Molecular Cloning, Characterization and Expression of the Phenylalanine Ammonia-Lyase Gene from Juglans regia
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Full-Length cDNA Sequence of JrPAL Gene and Deduced JrPAL Protein
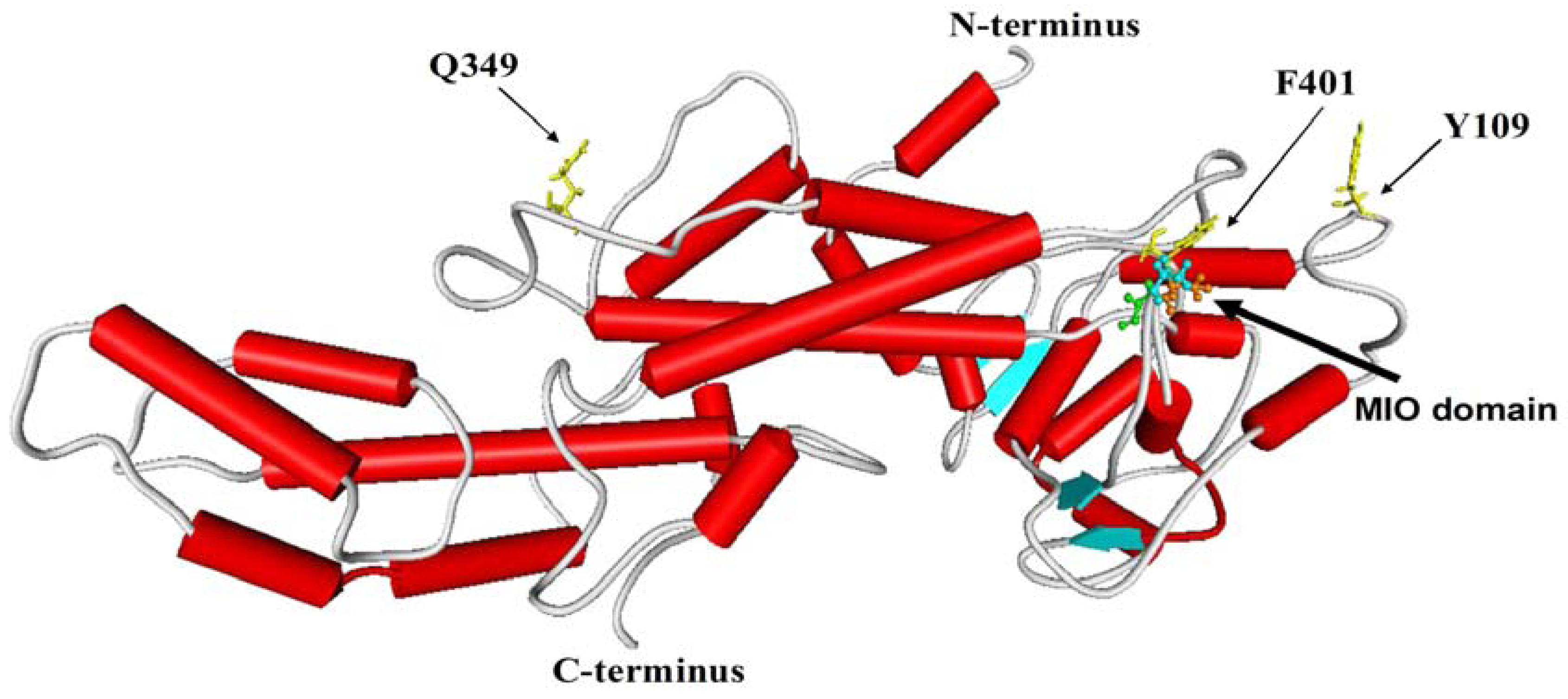
2.2. Three-Dimensional Model Analysis

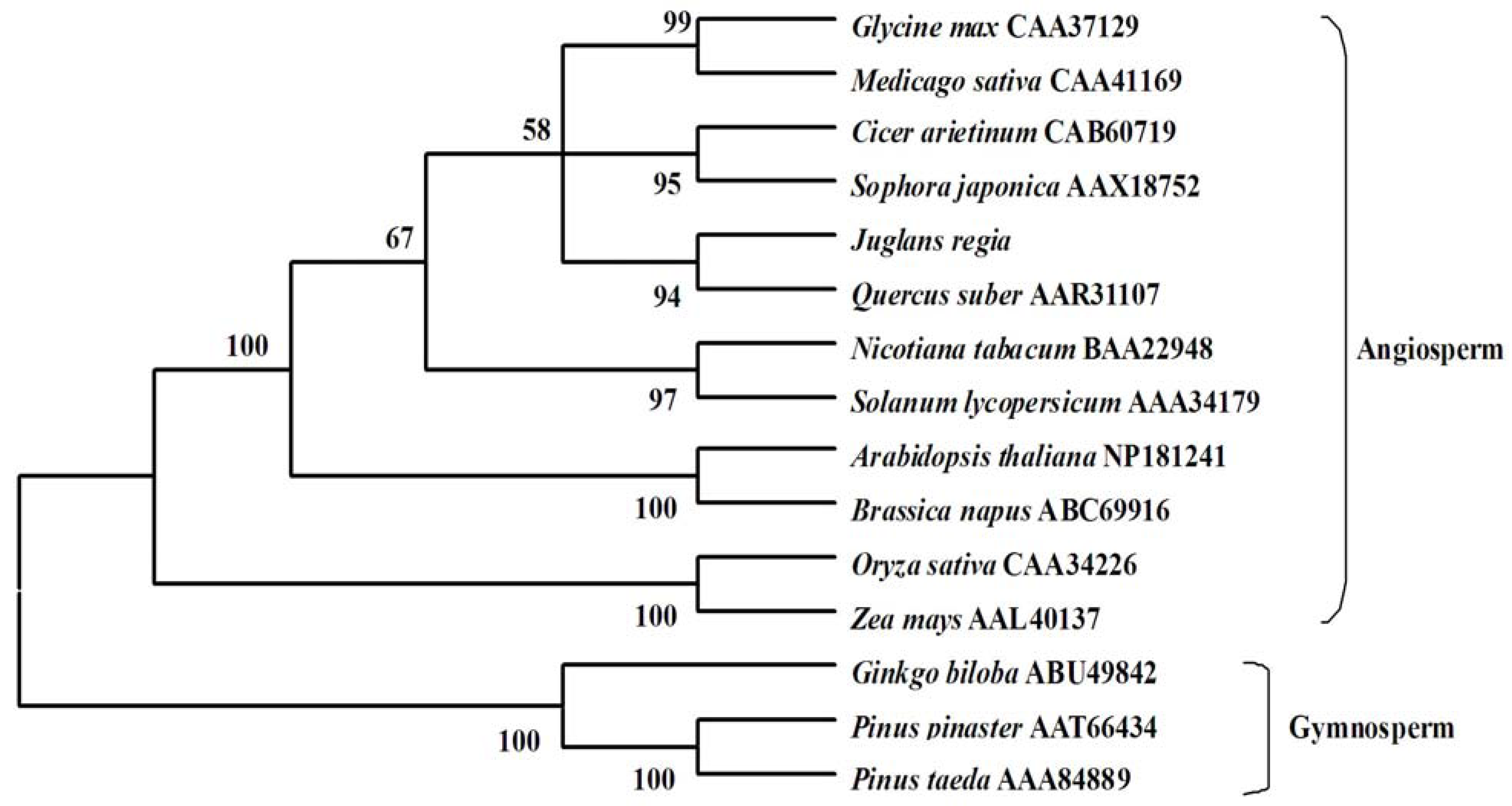
2.3. Molecular Evolution Analysis
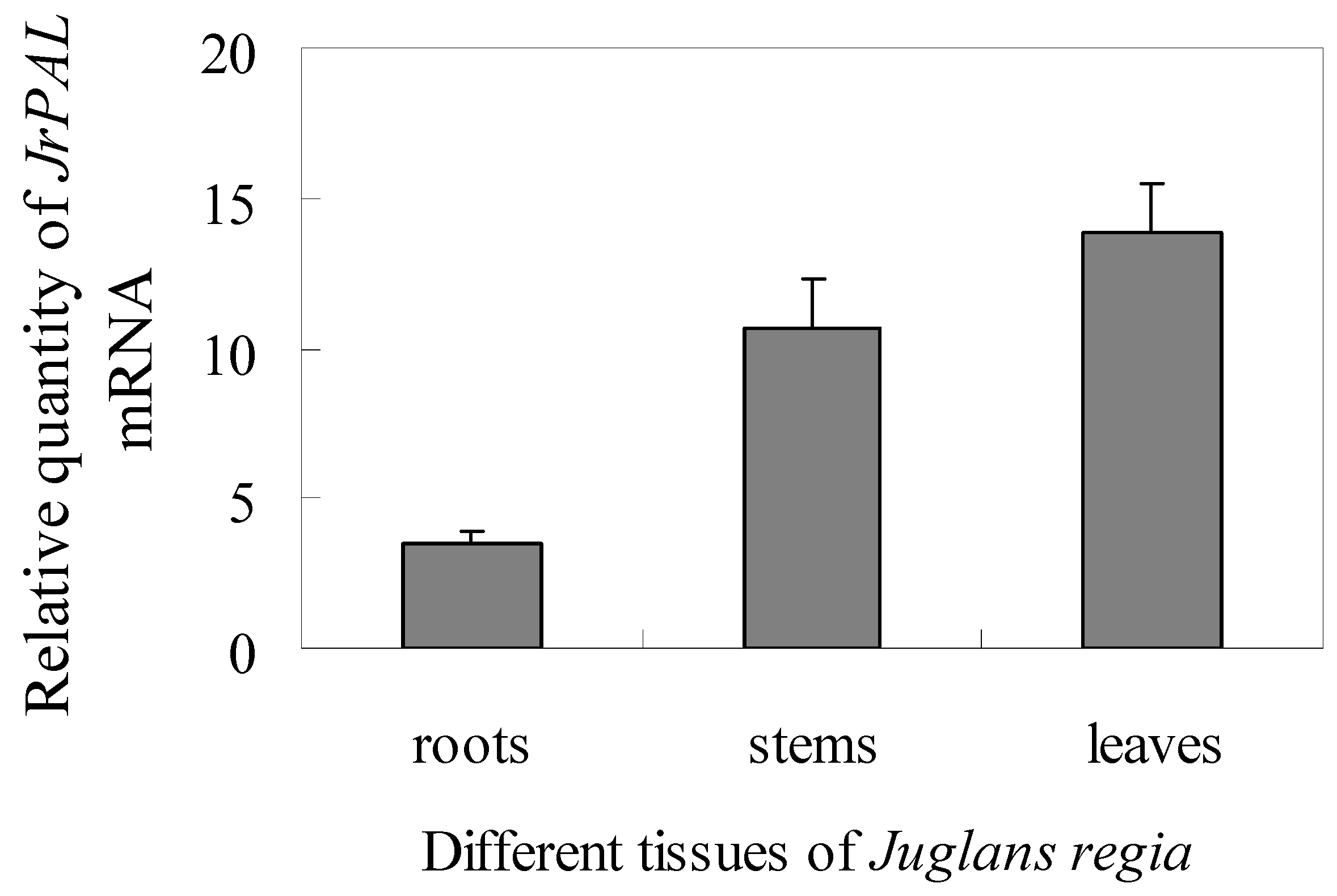
2.4. Transcript Accumulation of JrPAL in Different Tissues and Under Different Stresses
3. Experimental
3.1. Plant Materials and Treatments
3.2. DNA and RNA Extraction

3.3. Cloning of the Conserved Fragment of JrPAL
| Primer Name | Primer Sequence |
|---|---|
| PALsP | 5′-GCHTCBGGTGATYTRGTY-3′ |
| PALaP | 5′-ACATCTTGGTTRTGYGCTC-3′ |
| JuPAL3 | 5′-CATCGGGCGACCTGGTTCCTCTGTCTTAC-3′ |
| JuPAL3N | 5′-GGCTGTAGGACCCAATGGAGAGT-3′ |
| JuPAL5 | 5′-GTCGCAGAGTGAGGTAATGTGTGGCAAGA-3′ |
| JuPAL5N | 5′-CCATTGCCAAAGATTCCAGAGTT-3′ |
| JrPALUTR | 5′-TCCAAGTTTTCCTTTGCGTTT-3′ |
| PALRT-S | 5′-CTGGGATCAATGGTGGGTTT-3′ |
| PALRT-A | 5′-TATCGGTCTTGTTTGGGCTT-3′ |
| PAL prob | 5′-GAGTTACAACCCAAAGAAG-3′ |
| 18S-S | 5′-GGTCAATCTTCTCGTTCCCTT-3′ |
| 18S-A | 5′-TCGCATTTCGCTACGTTCTT-3′ |
| 18S prob | 5′-AAACAAGGAGTAACCACGG-3′ |
3.4. 3′RACE and 5′RACE of JrPAL Gene
3.5. Generation of the Full-Length cDNA of JrPAL
3.6. Bioinformatic and Molecular Evolution Analyses
3.7. Quantification of Transcript Levels by Real-Time PCR Assay
3.8. Statistical Analyses
4. Conclusions
Acknowledgments
References
- Campos, R.; Nonogakil, H.; Suslow, T.; Saltveit, M.E. Isolation and characterization of a wound inducible phenylalanine ammonia-lyase gene (LsPAL1) from Romaine lettuce leaves. Physiol. Plant 2004, 121, 429–438. [Google Scholar] [CrossRef]
- Koukol, J.; Conn, E.E. The metabolism of aromatic compounds in higher plants. J. Biol. Chem. 1961, 236, 2692–2697. [Google Scholar]
- Hahlbrock, K.; Scheel, D. Physiology and molecular biology of phenylpropanoid metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 347–369. [Google Scholar] [CrossRef]
- Dixon, R.A.; Paiva, N.L. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085–1097. [Google Scholar]
- Ritter, H.; Schulz, G.E. Structural basis for the entrance into the phenylpropanoid metabolism catalyzed by phenylalanine ammonia-lyase. Plant Cell 2004, 16, 3426–3436. [Google Scholar] [CrossRef]
- Iriti, M.; Faoro, F. Chemical diversity and defence Metabolism: How plants cope with pathogens and ozone pollution. Int. J. Mol. Sci. 2009, 10, 3371–3399. [Google Scholar] [CrossRef]
- Schijlen, E.G.; Ric de Vos, C.H.; van Tunen, A.J.; Bovy, A.G. Modification of flavonoid biosynthesis in crop plants. Phytochemistry 2004, 65, 2631–2648. [Google Scholar]
- Sarma, A.D.; Sreelakshmi, Y.; Sharma, R. Differential expression and properties of phenylalanine ammonia-lyase isoforms in tomato leaves. Phytochemistry 1998, 49, 2233–2243. [Google Scholar]
- Sarma, A.D.; Sharma, R. Purification and characterization of UV-B induced phenylalanine ammonia-lyase from rice seeding. Phytochemistry 1999, 50, 729–737. [Google Scholar]
- Hisaminato, H.; Murata, M.; Homma, S. Relationship between the enzymatic browning and phenylalanine ammonia-lyase activity of cut lettuce, and the prevention of browning by inhibitors of polyphenol biosynthesis. Biosci. Biotechnol. Biochem. 2001, 65, 1016–1021. [Google Scholar] [CrossRef]
- Hsieh, L.S.; Hsieh, Y.L.; Yeh, C.S.; Cheng, C.Y.; Yang, C.C.; Lee, P.D. Molecular characterization of a phenylalanine ammonia-lyase gene (BoPAL1) from Bambusa oldhamii. Mol. Biol. Rep. 2011, 38, 283–290. [Google Scholar]
- Winkel-Shirley, B. Biosynthesis of flavonoids and effects of stress. Curr. Opin. Plant Biol. 2002, 5, 218–233. [Google Scholar] [CrossRef]
- Chaman, M.E.; Copaja, S.V.; Argandona, V.H. Relationships between salicylic acid content, phenylalanine ammonia-lyase (PAL) activity, and resistance of barley to aphid infestation. J. Agric. Food Chem. 2003, 51, 2227–2231. [Google Scholar] [CrossRef]
- Xu, F.; Cai, R.; Cheng, S.Y.; Du, H.W.; Wang, Y.; Cheng, S.H. Molecular cloning, characterization and expression of phenylalanine ammonia-lyase gene from Ginkgo biloba. Afr. J. Biotechnol. 2008, 7, 721–729. [Google Scholar]
- Kim, S.H.; Kronstad, J.W.; Ellis, B.E. Purification and characterization of phenylalanine ammonia-lyase from Ustilago maydis. Phytochemistry 1996, 43, 351–357. [Google Scholar]
- Hattori, T.; Nishiyawa, A.; Shimada, M. Induction of L-phenylalanine ammonia-lyase and suppression of veratryl alcohol biosynthesis by exogenously added L-phenylalanine in a whiterot fungus Phanerochaete chrysosporium. FEMS Microbiol. Lett. 1999, 179, 305–309. [Google Scholar]
- Moffitt, M.C.; Louie, G.V.; Bowman, M.E.; Pence, J.; Noel, J.P.; Moore, B.S. Discovery of the cyanobacterial phenylalanine ammonia-lyases: Kinetic and structural characterization. Biochemistry 2007, 46, 1004–1012. [Google Scholar]
- Xiang, L.; Moore, B.S. Biochemical characterization of a prokaryotic phenylalanine ammonia lyase. J. Bacteriol. 2005, 187, 4286–4289. [Google Scholar] [CrossRef]
- Ohl, S.; Hedrick, S.A.; Choy, J.; Lamb, C.J. Functional properties of a phenylalanine ammonia-lyase promoter from Arabidopsis. Plant Cell 1990, 2, 837–848. [Google Scholar]
- Logemann, E.; Parniske, M.; Hahlbrock, K. Modes of expression and common structural features of the complete phenylalanine ammonia-lyase gene family in parsley. Proc. Natl. Acad. Sci. USA 1995, 92, 5905–5909. [Google Scholar] [CrossRef]
- Cochrane, F.C.; Davin, L.B.; Lewis, N.G. The Arabidopsis phenylalanine ammonia-lyase gene family: Kinetic characterization of the four PAL isoforms. Phytochemistry 2004, 65, 1557–1564. [Google Scholar]
- Song, J.; Wang, Z. Molecular cloning, expression and characterization of a phenylalanine ammonia-lyase gene (SmPAL1) from Salvia miltiorrhiza. Mol. Biol. Rep. 2009, 36, 939–952. [Google Scholar] [CrossRef]
- Guo, J.; Wang, W.H. Characterization of the phenylalanine ammonia-lyase gene (SlPAL5) from tomato (Solanum lycopersicum L.). Mol. Biol. Rep. 2009, 36, 1579–1585. [Google Scholar]
- Hsieh, L.S.; Yeh, C.S.; Pan, H.C.; Cheng, C.Y.; Yang, C.C.; Lee, P.D. Cloning and expression of a phenylalanine ammonia-lyase gene(BoPAL2) from Bambusa oldhamii in Escherichia coli and Pichia pastoris. Protein Expr. Purif. 2010, 71, 224–230. [Google Scholar] [CrossRef]
- Havir, E.A.; Reid, P.D.; Marsh, H.V.Jr. L-phenylalanineammonia-lyase (maize). Plant Physiol. 1971, 48, 130–136. [Google Scholar] [CrossRef]
- Rösler, J.; Krekel, F.; Amrhein, N.; Schmid, J. Maize phenylalanineammonia-lyase has tyrosine ammonia-lyase activity. Plant Physiol. 1997, 113, 175–179. [Google Scholar]
- Albert, C.M.; Gaziano, J.M.; Willett, W.C.; Manson, J.A.E. Nut consumption and decreased risk of sudden cardiac death in the Physicians’ Health Study. Arch. Intern. Med. 2002, 162, 1382–1387. [Google Scholar] [CrossRef]
- Vinson, J.A.; Cai, Y.X. Nuts, especially walnuts, have both antioxidant quantity and efficacy and exhibit significant potential health benefits. Food Funct. 2012, 2, 134–140. [Google Scholar] [CrossRef]
- Davis, L.; Stonehouse, W.; Loots, D.T.; Mukuddem-Petersen, J.; van der Westhuizen, F.H.; Hanekom, S.M.; Jerling, J.C. The effects of high walnut and cashew nut diets on the antioxidant status of subjects with metabolic syndrome. Eur. J. Nutr. 2007, 46, 155–164. [Google Scholar] [CrossRef]
- Schulz, G.E.; Ritter, H. Structural basis for the entrance into the phenylpropanoid metabolism catalyzed by phenylalanine ammonia-lyase. Plant Cell 2004, 16, 3426–3436. [Google Scholar]
- Röther, D.; Poppe, L.; Morlock, G.; Viergutz, S.; Rétey, J. An active site homology model of phenylalanine ammonia-lyase from Petroselinum crispum. Eur. J. Biochem. 2002, 269, 3065–3075. [Google Scholar] [CrossRef]
- Schwede, T.; Kopp, J.; Guex, N. Peitsch M.C.; Peitsch, M.C. SWISS-MODEL: An automated protein homology-modeling server. Nucleic Acids Res. 2003, 31, 3381–3385. [Google Scholar]
- Wanner, L.A.; Li, G.; Ware, D.; Somssich, I.E.; Davis, K.R. The phenylalanine ammonia-lyase gene family in Arabidopsis thaliana. Plant Mol. Biol. 1995, 27, 327–338. [Google Scholar] [CrossRef]
- Kervinen, T.; Peltonen, S.; Utriainen, M.; Kangasjärvi, J.; Teeri, T.H. Karjalainen R Cloning and characterization of cDNA clones encoding phenylalanine ammonia-lyase in barley. Plant Sci. 1997, 123, 143–150. [Google Scholar] [CrossRef]
- Joos, H.J.; Hahlbrock, K. Phenylalanine ammonia-lyase in potato (Solanum tuberosum L.) Genomic complexity, structural comparison of two selected genes and modes of expression. Eur. J. Bichem. 1992, 204, 621–629. [Google Scholar]
- Dixon, R.A.; Maxwell, C.A.; Weiting, N.; Oommen, A.; Paiva, N.L. Genetic manipulation of ligin and phenylpropanoid compounds involved with microorganisms. In Genetic Engineering of Plant Secondary Metabolsim, 2nd; Ellis, B.E., Kuroki, G.W., Stafford, H.H., Eds.; Plenum Press: New York, NY, USA, 1994; Volume 28, pp. 153–178. [Google Scholar]
- Lu, B.B.; Du, Z.; Ding, R.X.; Zhang, L.; Yu, X.J.; Liu, C.H.; Chen, W.S. Cloning and characterization of a differentially expressed phenylalanine ammonialase gene (IiPAL) after genome duplication from Tetraploid Isatis indigotica Fort. J. Integr. Plant Biol. 2006, 48, 1439–1449. [Google Scholar] [CrossRef]
- Thulke, O.; Conrath, U. Salicylic acid has a dual role in activation of defence-related genes in parsley. Plant J. 1998, 14, 35–42. [Google Scholar] [CrossRef]
- Fraissinet-Tachet, L.; Baltz, R.; Chong, J.; Kauffmann, S.; Friting, B.; Saindrenan, P. Two tobacco genes induced by infection, elicitor and salicylic acid encode glucosyltransferases acting on phenylpropanoids and benzoic acid derivatives, including salicylic acid. FEBS Lett. 1998, 437, 319–323. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Joyce, D.C. ABA effects on ethylene production, PAL activity, anthocyanin and phenolic contents of strawberry fruit. Plant Growth Regul. 2003, 39, 171–174. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, H.; Wang, W.; Qian, Z.; Li, L.; Wang, J.; Zhou, G.; Kai, G. Molecular characterization and expression analysis of a new cDNA encoding strictosidine synthase from Ophiorrhiza japonica. Mol. Biol. Rep. 2009, 36, 1845–1852. [Google Scholar] [CrossRef]
- Wen, P.F.; Chen, J.Y.; Kong, W.F.; Pan, Q.H.; Wan, S.B.; Huang, W.D. Salicylic acid induced the expression of phenylalanine ammonia-lyase gene in grape berry. Plant Sci. 2005, 169, 928–934. [Google Scholar] [CrossRef]
- Sanchez-Ballesta, M.T.; Lafuente, M.T.; Zacarias, L.; Granell, A. Involvement of phenylalanine ammonia-lyase in the response of Fortune mandarin fruits to cold temperature. Physiol. Plant 2000, 108, 382–389. [Google Scholar] [CrossRef]
- Hyodo, H. Phenylalanine ammonia-lyase in strawberry fruits. Plant Cell Physiol. 1971, 12, 989–991. [Google Scholar]
- Given, N.K.; Venis, M.A.; Grierson, D. Phenylalanine ammonia-lyase activity and anthocyanin synthesis in ripening strawberry fruit. J. Plant Physiol. 1988, 133, 25–30. [Google Scholar] [CrossRef]
- Zhang, F.; Wan, X.Q. A new and simple method for isolating genomic DNA from Julandaceae for genetic diversity analysis. World J. Agric. Sci. 2009, 5, 746–750. [Google Scholar]
- Xu, F.; Cai, R.; Chen, L.J.; Chang, J.; Cheng, S.Y. A method for isolation of high quality RNA from various Juglans regia tissues. J. Fruit Sci. 2008, 25, 435–439. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kurmar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) Software Version 4. 0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 2876–4882. [Google Scholar]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Sample Availability: The samples of Juglans regia and all primers used in this study are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, F.; Deng, G.; Cheng, S.; Zhang, W.; Huang, X.; Li, L.; Cheng, H.; Rong, X.; Li, J. Molecular Cloning, Characterization and Expression of the Phenylalanine Ammonia-Lyase Gene from Juglans regia. Molecules 2012, 17, 7810-7823. https://doi.org/10.3390/molecules17077810
Xu F, Deng G, Cheng S, Zhang W, Huang X, Li L, Cheng H, Rong X, Li J. Molecular Cloning, Characterization and Expression of the Phenylalanine Ammonia-Lyase Gene from Juglans regia. Molecules. 2012; 17(7):7810-7823. https://doi.org/10.3390/molecules17077810
Chicago/Turabian StyleXu, Feng, Guang Deng, Shuiyuan Cheng, Weiwei Zhang, Xiaohua Huang, Linling Li, Hua Cheng, Xiaofeng Rong, and Jinbao Li. 2012. "Molecular Cloning, Characterization and Expression of the Phenylalanine Ammonia-Lyase Gene from Juglans regia" Molecules 17, no. 7: 7810-7823. https://doi.org/10.3390/molecules17077810
APA StyleXu, F., Deng, G., Cheng, S., Zhang, W., Huang, X., Li, L., Cheng, H., Rong, X., & Li, J. (2012). Molecular Cloning, Characterization and Expression of the Phenylalanine Ammonia-Lyase Gene from Juglans regia. Molecules, 17(7), 7810-7823. https://doi.org/10.3390/molecules17077810




