Glutathione and the Antioxidant Potential of Binary Mixtures with Flavonoids: Synergisms and Antagonisms
Abstract
:1. Introduction
| Compound | Structure |
|---|---|
| Quercetin | 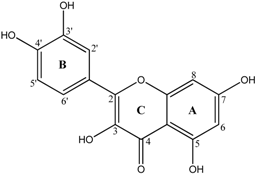 |
| Quercetagetin | 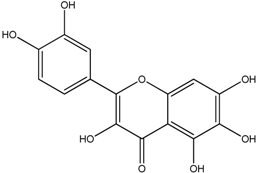 |
| Isorhamnetin |  |
| Fisetin | 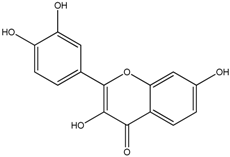 |
| Galangin | 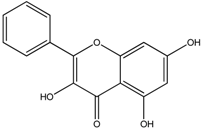 |
| Myricetin | 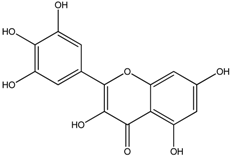 |
| Luteolin | 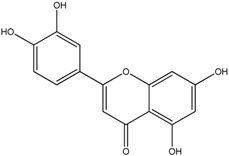 |
| Luteolin-7-O-glucoside | 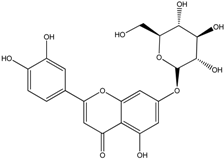 |
| Taxifolin |  |
| (+)-Catechin | 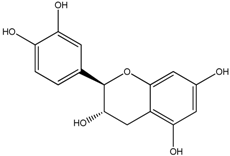 |
2. Results and Discussion
2.1. DPPH• Scavenging Activity of Flavonoids
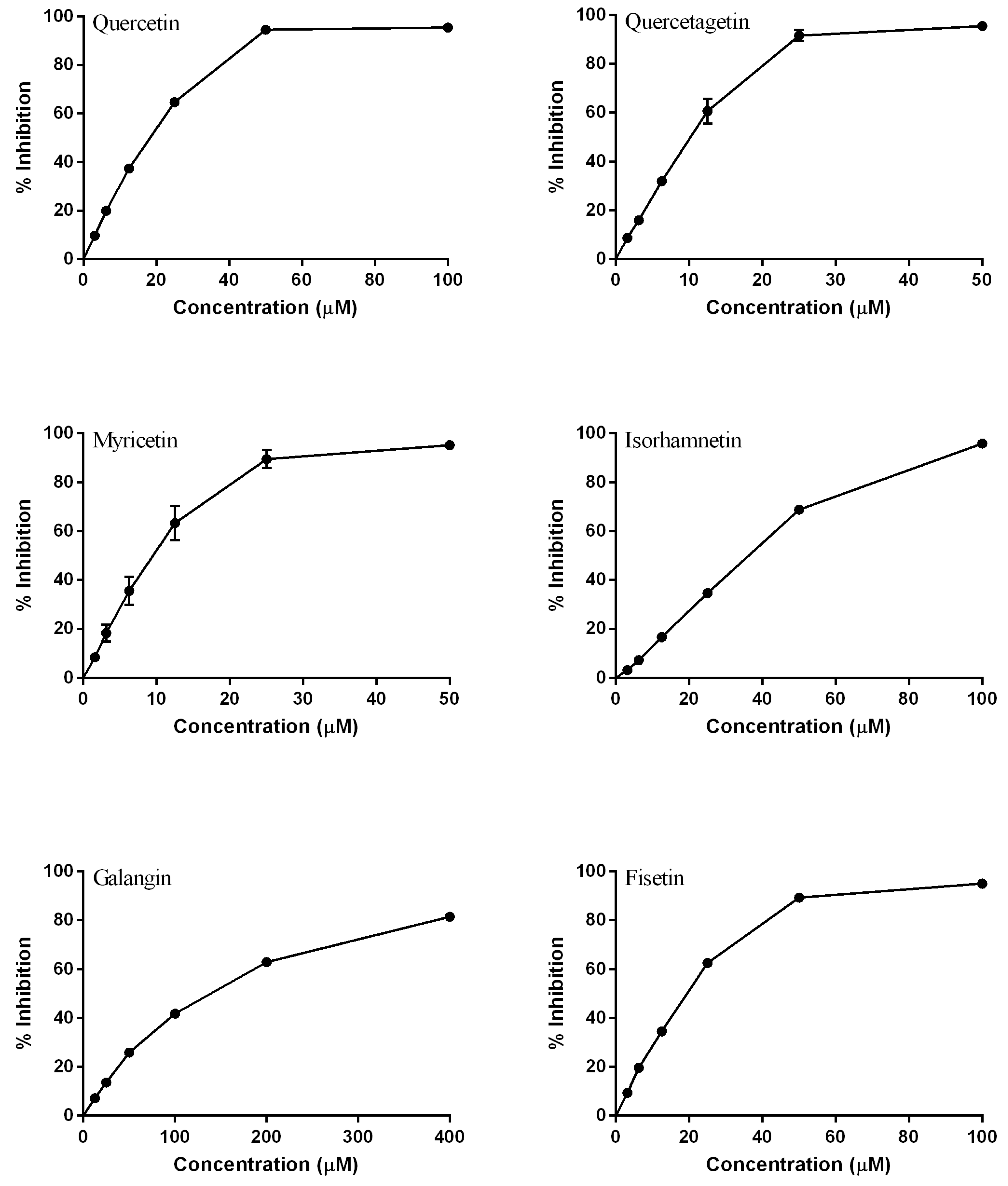
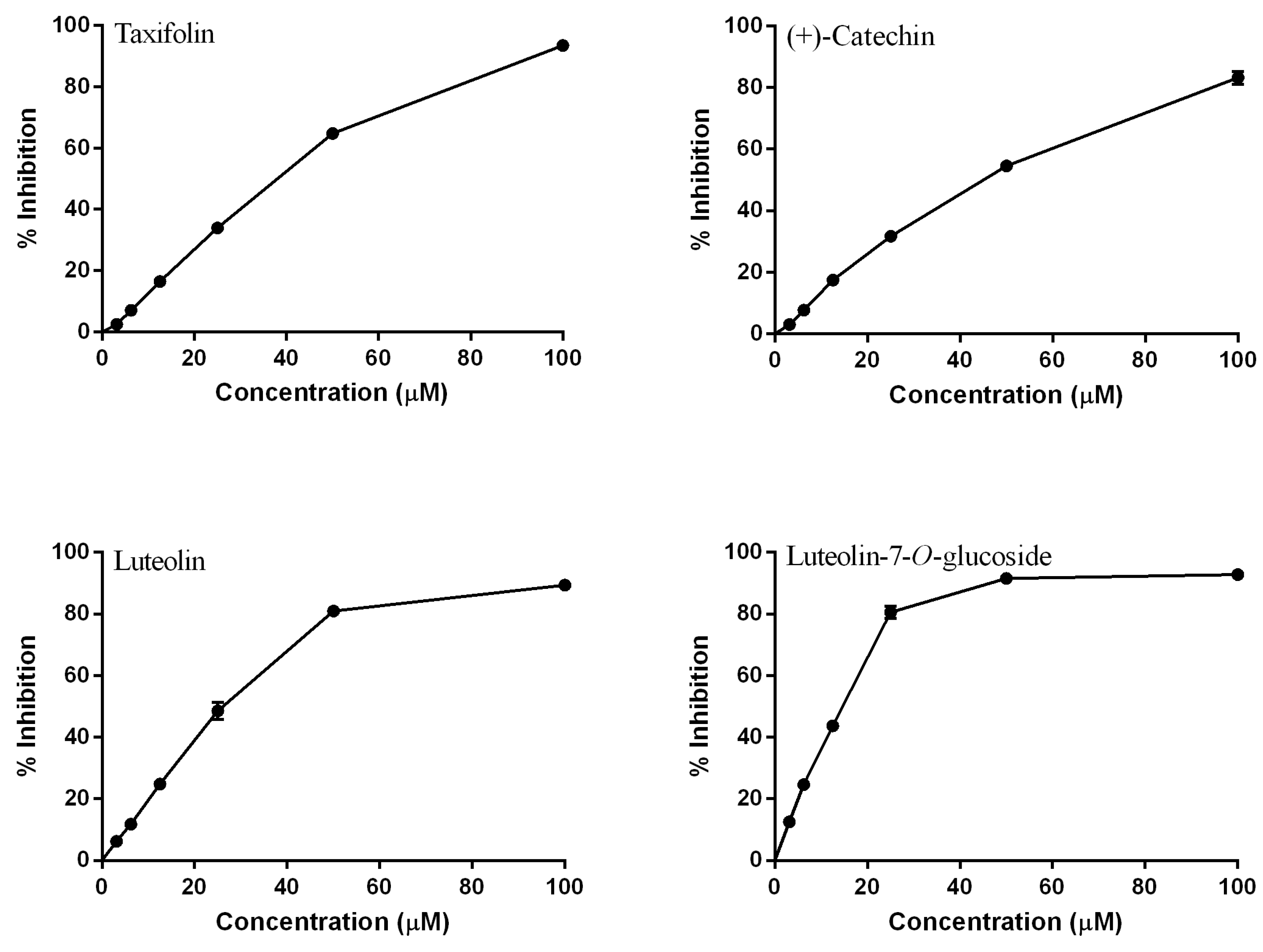
2.1.1. Modifications in A Ring
2.1.2. Modifications in the B Ring
2.1.3. Modifications in C Ring
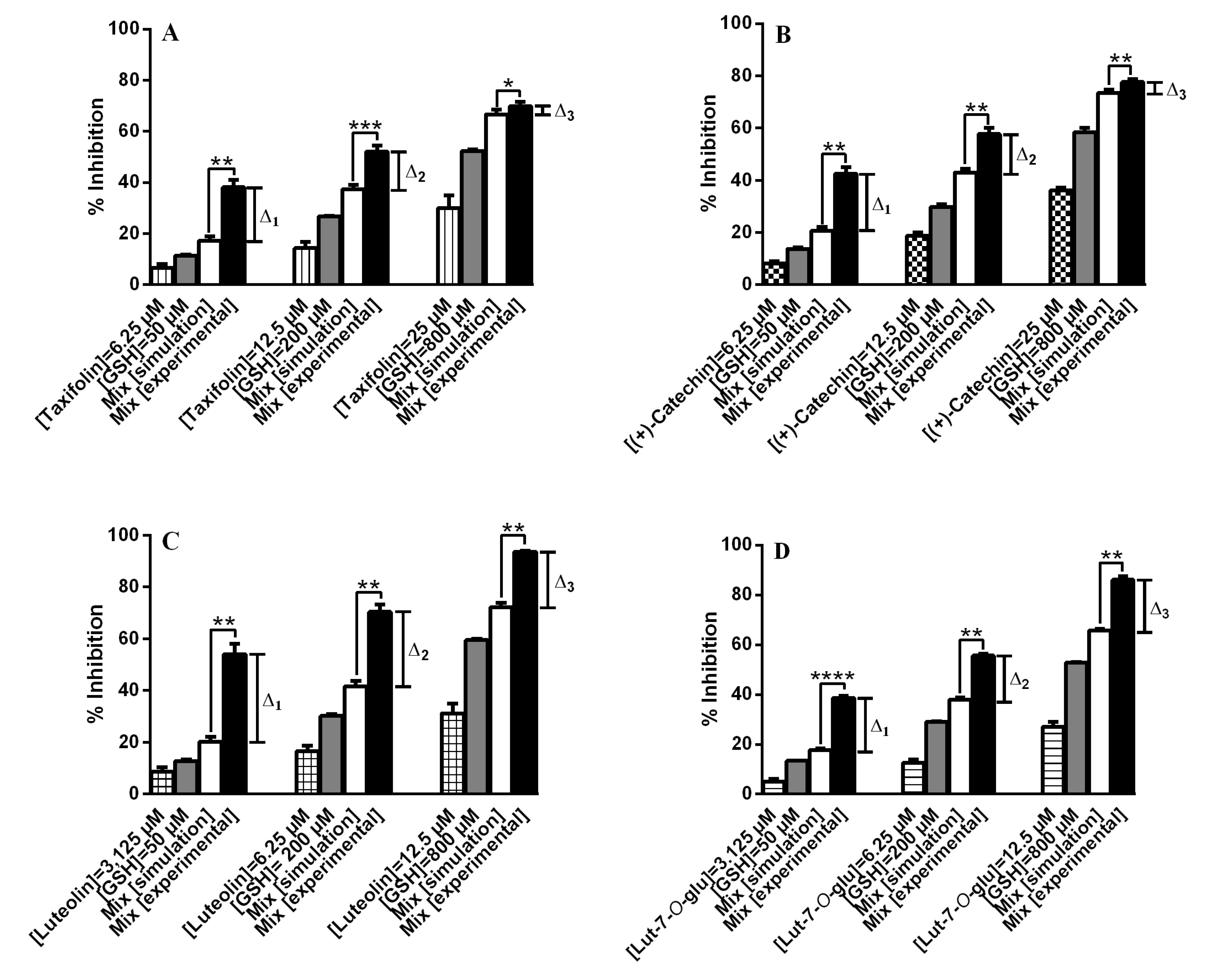
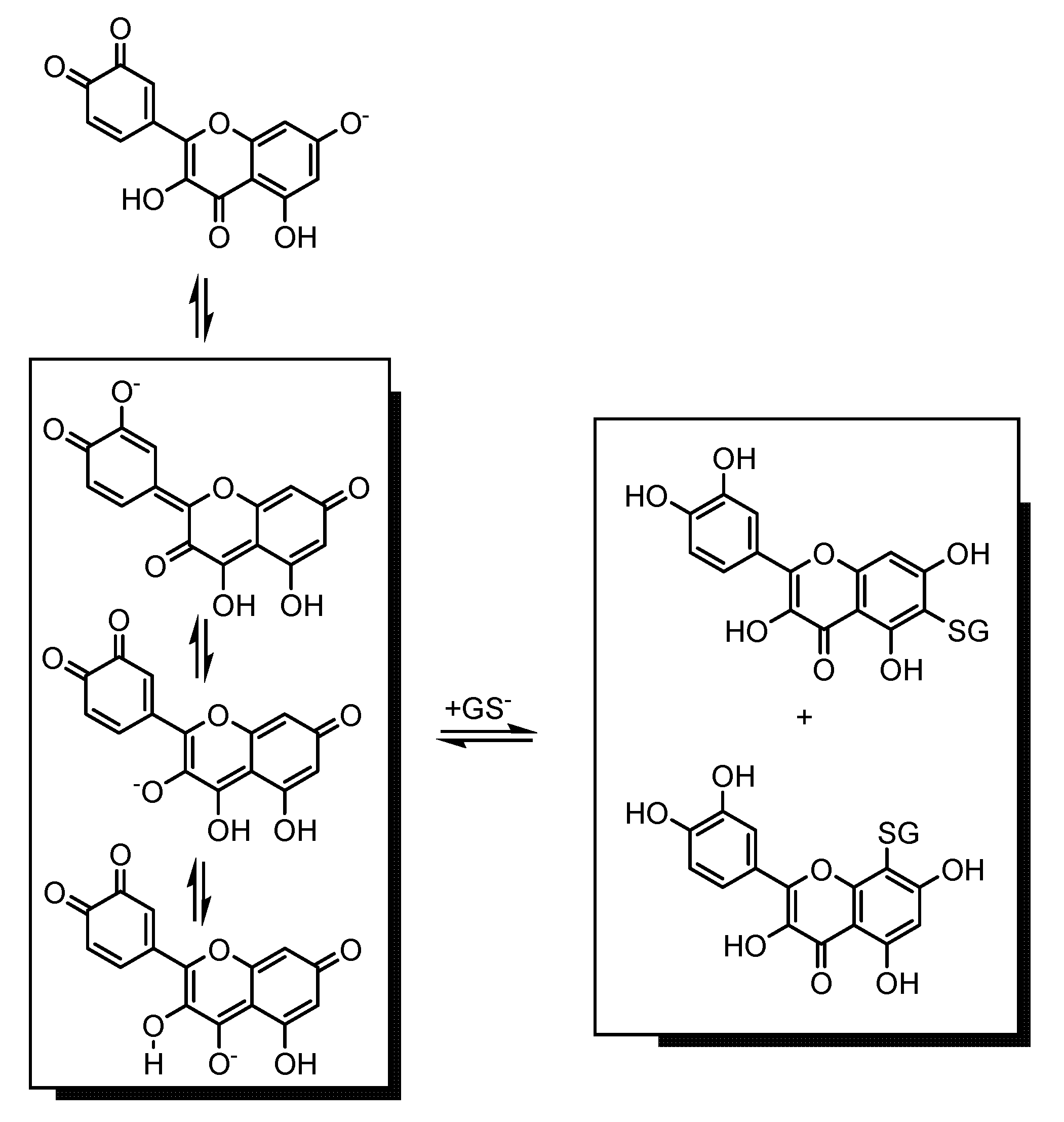
2.2. DPPH• Scavenging Activity of Flavonoids in Mixtures with GSH

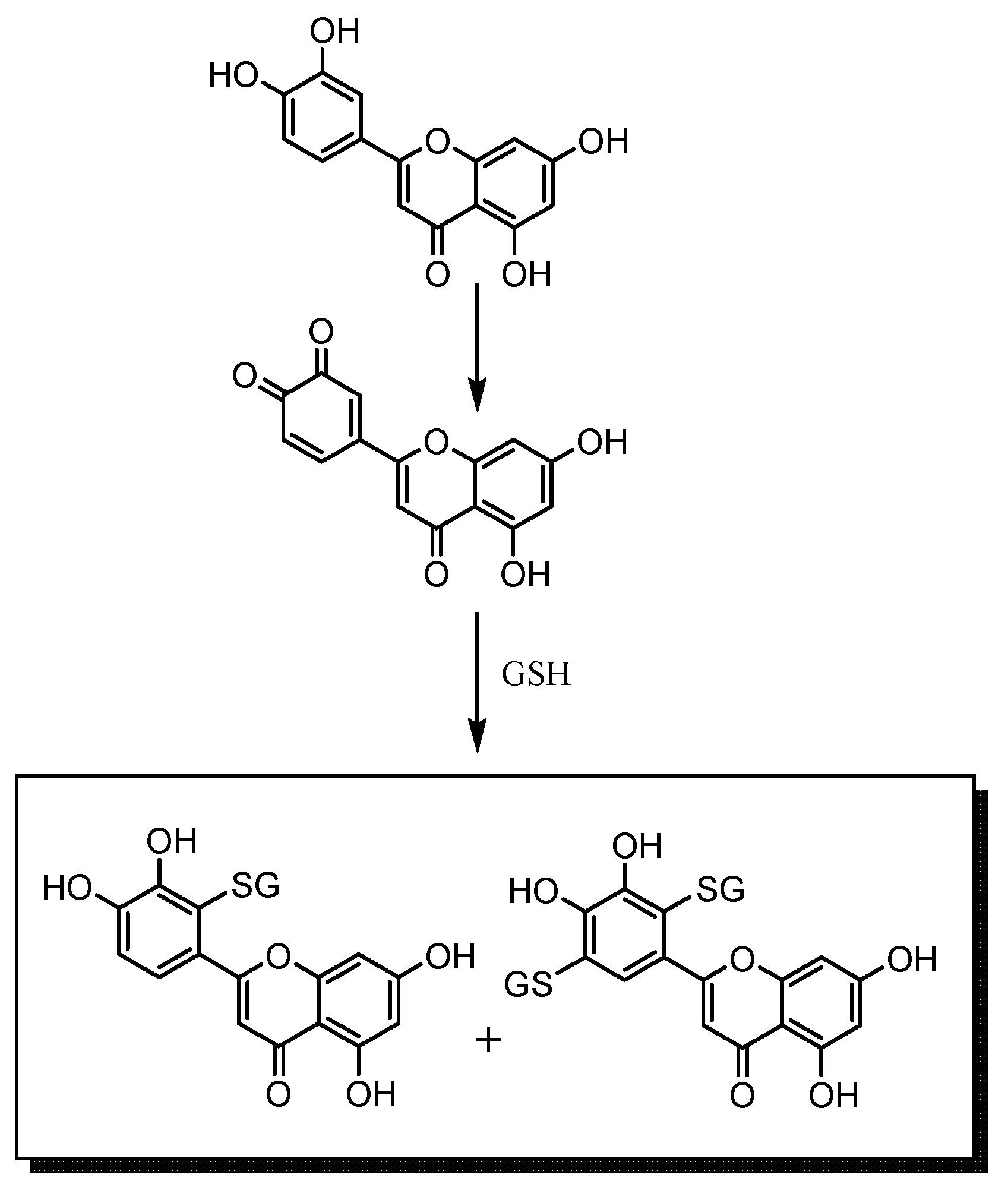
3. Experimental
3.1. Reagents
3.2. Compounds Solutions and Binary Mixtures
3.3. Antioxidant Activity
3.3.1. DPPH• Scavenging Activity
3.3.2. Determination of Mixture Effect (Simulation)
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Thomas, J.A.; Poland, B.; Honzatko, R. Protein sulfhydryls and their role in the antioxidant function of protein S-thiolation. Arch. Biochem. Biophys. 1995, 319, 1–9. [Google Scholar] [CrossRef]
- Huang, K.P.; Huang, F.L. Glutathionylation of proteins by glutathione disulfide S-oxide. Biochem. Pharmacol. 2002, 64, 1049–1056. [Google Scholar] [CrossRef]
- Hayes, J.D.; Pulford, D.J. The glutathione S-transferase supergene family: Regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 445–600. [Google Scholar] [CrossRef]
- Wild, A.C.; Mulcahy, R.T. Regulation of γ-glutamylcysteine synthetase subunit gene expression: Insights into transcriptional control of antioxidant defenses. Free Radic. Res. 2000, 32, 281–301. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Bolwell, P.G.; Bramley, P.M.; Pridham, J.B. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res. 1995, 22, 375–383. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Milella, L.; Caruso, M.; Galgano, F.; Favati, F.; Padula, M.C.; Martelli, G. Role of the cultivar in choosing Clementine fruits with a high level of health-promoting compounds. J. Agric. Food Chem. 2011, 59, 5293–5298. [Google Scholar] [CrossRef]
- Walle, T. Absorption and metabolism of flavonoids. Free Radic. Biol. Med. 2004, 36, 829–837. [Google Scholar] [CrossRef]
- Gaikwad, P.; Barik, A.; Priyadarsini, K.I.; Rao, B.S.M. Antioxidant activities of phenols in different solvents using DPPH assay. Res. Chem. Intermed. 2010, 36, 1065–1072. [Google Scholar] [CrossRef]
- Beecher, C.W.W. Cancer preventive properties of varieties of Brassica oleracea—A review. Am. J. Clin. Nutr. 1994, 59, 1166S–1170S. [Google Scholar]
- Van Poppel, G.; Verhoeven, D.T.H.; Verhagen, H.; Goldbohm, R.A. Brassica vegetables and cancer prevention—Epidemiology and mechanisms. Adv. Nutr. Cancer 1999, 2, 159–168. [Google Scholar]
- Millic, B.L.; Diljas, S.M.; Canadanovic-Brunet, J.M. Antioxidative activity of phenolic compounds on the metal-ion breakdown of lipid peroxidation system. Food Chem. 1998, 61, 443–447. [Google Scholar] [CrossRef]
- Karadag, A.; Ozcelik, B.; Saner, S. Review of methods to determine antioxidant capacities. Food Anal. Methods 2009, 2, 41–60. [Google Scholar] [CrossRef]
- Menezes, J.C.; Kamat, S.P.; Cavaleiro, J.A.; Gaspar, A.; Garrido, J.; Borges, F. Synthesis and antioxidant activity of long chain alkyl hydroxycinnamates. Eur. J. Med. Chem. 2011, 46, 773–777. [Google Scholar] [CrossRef]
- Arnao, M.B. Some methodological problems in the determination of antioxidant activity using chromogen radicals: A practical case. Trends Food Sci. Technol. 2000, 11, 419–421. [Google Scholar] [CrossRef]
- Villaño, D.; Fernández-Pachon, M.S.; Troncoso, A.M.; Garcia-Parrilla, M.C. The antioxidant activity of wines determined by the ABTS•+ method: Influence of sample dilution and time. Talanta 2004, 64, 501–509. [Google Scholar] [CrossRef]
- Marais, J.P.J.; Deavours, B.; Dixon, R.A.; Ferreira, D. The Stereochemistry of Flavonoids. In The Science of Flavonoids; Grotewold, E., Ed.; Springer Press: New York, NY, USA, 2007; pp. 1–35. [Google Scholar]
- Boots, A.W.; Kubben, N.; Haenen, G.R.; Bast, A. Oxidized quercetin reacts with thiols rather than with ascorbate: Implication for quercetin supplementation. Biochem. Biophys. Res. Commun. 2003, 308, 560–565. [Google Scholar] [CrossRef]
- Kessler, M.; Ubeaud, G.; Jung, L. Anti- and pro-oxidant activity of rutin and quercetin derivatives. J. Pharm. Pharmacol. 2003, 55, 131–142. [Google Scholar] [CrossRef]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomás-Barberán, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Heimann, W.; Reiff, F. Beziehung zwischen chemischer konstitution und antioxygener wirkung bei flavonolen. (In German)Fette Seifen Anstr-Mittel. 1953, 55, 451–458. [Google Scholar] [CrossRef]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as antioxidants: Determination of radical scavenging efficiencies. Methods Enzymol. 1990, 186, 343–355. [Google Scholar] [CrossRef]
- Sichel, G.; Corsaro, C.; Scalia, M.; di Bilio, A.J.; Bonomo, R.P. In vitro scavenger activity of some flavonoids and melanins against O2•−. Free Radic. Biol. Med. 1991, 11, 1–8. [Google Scholar] [CrossRef]
- Rastija, V.; Medić-Sarić, M. QSAR study of antioxidant activity of wine polyphenols. Eur. J. Med. Chem. 2009, 44, 400–408. [Google Scholar] [CrossRef]
- Harborne, J.B.; Mabry, T.J.; Mabry, H. The Flavonoids; Chapman and Hall: London, UK, 1975. [Google Scholar]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef]
- Ratty, A.K.; Das, N.P. Effects of flavonoids on nonenzymatic lipid peroxidation: Structure-activity relationship. Biochem. Med. Metab. Biol. 1988, 39, 69–79. [Google Scholar] [CrossRef]
- Matthiesen, L.; Malterud, K.E.; Sund, R.B. Hydrogen bond formation as basis for radical scavenging activity: A structure-activity study of C-methylated dihydrochalcones from Myrica gale and structurally related acetophenones. Free Radic. Biol. Med. 1997, 2, 307–311. [Google Scholar] [CrossRef]
- Wu, M.J.; Huang, C.L.; Lian, T.W.; Kou, M.C.; Wang, L. Antioxidant activity of Glossogyne tenuifolia. J. Agric. Food Chem. 2005, 53, 6305–6312. [Google Scholar] [CrossRef]
- Chou, T.C.; Talalay, P. Quantitative analysis of dose-effect relationships: The combined effects of multiple drugs or enzyme inhibitors. Adv. Enzyme Regul. 1984, 22, 27–55. [Google Scholar] [CrossRef]
- Webb, J.L. Effect of More than One Inhibitor. In Enzyme and Metabolic Inhibitors; Academic Press: New York, NY, USA and London, UK, 1963; Volume 1, pp. 66–79, 488–512. [Google Scholar]
- Awad, H.M.; Boersma, M.G.; Boeren, S.; van Bladeren, P.J.; Vervoort, J.; Rietjens, I.M. The regioselectivity of glutathione adduct formation with flavonoid quinone/quinone methides is pH-dependent. Chem. Res. Toxicol. 2002, 15, 343–351. [Google Scholar] [CrossRef]
- Hodnick, W.F.; Kung, F.S.; Roettger, W.J.; Bohmont, C.W.; Pardini, R.S. Inhibition of mitochondrial respiration and production of toxic oxygen radicals by flavonoids: A structure-activity study. Biochem. Pharmacol. 1986, 35, 2345–2357. [Google Scholar] [CrossRef]
- Frey-Schroder, G.; Barz, W. Isolation and characterization of flavonol converting enzymes from Mentha piperita plants and from Mentha arvensis cell suspension cultures. Z. Naturforsch. 1979, 34C, 200–209. [Google Scholar]
- Dangles, O.; Fargeix, G.; Dufour, C. One-electron oxidation of quercetin derivatives in protic and non protic media. J. Chem. Soc. Perkin Trans. 1999, 2, 1387–1395. [Google Scholar]
- Van der Woude, H.; Boersma, M.G.; Alink, G.M.; Vervoort, J.; Rietjens, I.M. Consequences of quercetin methylation for its covalent glutathione and DNA adduct formation. Chem. Biol. Interact. 2006, 160, 193–203. [Google Scholar] [CrossRef]
- Awad, H.M.; Boersma, M.G.; Boeren, S.; van Bladeren, P.J.; Vervoort, J.; Rietjens, I.M.C.M. Structure-activity study on the quinone/quinone methide chemistry of flavonoids. Chem. Res. Toxicol. 2001, 14, 398–408. [Google Scholar] [CrossRef]
- Moridani, M.Y.; Scobie, H.; Salehi, P.; O’Brien, P.J. Catechin metabolism: Glutathione conjugate formation catalyzed by tyrosinase, peroxidase, and cytochrome p450. Chem. Res. Toxicol. 2001, 14, 841–848. [Google Scholar] [CrossRef]
- Vrchovská, V.; Sousa, C.; Valentão, P.; Ferreres, F.; Pereira, J.A.; Seabra, R.M.; Andrade, P.B. Antioxidative properties of tronchuda cabbage (Brassica oleracea L. var. costata DC) external leaves against DPPH•, superoxide radical, hydroxyl radical and hypochlorous acid. Food Chem. 2006, 98, 416–425. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pereira, R.B.; Sousa, C.; Costa, A.; Andrade, P.B.; Valentão, P. Glutathione and the Antioxidant Potential of Binary Mixtures with Flavonoids: Synergisms and Antagonisms. Molecules 2013, 18, 8858-8872. https://doi.org/10.3390/molecules18088858
Pereira RB, Sousa C, Costa A, Andrade PB, Valentão P. Glutathione and the Antioxidant Potential of Binary Mixtures with Flavonoids: Synergisms and Antagonisms. Molecules. 2013; 18(8):8858-8872. https://doi.org/10.3390/molecules18088858
Chicago/Turabian StylePereira, Renato B., Carla Sousa, Andreia Costa, Paula B. Andrade, and Patrícia Valentão. 2013. "Glutathione and the Antioxidant Potential of Binary Mixtures with Flavonoids: Synergisms and Antagonisms" Molecules 18, no. 8: 8858-8872. https://doi.org/10.3390/molecules18088858
APA StylePereira, R. B., Sousa, C., Costa, A., Andrade, P. B., & Valentão, P. (2013). Glutathione and the Antioxidant Potential of Binary Mixtures with Flavonoids: Synergisms and Antagonisms. Molecules, 18(8), 8858-8872. https://doi.org/10.3390/molecules18088858







