1,2,3-Triazole-Substituted Oleanolic Acid Derivatives: Synthesis and Antiproliferative Activity
Abstract
:1. Introduction
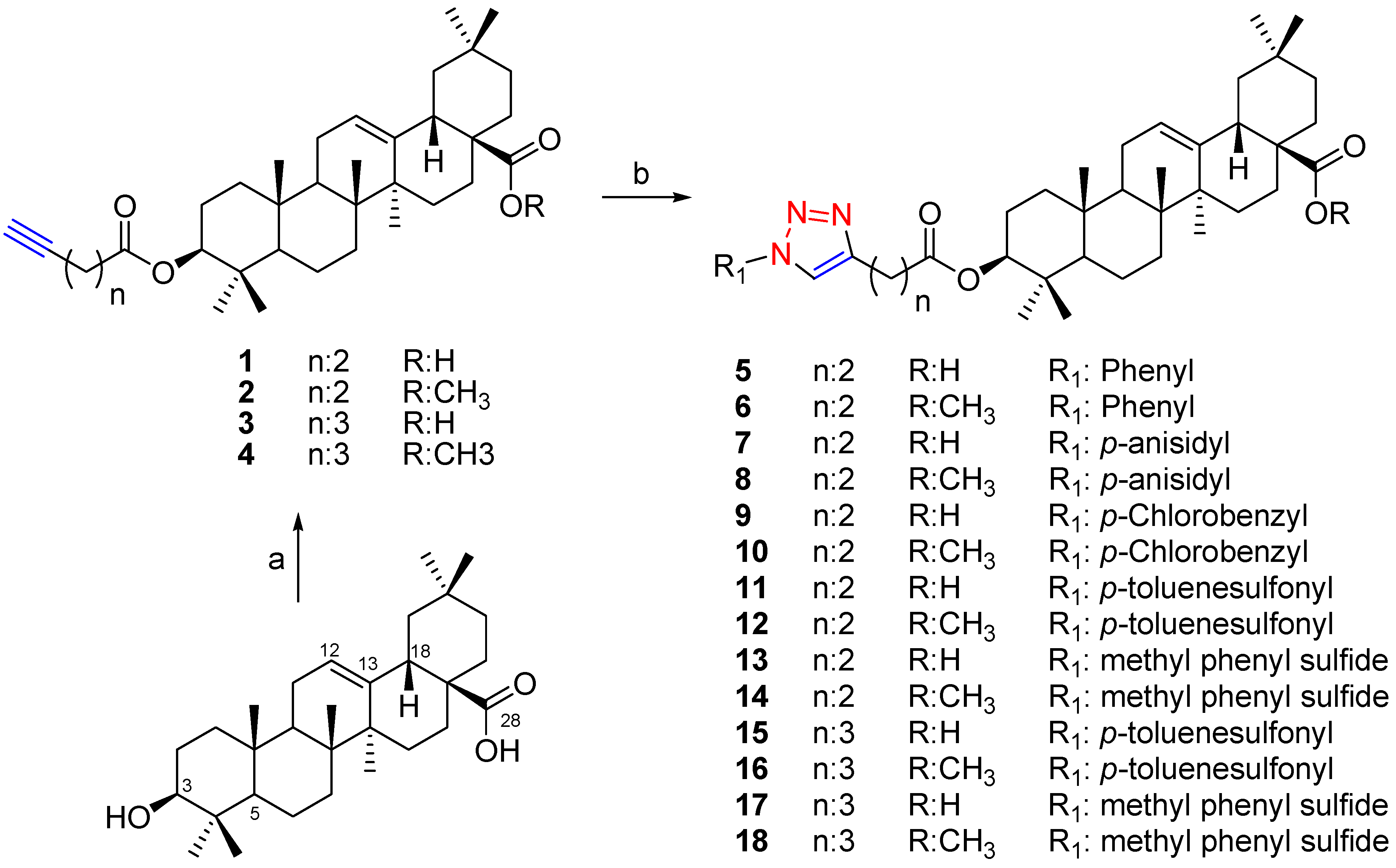
2. Results and Discussion
| Compound | (IC50 ± SD, µM) b | ||||
|---|---|---|---|---|---|
| MRC-5 | AGS | SK-MES-1 | J82 | HL-60 | |
| 1 | 21.3 ± 0.9 | 19.1 ± 1.0 | 22.4 ± 1.9 | 23.1 ± 1.6 | 19.6 ± 0.1 |
| 2 | >100 | >100 | >100 | >100 | >100 |
| 3 | 14.1 ± 1.2 | 22.1 ± 1.5 | 23.4 ± 1.8 | 56.2 ± 4.3 | 22.4 ± 0.1 |
| 4 | >100 | >100 | >100 | >100 | >100 |
| 5 | >100 | >100 | >100 | >100 | >100 |
| 6 | >100 | >100 | >100 | >100 | >100 |
| 7 | >100 | 95.1 ± 5.8 | >100 | >100 | >100 |
| 8 | >100 | >100 | >100 | >100 | >100 |
| 9 | >100 | 8.9 ± 0.4 | 50.4 ± 3.5 | 35.4 ± 2.8 | 35.8 ± 4.1 |
| 10 | >100 | >100 | >100 | >100 | >100 |
| 11 | 14.1 ± 0.8 | 10.8± 0.6 | 21.4 ± 1.3 | 47.1 ± 2.9 | 15.9 ± 1.5 |
| 12 | 76.0 ± 3.8 | 63.0 ± 3.8 | 76.3 ± 5.1 | >100 | 22.4 ± 1.8 |
| 13 | >100 | 31.6 ± 1.6 | 65.5 ± 4.4 | >100 | 18.1 ± 1.6 |
| 14 | >100 | >100 | >100 | >100 | >100 |
| 15 | 17.1 ± 1.0 | 22.2 ± 1.6 | 11.9 ± 0.6 | 14.3 ± 0.6 | 11.5 ± 1.0 |
| 16 | >100 | >100 | >100 | >100 | 84.2 ± 7.5 |
| 17 | 46.7 ± 2.4 | 23.7 ± 1.4 | 61.8 ± 3.1 | >100 | 11.8 ± 0.9 |
| 18 | >100 | >100 | >100 | >100 | >100 |
| Etoposide c | 0.33 ± 0.02 | 0.58 ± 0.02 | 1.83 ± 0.09 | 3.49 ± 0.16 | 2.23 ± 0.09 |
3. Experimental
3.1. General Procedures
3.2. General Procedure for the Synthesis of Compounds 1–18
3.2.1. Preparation of Alkynyl Esters 1, 3
3.2.2. General Procedure for the Synthesis of Triazoles 5, 7, 9, 11, 13, 15 and 17
3.2.3. Preparation of Methyl Esters 2, 4, 6, 8, 10, 12, 14, 16 and 18
3.3. Antiproliferative Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Kashiwada, Y.; Hashimoto, F.; Cosentino, L.M.; Chen, C.-H.; Garrett, P.E.; Lee, K.-H. Betulinic acid and dihydrobetulinic acid derivatives as potent anti-HIV agents. J. Med. Chem. 1996, 39, 1016–1017. [Google Scholar] [CrossRef]
- Jeong, H.-J.; Chai, H.-B.; Park, S.-Y.; Kim, D.S.H.L. Preparation of amino acid conjugates of betulinic acid with activity against human melanoma. Bioorg. Med. Chem. Lett. 1999, 9, 1201–1204. [Google Scholar] [CrossRef]
- Gauthier, C.; Legault, S.; Rondeau, S.; Pichette, A. Synthesis of betulinic acid acyl glucuronide for application in anticancer prodrug monotherapy. Tetrahedron Lett. 2009, 50, 988–991. [Google Scholar] [CrossRef]
- Chen, J.; Liu, J.; Zhang, L.; Wu, G.; Hua, W. Pentacyclic triterpenes. Part 3: Synthesis and biological evaluation of oleanolic acid derivatives as novel inhibitors of glycogen phosphorylase. Bioorg. Med. Chem. Lett. 2006, 16, 2915–2919. [Google Scholar] [CrossRef]
- Sánchez, M.; Theoduloz, C.; Schmeda-Hirschmann, G.; Razmilic, I.; Yáñez, T.; Rodríguez, J.A. Gastroprotective and ulcer-healing activity of oleanolic acid derivatives: In vitro–in vivo relationships. Life Sci. 2006, 79, 1349–1356. [Google Scholar] [CrossRef]
- Laszczyk, M.N. Pentacyclic triterpenes of the lupane, oleanane and ursane group as tools in cancer therapy. Planta Med. 2009, 75, 1549–1560. [Google Scholar] [CrossRef]
- Li, J.-F.; Zhao, Y.; Cai, M.-M.; Li, X.-F.; Li, J.-X. Synthesis and evaluation of a novel series of heterocyclic oleanolic acid derivatives with anti-osteoclast formation activity. Eur. J. Med. Chem. 2009, 44, 2796–2806. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, Y.; Kong, X.; Peng, S.; Tian, J. Synthesis and biological evaluation of nitric oxide-releasing derivatives of oleanolic acid as inhibitors of HepG2 cell apoptosis. Bioorg. Med. Chem. Lett. 2007, 17, 2979–2982. [Google Scholar] [CrossRef]
- Hao, J.; Liu, J.; Wen, X.; Sun, H. Synthesis and cytotoxicity evaluation of oleanolic acid derivatives. Bioorg. Med. Chem. Lett. 2013, 23, 2074–2077. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, Z-F.; Meng, F.; Xu, C-S.; Zhang, Y-H. Synthesis and cytotoxicity of oleanolic acid/N-aryl-N’-hydroxyguanidine hybrids. Chin. J. Nat. Med. 2010, 8, 436–440. [Google Scholar]
- Whiting, M.; Muldoon, J.; Lin, Y.C.; Silverman, S.M.; Lindstron, W.; Olson, A.J.; Kolb, H.C.; Finn, M.G.; Sharpless, K.B.; Elder, J.H.; et al. Inhibitors of HIV-1 protease by usingin situ click chemistry. Angew. Chem Int. Ed. 2006, 45, 1435–1439. [Google Scholar] [CrossRef]
- Tornøe, C.W.; Sanderson, S.J.; Mottram, J.C.; Coombs, G.H.; Meldal, M. Combinatorial library of peptidotriazoles: identification of [1,2,3]-triazole inhibitors against a recombinant leishmania mexicana cysteine protease. J. Comb. Chem. 2004, 6, 312–324. [Google Scholar] [CrossRef]
- De las Heras, F.G.; Alonso, R.; Alonso, G.J. Synthesis and cytostatic activity of N-glycosyl(halomethyl)-1,2,3-triazoles. A new type of alkylating agent. J. Med. Chem. 1979, 22, 496–501. [Google Scholar] [CrossRef]
- Alonso, R.; Camarasa, M.J.; Alonso, G.; De Las Heras, F.G. Synthesis and cytostatic activity of N-ribosylhalomethyl-1,2,3 triazoles. Eur. J. Med. Chem. 1980, 15, 105–109. [Google Scholar]
- Cho, S.; Oh, S.; Uma, Y.; Jung, J.-H.; Hamc, J.; Shin, W.-S.; Lee, S. Synthesis of 10-substituted triazolyl artemisinins possessing anticancer activity via Huisgen 1,3-dipolar cylcoaddition. Bioorg. Med. Chem. Lett. 2009, 19, 382–385. [Google Scholar]
- Cafici, L.; Pirali, T.; Condorelli, F.; Del Grosso, E.; Massarotti, A.; Sorba, G.; Canonico, P.L.; Tron, G.C.; Genazzani, A.A. Solution-phase parallel synthesis and biological evaluation of combretatriazoles. J. Comb. Chem. 2008, 10, 732–740. [Google Scholar] [CrossRef]
- Kádár, Z.; Molnár, J.; Schneider, G.; Zupkó, I.; Frank, E. A facile ‘click’ approach to novel 15β-triazolyl-5α-androstane derivatives, and an evaluation of their antiproliferative activities in vitro. Bioorg. Med. Chem. 2012, 20, 1396–1402. [Google Scholar] [CrossRef]
- Duan, Y.-C.; Ma, Y.-C.; Zhang, E.; Shi, X.-J.; Wang, M.-M.; Ye, X.-W.; Liu, H.-M. Design and synthesis of novel 1,2,3-triazole-dithiocarbamate hybrids as potential anticancer agents. Eur. J. Med. Chem. 2013, 62, 11–19. [Google Scholar] [CrossRef]
- Wang, S.; Wang, Q.; Wang, Y.; Liu, L.; Weng, X.; Li, G.; Zhang, X.; Zhou, X. Novel anthraquinone derivatives: Synthesis via click chemistry approach and their induction of apoptosis in BGC gastric cancer cells via reactive oxygen species (ROS)-dependent mitochondrial pathway. Bioorg. Med. Chem. Lett. 2008, 18, 6505–6508. [Google Scholar] [CrossRef]
- Rao, P.S.; Kurumurthy, C.; Veeraswamy, B.; Kumar, G.S.; Rao, P.S.; Pamanji, R.; Rao, J.V.; Narsaiaha, B. Synthesis of novel 2-alkyl triazole-3-alkyl substituted quinoline derivatives and their cytotoxic activity. Bioorg. Med. Chem. 2013, 23, 1225–1227. [Google Scholar] [CrossRef]
- Gupta, S.S.; Kuzelka, J.; Singh, P.; Lewis, W.G.; Manchester, M.; Finn, M.G. Accelerated bioorthogonal conjugation: A practical method for the ligation of diverse functional molecules to a polyvalent virus scaffold. Bioconjugate Chem. 2005, 16, 1572–1579. [Google Scholar] [CrossRef]
- Kume, M.; Kubota, T.; Kimura, Y.; Nakashimizu, K.; Motokawa, M.; Nakano, M. Synthesis and structure-activity relationship of new 7-beta-[(Z)-2-(2-aminothiazol-4-yl)-2-hydroxyminoacetamido]-cephalosporins with 1,2,3-triazole in C-3 side chain. J. Antibiot. 1993, 46, 177–192. [Google Scholar] [CrossRef]
- Bock, V.D.; Hiemstra, H.; Van Maarseveen, J.H. Eur. Cu(I)-catalyzed alkyne-azide click cycloadditions from a mechanistic and synthetic perspective. J. Org. Chem. 2006, 1, 51–68. [Google Scholar]
- Baraniak, D.; Kacprzak, K.; Celewicz, L. Synthesis of 3'-azido-3'-deoxythymidine (AZT)-Cinchona alkaloid conjugates via click chemistry: Toward novel fluorescent markers and cytostatic agents. Bioorg. Med. Chem. Lett. 2011, 21, 723–726. [Google Scholar] [CrossRef]
- Kacprzak, K.M.; Maier, N.M.; Lindner, W. Highly efficient immobilization of Cinchona alkaloid derivatives to silica gel via click chemistry. Tetrahedron Lett. 2006, 47, 8721–8726. [Google Scholar] [CrossRef]
- Olomola, T.O.; Klein, R.; Mautsa, N.; Sayed, Y.; Kayea, P.T. Synthesis and evaluation of coumarin derivatives as potential dual-action HIV-1 protease and reverse transcriptase inhibitors. Bioorg. Med. Chem. 2013, 21, 1964–1971. [Google Scholar] [CrossRef]
- Perez-Labrada, K.; Brouarda, I.; Morera, C.; Estevez, F.; Bermejoa, J.; Rivera, D.G. Synthesis and conformational study of triazole-linked bis-spirostanic conjugates. Tetrahedron 2011, 67, 7713–7727. [Google Scholar] [CrossRef]
- Suh, B.-C.; Jeon, HB.; Posner, G.H.; Silverman, S.M. Vitamin D side chain triazole analogs via cycloaddition ‘click’ chemistry. Tetrahedron Lett. 2004, 45, 4623–4625. [Google Scholar] [CrossRef]
- Vasilevsky, S.F.; Govdi, A.I.; Sorokina, I.V.; Tolstikova, T.G.; Baev, D.S.; Tolstikov, G.A.; Mamatuyk, V.I.; Alabugin, G.V. Rapid access to new bioconjugates of betulonic acid via click chemistry. Bioorg. Med. Chem. Lett. 2011, 21, 62–65. [Google Scholar] [CrossRef]
- Bori, I.D.; Hung, H.-Y.; Qian, K.; Chen, C.-H.; Morris-Natschke, S.L.; Lee, K.-H. Anti-AIDS agents 88. Anti-HIV conjugates of betulin and betulinic acid with AZT prepared via click chemistry. Tetrahedron Lett. 2012, 53, 1987–1989. [Google Scholar] [CrossRef]
- Majeed, R.; Sangwan, P.L.; Chinthakindi, P.K.; Khan, I.; Dangroo, N.A.; Thota, N.; Hamid, A.; Sharma, P.R.; Saxena, A.K.; Koul, S. Synthesis of 3-O-propargylated betulinic acid and its 1,2,3-triazoles as potential apoptotic agents. Eur. J. Med. Chem. 2013, 63, 782–792. [Google Scholar] [CrossRef]
- Cheng, K.; Liu, J.; Liu, X.; Li, H.; Sun, H.; Xie, J. Synthesis of glucoconjugates of oleanolic acid as inhibitors of glycogen phosphorylase. Carbohyd. Res. 2009, 344, 841–850. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–18 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pertino, M.W.; Lopez, C.; Theoduloz, C.; Schmeda-Hirschmann, G. 1,2,3-Triazole-Substituted Oleanolic Acid Derivatives: Synthesis and Antiproliferative Activity. Molecules 2013, 18, 7661-7674. https://doi.org/10.3390/molecules18077661
Pertino MW, Lopez C, Theoduloz C, Schmeda-Hirschmann G. 1,2,3-Triazole-Substituted Oleanolic Acid Derivatives: Synthesis and Antiproliferative Activity. Molecules. 2013; 18(7):7661-7674. https://doi.org/10.3390/molecules18077661
Chicago/Turabian StylePertino, Mariano Walter, Cecilia Lopez, Cristina Theoduloz, and Guillermo Schmeda-Hirschmann. 2013. "1,2,3-Triazole-Substituted Oleanolic Acid Derivatives: Synthesis and Antiproliferative Activity" Molecules 18, no. 7: 7661-7674. https://doi.org/10.3390/molecules18077661
APA StylePertino, M. W., Lopez, C., Theoduloz, C., & Schmeda-Hirschmann, G. (2013). 1,2,3-Triazole-Substituted Oleanolic Acid Derivatives: Synthesis and Antiproliferative Activity. Molecules, 18(7), 7661-7674. https://doi.org/10.3390/molecules18077661




