Characterization of Different Capsicum Varieties by Evaluation of Their Capsaicinoids Content by High Performance Liquid Chromatography, Determination of Pungency and Effect of High Temperature
Abstract
:1. Introduction
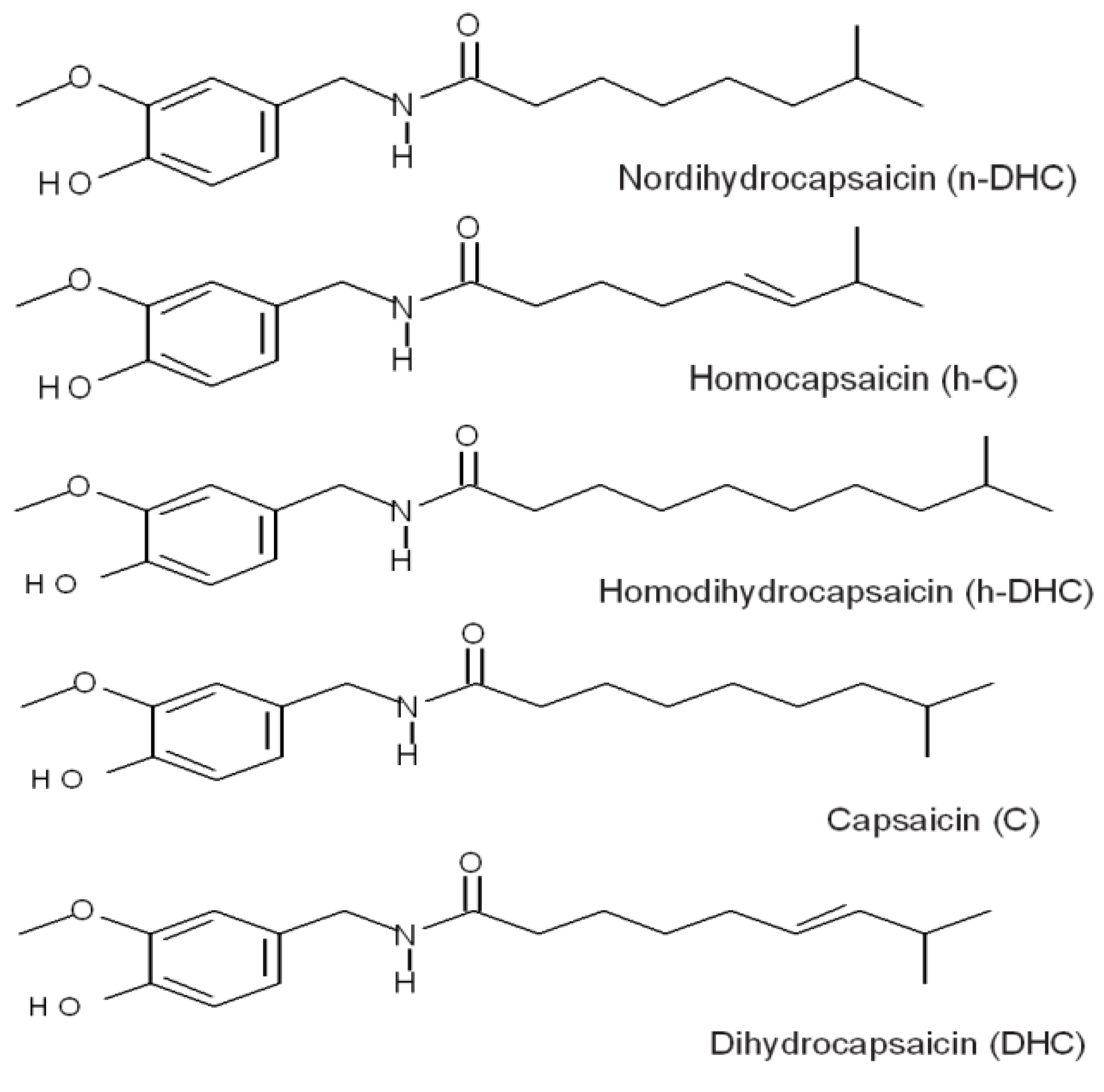
2. Results and Discussion

2.1. Quantification of Individual Compounds
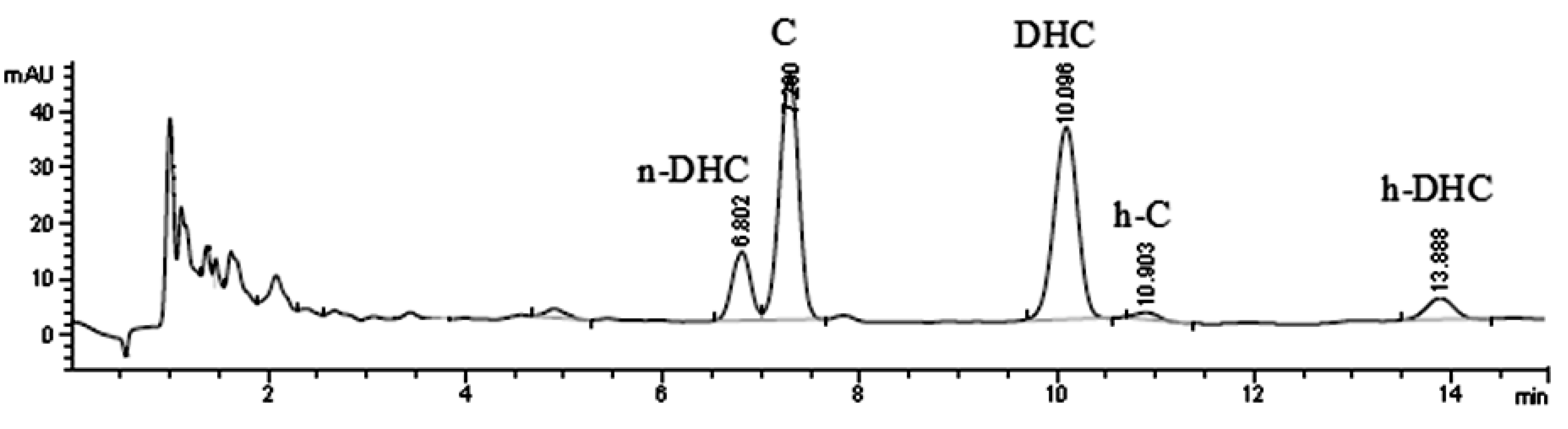
2.2. Effect of High Temperature on Capsaicinoids Content
| Sample | n-DHC | C | DHC | C:DHC ratio | h-C | h-DHC | Total capsaicinoids | SHU | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | |||
| dry weight | dry weight | dry weight | dry weight | dry weight | dry weight | ||||||||
| Ancho cv. Don Matias | 0.25 ± 0.01 ab | 13 | 0.29 ± 0.03 a | 15 | 0.77 ± 0.04 a | 41 | 1:2.7 | ND | − | 0.58 ± 0.12 | 31 | 1.88 ± 0.05 a | 19,335.7 ± 1,133.7 a |
| De árbol | 1.07 ± 0.01 c | 8 | 5.22 ± 0.16 b | 37 | 6.25 ± 0.30 c | 45 | 1:1.2 | 0.13 ± 0.11 | 1 | 1.29 ± 0.77 | 9 | 13.96 ± 0.29 c | 194,591.9 ± 6,852.6 c |
| Bell pepper cv. Cardinal | ND | − | ND | − | ND | − | ND | ND | − | ND | − | ND | ND |
| Chiltepín | 2.17 ± 0.21 d | 7 | 15.36 ± 1.67 d | 48 | 13.39 ± 1.60 e | 42 | 1:0.9 | 0.22 ± 0.07 | 1 | 0.69 ± 0.19 | 2 | 31.84 ± 0.75 e | 483,089.3 ± 54,336.8 e |
| Guajillo cv. San Luis | 0.12 ± 0.03 a | 8 | 0.17 ± 0.01 a | 12 | 0.61 ± 0.11 a | 42 | 1:3.6 | ND | − | 0.56 ± 0.16 | 38 | 1.46 ± 0.08 a | 13,704.8 ± 2,275.7 a |
| Jalapeño cv. Don Julio | 2.48 ± 0.28 d | 12 | 8.03 ± 0.37 c | 38 | 9.39 ± 0.41 d | 45 | 1:1.2 | 0.16 ± 0.13 | 1 | 0.97 ± 0.14 | 5 | 21.03 ± 0.27 d | 303,602.5 ± 8,722.0 d |
| Puya | 0.55 ± 0.06 b | 11 | 1.18 ± 0.07 a | 25 | 2.32 ± 0.22 ab | 48 | 1:2.0 | 0.08 ± 0.01 | 2 | 0.68 ± 0.04 | 14 | 4.80 ± 0.08 ab | 61,526.0 ± 5,117.4 ab |
| Serrano cv. Don Diego | 0.53 ± 0.04 b | 8 | 1.52 ± 0.01 a | 24 | 3.54 ± 0.01 b | 57 | 1:2.3 | ND | − | 0.64 ± 0.09 | 10 | 6.25 ± 0.04 b | 86,427.2 ± 635.4 b |
| Sample | n-DHC | C | DHC | C:DHC ratio | h-C | h-DHC | Total capsaicinoids | SHU | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | % | mg g−1 | |||
| dry weight | dry weight | dry weight | dry weight | dry weight | dry weight | ||||||||
| Ancho cv. Don Matias | 0.44 ± 0.26 ab | 22 | 0.38 ± 0.05 a | 18 | 0.63 ± 0.00 a | 31 | 1:1.7 | ND | − | 0.60 ± 0.04 | 29 | 2.05 ± 0.09 a | 20,275.2 ± 2815.5 d |
| De árbol | 0.64 ± 0.05 ab | 7 | 4.20 ± 0.07 d | 44 | 4.19 ± 0.04 c | 45 | 1:1.0 | 0.04 ± 0.02 | 0.5 | 0.33 ± 0.06 | 4 | 9.40 ± 0.50 c | 141,072.7 ± 2,091.9 d |
| Bell pepper cv. Cardinal | ND | − | ND | − | ND | − | ND | ND | − | 0.41 ± 0.08 | 100 | 0.41 ± 0.08 a | ND |
| Guajillo cv. San Luis | 0.27 ± 0.02 a | 6 | 1.32 ± 0.16 b | 28 | 2.83 ± 0.27 b | 60 | 1:2.2 | ND | − | 0.33 ± 0.08 | 7 | 4.74 ± 0.13 b | 69,210.5 ± 7,228.8 b |
| Jalapeño cv. Don Julio | 1.19 ± 0.08 c | 15 | 2.35 ± 0.11 c | 29 | 4.18 ± 0.27 c | 51 | 1:1.8 | ND | − | 0.40 ± 0.13 | 5 | 8.11 ± 0.15 c | 116,158.5 ± 6,534.7 c |
| Puya | 0.90 ± 0.04 bc | 15 | 1.54 ± 0.26 b | 27 | 3.37 ± 0.82 bc | 58 | 1:2.2 | ND | − | ND | − | 5.81 ± 0.37 b | 87,440.1 ± 19,221.8 bc |
| Serrano cv. Don Diego | 2.47 ± 0.08 d | 14 | 4.76 ± 0.22 e | 26 | 10.14 ± 0.56 d | 56 | 1:2.1 | 0.36 ± 0.03 | 2 | 0.31 ± 0.14 | 2 | 18.05 ± 0.21 d | 262,916.3 ± 13,306.4 e |
3. Experimental
3.1. Chemicals
3.2. Samples and Cultivar Conditions
3.3. Capsaicinoids Extraction
3.4. Capsaicinoids analysis by HPLC-DAD
3.5. Pungency
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hernández, V.; Dávila, S.A.P.; Oyama, K. Síntesis del conocimiento taxonómico, origen y domesticación del género Capsicum. Bol. Soc. Bot. Mex. 1999, 64, 65–84. [Google Scholar]
- Pino, J.; Sauri-Duch, E.; Marbot, R. Change in volatile compounds of Habanero chile pepper (Capsicum chinense Jack. Cv. Habanero) at two ripening stages. Food Chem. 2006, 94, 394–398. [Google Scholar] [CrossRef]
- Marti, M.C.; Camejo, D.; Vallejo, F.; Romojaro, F.; Bacarizo, S.; Palma, J.M.; Sevilla, F.; Jiménez, A. Influence of fuit ripening stage and harvest period on the antioxidant content of sweet pepper cultivars. Plant Food Hum. Nutr. 2011, 66, 416–423. [Google Scholar] [CrossRef]
- Ben-Chaim, A.; Borovsky, Y.; Falise, M.; Mazourek, M.; Kang, B.-C.; Paran, I.; Jahn, M. QTL analysis for capsaicinoid content in Capsicum. Theor. Appl. Genet. 2006, 113, 1481–1490. [Google Scholar]
- Zewdie, Y.; Bosland, P.W. Evaluation of genotype, environment, and genotype-by-environment interaction for capsaicinoids in Capsicum annuum L. Euphytica 2000, 111, 185–190. [Google Scholar] [CrossRef]
- Kosuge, S.; Furata, M. Studies on the pungent principle of Capsicum. Part XIV. Chemical constitution of the pungent principle. Agr. Biol. Chem. Tokyo 1970, 34, 248–256. [Google Scholar] [CrossRef]
- Laskaridou-Monnerville, A. Determination of capsaicin and dihydrocapsaicin by micellar electrokinetic capillary chromatography and its application to various species of Capsicum, Solanaceae. J. Chromatogr. A 1999, 836, 293–302. [Google Scholar] [CrossRef]
- Djamgoz, M.B.A.; Isbilen, B. Dietary compounds as anti-cancer agents: A preliminary evaluation of ion channels and membrane excitability as possible target mechanisms. Turk. J. Biochem. 2006, 31, 57–68. [Google Scholar]
- Mori, A.; Lehmann, S.; O’Kelly, J.; Kumagai, T.; Desmond, J.C.; Pervan, M.; McBride, W.H.; Kizaki, M.; Koeffler, H.P. Capsaicin, a component of red peppers, inhibits the growth of androgen-independent, p53 mutant prostate cancer cells. Cancer Res. 2006, 66, 3222–3229. [Google Scholar] [CrossRef]
- Antonious, G.F.; Kochhar, T.S.; Jarret, R.L.; Snyder, J.C. Antioxidants in hot pepper: Variation among accessions. J. Environm. Sci. Heal. B 2006, 41, 1237–1243. [Google Scholar] [CrossRef]
- Cisneros-Pineda, O.; Torres-Tapia, L.W.; Gutiérrez-Pacheco, L.C.; Contreras-Martín, F.; González-Estrada, T.; Peraza-Sánchez, S.R. Capsaicinoids quantification in chili peppers cultivated in the state of Yucatan, Mexico. Food Chem. 2007, 104, 1755–1760. [Google Scholar]
- Zhang, Q.; Hu, J.; Sheng, L.; Li, Y. Simultaneous quantification of capsaicin and dihydrocapsaicin in rat plasma using HPLC coupled with tandem mass spectrometry. J. Chromatogr. B 2010, 878, 2292–2297. [Google Scholar] [CrossRef]
- Liu, L.; Chen, X.; Liu, J.; Deng, X; Duan, W; Tan, S. Determination of capsaicin and dihydrocapsaicin in Capsicum anuum and related products by capillary electrophoresis with a mixed surfactant system. Food Chem. 2010, 119, 1228–1232. [Google Scholar] [CrossRef]
- Ritota, M.; Marini, F.; Sequi, P.; Valentine, M. Metabolomic characterization of Italian sweet pepper (Capsicum annum L.) by means of HRMAS-NMR spectroscopy and multivariate analysis. J. Agric. Food Chem. 2010, 58, 9675–9684. [Google Scholar] [CrossRef]
- Orellana-Escobedo, L.; Garcia-Amezquita, L.E.; Olivas, G.I.; Ornelas-Paz, J.J.; Sepulveda, D.R. Capsaicinoids content and proximate composition of Mexican chili peppers (Capsicum spp.) cultivated in the State of Chihuahua. Cienc. Tecnol. Aliment. 2013, 11, 179–184. [Google Scholar]
- SIAP. Servicio de Información Agroalimentaria y Pesquera. Producción agrícola por cultivo. Available online: http://www.siap.gob.mx/ (accessed on 25 June 2013).
- Laborde, J.A.; Pozo, C. Presente y Pasado del Chile en México; Secretaría de Agricultura y Recursos Hidráulicos, Instituto Nacional de Investigaciones Agrícolas: DF, Mexico, 1984; p. 80. [Google Scholar]
- Jarret, R.L.; Baldwin, E.; Perkins, B.; Bushway, R.; Guthrie, K. Diversity of fruit quality characteristics in Capsicum frutescens. HortScience 2007, 42, 16–19. [Google Scholar]
- Alvarez-Parrilla, E.; de la Rosa, L.A.; Amaro wicz, R.; Shahidi, F. Antioxidant activity of fresh and processed Jalapeño and Serrano peppers. J. Agric. Food Chem. 2011, 59, 163–173. [Google Scholar]
- Weiss, E.A. Spice Crops; CABI Publishing International: New York, NY, USA, 2002; p. 411. [Google Scholar]
- Harvell, K.P.; Bosland, P.W. The environment produces a significant effect on pungency of chiles. HortScience 1997, 32, 1292. [Google Scholar]
- Wilson, D.R.; Muchow, R.C.; Murgatroyd, C.J. Model analysis of temperature and solar radiation limitations to maize potential productivity in cool climate. Field. Crop. Res. 1995, 43, 1–18. [Google Scholar] [CrossRef]
- CNA. Comisión Nacional del Agua. Available online: http://www.cna.gob.mx/ (accessed on 25 June 2013).
- López, J.D.; Vázquez, C.; Salazar, E.; Zuñiga, R.; Trejo, H.I. Use of soil water content in deciding the sowing time in dry land agriculture. J. Hortic. For. 2010, 2, 122–126. [Google Scholar]
- Morán-Bañuelos, S.H.; Aguilar-Rincón, V.H.; Corona-Torres, T.; Castillo-González, F.; Soto-Hernández, R.M.; San Miguel-Chávez, R. Capsaicinoides en chiles nativos de Puebla, México. Agrociencia-Mexico 2008, 42, 807–816. [Google Scholar]
- Orellana-Escobedo, L.; Ornelas-Paz, J.J.; Olivas, G.I.; Guerrero-Beltran, J.A.; Jimenez-Castro, J.; Sepulveda, D.R. Determination of absolute threshold and just noticeable difference in the sensory perception of pungency. J. Food Sci. 2012, 77, 5135–5139. [Google Scholar]
- International Conference on Harmonisation of Technical Requeriments for Registration of Pharmaceuticals for Human Use. Validation of Analytical Procedures: Text and Methodology Q2(R1); International Conference on Harmonisation of Technical Requeriments for Registration of Pharmaceuticals for Human Use: Geneva, Switzerland, 2005; p. 13. [Google Scholar]
- Collins, M.D.; Wasmund, L.M.; Bosland, P.W. Improved method for quantifying capsaicinoids in Capsicum using high performance liquid chromatography. HortScience 1995, 30, 137–139. [Google Scholar]
- Kurian, A.L.; Starks, A.N. HPLC analysis of capsaicinoids extracted from whole orange Habañero chili peppers. J. Food Sci. 2002, 67, 956–962. [Google Scholar] [CrossRef]
- Barbero, G.F.; Liazid, A.; Palma, M.; Barroso, C.G. Fast determination of capsaicinoids from peppers by High-Performance Liquid Chromatography using a reversed phase monolithic column. Food Chem. 2008, 107, 1276–1282. [Google Scholar] [CrossRef]
- Giuffrida, D.; Dugo, P.; Torre, G.; Bignardi, C.; Cavazza, A.; Corradini, C.; Dugo, G. Characterization of 12 Capsicum varieties by evaluation of their carotenoid profile and pungency determination. Food Chem. 2013, 140, 794–802. [Google Scholar] [CrossRef]
- Govindarajan, V.S.; Sathyanarayana, M.N. Capsicum-production, technology, chemistry, and quality. Part V. Impact on physiology, pharmacology, nutrition and metabolism; structure, pungency, pain and desensitization sequences. Crit. Rev. Food Sci. 1991, 29, 435–473. [Google Scholar] [CrossRef]
- Manirakiza, P.A.; Covaci, A.; Schepens, P. Pungency principles. In Capsicum. The genus Capsicum; de, A.K., Ed.; Taylor and Francis: London, UK, 2003; pp. 71–86. [Google Scholar]
- Garcés-Claver, A.; Arnedo-Andrés, M.S.; Abadía, J.; Gil-Ortega, R.; Alvarez-Fernández, A. Determination of capsaicin and dihydrocapsaicin in Capsicum fruits by liquid chromatography-electrospray/time of flight mass spectrometry. J. Agric. Food Chem. 2006, 54, 9303–9311. [Google Scholar] [CrossRef]
- Collins, M.; Bosland, P. Rare and novel capsaicinoid profiles in Capsicum. Capsicum Eggplant Newsl. 1994, 13, 48–51. [Google Scholar]
- Cruz-Pérez, A.B.; González-Hernández, V.A.; Soto-Hernández, R.M.; Gutiérrez-Espinoza, M.A.; Gardea-Béjar, A.A.; Pérez-Grajalez, M. Capsaicinoides, vitamina C y heterosis durante el desarrollo del fruto de chile manzano. Agrociencia-Mexico 2007, 41, 627–635. [Google Scholar]
- Alpizar-Lara., E.; Trujillo-Aguirre, J.J.G.; Herrera-Rodríguez, F.J. Determinación de capsaicinoides en chile habanero (Capsicum chinense Jaq.), colectados en Yucatán. In Proceedings 16th International Pepper Conference, Tamaulipas, México, 10–12 November 2002; p. 43.
- Cázarez-Sánchez, E.; Ramírez-Vallejo, P.; Castillo-González, F.; Soto Hernández, R.M.; Rodríguez-González, M.T.; Chávez-Servia, J.L. Capsaicinoides y preferencia de uso en diferentes morfotipos de chile (Capsicum annuum L.) del centro-Oriente de Yucatán. Agrociencia 2005, 39, 627–638. [Google Scholar]
- Casanova, C.; Gutiérrez, P.; Torres, T.; Peraza, S.; González, E. Caracterización química y molecular de la pungencia de los chiles (Capsicum spp.) de Yucatán. In Proceedings Third World Pepper Convention, Chihuahua, México, 9–11 July 2006; p. 27.
- Bosland, P.W.; Baral, J.B. “Buhut Jolokia”—The world’s hottest known chile pepper is a putative naturally occurring interspecific hybrid. Hortscience 2007, 42, 222–224. [Google Scholar]
- Canto-Flick, A.; Balam-Uc, E.; Bello-Bello, J.J.; Lecona-Guzmán, C.; Solís-Marroquín, D.; Avilés-Viñas, S.; Gómez-Uc, E.; López-Puc, G.; Santana-Buzzy, N.; Iglesias-Andreu, L.G. Capsaicinoids content in Habanero pepper (Capsicum chinense Jacq.): Hottest known cultivars. Hortscience 2008, 43, 1344–1349. [Google Scholar]
- Barbero, G.F.; Palma, M.; Barroso, C.G. Determination of capsaicinoids in peppers by microwave-assisted extraction-high-performance liquid chromatography with fluorescence detection. Anal. Chim. Acta 2006, 578, 227–233. [Google Scholar] [CrossRef]
- Wahid, A. Physiological implications of metabolites biosynthesis in net assimilation and heat stress tolerance of sugarcane (Saccharum officinarum) sprouts. J. Plant Res. 2007, 120, 219–228. [Google Scholar] [CrossRef]
- Zróbek-Sokolnik, A. Temperature Stress and Response of Plants. In Environmental Adaptations and Stress Tolerance of Plant in the Era of Climate Change; Ahmad, P., Prasad, M.N.V., Eds.; Springer: New York, NY, USA, 2012; pp. 113–134. [Google Scholar]
- Mittler, R. Abiotic stress, the field environment and stress combitation. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef]
- Mateos, R.M.; Jiménez, A.; Román, P.; Romojaro, F.; Bacarizo, S.; Leterrier, M.; Gómez, M.; Sevilla, F.; del Río, L.A.; Corpas, F.J.; Plama, J.M. Antioxidant systems from pepper (Capsicum annuum L.): Involvement in the response to temperature changes in ripe fruits. Int. J. Mol. Sci. 2013, 14, 9556–9580. [Google Scholar] [CrossRef]
- Rahman, M.J.; Inden, H. Effect of nutrient solution and temperature on capsaicin content and yield contributing characteristics in six sweet pepper (Capsicum annuum L.) cultivars. J. Food Agric. Environ. 2012, 10, 524–529. [Google Scholar]
- Tiwari, A.; Kaushil, M.P.; Pandey, K.S.; Dangi, R.S. Adaptability and production of hottest chilli variety under Gwalior agro-climatic conditions. Curr. Sci. 2005, 88, 1545–1546. [Google Scholar]
- Estrada, B.; Pomar, F.; Díaz, J.; Merino, F.; Bernal, M.A. Pungency level in fruits of the Padrón pepper with different water supply. Sci. Hortic. 1999, 81, 385–396. [Google Scholar] [CrossRef]
- Johnson, T.S.; Ravishankar, G.A.; Venkataraman, L.V. Biotransformation of ferulic acid and vanillylamine to capsaicin and vanillin in immobilized cell cultures of Capsicum frutescens. Plant Cell Tiss. Org. 1996, 44, 117–121. [Google Scholar] [CrossRef]
- Narasimha, B.C.; Bhaskar, H.; Kumar, V.; Giridhar, P.; Parimalan, R.; Sharma, A.; Ashwathanarayana, G. Influence of 8-Methyl-nonenoic Acid on capsaicin biosynthesis in in vivo and in-vitro cell cultures of Capsicum spp. J. Agric. Food Chem. 2006, 54, 1854–1859. [Google Scholar] [CrossRef]
- Diaz, J.; Bernal, M.A.; Merino, F. Phenolic metabolism in Capsicum annuum. Recent Res. Dev. Phytochem. 1998, 2, 155–169. [Google Scholar]
- Sukrasno, N.; Yeoman, M.M. Phenylpropanoid metabolism during growth and development of Capsicum frutescens fruits. Phytochemistry 1993, 32, 839–844. [Google Scholar] [CrossRef]
- Lee, Y.; Howard, L.R.; Villalón, B. Flavonoids and antioxidant activity of fresh pepper (Capsicum annuum) cultivars. J. Food Sci. 1995, 60, 473–476. [Google Scholar] [CrossRef]
- Stewart, C., Jr.; Kang, B.-C.; Liu, K.; Mazourek, M.; Moore, S.L.; Yoo, E.Y.; Kim, B.-D.; Paran, I.; Jahn, M.M. The Pun1 gene for pungency in pepper encodes a putative acyltransferase. Plant J. 2005, 42, 675–688. [Google Scholar] [CrossRef]
- Kirschbaum-Titze, P.; Hiepler, C.; Mueller-Seitz, E.; Petz, M. Pungency in Paprika (Capsicum annuum): 1. Decrease in capsaicinoids content following cellular disruption. J. Agric. Food Chem. 2002, 50, 1260–1263. [Google Scholar] [CrossRef]
- Bernal, M.A.; Calderón, A.A.; Pedreño, M.A.; Muñoz, R.; Barceló, A.R.; de Cáceres, F.M. Capsaicin oxidation by peroxidase from Capsicum annuum (var. annuum) fruits. J. Agric. Food Chem. 1993, 41, 1041–1044. [Google Scholar] [CrossRef]
- Contreras-Padilla, M.; Yahia, E.M. Changes in capsacinoids during development, maturation, and senescence of chilie peppers and relation with peroxidase activity. J. Agric. Food Chem. 1998, 46, 2075–2079. [Google Scholar] [CrossRef]
- Saha, S.R.; Hossain, M.M.; Rahman, M.M.; Kuo, C.G.; Abdullah, S. Effect of high temperature stress on the performance of twelve sweet pepper genotypes. Bangladesh J. Agric. Res. 2010, 35, 525–534. [Google Scholar]
- Ruiz-Lau, N.; Medina-Lara, F.; Minero-García, Y.; Zamudio-Moreno, E.; Guzmán-Antonio, A.; Echavarría-Machado, I.; Martínez-Estévez, M. Water deficit affects the accumulation of capsaicinoids in fruits of Capsicum chinense Jacq. Hortscience 2011, 46, 487–492. [Google Scholar]
- Al Othman, Z.A.; Badjah, Y.; Habila, M.A.; Ghafar, A.A. Determination of capsaicin and dihydrocapsaicin in Capsicum fruit samples using high performance liquid chromatography. Molecules 2011, 16, 8919–8929. [Google Scholar] [CrossRef]
- Parrish, M. Liquid chromatographic method of determining capsaicinoids in capsicums and their extractives: Collaborative study. J. Assoc. Off. Anal. Chem. 1996, 79, 738–745. [Google Scholar]
- Todd, P., Jr.; Bensinger, M.G.; Biftu, T. Determination of pungency due to Capsicum by Gas-Liquid Chromatography. J. Food Sci. 1977, 42, 660–665. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
González-Zamora, A.; Sierra-Campos, E.; Luna-Ortega, J.G.; Pérez-Morales, R.; Ortiz, J.C.R.; García-Hernández, J.L. Characterization of Different Capsicum Varieties by Evaluation of Their Capsaicinoids Content by High Performance Liquid Chromatography, Determination of Pungency and Effect of High Temperature. Molecules 2013, 18, 13471-13486. https://doi.org/10.3390/molecules181113471
González-Zamora A, Sierra-Campos E, Luna-Ortega JG, Pérez-Morales R, Ortiz JCR, García-Hernández JL. Characterization of Different Capsicum Varieties by Evaluation of Their Capsaicinoids Content by High Performance Liquid Chromatography, Determination of Pungency and Effect of High Temperature. Molecules. 2013; 18(11):13471-13486. https://doi.org/10.3390/molecules181113471
Chicago/Turabian StyleGonzález-Zamora, Alberto, Erick Sierra-Campos, J. Guadalupe Luna-Ortega, Rebeca Pérez-Morales, Juan Carlos Rodríguez Ortiz, and José L. García-Hernández. 2013. "Characterization of Different Capsicum Varieties by Evaluation of Their Capsaicinoids Content by High Performance Liquid Chromatography, Determination of Pungency and Effect of High Temperature" Molecules 18, no. 11: 13471-13486. https://doi.org/10.3390/molecules181113471
APA StyleGonzález-Zamora, A., Sierra-Campos, E., Luna-Ortega, J. G., Pérez-Morales, R., Ortiz, J. C. R., & García-Hernández, J. L. (2013). Characterization of Different Capsicum Varieties by Evaluation of Their Capsaicinoids Content by High Performance Liquid Chromatography, Determination of Pungency and Effect of High Temperature. Molecules, 18(11), 13471-13486. https://doi.org/10.3390/molecules181113471




