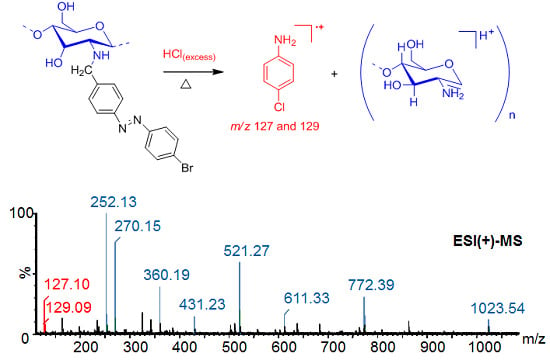
ESI(+)-MS and GC-MS Study of the Hydrolysis of N-Azobenzyl Derivatives of Chitosan
Abstract
:1. Introduction
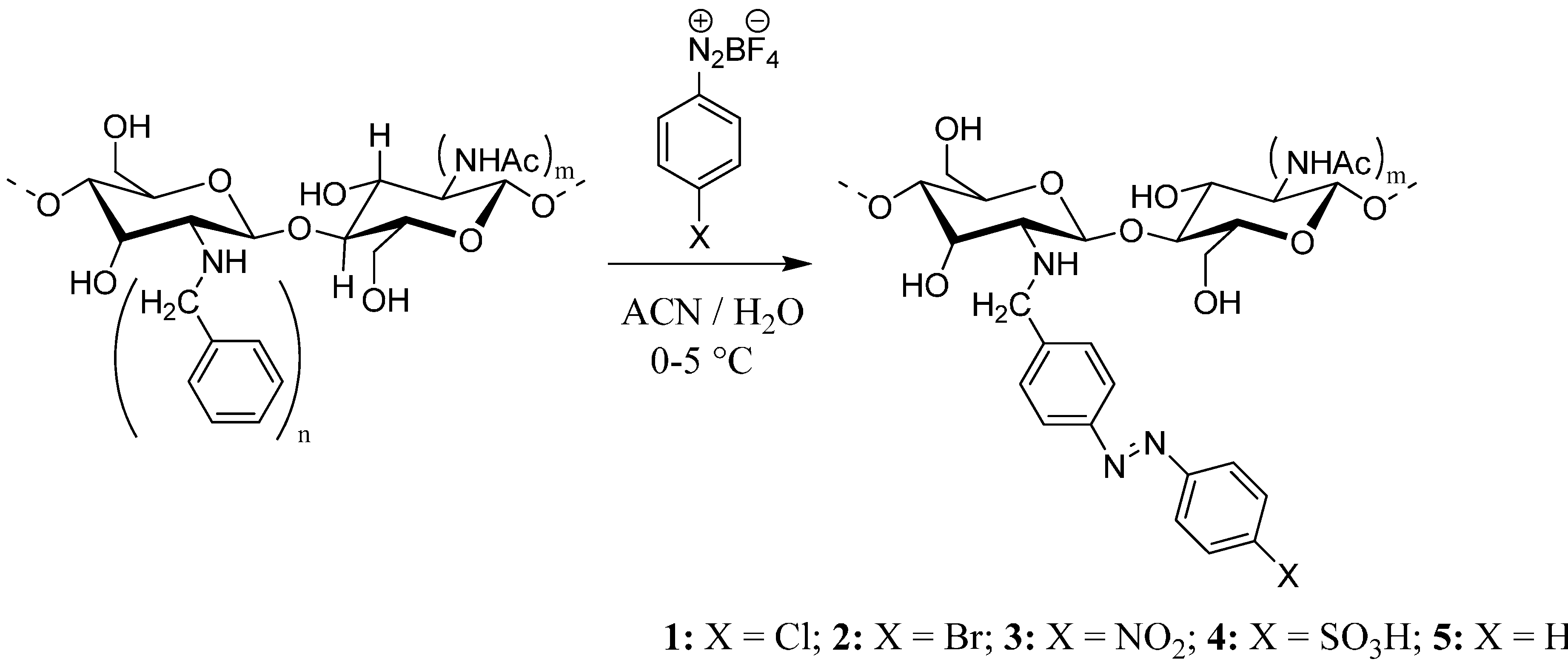
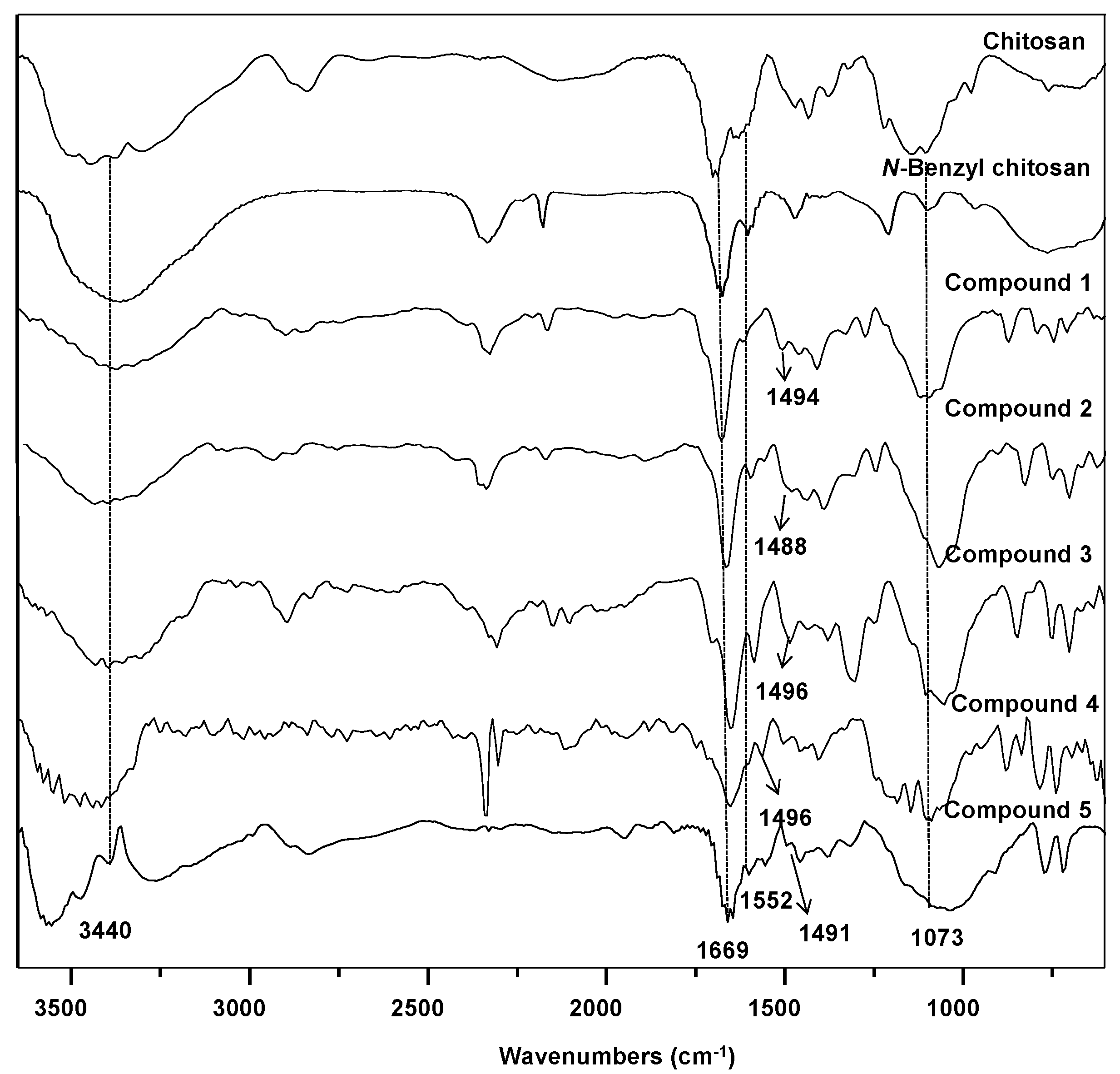
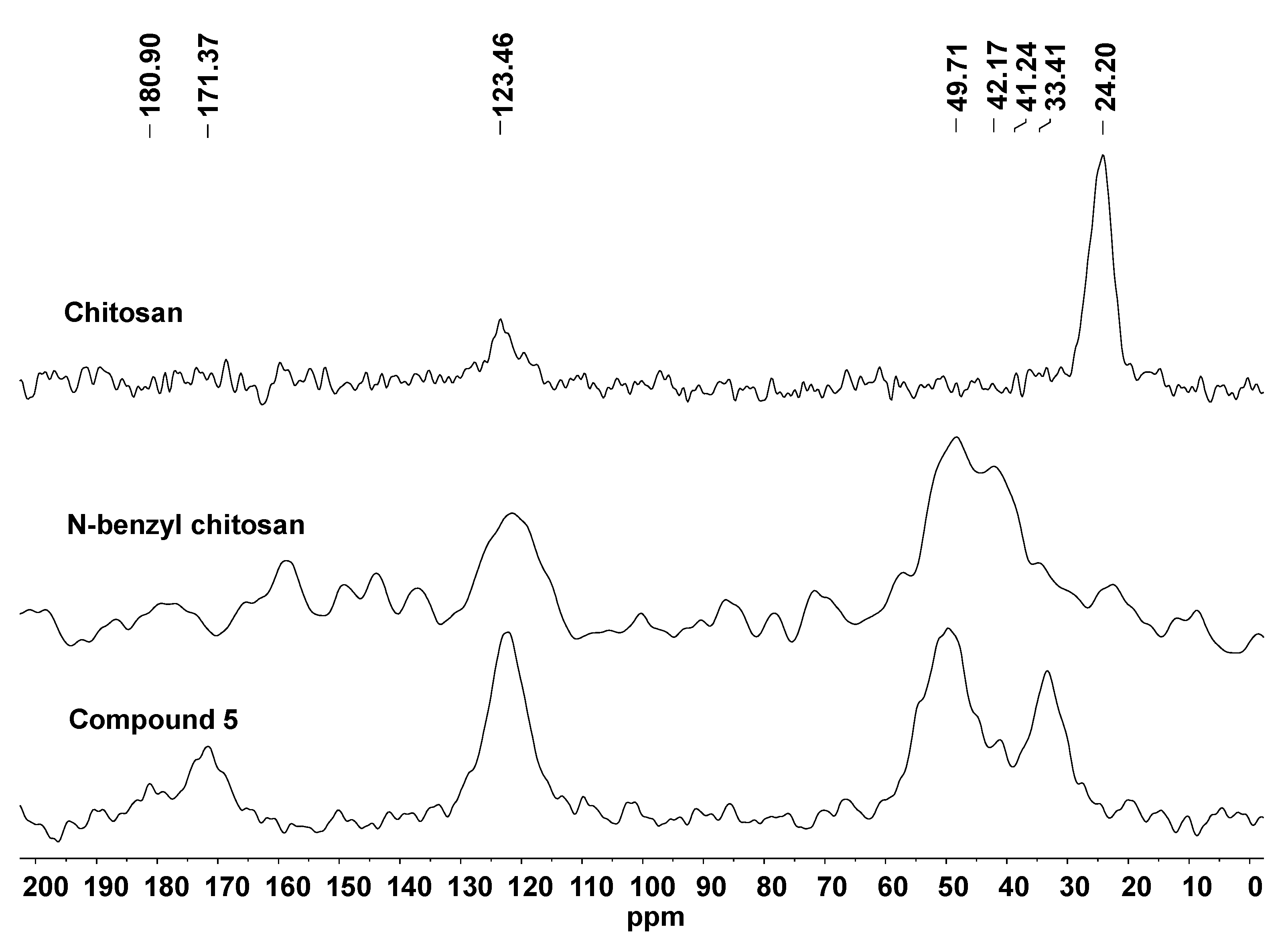
2. Results and Discussion
| Oligomers | Calcd. m/z [M + H]+ | Found m/z [M + H]+ | Found m/z [M-H2O + H]+ |
|---|---|---|---|
| GlcN | 180.06 | 180.16 | 162.14 |
| GlcN2 | 341.06 | 341.29 | 323.29 |
| GlcN3 | 502.06 | 502.82 | 484.82 |
| GlcN4 | 663.06 | 663.57 | 645.28 |
| GlcN5 | 824.06 | 824.70 | - |
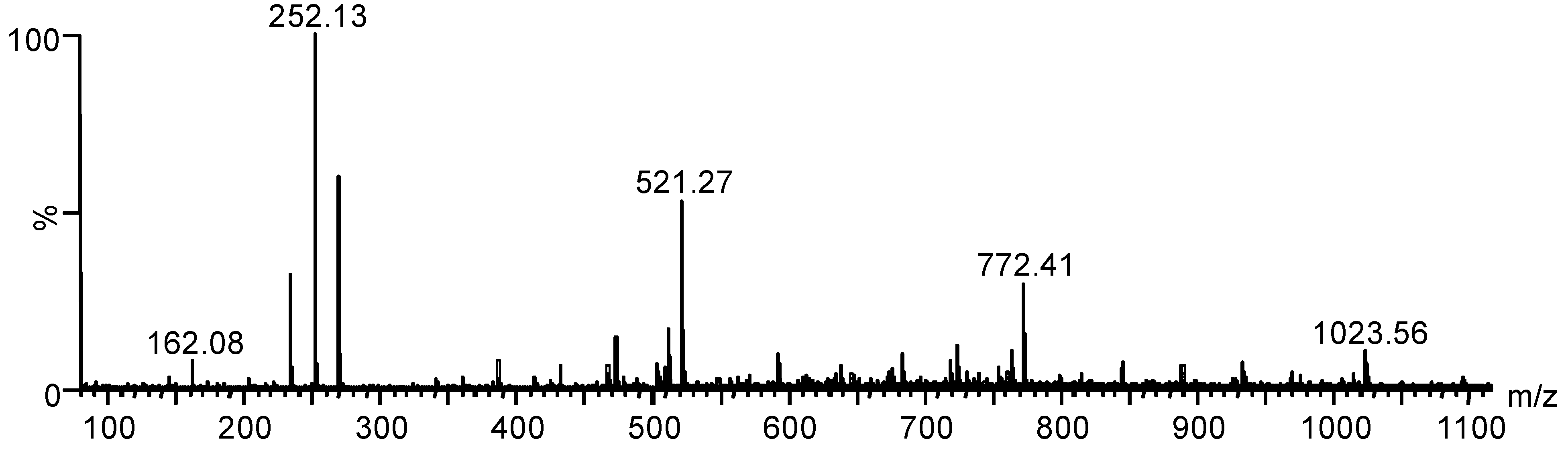
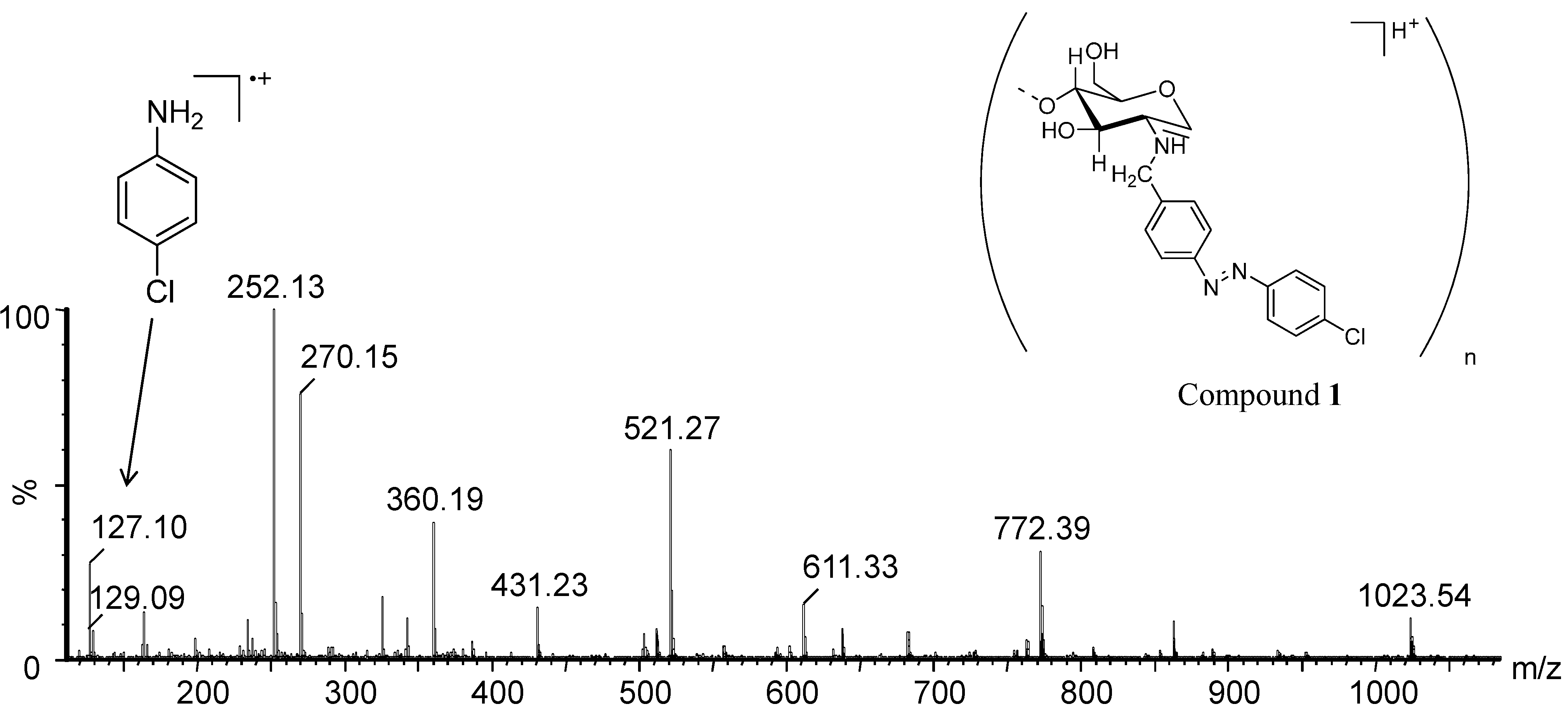
| Ion | m/z Calcd. | m/z Found |
|---|---|---|
| [GlcN-H2O + H]+ | 162.06 | 162.08 |
| [GlcNBn-H2O + H]+ | 252.06 | 252.13 |
| [(GlcNBn)2 + H]+ | 521.06 | 521.27 |
| [(GlcNBn)3 + H]+ | 772.06 | 772.39 |
| [(GlcNBn)4 + H]+ | 1023.06 | 1023.54 |
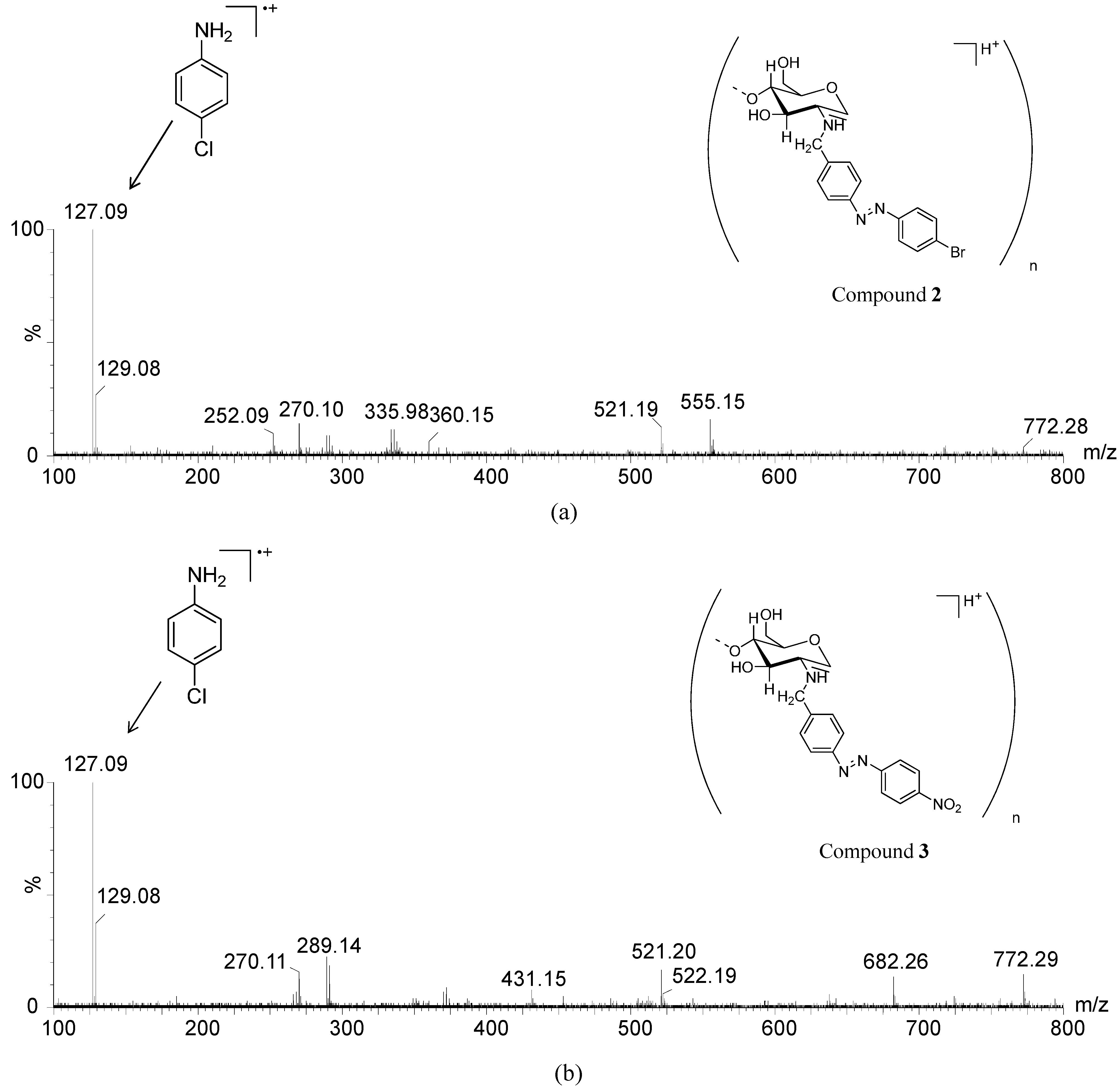
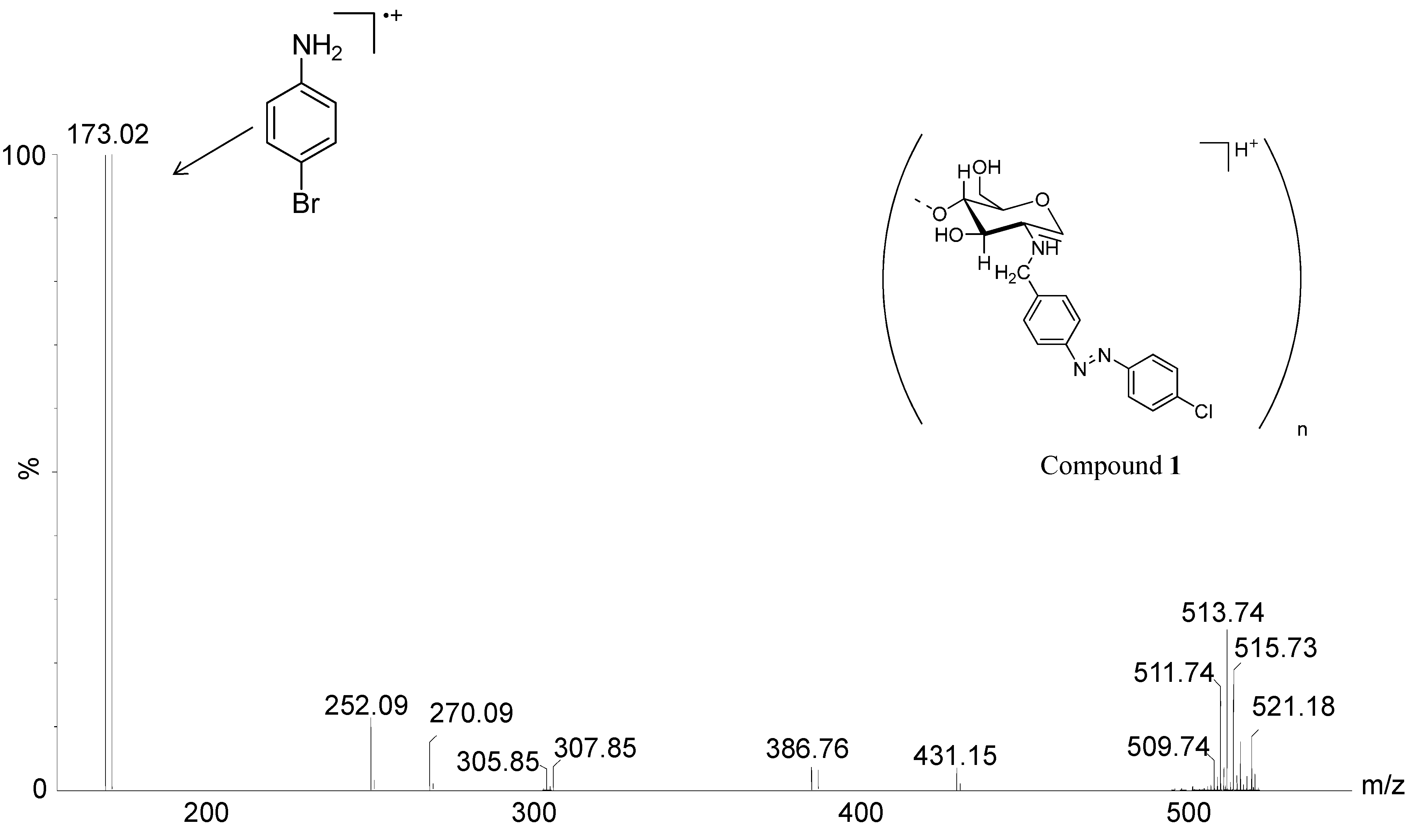

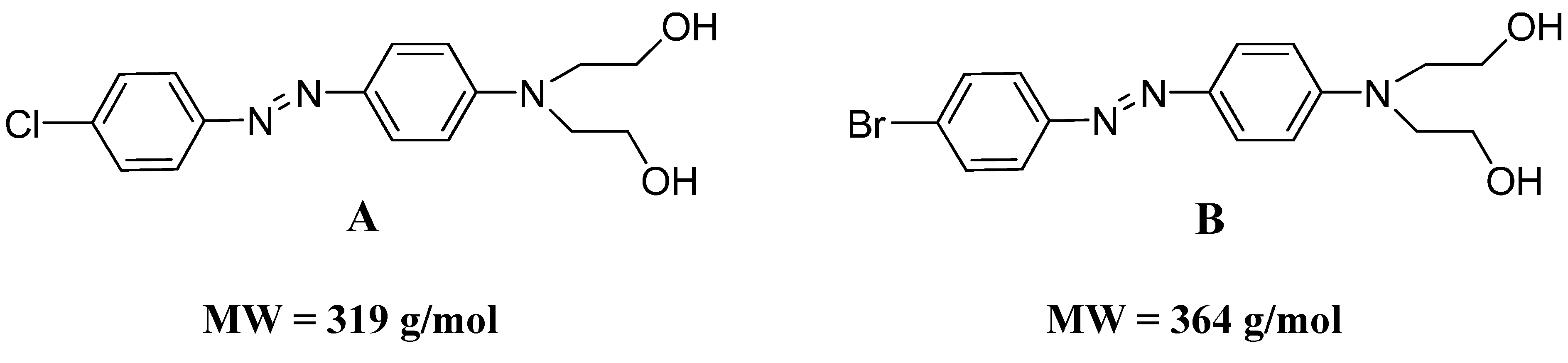
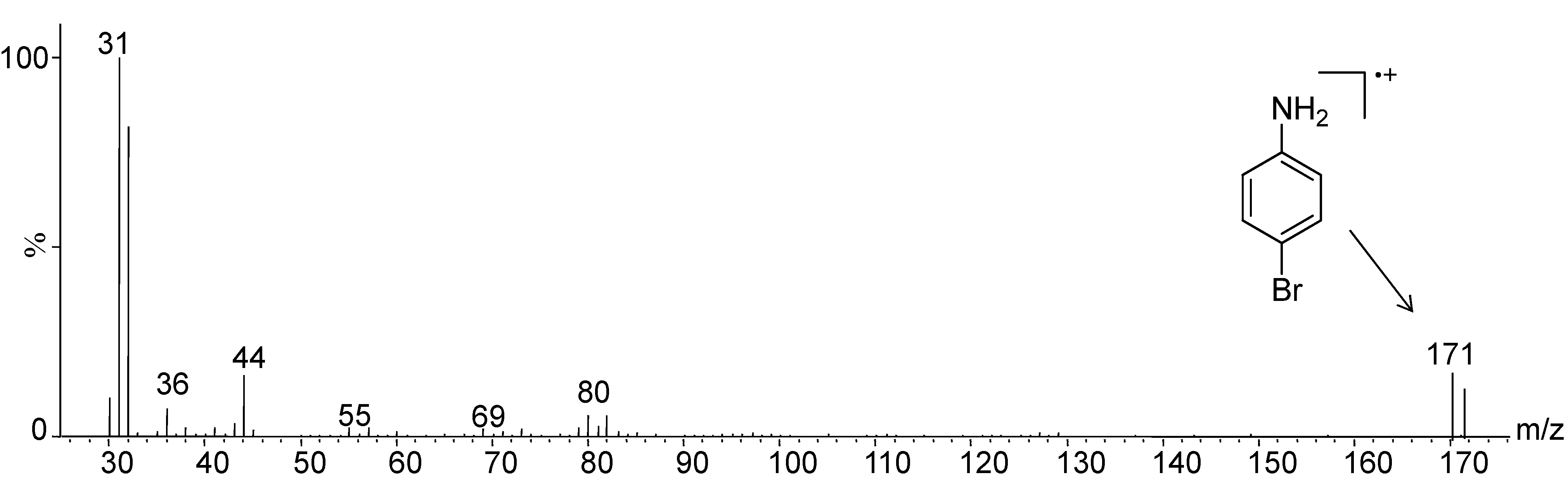
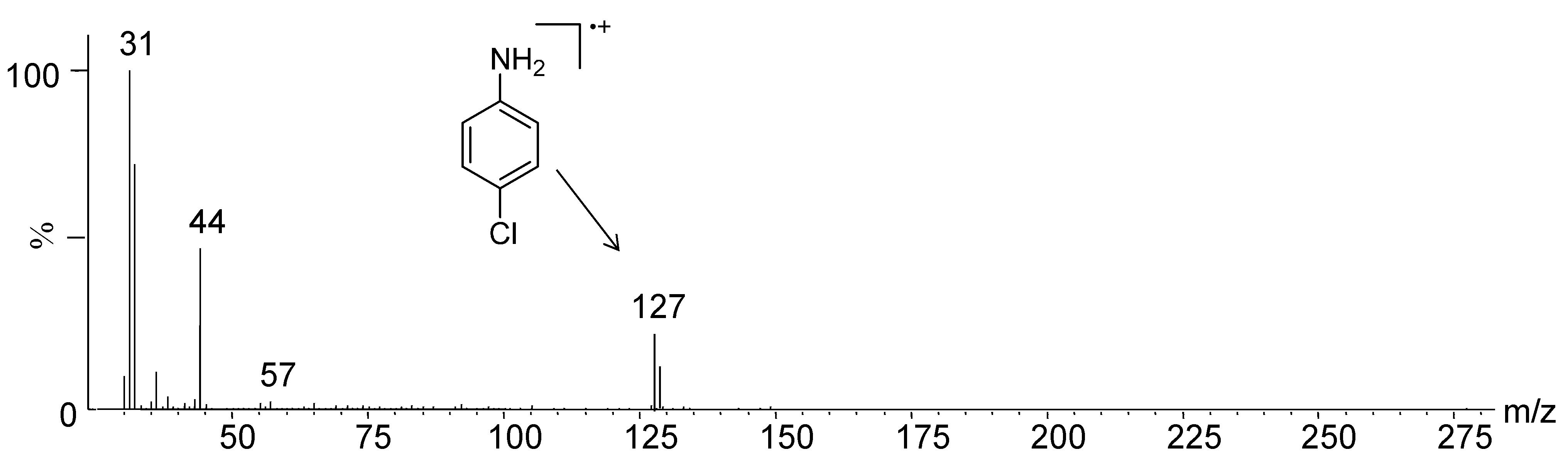
| Compounds | % of Cu2+ Chelated | mg/g of Cu2+ Chelated |
|---|---|---|
| Chitosan | 75 | 120 |
| N-benzyl chitosan | 14 | 23 |
| 1 | 14 | 23 |
| 2 | 11 | 18 |
| 3 | 9 | 15 |
| 4 | 10 | 16 |
| 5 | 9 | 15 |
3. Experimental Section
3.1. General Information
3.2. Reductive N-Alkylation of Chitosan
3.3. General Procedure for Azo-Coupling
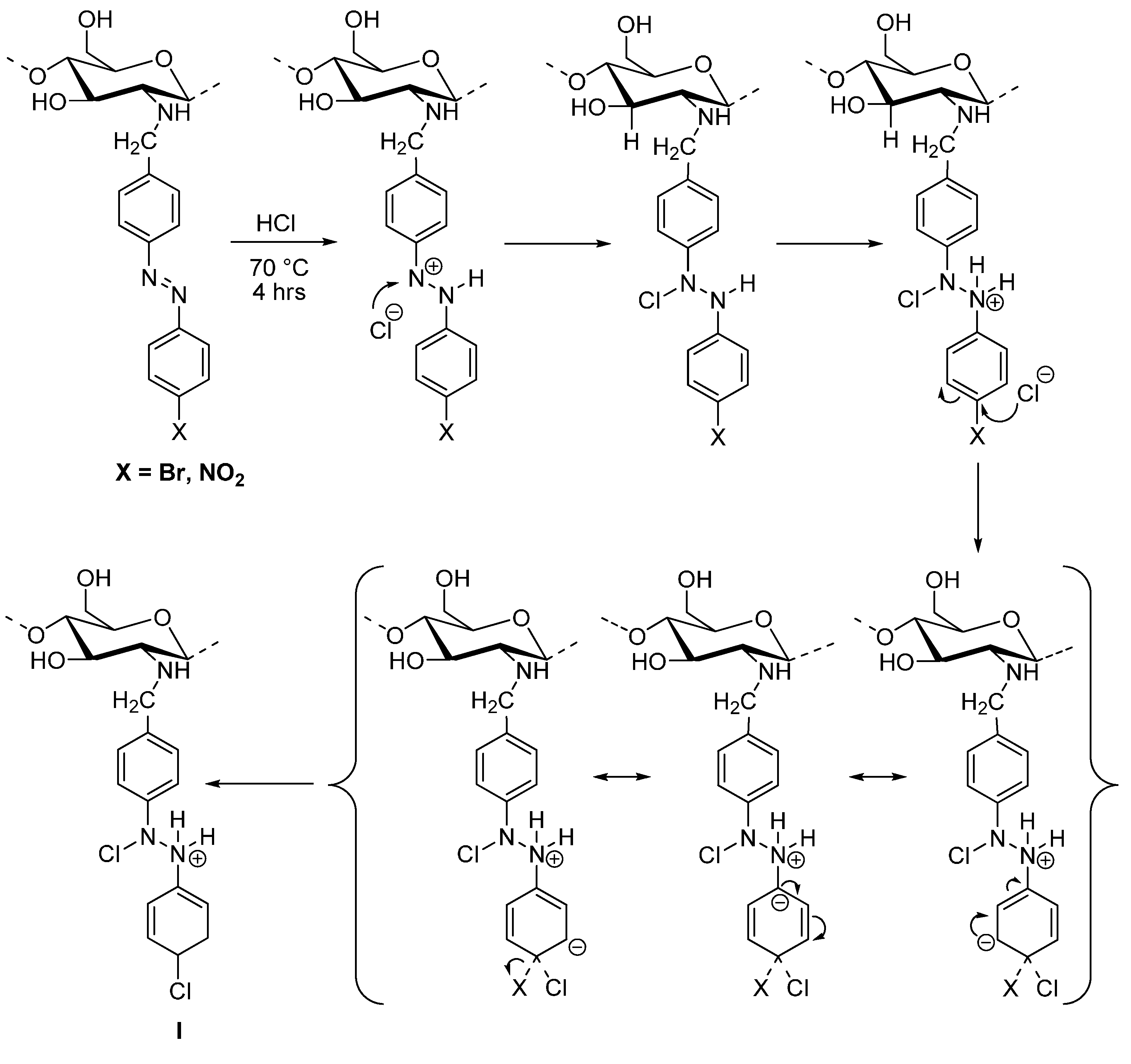
3.4. General Procedure for Acid Hydrolysis of Chitosan and Derivatives
3.5. General Procedure for Copper Complexes Formation
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fernández, R.; Ramos, J.A.; Espósito, L.; Tercjak, A.; Mondragon, I. Reversible Optical Storage Properties of Nanostructured Epoxy-Based Thermosets Modified with Azobenzene Units. Macromolecules 2011, 44, 9738–9746. [Google Scholar]
- Manickasundaram, S.; Kannan, P.; Hassan, Q.M.A.; Palanisamy, P.K. Azo dye based poly(alkyloxymethacrylate)s and their spacer effect on optical data storage. J. Mater. Sci.-Mater. Electron. 2008, 19, 1045–1053. [Google Scholar]
- Natansohn, A.; Rochon, P.; Gosselin, J.; Xie, S. Azo Polymers for Reversible Optical Storage. 1. Poly[4'-[[2-(Acryloyloxy)Ethyl]Ethylamino]-4-Nitroazobenzene]. Macromolecules 1992, 25, 2268–2273. [Google Scholar]
- Shihab, N.L.; Intedhar, K.M. Synthesis of some novel heterocyclic azo dyes for a cridine derivatives and evaluation of their antibacterial activities. J. Chem. Pharm. Res. 2013, 5, 345–354. [Google Scholar]
- Pandey, V.K.; Negi, H.S. Synthesis and Biological activity of some new benzophenothiazines. Indian Chem. Soc. 2003, 42, 206–210. [Google Scholar]
- Dhingra, V.; Bhatwadekar, R.; Pendse, S. Synthesis and Biological Activity of Some 1-Isonicotinoyl- 3,5-Diphenyl-4-4Substituted Phenyl azo Pyrazoles. Asian J. Chem. 1993, 5, 515–522. [Google Scholar]
- Suvarna, K.; Swain, S.P.; Gandhi, A.M. Synthesis and evaluation of novel benzothiazole derivatives against human cervical cancer cell lines. Indian J. Pharm. Sci. 2007, 69, 46–50. [Google Scholar]
- Racane, L.; Stojkovic, R.; Kulenovic, V.T.; Zamola, G.K. Synhesis and antitumor evaluation of novel derivatives of 6-amino-2-phenylbenzothiazoles. Molecules 2006, 11, 325–333. [Google Scholar]
- Bradshaw, T.D.; Stevens, M.F.G.; Westwall, A.D. The discovery of the potent and selective antitumour agent 2-(4-amino-3-thylphenyl)benzothiazole (DF 203) and related compounds. Curr. Med. Chem. 2001, 8, 203–210. [Google Scholar]
- Venkatesh, P.; Pandeya, S.N. Synthesis, characterisation and anti-inflammatory activity of some 2-amino benzothiazole derivatives. Int. J. ChemTech. Res. 2009, 1, 1354–1358. [Google Scholar]
- Chavan, M.S.; Sant, V.P.; Nagarsenker, M.S. Azo-containing urethane analogues for colonic drug delivery: Synthesis, characterization and in-vitro evaluation. J. Pharm. Pharmacol. 2001, 53, 895–900. [Google Scholar]
- Wang, H.; Bao, C.; Li, F.; Kong, X.; Xu, J. Preparation and application of 4-amino-4'-nitro azobenzene modified chitosan as a selective adsorbent for the determination of Au(III) and Pd(II). Microchim. Acta 2010, 168, 99–105. [Google Scholar]
- Wang, H.; Li, C.; Bao, C.; Liu, L.; Liu, X. Adsorption and Determination of Pd(II) and Pt(IV) onto 3'-Nitro-4-amino Azobenzene Modified Chitosan. J. Chem. Eng. Data 2011, 56, 4203–4207. [Google Scholar]
- Guibal, E. Heterogeneous catalysis on chitosan-based materials: A review. Prog. Polym. Sci. 2005, 30, 71–109. [Google Scholar]
- Antony, R.; David, T.S.; Saravanan, K.; Karuppasamy, K.; Balakumar, S. Synthesis, spectrochemical characterisation and catalytic activity of transition metal complexes derived from schiff base modified chitosan. Spectrochim. Acta A 2013, 103, 423–430. [Google Scholar]
- Chang, Y.; Wang, Y.; Zha, F.; Wang, R. Preparation and catalytic properties of chitosan bound schiff base copper complexes. Polym. Adv. Technol. 2004, 15, 284–286. [Google Scholar]
- Wang, X.; Du, Y.; Fan, L.; Liu, H.; Hu, Y. Chitosan-metal complexes as antimicrobial agent: Synthesis, characterization and structure-activity study. Polym. Bull. 2005, 55, 105–113. [Google Scholar]
- Mekahlia, S.; Bouzid, B. Chitosan-copper (II) complex as antibacterial agent: Synthesis, characterization and coordinating bond-activity correlation study. Phys. Procedia 2009, 2, 1045–1053. [Google Scholar]
- Zheng, Y.; Yi, Y.; Qi, Y.; Wang, Y.; Zang, W.; Du, M. Preparation of chitosan-copper complexes and their antitumor activity. Bioorg. Med. Chem. Lett. 2006, 16, 4127–4129. [Google Scholar]
- Brugnerotto, J.; Lizardi, J.; Goycoolea, F.M.; Arguelles-Monal, W.; Desbrière, J.; Rinaudo, M. An infrared investigation in relation with chitin and chitosan characterization. Polymer 2001, 42, 3569–3580. [Google Scholar]
- Kim, S. Chitin, Chitosan, Oligosaccharides and Their Derivatives, Biological Activities and Applications; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Heux, L.; Brugnerotto, J.; Desbrieres, J.; Versali, M.-F.; Rinaudo, M. Solid state NMR for determination of degree of acetylation of chitin and chitosan. Biomacromolecules 2000, 1, 746–751. [Google Scholar]
- Kumirska, J.; Czerwicka, M.; Kaczyński, Z.; Bychowska, A.; Brzozowski, K.; Thöming, J.; Stepnowski, P. Application of Spectroscopic Methods for Structural Analysis of Chitin and Chitosan. Mar. Drugs 2010, 8, 1567–1636. [Google Scholar]
- Sajomsang, W.; Tantayanon, S.; Tangpasuthadol, V.; Thatte, M.; Daly, W.H. Synthesis and characterization of N-benzyl chitosan derivatives. Polym. Prepr. 2006, 47, 294–295. [Google Scholar]
- Oakes, J.; Gratton, P. Kinetic investigations of the oxidation of Methyl Orange and substituted arylazonaphthol dyes by peracids in aqueous solution. J. Chem. Soc. Perkin Trans. 2 1998, 2, 2563–2568. [Google Scholar]
- Mielczarski, J.A.; Atenas, G.M.; Mielczarski, E. Role of iron surface oxidation layers in decomposition of azo-dye water pollutants in weak acidic solutions. Appl. Catal. B Environ. 2005, 56, 289–303. [Google Scholar]
- Tjosaas, F.; Fiksdahl, A. A simple synthetic route to methyl 3-fluoropyridine-4-carboxylate by nucleophilic aromatic substitution. Molecules 2006, 11, 130–133. [Google Scholar]
- Vlasov, V.M. Nucleophilic substitution of the nitro group, fluorine and chlorine in aromatic compounds. Russ. Chem. Rev. 2003, 72, 681–703. [Google Scholar]
- Kanwal, F.; Gul, A.; Jamil, T. Synthesis of Acid Doped Conducting Polyaniline. J. Chem. Soc. Pak. 2007, 29, 553–557. [Google Scholar]
- Stejskal, J.; Gilbert, R.G. Polyaniline. Preparation of a conducting polymer (IUPAC technical report) IUPAC. Pure Appl. Chem. 2002, 74, 857–867. [Google Scholar]
- Vivekanandan, J.; Ponnusamy, V.; Mahudeswaran, A.; Vijayanand, P.S. A Synthesis, characterization and conductivity study of polyaniline prepared by chemical oxidative and electrochemical methods. Arch. Appl. Sci. Res. 2011, 3, 147–153. [Google Scholar]
- Cabrera, J.C.; Cutsem, P.V. Preparation of chitooligosaccharides with degree of polymerization higher than 6 by acid or enzymatic degradation of chitosan. Biochem. Eng. J. 2005, 25, 165–172. [Google Scholar]
- Sample Availability: Samples of the N-Benzyl chitosan and compounds 1–5 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, F.S.; Nascimento, H.D.L.; Magalhães, A.; Peter, M.G.; Bataglion, G.A.; Eberlin, M.N.; González, E.R.P. ESI(+)-MS and GC-MS Study of the Hydrolysis of N-Azobenzyl Derivatives of Chitosan. Molecules 2014, 19, 17604-17618. https://doi.org/10.3390/molecules191117604
Pereira FS, Nascimento HDL, Magalhães A, Peter MG, Bataglion GA, Eberlin MN, González ERP. ESI(+)-MS and GC-MS Study of the Hydrolysis of N-Azobenzyl Derivatives of Chitosan. Molecules. 2014; 19(11):17604-17618. https://doi.org/10.3390/molecules191117604
Chicago/Turabian StylePereira, Fernanda S., Heliara D. L. Nascimento, Alviclér Magalhães, Martin G. Peter, Giovana Anceski Bataglion, Marcos N. Eberlin, and Eduardo R. P. González. 2014. "ESI(+)-MS and GC-MS Study of the Hydrolysis of N-Azobenzyl Derivatives of Chitosan" Molecules 19, no. 11: 17604-17618. https://doi.org/10.3390/molecules191117604
APA StylePereira, F. S., Nascimento, H. D. L., Magalhães, A., Peter, M. G., Bataglion, G. A., Eberlin, M. N., & González, E. R. P. (2014). ESI(+)-MS and GC-MS Study of the Hydrolysis of N-Azobenzyl Derivatives of Chitosan. Molecules, 19(11), 17604-17618. https://doi.org/10.3390/molecules191117604