Ganoderma tsugae Hepatoprotection against Exhaustive Exercise-Induced Liver Injury in Rats
Abstract
:1. Introduction
2. Results
2.1. Body Weight and Daily Intake
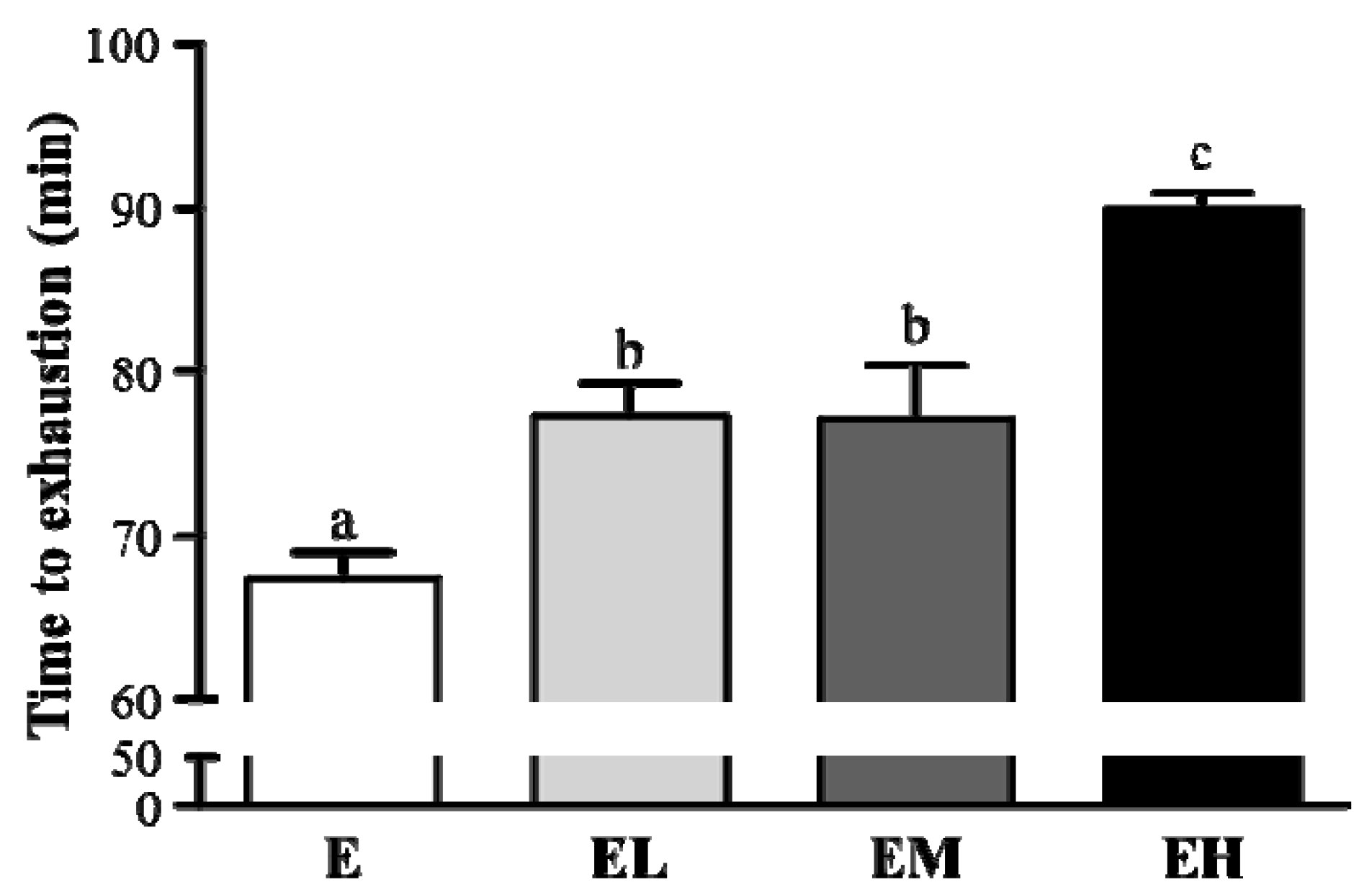
2.2. G. tsugae Effect on Exercise Performance in an Exhaustive Treadmill Exercise Test
2.3. G. tsugae Effect on Clinical Biochemistry Tests after an Exhaustive Treadmill Exercise Challenge
2.4. G. tsugae Effects on Oxidative Stress after an Exhaustive Treadmill Exercise Challenge
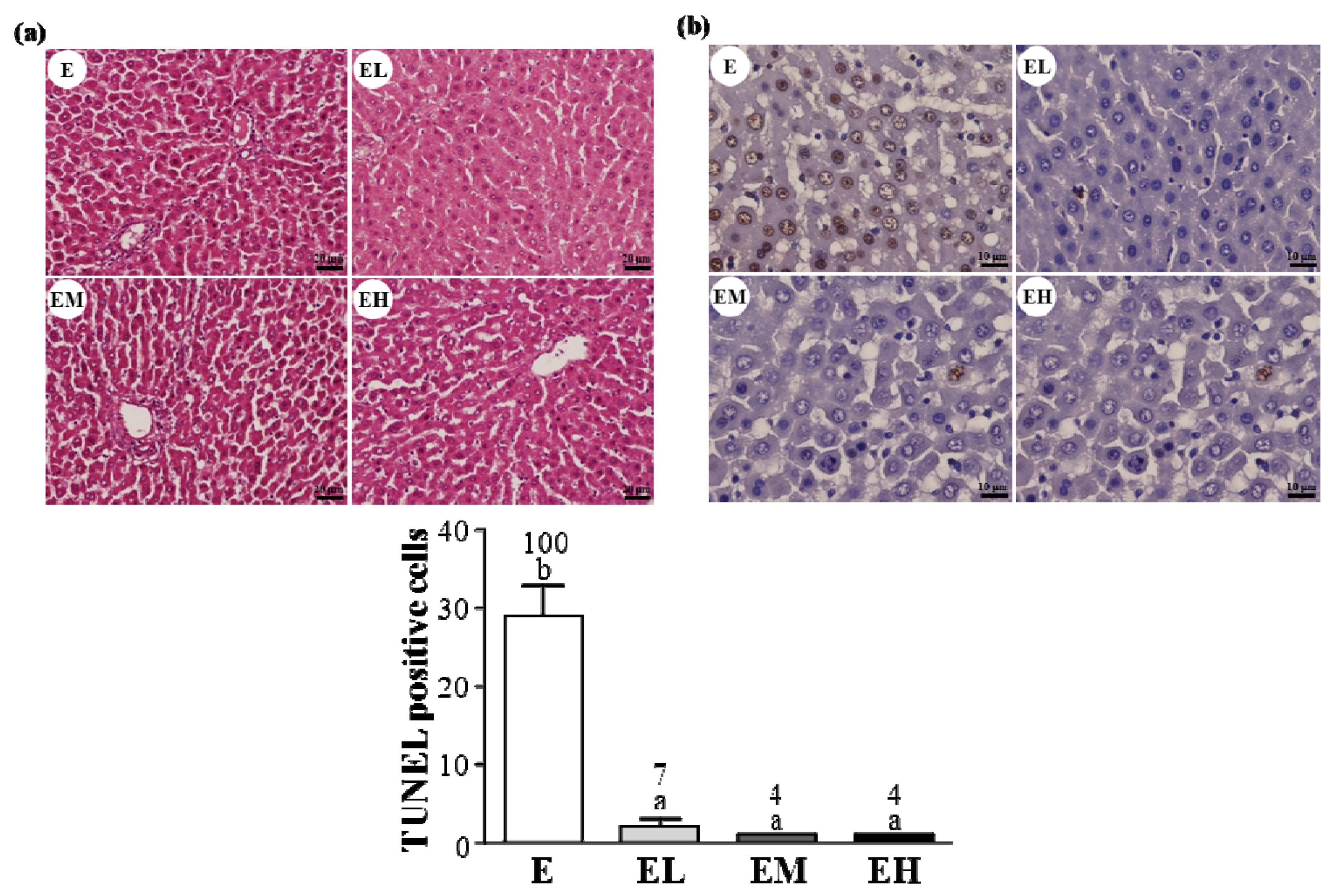
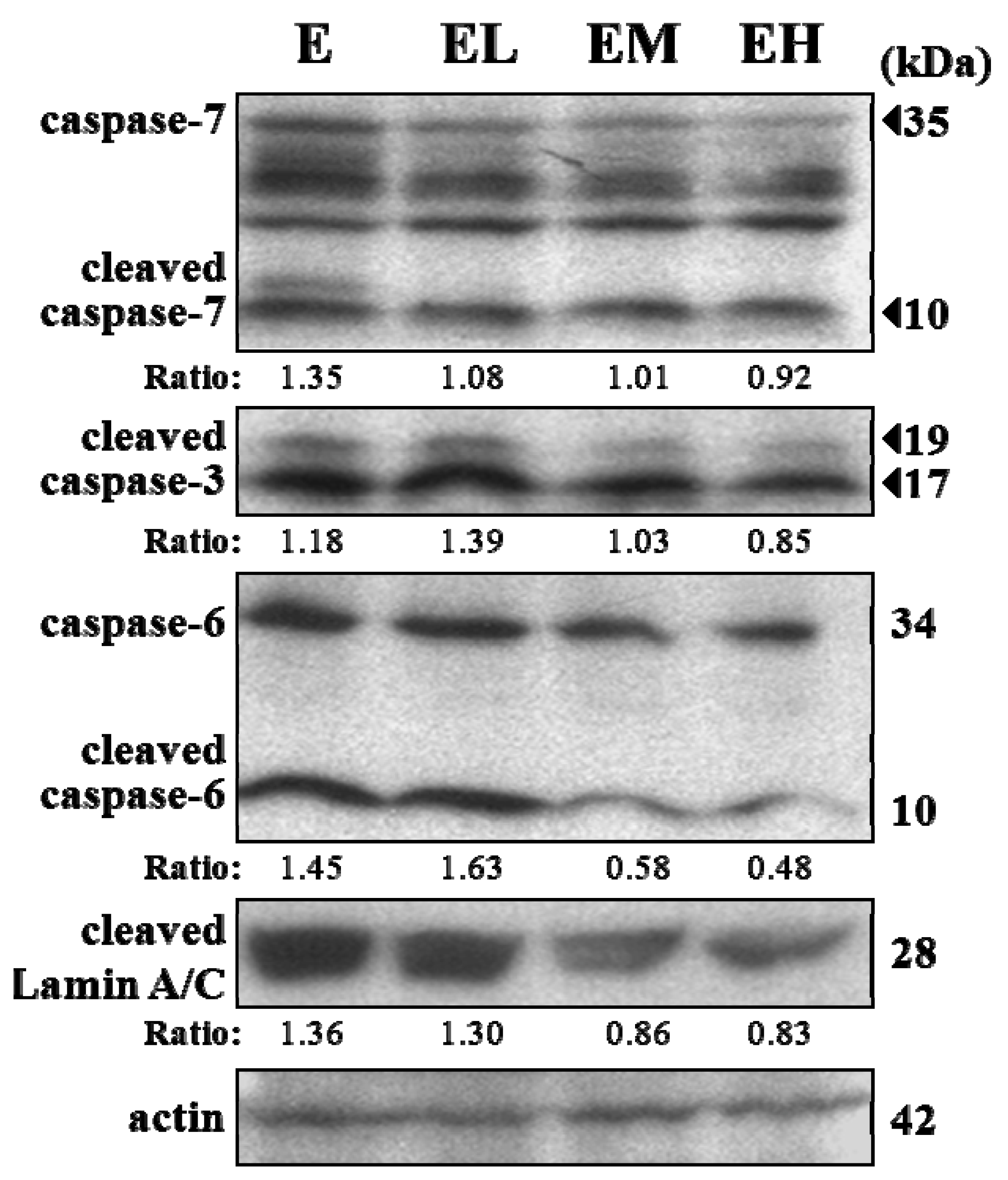
2.5. G. tsugae Effect on Hepatic Apoptosis after an Exhaustive Treadmill Exercise Challenge
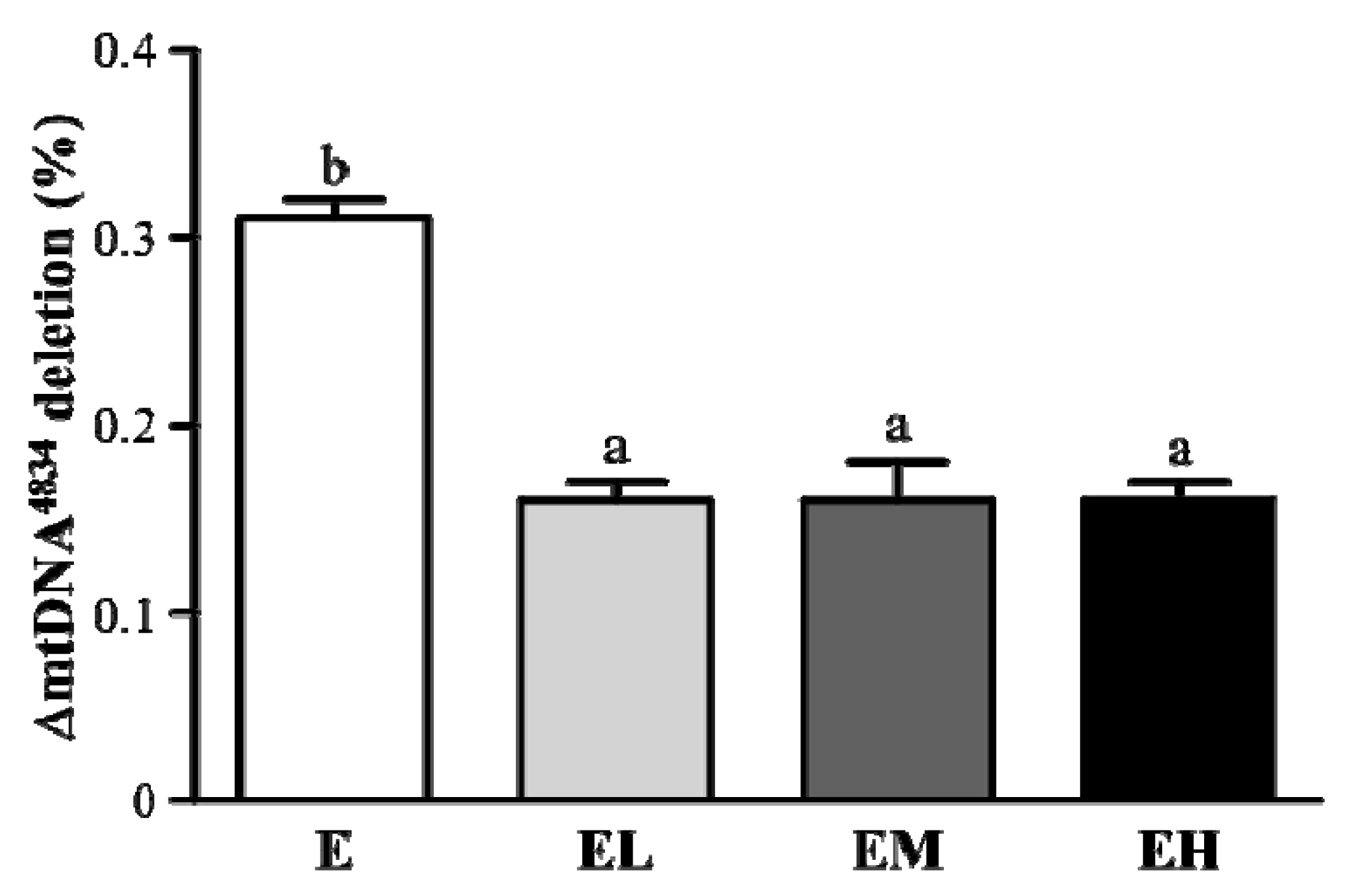
2.6. G. tsugae Effect on ΔmtDNA4834 Deletion of Liver Tissues after an Exhaustive Treadmill Exercise Challenge
3. Discussion
4. Experimental
4.1. Herb Material
4.2. Animals, Treatment, and Exhaustive Exercise
4.3. Determination of Blood Biochemical Variables
4.4. The Glutathione Peroxidase (GPX) Activities
4.5. The Glutathione Reductase (GRD) Activities
4.6. The Superoxide Dismutase (SOD) Activities
4.7. Thiobarbituric Acid Reactive Substances (TBARS) Analysis
4.9. Immunoblotting
4.10. TUNEL Assay and H&E Staining of Liver Tissue
4.11. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Pedersen, B.K.; Saltin, B. Evidence for prescribing exercise as therapy in chronic disease. Scand. J. Med. Sci. Sports 2006, 1, 3–63. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.J.; Quintanilha, A.T.; Brooks, G.A.; Packer, L. Free radicals and tissue damage produced by exercise. Biochem. Biophys. Res. Commun. 1982, 107, 1198–1205. [Google Scholar] [CrossRef]
- Viña, J.; Gomez-Cabrera, M.C.; Lloret, A.; Marquez, R.; Miñana, J.B.; Pallardó, F.V.; Sastre, J. Free radicals in exhaustive physical exercise: Mechanism of production, and protection by antioxidants. IUBMB Life 2000, 50, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Lin, T.J.; Lu, Y.F.; Chen, C.C.; Huang, C.Y.; Lin, W.T. Protective effects of L-arginine supplementation against exhaustive exercise-induced oxidative stress in young rat tissues. Chin. J. Physiol. 2009, 52, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Lin, T.J.; Chen, C.C.; Lin, W.T. Endurance training accelerates exhaustive exercise-induced mitochondrial DNA deletion and apoptosis of left ventriclemyocardium in rats. Eur. J. Appl. Physiol. 2009, 107, 697–706. [Google Scholar] [CrossRef] [PubMed]
- McLeay, Y.; Barnes, M.J.; Mundel, T.; Hurst, S.M.; Hurst, R.D.; Stannard, S.R. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Qian, Z.; Guo, J.; Hu, D.; Bao, J.; Xie, J.; Xu, W.; Lu, J.; Chen, X.; Wang, Y. Ganoderma lucidum extract induces G1 cell cycle arrest, and apoptosis in human breast cancer cells. Am. J. Chin. Med. 2012, 40, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.H.; Kuo, H.P.; Hsieh, H.H.; Li, J.W.; Hsu, W.H.; Chen, S.J.; Su, M.H.; Liu, S.H.; Cheng, Y.C.; Chen, C.Y.; et al. Ganoderma tsugae induces S phase arrest and apoptosis in doxorubicin-resistant lung adenocarcinoma H23/0.3 cells via modulation of the PI3K/Akt signaling pathway. Evid. Based Complement. Alternat. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Jinn, T.R.; Wu, C.M.; Tu, W.C.; Ko, J.L.; Tzen, J.T. Functional expression of FIP-gts, a fungal immunomodulatory protein from Ganoderma tsugae in Sf21 insect cells. Biosci. Biotechnol. Biochem. 2006, 70, 2627–2634. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Q.; Li, J.W.; Wang, Z.; Pan, H.X.; Chen, J.X.; Ning, Z.X. Optimization of alkaline extraction of polysaccharides from Ganoderma lucidum and their effect on immune function in mice. Molecules 2010, 15, 3694–3708. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.C.; Ou, C.C.; Chuang, T.C.; Li, J.W.; Lee, Y.J.; Wang, V.; Liu, J.Y.; Chen, C.S.; Lin, S.C.; Kao, M.C. Ganoderma tsugae extract inhibits expression of epidermal growth factor receptor and angiogenesis in human epidermoid carcinoma cells: In vitro and in vivo. Cancer Lett. 2009, 281, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Xu, S.; Cai, Y.; Zhou, M.; Zuo, X.; Chan, P. Ganoderma lucidum protects dopaminergic neuron degeneration through inhibition of microglial activation. Evid. Based Complement. Alternat. Med. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.W.; Chen, K.D.; Lin, W.C. Effect of Ganoderma tsugae on chronically carbon tetrachloride-intoxicated rats. Am. J. Chin. Med. 2004, 32, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Sudheesh, N.P.; Ajith, T.A.; Mathew, J.; Nima, N.; Janardhanan, K.K. Ganoderma lucidum protects liver mitochondrial oxidative stress and improves the activity of electron transport chain in carbon tetrachloride intoxicated rats. Hepatol. Res. 2012, 42, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Mau, J.L.; Lin, H.C.; Chen, C.C. Antioxidant properties of several medicinal mushrooms. J. Agric. Food Chem. 2002, 50, 6072–6077. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, Y.; Liu, X.; Yuan, Y.; Yue, T. Free radical scavenging and immunomodulatory activities of Ganoderma lucidum polysaccharides derivatives. Carbohydr. Polym. 2013, 91, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Chen, X.; Zhong, Z.; Chen, L.; Wang, Y. Ganoderma lucidum polysaccharides: Immunomodulation and potential anti-tumor activities. Am. J. Chin. Med. 2011, 39, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.H.; Chen, W.K.D. Determination of ganoderic acids in triterpenoid constituents of Ganoderma tsugae. J. Food Drug Anal. 2003, 11, 195–200. [Google Scholar]
- Pan, D.; Wang, L.; Chen, C.; Teng, B.; Wang, C.; Xu, Z.; Hu, B.; Zhou, P. Structure characterization of a novel neutral polysaccharide isolated from Ganoderma lucidum fruiting bodies. Food Chem. 2012, 135, 1097–1103. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, S.; Palani, P.; Nishanthi, R.; Kaviyarasan, V. Studies on screening, isolation and purification of a fibrinolytic protease from an isolate (VK12) of Ganoderma lucidum and evaluation of its antithrombotic activity. Med. Mycol. J. 2011, 52, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Oluba, O.M.; Olusola, A.O.; Eidangbe, G.O.; Babatola, L.J.; Onyeneke, E.C. Modulation of lipoprotein cholesterol levels in plasmodium berghei malarial infection by crude aqueous extract of Ganoderma lucidum. Cholesterol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Teng, B.S.; Wang, C.D.; Zhang, D.; Wu, J.S.; Pan, D.; Pan, L.F.; Yang, H.J.; Zhou, P. Hypoglycemic effect and mechanism of a proteoglycan from Ganoderma lucidum on streptozotocin-induced type 2 diabetic rats. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 166–175. [Google Scholar] [PubMed]
- Karwa, A.; Gaikwad, S.; Rai, M.K. Mycosynthesis of silver nanoparticles using Lingzhi or Reishi medicinal mushroom, Ganoderma lucidum (W. Curt.:Fr.) P. Karst. and their role as antimicrobials and antibiotic activity enhancers. Int. J. Med. Mushrooms 2011, 13, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Radak, Z.; Chung, H.Y.; Goto, S. Systemic adaptation to oxidative challenge induced by regular exercise. Free Radic. Biol. Med. 2008, 44, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.S.; Huang, Y.H. Effects of exercise intensity on lymphocyte apoptosis induced by oxidative stress in men. Eur. J. Appl. Physiol. 2005, 95, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Phaneuf, S.; Leeuwenburgh, C. Apoptosis and exercise. Med. Sci. Sports Exerc. 2001, 33, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Igney, F.H.; Krammer, P.H. Death and anti-death: Tumor resistance to apoptosis. Nat. Rev. Cancer 2002, 2, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, A.; Weiss, D.G.; Jonas, L.; Kriehuber, R. Oxidative stress-induced cytotoxic and genotoxic effects of nano-sized titanium dioxide particles in human HaCaT keratinocytes. Toxicology 2012, 296, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Yang, Y.; Hu, Y.; Sun, Y.; Zhang, S.; Peng, W.; Zhong, Y.; Huang, X.; Kong, W. A long-term high-fat diet increases oxidative stress, mitochondrial damage and apoptosis in the inner ear of D-galactose-induced aging rats. Hear Res. 2012, 287, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.F.; Yu, C.C.; Huang, R.F.S. Changes in mitochondrial DNA deletion, content, and biogenesis in folate-deficient tissues of young rats depend on mitochondrial folate and oxidative DNA injuries. J. Nutr. 2007, 137, 2036–2042. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.T.; Yang, S.C.; Tsai, S.C.; Huang, C.C.; Lee, N.Y. L-Arginine attenuates xanthine oxidase and myeloperoxidase activities in hearts of rats during exhaustive exercise. Br. J. Nutr. 2006, 95, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Lin, K.J.; Cheng, Y.W.; Hsu, C.A.; Yang, S.S.; Shyur, L.F. Hepatoprotective effect and mechanistic insights of deoxyelephantopin, a phyto-sesquiterpene lactone, against fulminant hepatitis. J. Nutr. Biochem. 2012. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |
| Parameters | E | EL | EM | EH |
|---|---|---|---|---|
| Initial BW (g) | 236 ± 5 | 238 ± 3 | 243 ± 7 | 236 ± 4 |
| Final BW (g) | 298 ± 12 a | 327 ± 15 ab | 356 ± 13 b | 335 ± 11 ab |
| Daily intake (g) | 18.6 ± 0.7 a | 19.9 ± 0.7 ab | 20.8 ± 0.6 b | 20.9 ± 0.6 b |
| Parameters | E | EL | EM | EH |
|---|---|---|---|---|
| AST (U/L) | 178 ± 17 | 159 ± 16 | 159 ± 21 | 156 ± 15 |
| AST (U/L) | 76.8 ± 10.2 | 61.3 ± 5.9 | 59.5 ± 2.3 | 72.2 ± 15.7 |
| CK (U/L) | 162.6 ± 12.9 | 139.6 ± 44.3 | 209.5 ± 21.3 | 161.8 ± 41.3 |
| LDH (U/L) | 669 ± 117 | 682 ± 152 | 820 ± 114 | 783 ± 206 |
| Glucose (mg/dL) | 53.9 ± 7.4 b | 51.3 ± 7.0 b | 29.3 ± 1.1 a | 36.3 ± 6.2 ab |
| Lactate (mg/dL) | 35.1 ± 5.0 | 29.6 ± 8.1 | 41.3 ± 7.0 | 45.0 ± 7.6 |
| FFA (mM) | 0.40 ± 0.04 b | 0.20 ± 0.06 a | 0.25 ± 0.05 ab | 0.39 ± 0.08 b |
| BUN (mg/dL) | 99.4 ± 16.8 | 67.3 ± 7.6 | 71.4 ± 8.1 | 83.6 ± 21.5 |
| Parameters | E | EL | EM | EH |
|---|---|---|---|---|
| SOD (U/mg Hb) | 9.2 ± 2.0 | 9.1 ± 1.1 | 10.0 ± 0.9 | 11.0 ± 2.9 |
| CAT (mU/mg Hb) | 47.1 ± 13.7 | 58.6 ± 18.1 | 63.7 ± 11.5 | 40.8 ± 13.2 |
| GRD (mU/mg Hb) | 0.51 ± 0.07 | 0.51 ± 0.16 | 0.69 ± 0.19 | 0.55 ± 0.14 |
| GPX (U/g Hb) | 230 ± 50 | 190 ± 37 | 248 ± 38 | 228 ± 60 |
| GSH/GSSG | 41.2 ± 14.6 | 59.3 ± 13.9 | 66.6 ± 14.6 | 36.9 ± 14.5 |
| TBARS (μM) | 4.0 ± 0.4 b | 3.8 ± 0.3 ab | 3.5 ± 0.2 ab | 3.1 ± 0.1 a |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, C.-C.; Huang, W.-C.; Yang, S.-C.; Chan, C.-C.; Lin, W.-T. Ganoderma tsugae Hepatoprotection against Exhaustive Exercise-Induced Liver Injury in Rats. Molecules 2013, 18, 1741-1754. https://doi.org/10.3390/molecules18021741
Huang C-C, Huang W-C, Yang S-C, Chan C-C, Lin W-T. Ganoderma tsugae Hepatoprotection against Exhaustive Exercise-Induced Liver Injury in Rats. Molecules. 2013; 18(2):1741-1754. https://doi.org/10.3390/molecules18021741
Chicago/Turabian StyleHuang, Chi-Chang, Wen-Ching Huang, Suh-Ching Yang, Chih-Chi Chan, and Wan-Teng Lin. 2013. "Ganoderma tsugae Hepatoprotection against Exhaustive Exercise-Induced Liver Injury in Rats" Molecules 18, no. 2: 1741-1754. https://doi.org/10.3390/molecules18021741
APA StyleHuang, C.-C., Huang, W.-C., Yang, S.-C., Chan, C.-C., & Lin, W.-T. (2013). Ganoderma tsugae Hepatoprotection against Exhaustive Exercise-Induced Liver Injury in Rats. Molecules, 18(2), 1741-1754. https://doi.org/10.3390/molecules18021741




