis, C. Direct TLC/MALDI–MS coupling for modified polyamidoamine dendrimers analyses. Anal. Chim. Acta 2014, 808, 144–150. [Google Scholar]
- Pringle, S.D.; Giles, K.; Wildgoose, J.L.; Williams, J.P.; Slade, S.E.; Thalassinos, K.; Bateman, R.H.; Bowers, M.T.; Scrivens, J.H. An investigation of the mobility separation of some peptide and protein ions using a new hybrid quadrupole/travelling wave IMS/oa-ToF instrument. Int. J. Mass Spectrom. 2007, 261, 1–12. [Google Scholar]
- Sample Availability: All the PAMAMs were synthesized in the laboratory in the mg scale and are not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leriche, E.-D.; Hubert-Roux, M.; Afonso, C.; Lange, C.M.; Grossel, M.C.; Maire, F.; Loutelier-Bourhis, C. Investigation of Dendriplexes by Ion Mobility-Mass Spectrometry. Molecules 2014, 19, 20731-20750. https://doi.org/10.3390/molecules191220731
Leriche E-D, Hubert-Roux M, Afonso C, Lange CM, Grossel MC, Maire F, Loutelier-Bourhis C. Investigation of Dendriplexes by Ion Mobility-Mass Spectrometry. Molecules. 2014; 19(12):20731-20750. https://doi.org/10.3390/molecules191220731
Chicago/Turabian StyleLeriche, Emma-Dune, Marie Hubert-Roux, Carlos Afonso, Catherine M. Lange, Martin C. Grossel, Florian Maire, and Corinne Loutelier-Bourhis. 2014. "Investigation of Dendriplexes by Ion Mobility-Mass Spectrometry" Molecules 19, no. 12: 20731-20750. https://doi.org/10.3390/molecules191220731
APA StyleLeriche, E.-D., Hubert-Roux, M., Afonso, C., Lange, C. M., Grossel, M. C., Maire, F., & Loutelier-Bourhis, C. (2014). Investigation of Dendriplexes by Ion Mobility-Mass Spectrometry. Molecules, 19(12), 20731-20750. https://doi.org/10.3390/molecules191220731






