Fumigant Antifungal Activity of Myrtaceae Essential Oils and Constituents from Leptospermum petersonii against Three Aspergillus Species
Abstract
:1. Introduction
2. Results and Discussion
2.1. Fumigant Antifungal Activity of Plant Essential Oils
| Plant species | Conc., ×10−3 mg/mL air | Inhibition rate (Mean ± S.E., %) | ||
|---|---|---|---|---|
| Aspergillus ochraceus | Aspergillus flavus | Aspergillus niger | ||
| Eucalyptus citriodora | 56 | 0d 1 | 23.5 ± 2.6c | 0c |
| Eucalyptus polybractea | 56 | 0d | 0d | 0c |
| Eucalyptus smithii | 56 | 0d | 0d | 0c |
| Eucalyptus radiata | 56 | 0d | 0d | 0c |
| Eucalyptus dives | 56 | 0d | 0d | 0c |
| Eucalyptus globulus | 56 | 26.1 ± 0.4b | 0d | 0c |
| Melaleuca dissitiflora | 56 | 21.5 ± 0.6c | 22.9 ± 1.9c | 0c |
| Melaleuca quinquenervia | 56 | 26.5 ± 0.4b | 34.4 ± 7.0b | 0c |
| Melaleuca uncinata | 56 | 0d | 0d | 0c |
| Melaleuca linariifolia | 56 | 0d | 0d | 0c |
| Leptospermum petersonii | 56 | 100a | 100a | 100a |
| 28 | 100a | 100a | 100a | |
| 14 | 0d | 20.2 ± 4.3c | 18.8 ± 13.4b | |
| F13,42 = 0.25 | F13,42 = 5.68 | F13,42 = 12.84 | ||
| p < 0.0001 | p < 0.0001 | p < 0.0001 | ||
2.2. Chemical Components of Plant Essential Oils
| No. | Compounds | RI 2 | Amount (w/w, %) | |
|---|---|---|---|---|
| DB-1 | FFAP | |||
| 1 | α-Pinene | 928 | 1014 | 0.54 |
| 2 | 6-Methyl-5-heptene-2-one | 964 | 1330 | 0.77 |
| 3 | β-Pinene | 967 | 1098 | 0.24 |
| 4 | β-Myrcene | 981 | 1155 | 1.26 |
| 5 | p-Cymene | 1012 | 1261 | 0.07 |
| 6 | 1,8-Cineole | 1018 | 1196 | 0.09 |
| 7 | Limonene | 1020 | 1189 | 0.11 |
| 8 | Linalool | 1084 | 1536 | 3.25 |
| 9 | Isopulegol | 1128 | 1559 | 5.20 |
| 10 | Citronellal | 1131 | 1470 | 16.72 |
| 11 | Citronellol | 1211 | 1753 | 6.00 |
| 12 | Neral | 1216 | 1670 | 22.83 |
| 13 | Geraniol | 1236 | 1834 | 3.63 |
| 14 | Geranial | 1245 | 1721 | 29.91 |
| 15 | β-Caryophyllene | 1414 | 1583 | 0.14 |
| 16 | Aromadendrene | 1435 | 1597 | 0.08 |
| Sum of identified compounds | 90.85 | |||
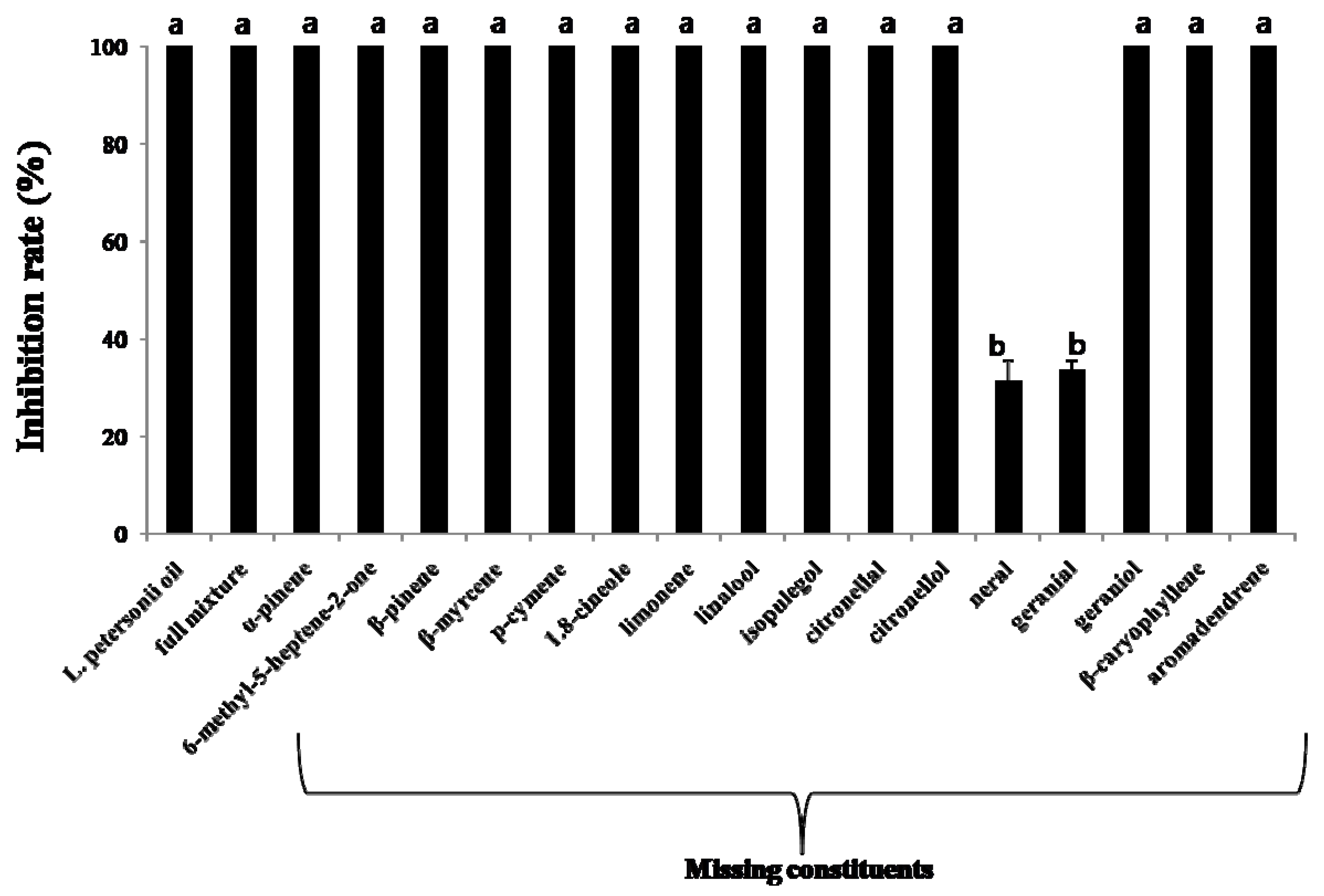
2.3. Antifungal Activities of Individual Compounds
| Compounds | Conc., ×10−3 mg/mL air | Inhibition rate (Mean±S.E., %) | ||
|---|---|---|---|---|
| Aspergillus ochraceus | Aspergillus flavus | Aspergillus niger | ||
| α-Pinene | 56 | 0f 1 | 0d | 0e |
| 6-Methyl-5-heptene-2-one | 56 | 0f | 0d | 0e |
| β-Pinene | 56 | 0f | 0d | 0e |
| β-Myrcene | 56 | 0f | 0d | 0e |
| p-Cymene | 56 | 0f | 0d | 0e |
| 1,8-Cineole | 56 | 0f | 0d | 0e |
| Limonene | 56 | 0f | 0d | 0e |
| Linalool | 56 | 36.6 ± 0.8d | 0d | 0e |
| Isopulegol | 56 | 34.6 ± 3.2d | 0d | 33.4 ± 2.2cd |
| Citronellal | 56 | 0f | 0d | 16.2 ± 11.8de |
| Citronellol | 56 | 83.7 ± 3.3b | 90 ± 0.7a | 81.1 ± 9.0b |
| 28 | 29.4 ± 3.0de | 37.7 ± 4.1b | 35.3 ± 7.0c | |
| Neral | 56 | 100a | 100a | 100a |
| 28 | 100a | 26.9 ± 2.1bc | 0e | |
| 14 | 100a | -2 | - | |
| 7 | 34.1 ± 4.2d | - | - | |
| Geraniol | 56 | 64.6 ± 7.5c | 36.7 ± 11.3b | 77.1 ± 9.8b |
| 28 | 38.4 ± 4.4d | - | 0e | |
| Geranial | 56 | 100a | 100a | 100a |
| 28 | 100a | 29.8±3.3bc | 0e | |
| 14 | 20.1 ± 3.8e | 22.3 ± 3.5c | - | |
| β-Caryophyllene | 56 | 0f | 0d | 0e |
| Aromadendrene | 56 | 0f | 0d | 0e |
| F22,69 = 6.14 | F19,60 = 8.77 | F19,60 = 18.71 | ||
| p < 0.0001 | p < 0.0001 | p < 0.0001 | ||
2.4. Antifungal Activity of Artificial Blends
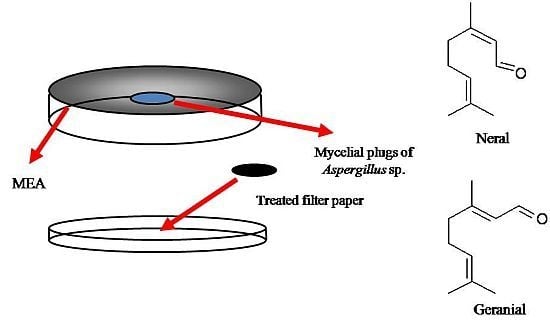
3. Experimental
3.1. Fungal Strains and Culture Conditions
3.2. Essential Oils and Chemicals
3.3. Chemical Analysis of L. petersonii Oil
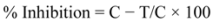
3.5. Antifungal Activity of Artificial Blends
3.6. Stastistical Analysis
4. Conclusions
References
- Abarc, M.L.; Bragulat, M.R.; Castella, G.; Cabanes, F.J. Mycoflora and aflatoxin-producing strains in animal mixed feeds. J. Food. Protect. 1994, 57, 256–258. [Google Scholar]
- Fente, C.A.; Jaimez, O.J.; Vizquez, B.I.; Franco, C.M.; Cepeda, A. A new additive for culture media for rapid determination of aflatoxin-producing Aspergillus strains. Appl. Environ. Microbiol. 2001, 67, 4858–4862. [Google Scholar] [CrossRef]
- Dvorackova, I. Aflatoxins and Human Health; CRC Press: Boca Raton, FL, USA, 1990; p. 154. [Google Scholar]
- Höhler, D. Ochratoxin A in food and feed: Occurrence, legislation and mode of action. Z. Ernährungswiss. 1998, 37, 2–12. [Google Scholar] [CrossRef]
- Park, I.K.; Choi, K.S.; Kim, D.H.; Choi, I.H.; Kim, L.S.; Bak, W.C.; Choi, J.W.; Shin, S.C. Fumigant activity of plant essential oils and components from horseradish, anise and garlic oils against Lycoriella ingenua (Diptera: Sciaridae). Pest Manag. Sci. 2006, 62, 723–728. [Google Scholar] [CrossRef]
- Park, H.M.; Kim, J.; Chang, K.S.; Kim, B.S.; Yang, Y.J.; Kim, G.H.; Shin, S.C.; Park, I.K. Larvicidal activity of Myrtaceae essential oils and their components against Aedes aetypti, acute toxicity on Daphnia magna, and aqueous residue. J. Med. Entomol. 2011, 48, 405–410. [Google Scholar] [CrossRef]
- Varma, J.; Dubey, N.K. Efficacy of essential oils of Caesulia axillaris and Mentha arvensis against some storage pests causing biodeterioration of food commodities. Int. J. Food Microbiol. 2001, 68, 207–210. [Google Scholar] [CrossRef]
- Soliman, K.M.; Badeaa, R.I. Effect of oil extracted from some medicinal plants on different mycotoxigenic fungi. Food Chem. Toxicol. 2002, 40, 1669–1675. [Google Scholar] [CrossRef]
- Nguefack, J.; Leth, V.; Amvam Zollo, P.H.; Mathur, S.B. Evaluation of five essential oils from aromatic plants of Cameroon for controlling food spoilage and mycotoxin producing fungi. Inter. J. Food Microbiol. 2004, 94, 329–334. [Google Scholar] [CrossRef]
- Rassoli, I.; Abyaneh, M.R. Inhibitory effects of thyme oils on growth and aflatoxin production by Aspergillus parasiticus. Food Control 2004, 15, 479–483. [Google Scholar] [CrossRef]
- Gowda, N.K.S.; Malathi, V.; Suganthi, R.U. Effect of some chemical and herbal compounds on growth of Aspergillus parasiticus and aflatoxin production. Anim. Feed Sci. Technol. 2004, 116, 281–291. [Google Scholar] [CrossRef]
- Rasooli, I.; Owlia, P. Chemoprevention by thyme oils of Aspergillus parasiticus growth and aflatoxin production. Phytochemistry 2005, 66, 2851–2856. [Google Scholar] [CrossRef]
- Rasooli, I.; Rezaei, M.B.; Allameh, A. Growth inhibition and morphological alternations of Aspergillus niger by essential oils from Thymus eriocalyx and Thymus x-porlock. Food Control 2006, 17, 359–364. [Google Scholar] [CrossRef]
- Lee, B.H.; Annis, P.C.; Tumaalii, F.; Choi, W.S. Fumigant toxicity of essential oils from the Myrtaceae family and 1,8-cineole against 3 major stored-grain insects. J. Stored Prod. Res. 2004, 40, 553–564. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kim, J.; Shin, S.C.; Lee, S.G.; Park, I.K. Antifungal activity of Myrtaceae essential oils and their components against three phytopathogenic fungi. Flavour Frag. J. 2008, 23, 23–28. [Google Scholar] [CrossRef]
- Park, I.K.; Park, J.Y.; Kim, K.H.; Choi, K.S.; Choi, I.H.; Kim, C.S.; Shin, S.C. Nematicidal activity of plant essential oils and components from garlic (Allium sativum) and cinnamon (Cinnamomum verum) oils against the pinewood nematode (Bursaphelenchus xylophilus). Nematology 2005, 7, 767–774. [Google Scholar] [CrossRef]
- Batish, D.R.; Singh, H.P.; Kohli, R.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. For. Ecol. Manag. 2008, 256, 2166–2174. [Google Scholar] [CrossRef]
- Boland, D.J.; Brophy, J.J.; House, A.P.N. Eucalyptus Leaf Oils: Use, Chemistry, Distillation and Marketing; Inkata Press: Melbourne/Sydney, Australia, 1991; p. 252. [Google Scholar]
- Faouzia, H.; Souad, F.T.; Tantaoui-Elaraki, A. Antimicrobial activity of twenty-one Eucalyptus essential oils. Fitoterapia 1993, 64, 71–77. [Google Scholar]
- Su, Y.C.; Ho, C.L.; Wang, E.I.-C.; Chang, S.T. Antifungal activities and chemical compositions of essential oils from leaves of four eucalypts. Taiwan J. For. Sci. 2006, 21, 49–61. [Google Scholar]
- Carson, C.F.; Riley, T.V. Antimicrobial activity of the major components of the essential oil of Melaleuca alternifolia. J. Appl. Bacteriol. 1995, 78, 264–269. [Google Scholar] [CrossRef]
- Ramezani, H.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Antifungal activity of the volatile oil of Eucalyptus citriodora. Fitoterapia 2002, 73, 261–262. [Google Scholar] [CrossRef]
- Hood, J.R.; Burton, D.; Wilkinson, J.M.; Cavanagh, H.M.A. Antifungal activity of Leptospermum petersonii oil volatiles against Aspergillus spp. in vitro and in vivo. J. Antimicrob. Chemother. 2010a, 65, 285–288. [Google Scholar] [CrossRef]
- Hood, J.R.; Burton, D.M.; Wilkinson, J.M.; Cavanagh, H.M.A. The effect of Leptospermum petersonii essential oil on Candida albicans and Aspergillus fumigates. Med. Mycol. 2012b, 48, 922–931. [Google Scholar]
- Park, M.J.; Gwak, K.S.; Yang, I.; Choi, W.S.; Jo, H.J.; Chang, J.W.; Jeung, E.B.; Choi, I.G. Antifungal activities of the essential oils in Syzygium aromaticum (L.) Merr. Et Perry and Leptospermum petersonii Bailey and their constituents against various dermatophytes. J. Microbiol. 2007, 45, 460–465. [Google Scholar]
- Demuner, A.J.; Barbosa, L.C.A.; Magalhaes, C.G.; Silva, C.J.; Maltha, C.R.A.; Pinheiro, A.L. Seasonal variation in the chemical composition and antimicrobial activity of volatile oils of three species of Leptospermum (Myrtaceae) grown in Brazil. Molecules 2011, 16, 1181–1191. [Google Scholar] [CrossRef]
- Yeom, H.J. Fumigant and contact toxicity of plant essential oils and their components againstGerman cockroach, Blattella germanica L.
- Azar, P.A.; Poraham-Darvasari, A.; Sharifan, A.; Saber-Tehrani, M.; Soleimani, A. Antibacterial activity and chemical constitutions of Melissa officinalis L. Asian J. Chem. 2011, 3, 1381–1383. [Google Scholar]
- López, M.A.; Stashenko, E.E.; Fuentes, J.L. Chemical and antigenotoxic properties of Lippia alba essential oils. Genet. Mol. Biol. 2011, 34, 479–488. [Google Scholar] [CrossRef]
- Si, L.; Chen, Y.; Han, X.; Zhan, Z.; Tian, S.; Cui, Q.; Wang, Y. Chemical composition of essential oils of Litsea cubeba harvested from its distribution areas in China. Molecules 2012, 17, 7057–7066. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Pereira, U.A.; Martinazzo, A.P.; Maltha, C.R.Á.; Teixeira, R.R.; de Castro Melo, E. Evaluation of the chemical composition of Brazilian commercial Cymbopogon citratus (D.C.) Stapf samples. Molecules 2008, 13, 1864–1874. [Google Scholar] [CrossRef]
- Shukla, R.; Kumar, A.; Singh, P.; Dubey, N.K. Efficcy of Lippia alba (Mill.) Brown essential oil and its monoterpene aldehyde constituents against fungi isolated from some edible legume seeds and alfatoxin B1 production. Int. J. Food Microbiol. 2009, 135, 165–170. [Google Scholar] [CrossRef]
- Mahmoud, A.L. Antifungal action and antiaflatoxigenic properties of some essential oil constituents. Lett. Appl. Microbiol. 1994, 19, 110–113. [Google Scholar] [CrossRef]
- Setzer, W.N.; Schmidt, J.M.; Eiter, L.C. The leaf oil composition of Zanthoxylum fagara (L.) Sarg. from monteverde, Costa Rica, and its biological activities. J. Essent. Oil Res. 2005, 17, 333–335. [Google Scholar] [CrossRef]
- Moleyar, V.; Narasimham, P. Antifungal activity of some essential oil components. Food Microbiol. 1986, 3, 331–336. [Google Scholar] [CrossRef]
- Jiang, Z.L.; Zhang, X.; Feng, J.T. A critical review of the essential oils and its application in plant protection. J. Shaanxi Agric. Sci. 2002, 1, 32–36. [Google Scholar]
- SAS Institute, SAS/STAT User’s Guide, Version 9.1.3; SAS Institute: Cary, NC, USA, 2004.
- Sample Availability: Samples of the compounds [L. petersonii oil, neral and geranial] are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kim, E.; Park, I.-K. Fumigant Antifungal Activity of Myrtaceae Essential Oils and Constituents from Leptospermum petersonii against Three Aspergillus Species. Molecules 2012, 17, 10459-10469. https://doi.org/10.3390/molecules170910459
Kim E, Park I-K. Fumigant Antifungal Activity of Myrtaceae Essential Oils and Constituents from Leptospermum petersonii against Three Aspergillus Species. Molecules. 2012; 17(9):10459-10469. https://doi.org/10.3390/molecules170910459
Chicago/Turabian StyleKim, Eunae, and Il-Kwon Park. 2012. "Fumigant Antifungal Activity of Myrtaceae Essential Oils and Constituents from Leptospermum petersonii against Three Aspergillus Species" Molecules 17, no. 9: 10459-10469. https://doi.org/10.3390/molecules170910459
APA StyleKim, E., & Park, I.-K. (2012). Fumigant Antifungal Activity of Myrtaceae Essential Oils and Constituents from Leptospermum petersonii against Three Aspergillus Species. Molecules, 17(9), 10459-10469. https://doi.org/10.3390/molecules170910459