Two New N-Oxide Alkaloids from Stemona cochinchinensis
Abstract
:1. Introduction
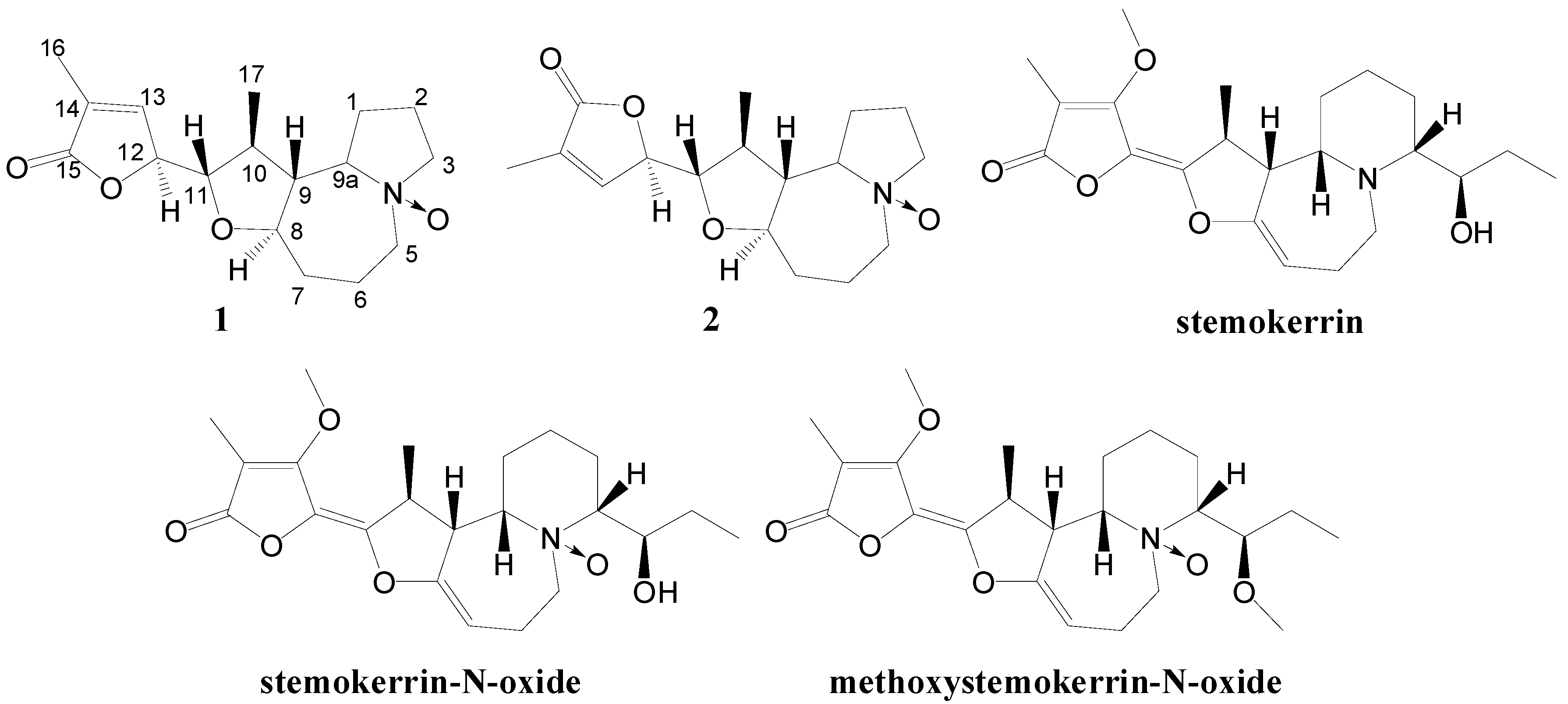
2. Results and Discussion
| 1 | 2 | |||
|---|---|---|---|---|
| δH, J (Hz) | δC | δH, J (Hz) | δC | |
| 1 | 1.82 m | 20.5 | 1.81 m | 20.6 |
| 1 | 1.57 m | 1.57 m | ||
| 2 | 1.91 m | 24.6 | 1.91 m | 24.8 |
| 2 | 1.37 m | 1.37 m | ||
| 3 | 3.48 m (2H) | 70.2 | 3.48 m (2H) | 70.4 |
| 5α | 3.95 m | 66.0 | 3.95 m | 66.0 |
| 5β | 3.60 m | 3.62 m | ||
| 6 | 1.54 m | 18.8 | 1.54 m | 18.9 |
| 6 | 1.37 m | 1.37 m | ||
| 7 | 2.06 m | 32.4 | 2.06 m | 32.1 |
| 7 | 1.27 m | 1.27 m | ||
| 8 | 3.94 m | 83.0 | 4.05 m | 82.8 |
| 9 | 2.11 m | 48.7 | 2.17 m | 49.0 |
| 9a | 3.89 m | 80.6 | 3.89 m | 80.5 |
| 10 | 2.22 m | 40.2 | 2.20 m | 39.6 |
| 11 | 3.72 m | 84.6 | 3.82 m | 84.6 |
| 12 | 4.88 m | 80.5 | 4.93 m | 80.2 |
| 13 | 6.98 q (1.7) | 145.8 | 7.07 q (1.7) | 145.8 |
| 14 | 131.1 | 131.3 | ||
| 15 | 174.2 | 173.9 | ||
| 16 | 1.92 d (1.7) | 10.8 | 1.92 d (1.7) | 10.7 |
| 17 | 1.15 d (6.1) | 14.9 | 1.09 d (6.1) | 16.3 |

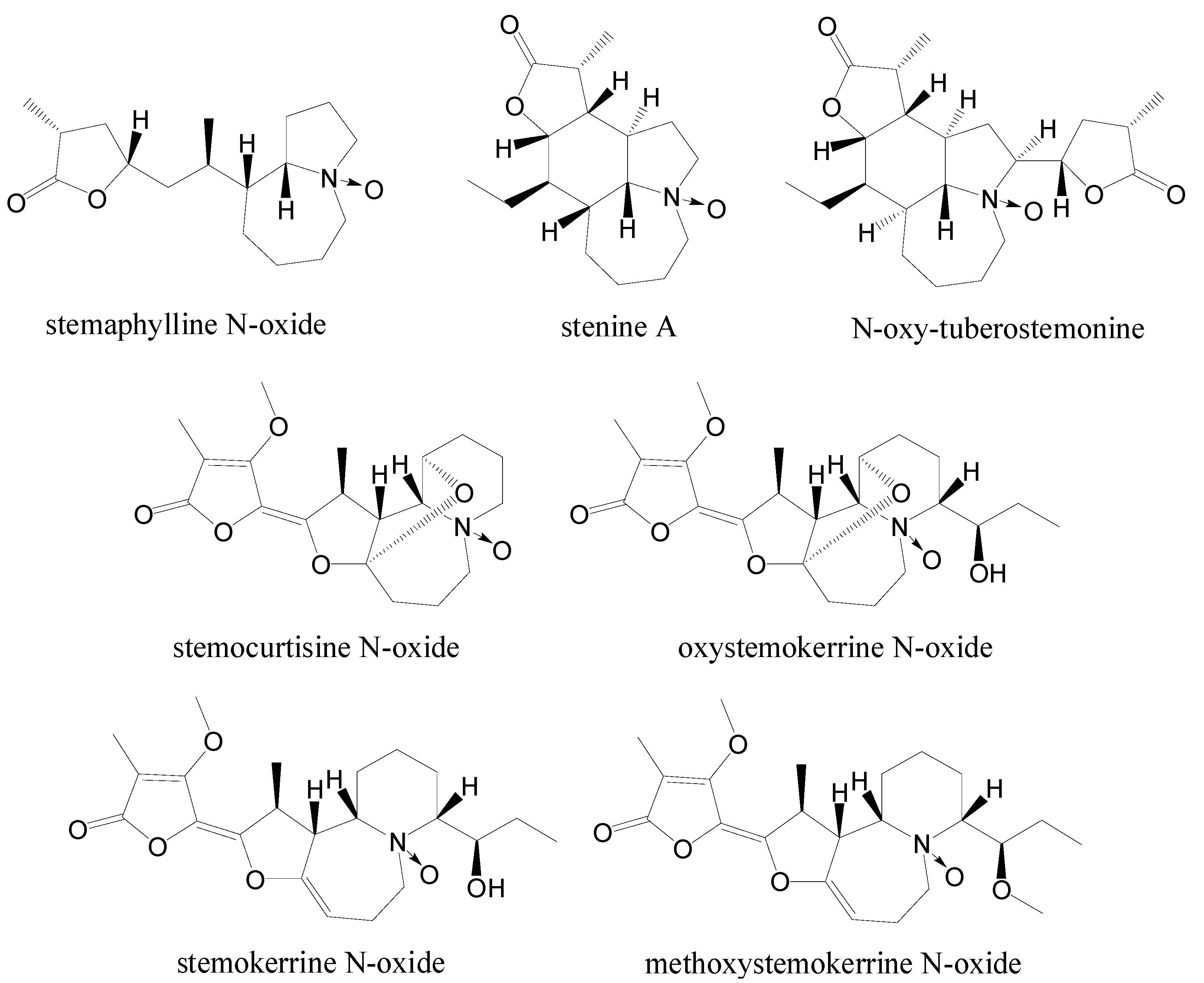
| Compound | δH | δC | Ref | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H-3 | Δ | H-5 | Δ | H-9a | Δ | C-3 | Δ | C-5 | Δ | C-9a | Δ | ||
| isoneostemocochinine N-oxide | 3.48 (2H) | 0.20 | 3.95/3.60 | 0.47/0.64 | 3.89 | 0.25 | 70.2 | 18.2 | 66.0 | 16.2 | 80.6 | 19.9 | [8] |
| isoneostemocochinine | 3.28 (2H) | 3.48/2.96 | 3.64 | 52.0 | 49.8 | 60.7 | |||||||
| neostemocochinine N-oxide | 3.48 (2H) | 0.20 | 3.95/3.62 | 0.46/0.64 | 3.89 | 0.25 | 70.4 | 18.3 | 66.0 | 16.1 | 80.5 | 19.8 | [8] |
| neostemocochinine | 3.28 (2H) | 3.49/2.98 | 3.64 | 52.1 | 49.9 | 60.7 | |||||||
| stemaphylline N-oxide | 3.57 (2H) | 0.56/1.06 | 3.33 (2H) | 0.39/0.82 | 4.65 | 1.72 | 71.0 | 16.7 | 67.2 | 14.9 | 81.6 | 16.8 | [11] |
| stemaphylline | 3.01/2.51 | 2.94/2.51 | 2.93 | 54.3 | 52.3 | 64.8 | [11] | ||||||
| stenine A | 3.79/3.69 | 0.14/0.22 | 3.87/3.54 | 0.74/0.58 | 3.69 | 1.62 | 72.2 | 19.5 | 67.7 | 15.4 | 84.3 | 19.0 | [12] |
| stenine B | 3.65/3.47 | 3.13/2.96 | 2.07 | 52.7 | 52.3 | 65.3 | [12] | ||||||
| N-oxytuberostemonine | 3.67 | 0.24 | 3.78/3.52 | 0.31/0.85 | 4.01 | 0.94 | 77.5 | 12.5 | 69.7 | 21.6 | 88.9 | 25.3 | [13] |
| tuberostemonine | 3.43 | 3.47/2.67 | 3.07 | 65.0 | 48.1 | 63.6 | [14] | ||||||
| H-4 | Δ | H-6 | Δ | H-10a | Δ | C-4 | Δ | C-6 | Δ | C-10a | Δ | ||
| stemocurtisine N-oxide | 3.44/3.29 | 0.42/0.42 | 4.12/3.01 | 0.74/0.05 | 3.66 | 0.22 | 69.6 | 16.0 | 66.4 | 13.4 | 82.3 | 20.3 | [9] |
| stemocurtisine | 3.02/2.87 | 3.38/2.96 | 3.44 | 53.6 | 53.0 | 62.0 | [15] | ||||||
| oxystemokerrine N-oxide | 3.31 | 0.72 | 4.04/3.55 | 0.68/0.49 | 3.93 | 0.43 | 79.4 | 13.6 | 61.9 | 17.7 | 85.4 | 19.0 | [4] |
| oxystemokerrine | 2.59 | 3.36/3.06 | 3.50 | 65.8 | 44.2 | 66.4 | [4] | ||||||
| stemokerrine N-oxide | 3.22 | 0.62 | 3.46/3.42 | 0/76/0.86 | 3.22 | 0.39 | 81.9 | 12.0 | 54.1 | 14.4 | 78.3 | 15.9 | [7] |
| methoxystemokerrine N-oxide | 3.26 | 0.66 | 3.36/2/63 | 0.66/0.07 | 3.21 | 0.38 | 84.3 | 14.4 | 56.2 | 16.5 | 78.5 | 16.1 | [4] |
| stemokerrine | 2.60 | 2.70/2.56 | 2.83 | 69.9 | 39.7 | 62.4 | [4] | ||||||
3. Experimental Section
3.1. General Information
3.2. HPLC Conditions
3.3. Plant Material
3.4. Extraction and Isolation
3.5. Anti-Tussive Activity of Stemokerrin
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Qin, G.W.; Xu, R.S. Recent advances on bioactive natural products from Chinese medicinal plants. Med. Res. Rev. 1998, 18, 375–382. [Google Scholar]
- Greger, H. Structural relationships, distribution and biological activities of stemona alkaloids. Planta Med. 2006, 72, 99–113. [Google Scholar]
- Pilli, R.A.; Rosso, G.B.; de Oliveira Mda, C. The chemistry of Stemona alkaloids: An update. Nat. Prod. Rep. 2010, 27, 1908–1937. [Google Scholar]
- Kaltenegger, E.; Brem, B.; Mereiter, K.; Kalchhauser, H.; Kahlig, H.; Hofer, O.; Vajrodaya, S.; Greger, H. Insecticidal pyrido[1,2-a]azepine alkaloids and related derivatives from Stemona species. Phytochemistry 2003, 63, 803–816. [Google Scholar]
- Mungkornasawakul, P.; Pyne, S.G.; Jatisatienr, A.; Supyen, D.; Jatisatienr, C.; Lie, W.; Ung, A.T.; Skelton, B.W.; White, A.H. Phytochemical and larvicidal studies on Stemona curtisii: Structure of a new pyrido[1,2-a]azepine Stemona alkaloid. J. Nat. Prod. 2004, 67. [Google Scholar]
- Lin, L.G.; Tang, C.P.; Dien, P.H.; Xu, R.S.; Ye, Y. Cochinchistemonine, a novel skeleton alkaloid from Stemona cochinchinensis. Tetrahedron Lett. 2007, 48, 1559–1561. [Google Scholar]
- Wang, Y.Z.; Tang, C.P.; Dien, P.H.; Ye, Y. Alkaloids from the roots of Stemona saxorum. J. Nat. Prod. 2007, 70, 1356–1359. [Google Scholar]
- Lin, L.G.; Dien, P.H.; Tang, C.P.; Ke, C.Q.; Yang, X.Z.; Ye, Y. Alkaloids from the roots of Stemona cochinchinensis. Helv. Chim. Acta 2007, 90, 2167–2175. [Google Scholar]
- Chaiyong, S.; Jatisatienr, A.; Mungkornasawakul, P.; Sastraruji, T.; Pyne, S.G.; Ung, A.T.; Urathamakul, T.; Lie, W. Phytochemical investigations of Stemona curtisii and synthetic studies on stemocurtisine alkaloids. J. Nat. Prod. 2010, 73, 1833–1838. [Google Scholar]
- Dien, P.H.; Lin, L.G.; Tang, C.P.; Ke, C.Q.; Ye, Y. Bisbenzopyrans and alkaloids from the roots of Stemona cochinchinensis. Nat. Prod. Res. 2008, 22, 915–920. [Google Scholar]
- Mungkornasawakul, P.; Chaiyong, S.; Sastraruji, T.; Jatisatienr, A.; Jatisatienr, C.; Pyne, S.G.; Ung, A.T.; Korth, J.; Lie, W. Alkaloids from the roots of Stemona aphylla. J. Nat. Prod. 2009, 72, 848–851. [Google Scholar]
- Lai, D.H.; Yang, Z.D.; Xue, W.W.; Sheng, J.; Shi, Y.; Yao, X.J. Isolation, characterization and acetylcholinesterase inhibitory activity of alkaloids from roots of Stemona sessilifolia. Fitoterapia 2013, 89, 257–264. [Google Scholar]
- Wenhan, L.; Hongzheng, F. Three new alkaloids from the roots of Stemona tuberosa lour. J. Chin. Pharm. Sci. 1999, 8, 1–7. [Google Scholar]
- Schinnerl, J.; Kaltenegger, E.; Pacher, T.; Vajrodaya, S.; Hofer, O.; Greger, H. New pyrrolo[1,2-a]azepine type alkaloids from Stemona and Stichoneuron (Stemonaceae). Monatsh. Chem. 2005, 136, 1671–1680. [Google Scholar]
- Mungkornasawakul, P.; Pyne, S.G.; Jatisatienr, A.; Supyen, D.; Lie, W.; Ung, A.T.; Skelton, B.W.; White, A.H. Stemocurtisine, the first pyrido[1,2-a]azapine Stemona alkaloid. J. Nat. Prod. 2003, 66, 980–982. [Google Scholar]
- Lin, L.G.; Zhong, Q.X.; Cheng, T.Y.; Tang, C.P.; Ke, C.Q.; Lin, G.; Ye, Y. Stemoninines from the roots of Stemona tuberosa. J. Nat. Prod. 2006, 69, 1051–1054. [Google Scholar]
- Sample Availability: Samples of the compounds stemokerrin and stemokerrin-N-oxide are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, L.; Bao, H.; Wang, A.; Tang, C.; Dien, P.-H.; Ye, Y. Two New N-Oxide Alkaloids from Stemona cochinchinensis. Molecules 2014, 19, 20257-20265. https://doi.org/10.3390/molecules191220257
Lin L, Bao H, Wang A, Tang C, Dien P-H, Ye Y. Two New N-Oxide Alkaloids from Stemona cochinchinensis. Molecules. 2014; 19(12):20257-20265. https://doi.org/10.3390/molecules191220257
Chicago/Turabian StyleLin, Ligen, Han Bao, Anqi Wang, Chunping Tang, Pham-Huu Dien, and Yang Ye. 2014. "Two New N-Oxide Alkaloids from Stemona cochinchinensis" Molecules 19, no. 12: 20257-20265. https://doi.org/10.3390/molecules191220257
APA StyleLin, L., Bao, H., Wang, A., Tang, C., Dien, P.-H., & Ye, Y. (2014). Two New N-Oxide Alkaloids from Stemona cochinchinensis. Molecules, 19(12), 20257-20265. https://doi.org/10.3390/molecules191220257







