Antitrypanosomal Alkaloids from the Marine Bacterium Bacillus pumilus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Collection and Identification of Biological Material
2.2. Extraction, Isolation and Structural Elucidation of Compounds
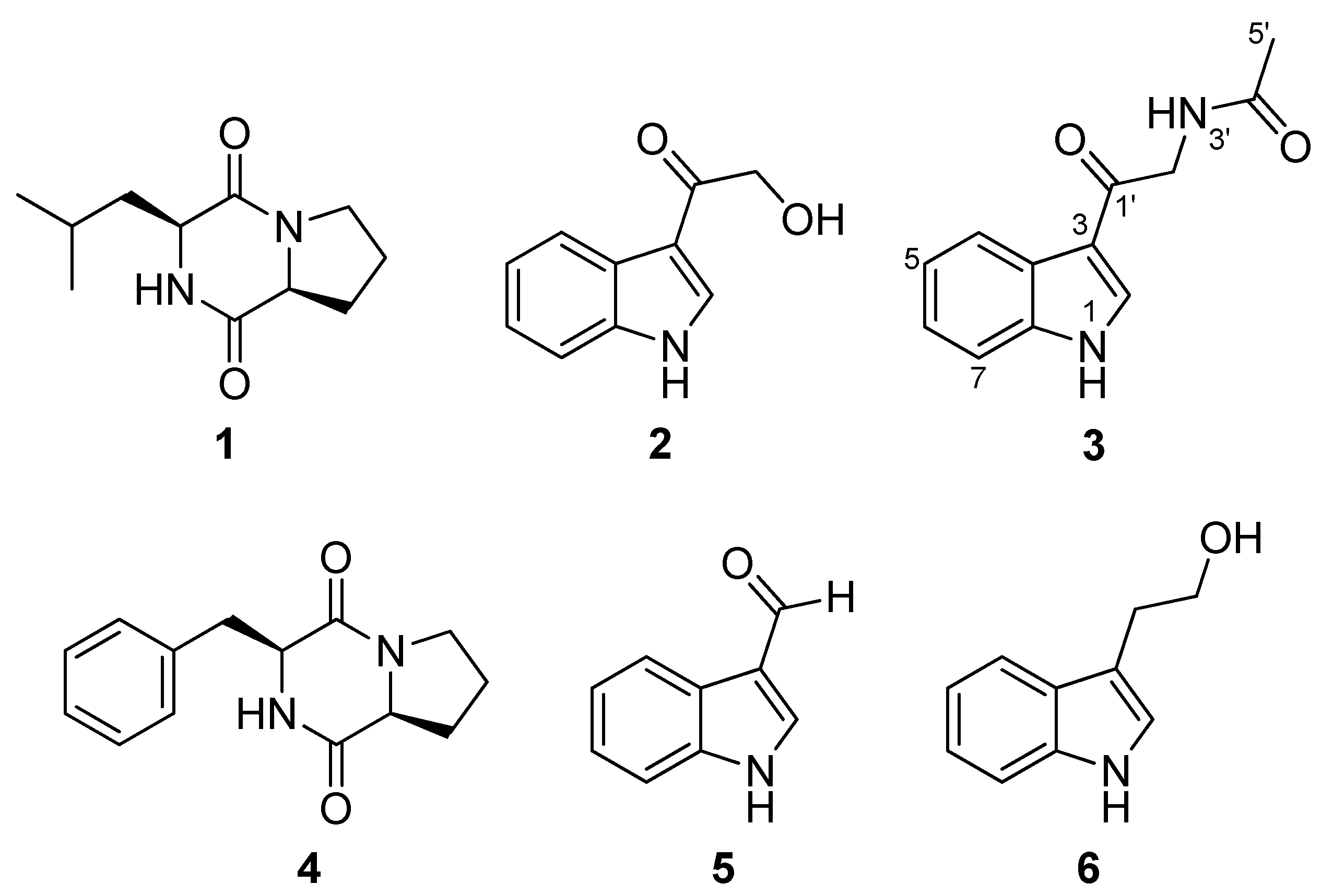
2.3. Antiparasitic Activity
| Compounds | L. donovani | P. falciparum | T. cruzia |
|---|---|---|---|
| Cyclo-(L-Leu-L-Pro) (1) | I | I | I |
| 3-Hydroxyacetylindole (2) | I | I | 20.6 |
| N-acetyl-β-oxotryptamine (3) | I | I | 19.4 |
| Cyclo-(L-Phe-L-Pro) (4) | I | I | I |
| 3-Formylindole (5) | I | I | 26.9 |
| Nifurtimox | 1.6 |
3. Experimental
3.1. General Procedures
3.2. Biological Material Collection and Identification
3.3. Fermentation and Extraction
3.4. Isolation of Alkaloids
3.5. Spectral Data
3.6. In Vitro Biological Activity
3.6.1. Plasmodium falciparum Bioassay
3.6.2. Leishmania donovani Bioassay
3.6.3. Trypanosoma cruzi Bioassay
3.6.4. Cytotoxicity Bioassay
4. Conclusions
Acknowledgments
References
- World Health Organization. Available online: http://www.who.int/mediacentre/factsheets/fs340/en/index.html (accessed on 7 September 2012).
- Rassi, A., Jr.; Rassi, A.; Marin-Neto, J.A. Chagas disease. Lancet 2010, 375, 1388–1402. [Google Scholar] [CrossRef]
- Andriani, G.; Chessler, A.D.; Courtemanche, G.; Burleigh, B.A.; Rodriguez, A. Activity in vivo of anti-Trypanosoma cruzi compounds selected from a high throughput screening. PLoS Negl. Trop. Dis. 2011, 5, e1298. [Google Scholar] [CrossRef]
- Izumi, E.; Ueda-Nakamura, T.; Filho, B.P.D.; Júnior, V.F.V.; Nakamura, C.V. Natural products and Chagas’ disease: A review of plant compounds studied for activity against Trypanosoma cruzi. Nat. Prod. Rep. 2011, 28, 809–823. [Google Scholar] [CrossRef]
- Rodriguez, I.A.; Fernandez-Amaral, A.C.; Santos-Rosa, M.S. Trypanosomatid enzymes as target for plant-derived compounds: New perspectives for phytotherapeutic approaches. Curr. Enzyme Inhib. 2011, 7, 32–41. [Google Scholar]
- Ioset, J.R. Natural products for neglected diseases: A review. Curr. Org. Chem. 2008, 12, 643–666. [Google Scholar] [CrossRef]
- Regalado, E.L.; Tasdemir, D.; Kaiser, M.; Cachet, N.; Amade, P.; Thomas, O.P. Antiprotozoal steroidal saponins from the marine sponge Pandaros acanthifolium. J. Nat. Prod. 2010, 73, 1404–1410. [Google Scholar] [CrossRef]
- Kossuga, M.H.; Nascimento, A.M.; Reimão, J.Q.; André, G.T.; Taniwaki, N.N.; Veloso, K.; Ferreira, A.G.; Cavalcanti, B.C.; Pessoa, C.; Moraes, M.O.; et al. Antiparasitic, antineuroinflammatory, and cytotoxic polyketides from the marine sponge plakortis angulospiculatus collected in Brazil. J. Nat. Prod. 2008, 71, 334–339. [Google Scholar]
- Gutiérrez, M.; Capson, T.L.; Guzmán, H.M.; González, J.; Ortega-Barría, E.; Quiñoá, E.; Riguera, R. Antiprotozoal activity against Plasmodium falciparum and Trypanosoma cruzi of aeroplysinin-1 isolated from the new sponge Aplysina chiriquiensis. Pharm. Biol. 2005, 43, 762–765. [Google Scholar] [CrossRef]
- Wright, A.D.; Goclik, E.; Konig, G.; Kaminsky, R. Lepadins D–F. Antiplasmodial and antitrypanosomal decahydroquinoline derivatives from the tropical marine tunicate Didemnum sp. J. Med. Chem. 2002, 45, 3067–3072. [Google Scholar] [CrossRef]
- Pontius, A.; Krick, A.; Kehraus, S.; Brun, R.; König, G.M. Antiprotozoal activities of heterocyclic-substituted xanthones from the marine-derived fungus Chaetomium sp. J. Nat. Prod. 2008, 71, 1579–1584. [Google Scholar] [CrossRef]
- Osterhage, C.; Kaminsky, R.; König, G.M.; Wright, A.D. Ascosalipyrrolidinone A, an antimicrobial alkaloid, from the obligate marine fungus Ascochyta salicorniae. J. Org. Chem. 2000, 65, 6412–6417. [Google Scholar] [CrossRef]
- Linington, R.G.; Edwards, D.J.; Shuman, C.F.; McPhail, K.L.; Matainaho, T.; Gerwick, W.H. Symplocamide A, a potent cytotoxin and chymotrypsin inhibitor from the marine cyanobacterium Symploca sp. J. Nat. Prod. 2008, 71, 22–27. [Google Scholar]
- Simmons, T.L.; Engene, N.; Ureña, L.D.; Romero, L.I.; Ortega-Barría, E.; Gerwick, L.; Gerwick, W.H. Viridamides A and B, lipodepsipeptides with antiprotozoal activity from the marine cyanobacterium Oscillatoria nigro-viridis. J. Nat. Prod. 2008, 71, 1544–1550. [Google Scholar] [CrossRef]
- Linington, R.G.; Gonzalez, J.; Ureña, L.D.; Romero, L.I.; Ortega-Barría, E.; Gerwick, W.H. Venturamides A and B: Antimalarial constituents of the panamanian marine cyanobacterium Oscillatoria sp. J. Nat. Prod. 2007, 70, 397–401. [Google Scholar] [CrossRef]
- Mondol, M.A.M.; Kim, J.H.; Lee, H.S.; Lee, Y.J.; Shin, H.J. Macrolactin W, a new macrolide from a marine Bacillus sp. Bioorg. Med. Chem. Lett. 2011, 21, 3832–3835. [Google Scholar] [CrossRef]
- Mondol, M.A.M.; Tareq, F.S.; Kim, J.H.; Lee, M.H.; Lee, Y.L.; Lee, J.S.; Shin, H.J. Cyclic ether-containing macrolactins, antimicrobial 24-membered isomeric macrolactones from a marine Bacillus sp. J. Nat. Prod. 2011, 74, 2582–2587. [Google Scholar] [CrossRef]
- Lu, X.L.; Xu, Q.Z.; Shen, Y.H.; Liu, X.Y.; Jiao, B.H.; Zhang, W.D.; Ni, K.Y. Macrolactin S, a novel macrolactin antibiotic from marine Bacillus sp. Nat. Prod. Res. 2008, 22, 342–347. [Google Scholar] [CrossRef]
- Wang, G.; Dai, S.; Chen, M.; Wu, H.; Xie, L.; Luo, X.; Li, X. Two diketopiperazine cyclo(pro-phe) isomers from the marine bacteria Bacillus subtilis sp. 13-2. Chem. Nat. Compd. 2010, 46, 583–585. [Google Scholar]
- Berrue, F.; Ibrahim, A.; Boland, P.; Kerr, R.G. Newly isolated marine Bacillus pumilus (sp21): A source of novel lipoamides and other antimicrobial agents. Pure Appl. Chem. 2009, 81, 1027–1031. [Google Scholar] [CrossRef]
- Mondol, M.A.M.; Kim, J.H.; Lee, M.A.; Tareq, F.S.; Lee, H.S.; Lee, Y.J.; Shin, H.J. Iedomycins A–D, antimicrobial fatty acids from a marine Bacillus sp. J. Nat. Prod. 2011, 74, 1606–1612. [Google Scholar] [CrossRef]
- Zhang, H.L.; Hua, H.M.; Pei, Y.H.; Yao, X.S. Three new cytotoxic cyclic acylpeptides from marine Bacillus sp. Chem. Pharm. Bull. 2004, 52, 1029–1030. [Google Scholar] [CrossRef]
- Oleinikova, G.K.; Dmitrenok, A.S.; Voinov, V.G.; Chaikina, E.L.; Shevchenko, L.S.; Kuznetsova, T.A. Bacillomycin D from the marine isolate of Bacillus subtilis KMM 1922. Chem. Nat. Comp. 2005, 41, 461–464. [Google Scholar] [CrossRef]
- Pettit, J.R.; Knight, J.C.; Herald, D.L.; Pettit, R.K.; Hogan, F.; Mukku, V.J.R.; Hamblin, J.S.; Dodson, M.J., II.; Chapuis, J.C. Antineoplastic agents. 570. Isolation and structure elucidation of bacillistatins 1 and 2 from a marine Bacillus silvestris. J. Nat. Prod. 2009, 72, 366–371. [Google Scholar]
- Boya, C.A.; Herrera, L.; Guzman, H.M.; Gutierrez, M. Antiplasmodial activity of bacilosarcin A isolated from the octocoral-associated bacterium Bacillus sp. collected in Panama. J. Pharm. Bioall. Sci. 2012, 4, 66–69. [Google Scholar] [CrossRef]
- Jeong, S.H.; Ishida, K.; Ito, I.; Okada, S.; Murakami, M. Bacillamide, a novel algicide from the marine bacterium, Bacillus sp. SY-1, against the harmful dinoflagellate, Cochlodinium polykrikoides. Tetrahedron Lett. 2003, 44, 8005–8007. [Google Scholar]
- Martínez-Luis, S.; Ballesteros, J.; Gutierrez, M. Antibacterial constituents from the octocoral-associated bacterium Pseudoalteromonas sp. Rev. Latinoamer. Quím. 2011, 39, 94–102. [Google Scholar]
- Bohlendorf, B.; Forche, E.; Bedorf, N.; Gerth, K.; Irschik, H.; Jansen, R.; Kunze, B.; Trowitzsch-Kienast, W.; Reichenbach, H.; Hofle, G. Indole and quinoline derivatives as metabolites of tryptophan in myxobacteria. Liebigs Ann. 1996, 1, 49–53. [Google Scholar]
- Chen, Y.; Zeeck, A.; Chen, Z.; Zähner, H. Metabolic products of microorganisms-222-β-oxotryptamine derivatives isolated from Streptomyces Ramulosus. J. Antibiot. 1983, 36, 913–915. [Google Scholar] [CrossRef]
- Bernart, M.; Gerwick, W.H. 3-(Hydroxyacetyl)indole, a plant growth regulator from the Oregon red alga Prionitis lanceolata. Phytochemistry 1990, 29, 3697–3698. [Google Scholar] [CrossRef]
- Orhan, I.; Sener, B.; Kaiser, M.; Brun, R.; Tasdemir, D. Inhibitory activity of marine sponge-derived natural products against parasitic protozoa. Mar. Drugs 2010, 8, 47–58. [Google Scholar] [CrossRef]
- Corbett, Y.; Herrera, L.; González, J.; Cubilla, L.; Capson, T.L.; Colley, P.D.; Kursar, T.A.; Romero, L.I.; Ortega-Barría, E. A novel DNA-based microfluorimetric method to evaluate antimalarial drug activity. Am. J. Trop. Med. Hyg. 2004, 70, 119–124. [Google Scholar]
- Williams, C.; Espinosa, O.A.; Montenegro, H.; Cubilla, L.; Capson, T.L.; Ortega-Barría, E.; Romero, L.I. Hydrosoluble formazan XTT: Its application to natural products drug discovery for Leishmania. J. Microbiol. Methods 2003, 55, 813–816. [Google Scholar] [CrossRef]
- Díaz-Chiguer, D.; Márquez-Navarro, A.; Nogueda-Torres, B.; León-Ávila, G.; Pérez-Villanueva, J.; Hernández-Campos, A.; Castillo, R.; Ambrosio, J.; Nieto-Meneses, R.; Yépez-Mulia, L.; et al. In vitro and in vivo trypanocidal activity of some benzimidazole derivatives against two strains of Trypanosoma cruzi. Acta Trop. 2012, 122, 108–112. [Google Scholar] [CrossRef]
- Solari, A.; Ortiz, S.; Soto, A.; Arancibia, C.; Campillay, R.; Contreras, M.; Salinas, P.; Rojas, A.; Schenone, H. Treatment of Trypanosoma cruzi infected-children with nifurtimox: A 3 year follow-up by PCR. J. Antimic. Chemother. 2001, 48, 515–519. [Google Scholar] [CrossRef]
- Manger, R.L.; Leja, L.S.; Lee, S.Y.; Hungerford, J.M.; Hokama, Y.; Dickey, R.W.; Granade, H.R.; Lewis, R.; Yasumoto, T.; Wekell, M.M. Detection of sodium channel toxins: directed cytotoxicity assays of purified ciguatoxins, brevetoxins, saxitoxins, and seafood extracts. J. AOAC Int. 1995, 78, 521–527. [Google Scholar]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Martínez-Luis, S.; Gómez, J.F.; Spadafora, C.; Guzmán, H.M.; Gutiérrez, M. Antitrypanosomal Alkaloids from the Marine Bacterium Bacillus pumilus. Molecules 2012, 17, 11146-11155. https://doi.org/10.3390/molecules170911146
Martínez-Luis S, Gómez JF, Spadafora C, Guzmán HM, Gutiérrez M. Antitrypanosomal Alkaloids from the Marine Bacterium Bacillus pumilus. Molecules. 2012; 17(9):11146-11155. https://doi.org/10.3390/molecules170911146
Chicago/Turabian StyleMartínez-Luis, Sergio, José Félix Gómez, Carmenza Spadafora, Héctor M. Guzmán, and Marcelino Gutiérrez. 2012. "Antitrypanosomal Alkaloids from the Marine Bacterium Bacillus pumilus" Molecules 17, no. 9: 11146-11155. https://doi.org/10.3390/molecules170911146
APA StyleMartínez-Luis, S., Gómez, J. F., Spadafora, C., Guzmán, H. M., & Gutiérrez, M. (2012). Antitrypanosomal Alkaloids from the Marine Bacterium Bacillus pumilus. Molecules, 17(9), 11146-11155. https://doi.org/10.3390/molecules170911146





