In Vitro Antioxidant Activity Potential of Lantadene A, a Pentacyclic Triterpenoid of Lantana Plants
Abstract
:1. Introduction
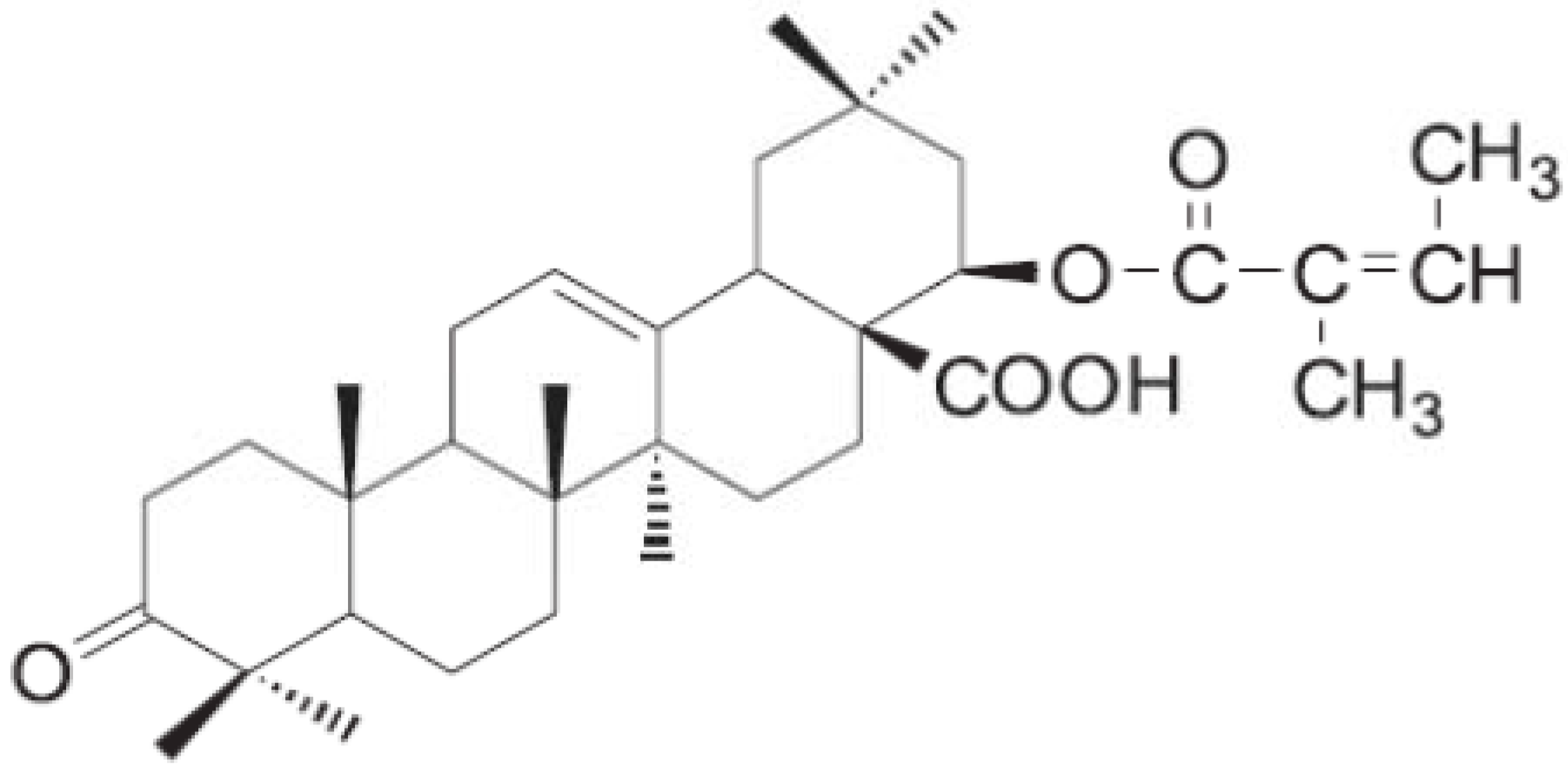
2. Results and Discussion
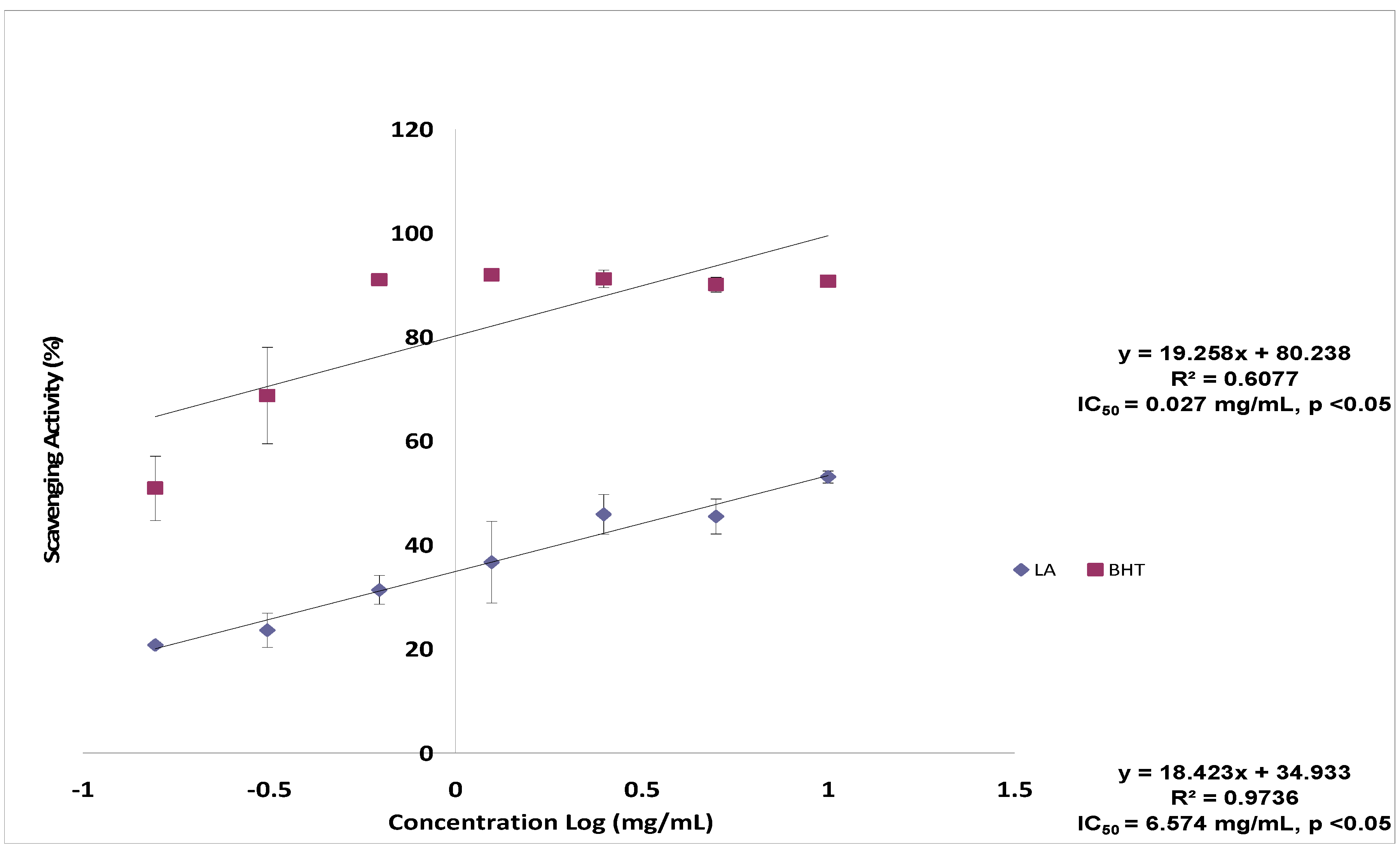
2.1. DPPH Radical Scavenging Activity
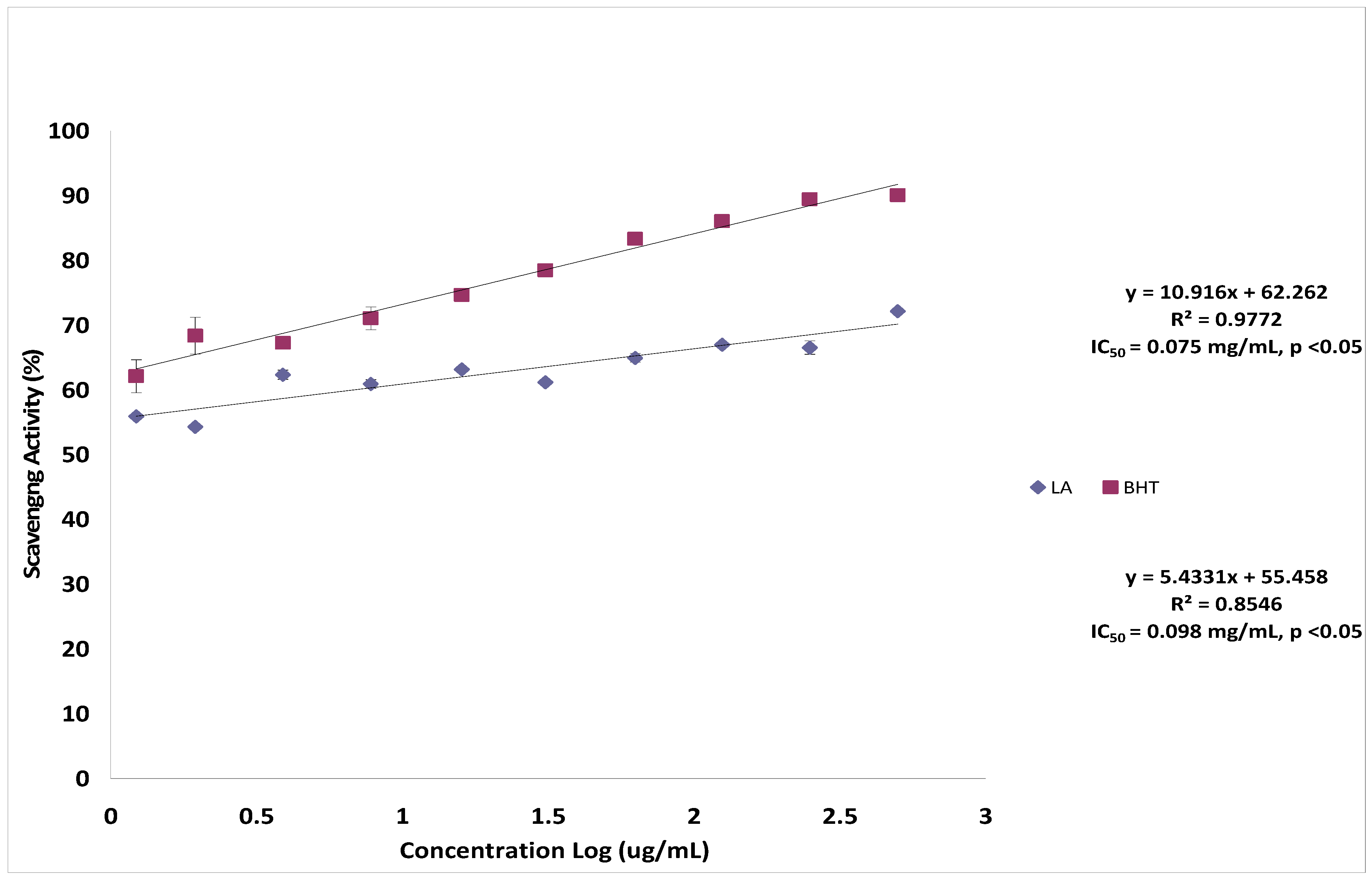
2.2. Hydroxyl Radical Scavenging Activity
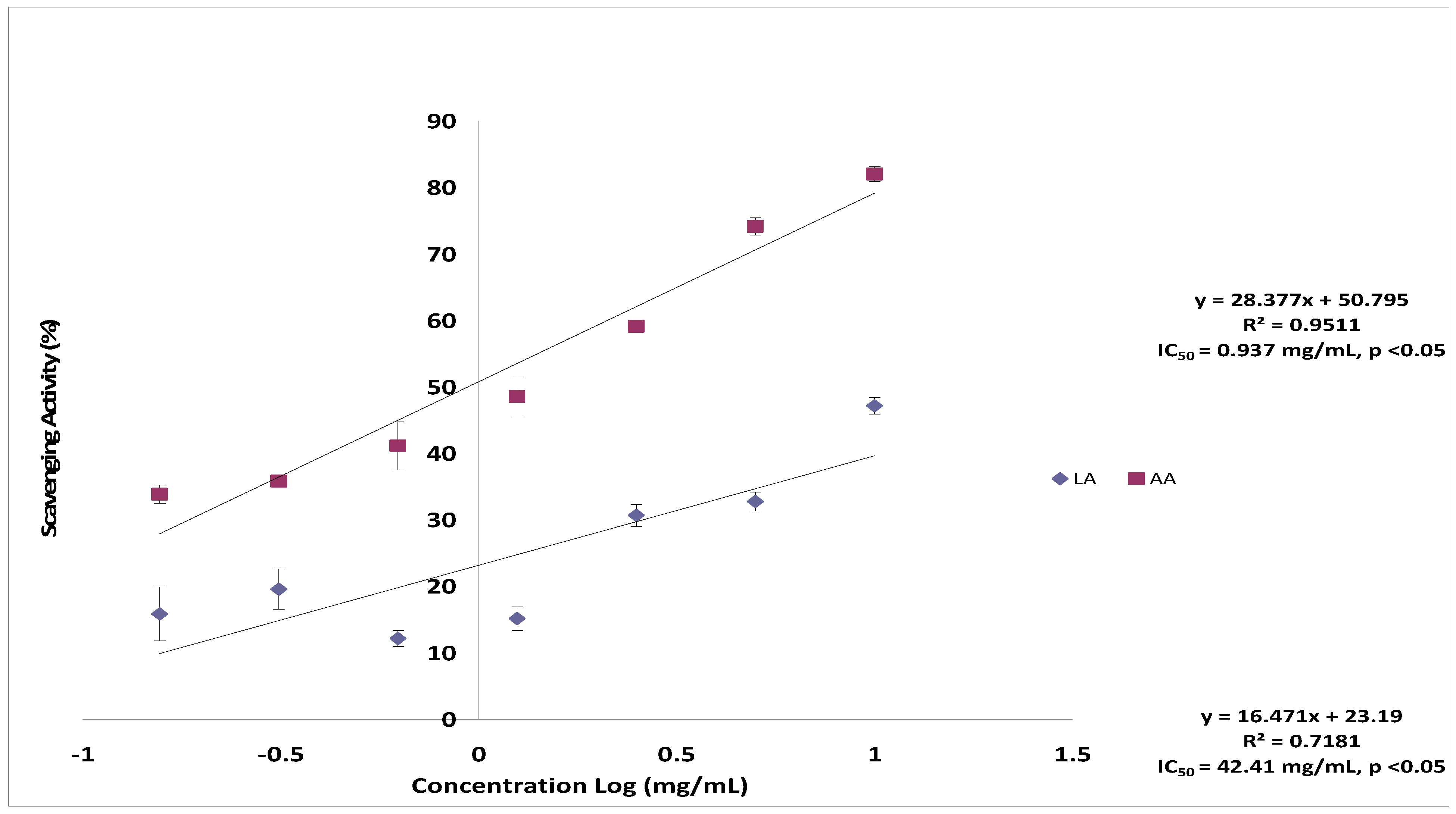
2.3. Superoxide Anion Radical Scavenging Activity

2.4. Nitric Oxide Scavenging Assay
2.5. Ferrous Ion Chelating Assay
2.6. Reducing Power
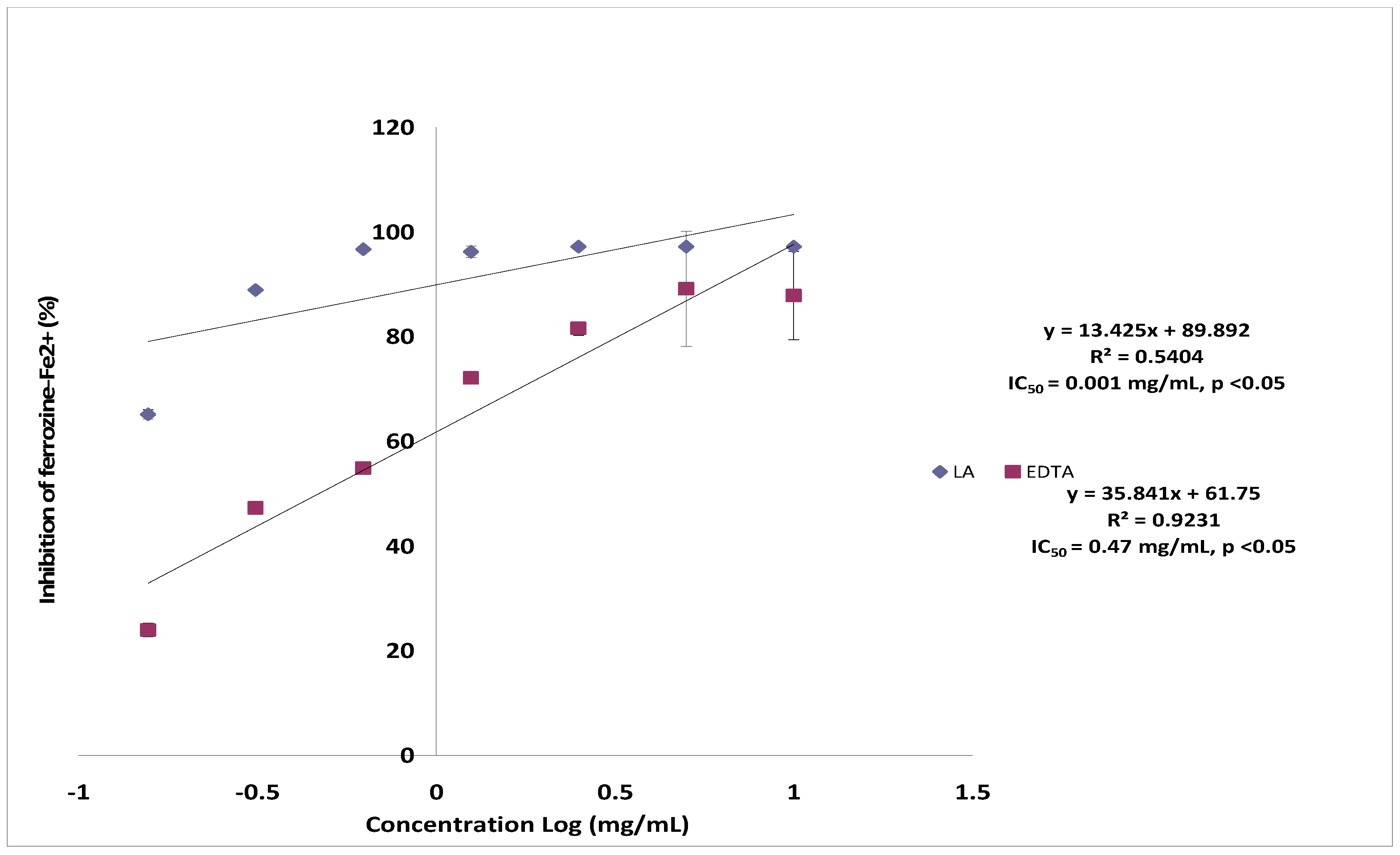
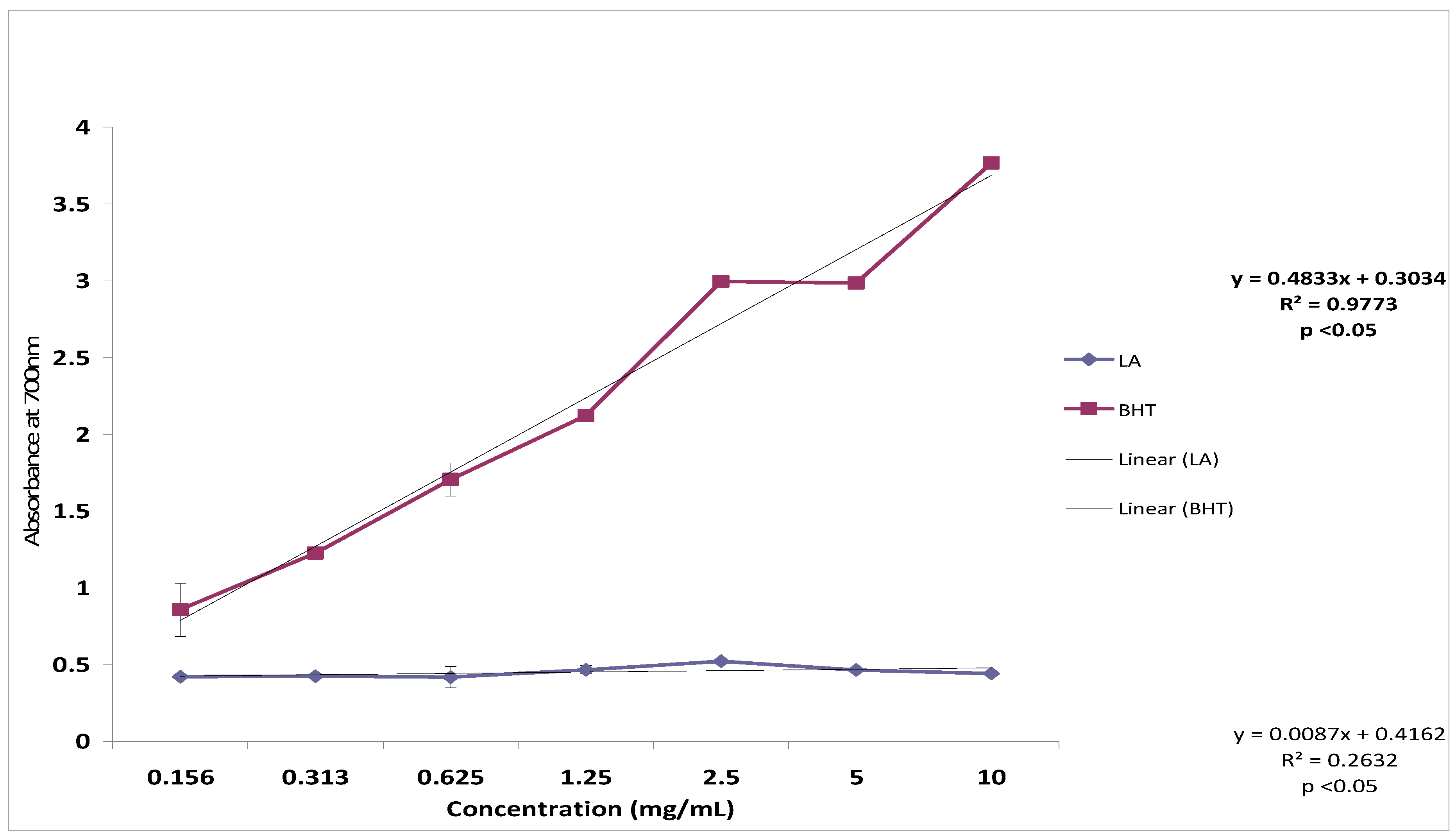
2.7. Toxicity and Possible Molecular Mechanisms of Lantadene A Action
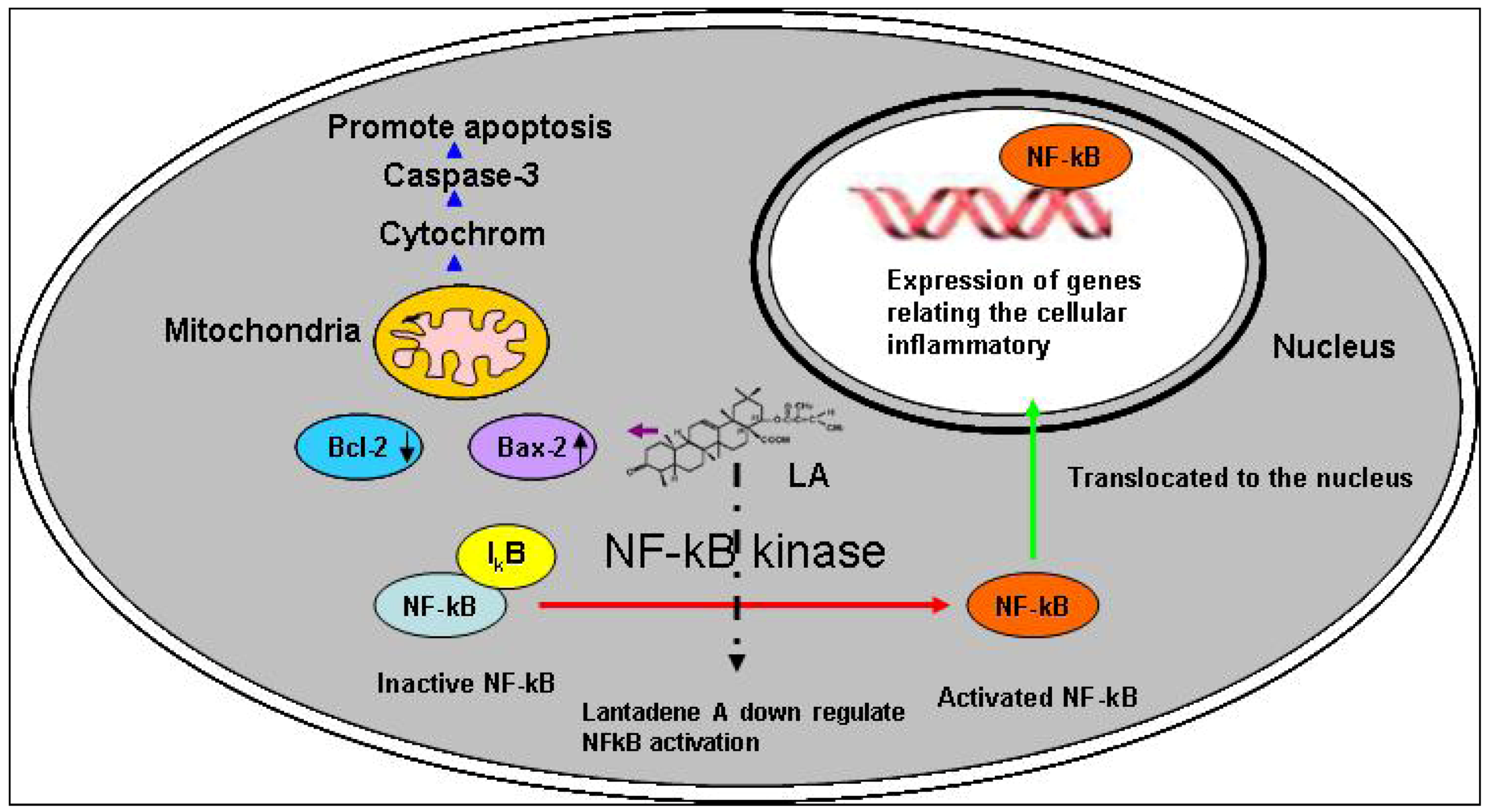
3. Experimental
3.1. Preparation of the Lantadene A
3.2. DPPH Radical-Scavenging Assay
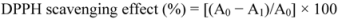
3.3. Reducing power Assay
3.4. Hydroxyl Radical Scavenging Assay
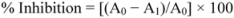
3.5. Nitric Oxide Scavenging Assay
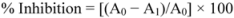
3.6. Ferrous Ion Chelating Assay
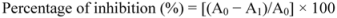
3.7. Determination of Superoxide Anions Scavenging Activity
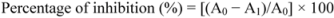
4. Conclusions
Acknowledgments
References
- Sheng, H.; Sun, H. Synthesis, biology and clinical significance of pentacyclic triterpenes: A multi-target approach to prevention and treatment of metabolic and vascular diseases. Nat. Prod. Rep. 2011, 28, 543–593. [Google Scholar] [CrossRef]
- Krasutsky, P.A. Birch barks research and development. Nat. Prod. Rep. 2006, 23, 919–942. [Google Scholar] [CrossRef]
- Chichewicz, R.; Kouzi, S.A. Chemistry, biological activity and chemotherapeutic potential of betulinic acid for the prevention and treatment of cancer and HIV infection. Med. Res. Rev. 2004, 24, 90–114. [Google Scholar] [CrossRef]
- Thurnher, D.; Turhani, D.; Pelzmann, M.; Wannemacher, B.; Knerer, B.; Formanek, M.; Wacheck, V.; Selzer, E. Betulinic acid: A new cytotoxic compound against malignant head and neck cancer cells. Head Neck 2003, 25, 732–740. [Google Scholar] [CrossRef]
- Rajic, A.; Kweifio-Okai, G.; Macrides, T.; Sandeman, R.M.; Chandler, D.S.; Polya, G.M. Inhibition of serine proteases by anti-inflammatory triterpenoids. Planta Med. 2000, 66, 206–210. [Google Scholar] [CrossRef]
- Patocka, J. Biologically active pentacyclic triterpenes and their current medicine signification. J. Appl. Biomech. 2003, 1, 7–12. [Google Scholar]
- Otuki, M.F.; Ferreira, J.; Lima, F.V.; Meyre-Silva, C.; Malheiros, A.; Muller, L.A.; Cani, G.S.; Santos, A.R.S.; Yunes, R.A.; Calixto, J.B. Antinociceptive properties of mixture of α-amyrin and β-amyrin triterpenes; evidence for participation of protein kinase A pathway. J. Pharmacol. Exp. Ther. 2005, 313, 310–318. [Google Scholar]
- Hart, N.K.; Lamberton, J.A.; Sioumis, A.A.; Suares, H. New triterpenoids of Lantana camara. A comparative study of the constituents of several taxa. Aust. J. Chem. 1976, 29, 655–671. [Google Scholar] [CrossRef]
- Sharma, O.P.; Makkar, H.P.S.; Dawra, R.K. A review of the noxious plant Lantana camara. Toxicon 1988, 26, 975–987. [Google Scholar] [CrossRef]
- Sharma, O.P.; Sharma, P.D. Natural products of the lantana plant—The present and prospects. J. Sci. Ind. Res. 1989, 48, 471–478. [Google Scholar]
- Pass, M.A. Current ideas on the pathophysiology and treatment of lantana poisoning of ruminants. Aust. Vet. J. 1986, 63, 169–171. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Parthasarathy, S.; Bin Azizi, J.; Ramanathan, S.; Ismail, S.; Sasidharan, S.; Said, M.I.M.; Mansor, S.M. Evaluation of Antioxidant and Antibacterial Activities of Aqueous, Methanolic and Alkaloid Extracts from Mitragyna Speciosa (Rubiaceae Family) Leaves. Molecules 2009, 14, 3964–3974. [Google Scholar] [CrossRef]
- Kaviarasan, S.; Naik, G.H.; Gangabhagirathi, R.; Anuradha, C.V.; Priyadarsini, K.I. In vitro studies on antiradical and antioxidant activities of fenugreek (Trigonella foenum graecum) seeds. Food Chem. 2007, 103, 31–37. [Google Scholar] [CrossRef]
- Liu, C.Z.; Yu, J.C.; Zhang, X.Z.; Wang, T.; Han, J.X. On changes of activity of antioxidases in hippocampus of rats with multi-infarct dementia and the intervention effects of acupuncture. China J. Trad. Chin. Med. Pharm. 2005, 20, 724–726. [Google Scholar]
- Halliwell, B.; Chirico, S. Lipid peroxidation: Its mechanism, measurement, and significance. Am. J. Clin. Nutr. 1993, 57, 715–724. [Google Scholar]
- Halliwell, B. Reactive oxygen species in living systems: Source, biochemistry, and role in human disease. Am. J. Med. 1991, 91, 14–22. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Riedl, K.M.; Jones, G.A.; Sovik, K.N.; Ritchard, N.T.; Hartzfeld, P.W. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem. 1998, 46, 1887–1892. [Google Scholar] [CrossRef]
- Marcocci, L.; Maguire, J.J.; Droy-Lefaix, M.T.; Packer, L. The nitric oxide-scavenging properties of Ginkgo biloba extract EGb 761. Biochem. Biophys. Res. Commun. 1994a, 15, 748–755. [Google Scholar]
- Marcocci, L.; Packer, L.; Droy-Lefaix, M.T. Antioxidant action of Ginkgo biloba extract EGB 761. Methods Enzymol. 1994b, 234, 462–475. [Google Scholar] [CrossRef]
- Levi, P.E. Mutagenesis. In A Textbook of Modern Toxicology; Hodgson, E., Levi, P.E., Eds.; Elsevier Science Publishing Co.: New York, NY, USA, 1987; pp. 154–159. [Google Scholar]
- Wink, D.A.; Kasprzak, K.S.; Maragos, C.M. DNA deaminating ability and genotoxicity of nitric oxide and its progenitors. Science 1991, 254, 1001–1003. [Google Scholar]
- Dinis, T.C.P.; Madeita, V.M.C.; Almeida, L.M. Action of phenolic derivatives (acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxylradical scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef]
- Ebrahimzadeh, M.A.; Pourmorad, F.; Bekhradnia, A.R. Iron chelating activity, phenol and flavonoid content of some medicinal plants from Iran. Afr. J. Biotechnol. 2008, 7, 3188–3192. [Google Scholar]
- Shinar, E.; Rachmilewitz, E.A. Oxidation denaturation of red blood cells in thalassemia. Semin. Hematol. 1990, 27, 70–91. [Google Scholar]
- Hebbel, R.P.; Leung, A.; Mohandas, N. Oxidation-induced changes in microheological properties of the red cell membrane. Blood 1990, 76, 1015–1022. [Google Scholar]
- Shimada, T.; Watanabe, N.; Ohtsuka, Y.; Endoh, M.; Kojima, K.; Hiraishi, H.; Terano, A. Polaprezinc down-regulates proinflammatory cytokine-induced nuclear factor-kappaB activiation and interleukin-8 expression in gastric epithelial cells. J. Pharmacol. Exp. Ther. 1999, 291, 345–352. [Google Scholar]
- Sen, C.K.; Packer, L. Antioxidant and redox regulation of gene transcription. Fed. Am. Soc. Exp. Biol. J. 1996, 10, 709–720. [Google Scholar]
- Pour, B.M.; Latha, L.Y.; Sasidharan, S. Cytotoxicity and Oral Acute Toxicity Studies of Lantana camara Leaf Extract. Molecules 2011, 16, 3663–3674. [Google Scholar] [CrossRef]
- Sasidharan, S.; Darah, I.; Mohd Jain Noordin, M.K. Free radical Scavenging Activity and Total Phenolic Compounds of Gracilaria changii. Int. J. Nat. Eng. Sci. 2007, 1, 115–117. [Google Scholar]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food Chem. 1995, 43, 27–32. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C.; Aruoma, O.I. The deoxyribose method: A simple test tube assay for determination of rate constants for hydroxyl radicals. Anal. Biochem. 1987, 156, 215–219. [Google Scholar]
- Elizabeth, K.; Rao, M.N.A. Oxygen scavenging activity of curcumin. Int. J. Pharm. 1990, 58, 237–240. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite and (15 N) nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar]
- Ebrahimzadeh, M.A.; Nabavi, S.F.; Nabavi, S.M. Antioxidant activities of methanol extract of Sambucus ebulus L. flower. Pakistan J. Biol. Sci. 2009d, 12, 447–450. [Google Scholar] [CrossRef]
- Nishikimi, M.; Rao, N.A.; Yagi, K. The occurrence of super oxide anion in the reaction of reduced Phenazine methosulphate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–853. [Google Scholar] [CrossRef]
- Kulkarni, R.D. Principles of Pharmacology in Ayurveda; Ram Sangam Graphics: Mumbai, India, 1997. [Google Scholar]
- Sample Availability: Samples of the compound lantadene A is available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Grace-Lynn, C.; Darah, I.; Chen, Y.; Latha, L.Y.; Jothy, S.L.; Sasidharan, S. In Vitro Antioxidant Activity Potential of Lantadene A, a Pentacyclic Triterpenoid of Lantana Plants. Molecules 2012, 17, 11185-11198. https://doi.org/10.3390/molecules170911185
Grace-Lynn C, Darah I, Chen Y, Latha LY, Jothy SL, Sasidharan S. In Vitro Antioxidant Activity Potential of Lantadene A, a Pentacyclic Triterpenoid of Lantana Plants. Molecules. 2012; 17(9):11185-11198. https://doi.org/10.3390/molecules170911185
Chicago/Turabian StyleGrace-Lynn, Chong, Ibrahim Darah, Yeng Chen, Lachimanan Yoga Latha, Subramanion L. Jothy, and Sreenivasan Sasidharan. 2012. "In Vitro Antioxidant Activity Potential of Lantadene A, a Pentacyclic Triterpenoid of Lantana Plants" Molecules 17, no. 9: 11185-11198. https://doi.org/10.3390/molecules170911185
APA StyleGrace-Lynn, C., Darah, I., Chen, Y., Latha, L. Y., Jothy, S. L., & Sasidharan, S. (2012). In Vitro Antioxidant Activity Potential of Lantadene A, a Pentacyclic Triterpenoid of Lantana Plants. Molecules, 17(9), 11185-11198. https://doi.org/10.3390/molecules170911185



