Kale Extract Increases Glutathione Levels in V79 Cells, but Does not Protect Them against Acute Toxicity Induced by Hydrogen Peroxide
Abstract
:1. Introduction

2. Results
2.1. Chemical Composition
2.1.1. HPLC-DAD Phenolic Compounds Qualitative Analysis
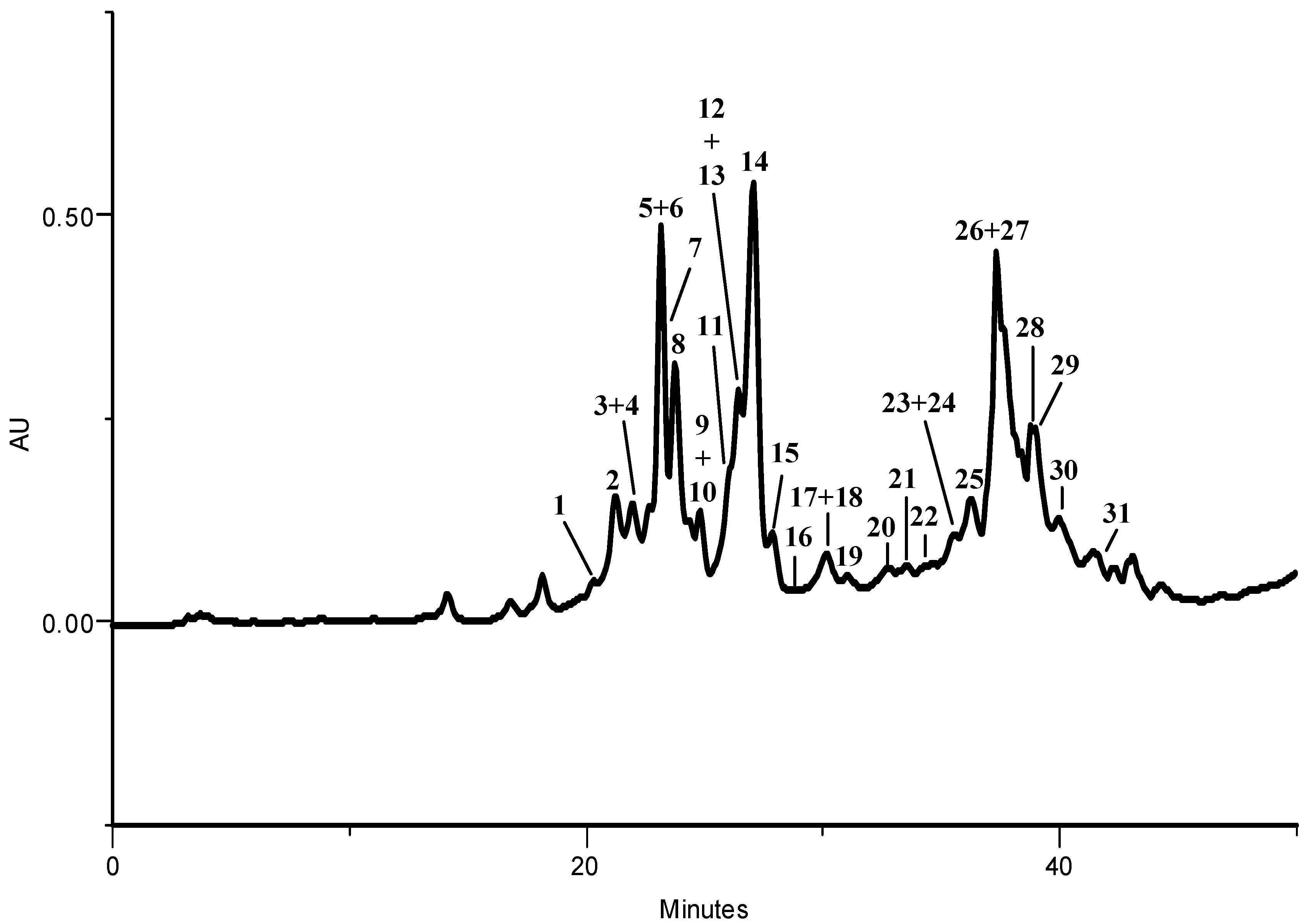

| Compound | Kale | Larvae | Excrements | |
|---|---|---|---|---|
| 1 | Quercetin-3-O-sophtr-7-O-gluc | 12.0 (0.1) | - | - |
| 2 | Quercetin-3-O-soph-7-O-gluc | 24.3 (1.0) | - | nq |
| 3 | Kaempferol-3-O-(methoxycaffeoyl)soph-7-O-gluc + | 244.1 (15.5) | - | - |
| 4 | Quercetin-3-O-soph-7-O-digluc | - | - | |
| 34 | Kaempferol-3-O-sophtr-7-O-gluc + | - | - | 1.4 (0.0) |
| 5 | Kaempferol-3-O-soph-7-O-gluc + | 310.9 (0.1) | - | |
| 6 | Kaempferol-3-O-(caffeoyl)soph-7-O-gluc + | - | - | |
| 7 | Quercetin-3-O-(sinapoyl)soph-7-O-gluc | - | - | |
| 35 | Kaempferol-3-O-sophtr-7-O-digluc + | - | - | 5.4 (0.1) |
| 8 | Kaempferol-3-O-soph-7-O-digluc | 75.1 (8.2) | - | |
| 9 | Isorhamnetin-3-O-soph-7-O-gluc + | 34.8 (2.6) | - | - |
| 10 | Quercetin-3-O-(feruloyl)soph-7-O-gluc | - | - | |
| 11 | Quercetin-3-O-(feruloyl)soph-7-O-soph + | 883.0 (97.8) | - | - |
| 12 | Kaempferol-3-O-(sinapoyl)soph-7-O-gluc + | - | 7.9 (0.1) | |
| 13 | Kaempferol-3-O-(sinapoyl)soph-7-O-soph | - | ||
| 14 | Kaempferol-3-O-(feruloyl)soph-7-O-gluc | 581.6 (0.7) | - | 10.5 (0.1) |
| 15 | Kaempferol-3-O-(feruloyl)soph-7-O-soph | 70.4 (8.8) | - | 1.1 (0.1) |
| 16 | Kaempferol-3-O-(p-coumaroyl)soph-7-O-gluc | nq | - | - |
| 17 | Kaempferol-3-O-(p-coumaroyl)soph-7-O-soph + | 159.5 (12.3) | - | - |
| 18 | Kaempferol-3-O-gent-7-O-gluc | - | - | |
| 19 | Kaempferol-3-O-gent-7-O-digluc + | 29.7 (4.7) | - | 3.8 (0.7) |
| 32 | Kaempferol-3-O-digluc sulfate | - | nq | |
| 20 | Isorhamnetin-3-O-gent-7-O-gluc | 101.6 (9.6) | - | - |
| 21 | Quercetin-3-O-(sinapoyl)soph | 16.8 (0.1) | - | - |
| 22 | Quercetin-3-O-(feruloyl)soph | 16.3 (3.6) | - | - |
| FA | Ferulic acid | - | 8.3 (0.9) | 3.0 (0.0) |
| 23 | Quercetin-3-O-soph + | 31.4 (0.9) | 16.4 (0.8) | 1.1 (0.0) |
| 24 | Kaempferol-3-O-(p-coumaroyl)gent-7-O-gluc | - | - | |
| SA | Sinapic acid | - | 191.0 (18.1) | - |
| 25 | Kaempferol-3-O-(sinapoyl)soph | 133.5 (0.9) | - | - |
| 26 | Kaempferol-3-O-soph + | 960.8 (35.4) | 55.8 (1.9) | 2.6 (0.2) |
| 27 | Kaempferol-3-O-(feruloyl)soph | - | - | |
| 28 | Disinapoyl-gent + | 92.2 (6.3) | - | - |
| 29 | Sinapoyl,feruloyl-gent | - | - | |
| 30 | Diferuloyl-gent | 1.6 (0.1) | - | - |
| 31 | Disinapoyl,feruloyl-gent | nq | - | - |
| 33 | Kaempferol-3-O-gluc sulfate | - | nq | 3.0 (0.0) |
| 36 | Kaempferol-3-O-(sinapoyl)sophtr + | - | - | 1.2 (0.3) |
| 37 | Kaempferol-3-O-sophtr | - | - | |
| 38 | Kaempferol-3-O-(feruloyl)sophtr | - | - | 2.4 (0.1) |
| 39 | Isorhamnetin-3-O-soph | - | - | 0.3 (0.0) |
| 40 | Kaempferol-3-O-(p-coumaroyl)sophtr | - | - | 0.1 (0.0) |
| 41 | Quercetin-3-O-gluc sulfate | - | - | nq |
| 42 | Kaempferol-3-O-gent | - | - | nq |
| 43 | Kaempferol-3-O-gluc | - | - | nq |
| 44 | Isorhamnetin-3-O-gent | - | - | nq |
| 45 | Kaempferol-3-O-(feruloyl)soph + | - | - | 5.5 (0.3) |
| 46 | Kaempferol-3-O-(p-coumaroyl)soph | - | - | |
| Σ | 3779.6 | 271.5 | 49.3 |
2.1.2. Phenolic Compounds Quantitative Analysis

2.2. Biological Activity
2.2.1. H2O2 Induced Toxicity in V79 Cells
2.2.2. Effects of Kale and P. brassicae Extracts on V79 Cells Viability
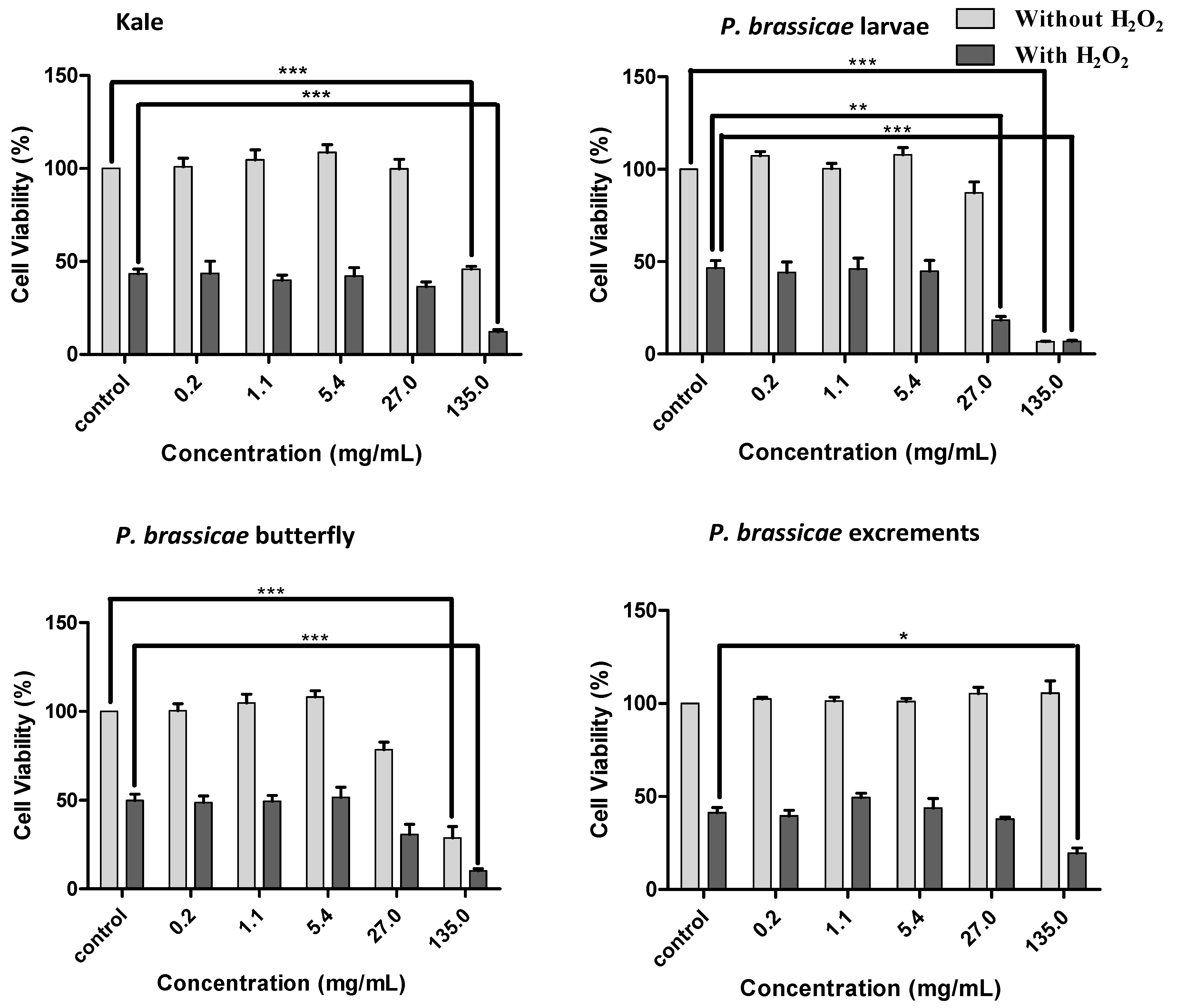
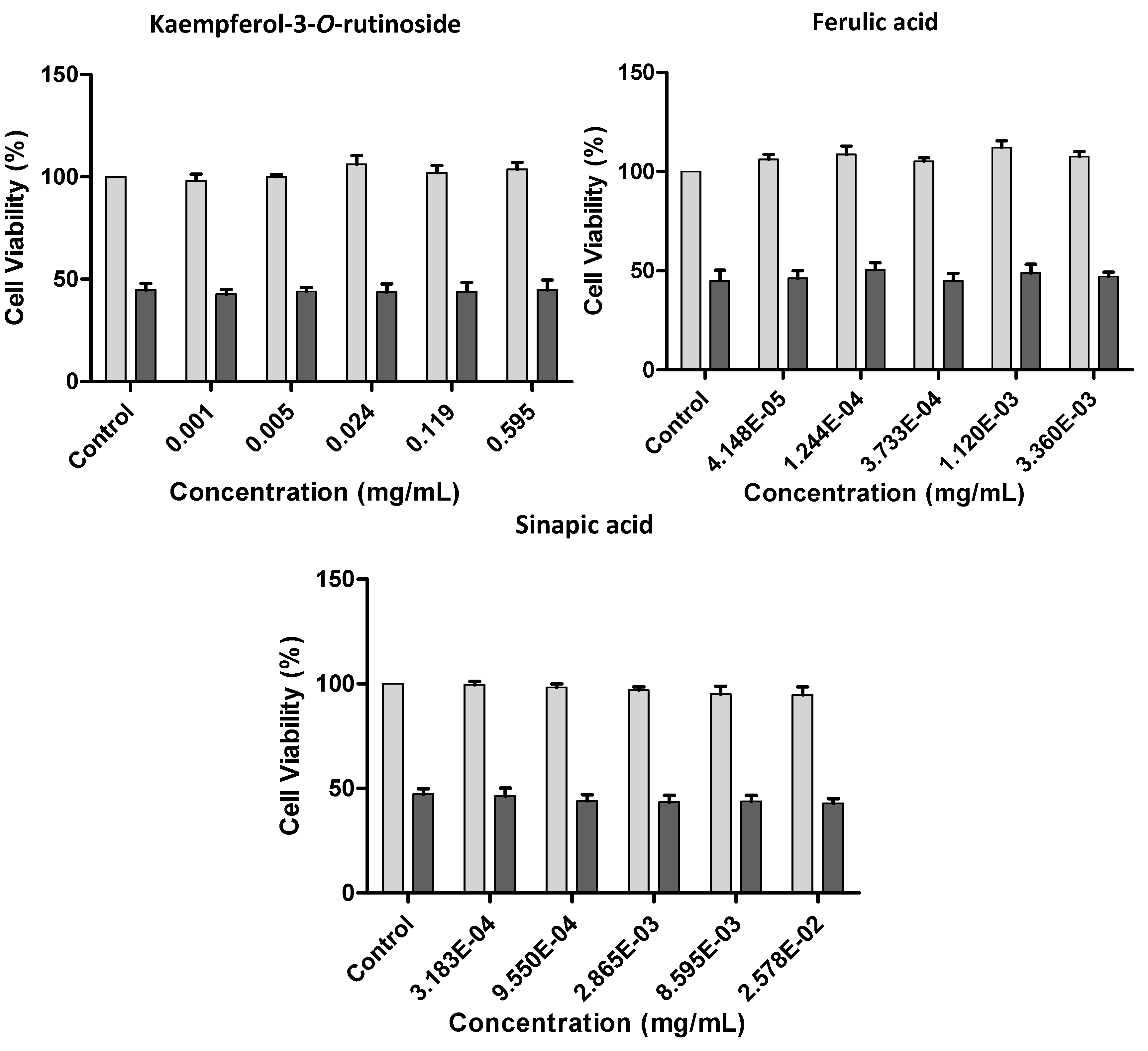
2.2.3. Effect of Kale and P. brassicae Extracts on Cellular Hydrogen Peroxide-Induced Toxicity
2.2.4. Effect of Kale and P. brassicae Extracts on Glutathione Homeostasis
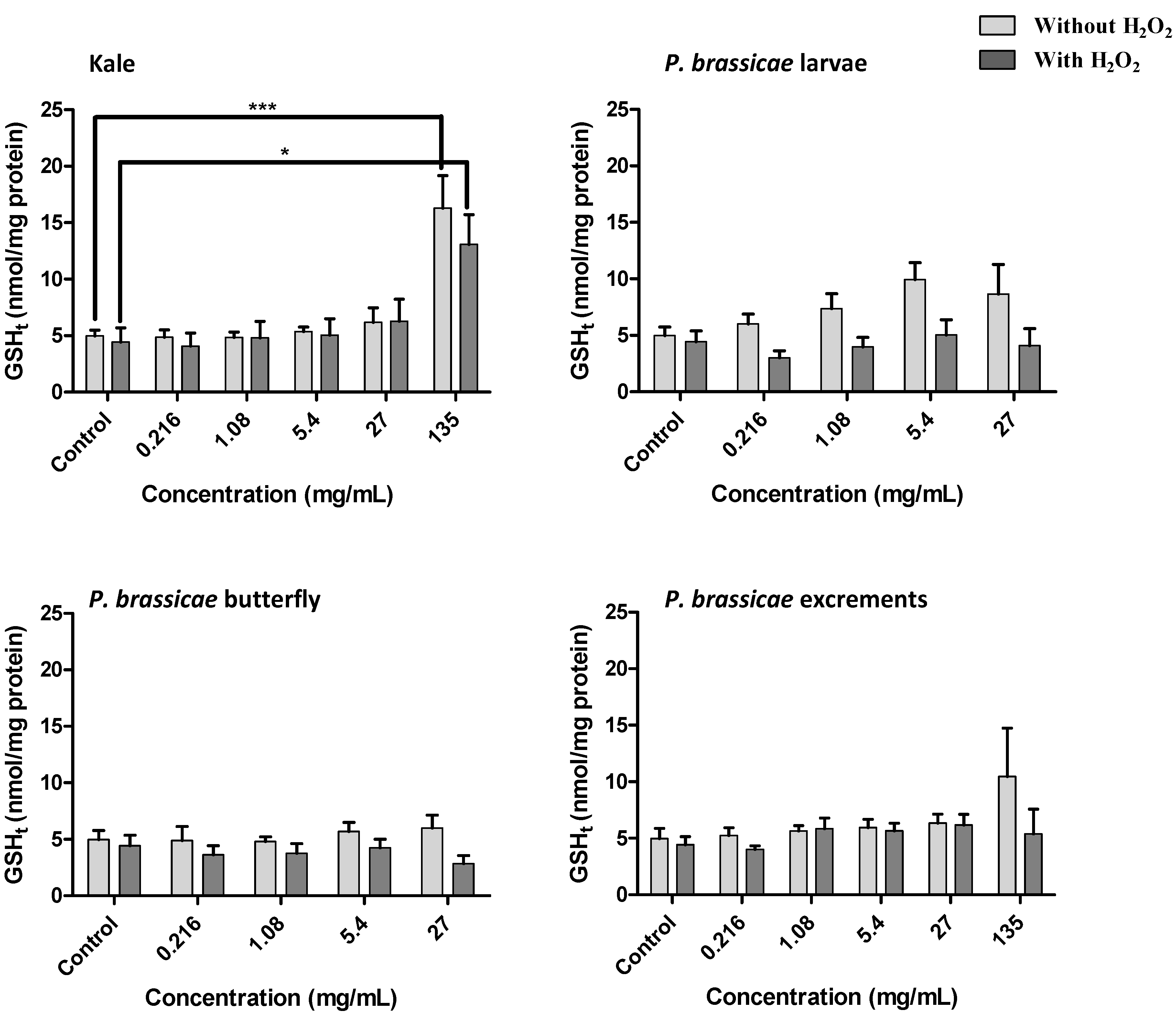
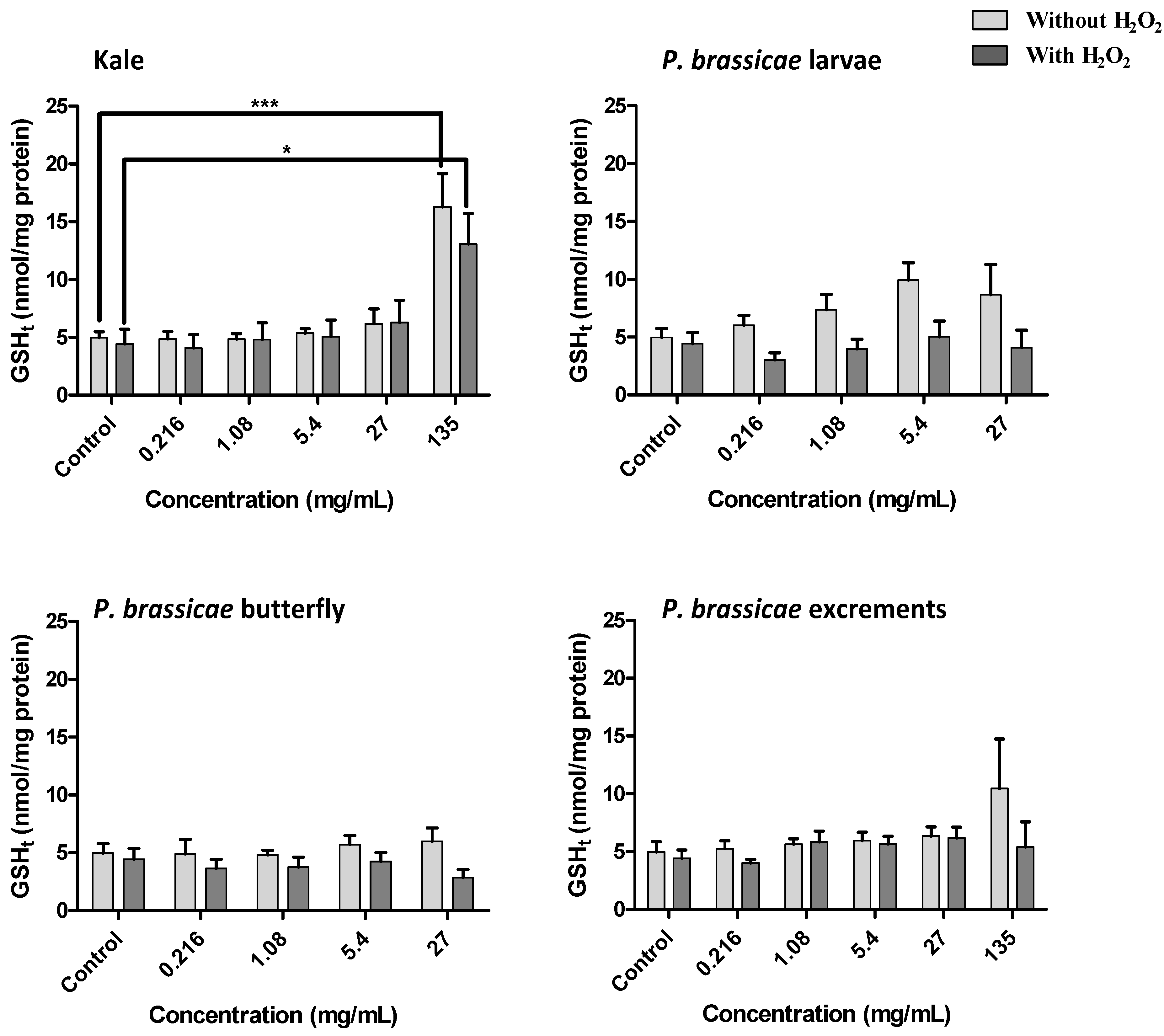
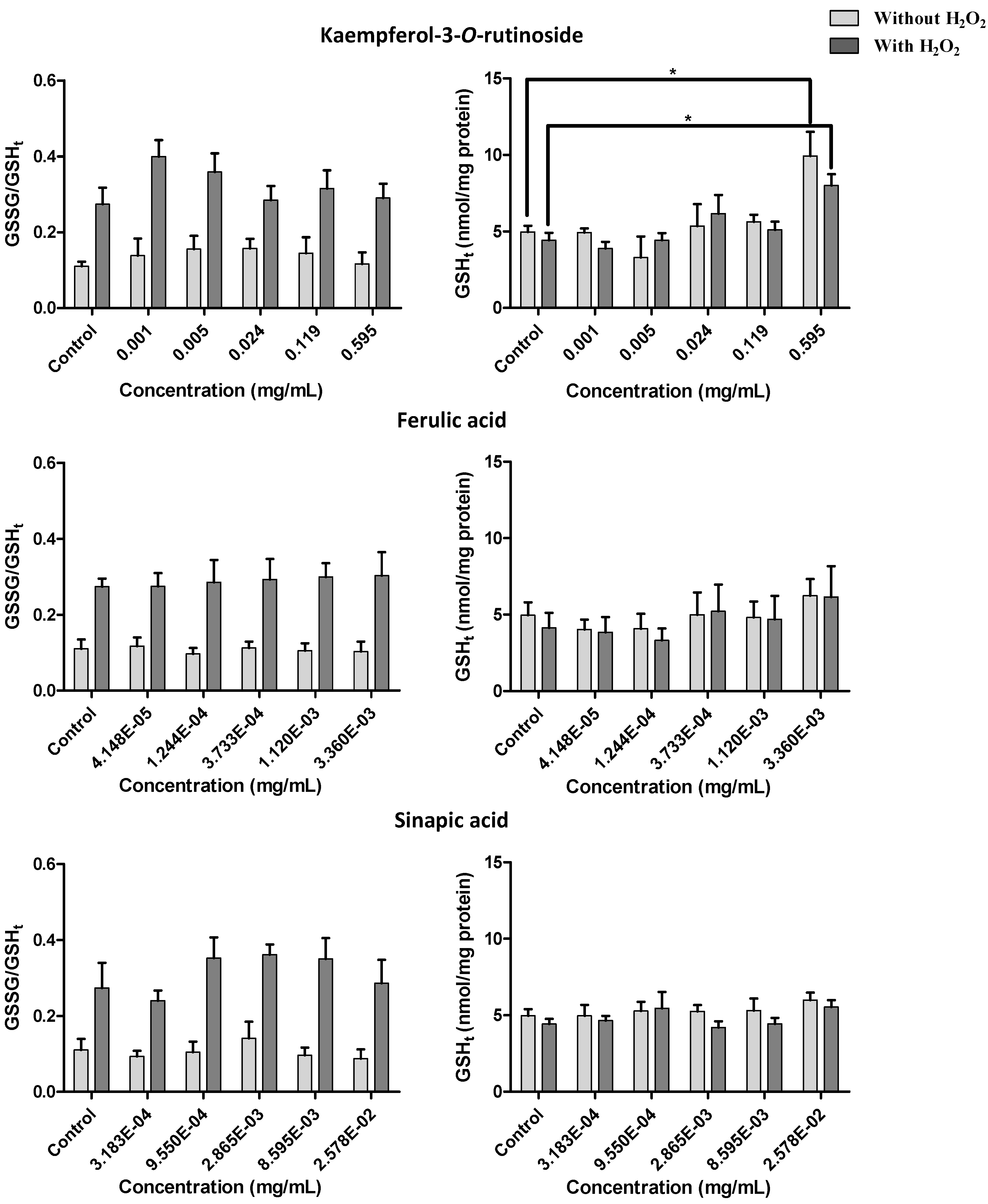
3. Discussion
4. Experimental
4.1. Reagents
4.2. Samples
4.3. Extract Preparation
4.4. HPLC-DAD Phenolic Compounds Analysis
4.5. Cell Culture and Treatments
4.6. Cell Viability
4.7. GSHt and GSSG Determination
4.8. Measurement of Protein Content
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Conflict of Interest
- Sample Availability: Samples of B. oleracea var. acephala leaves and P. brassicae butterfly, larvae, and its excrements are available from the authors.
References and Notes
- Verhoeven, D.T.H.; Goldbohm, R.A.; Van Poppel, G.; Verhagen, H.; Van Den Brandt, P.A. Epidemiological studies on Brassica vegetables and cancer risk. Cancer Epidemiol. Biomark. Prev. 1996, 5, 733–748. [Google Scholar]
- Podsedek, A. Natural antioxidants and antioxidant capacity of Brassica vegetables: A review. LWT-Food Sci. Technol. 2007, 40, 1–11. [Google Scholar] [CrossRef]
- Singh, M.; Arseneault, M.; Sanderson, T.; Murthy, V.; Ramassamy, C. Challenges for research on polyphenols from foods in Alzheimer’s disease: Bioavailability, metabolism, and cellular and molecular mechanisms. J. Agric. Food Chem. 2008, 56, 4855–4873. [Google Scholar] [CrossRef]
- Galati, G.; Sabzevari, O.; Wilson, J.X.; O’Brien, P.J. Prooxidant activity and cellular effects of the phenoxyl radicals of dietary flavonoids and other polyphenolics. Toxicology 2002, 177, 91–104. [Google Scholar] [CrossRef]
- Cartea, M.E.; Velasco, P.; Obregón, S.; Padilla, G.; de Haro, A. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain. Phytochemistry 2008, 69, 403–410. [Google Scholar] [CrossRef]
- Ferreres, F.; Fernandes, F.; Oliveira, J.; Valentão, P.; Pereira, J.A.; Seabra, R.M.; Andrade, P.B. Metabolic profiling and biological capacity of Pieris brassicae fed with kale (Brassica oleracea L. var. acephala). Food Chem. Toxicol. 2009, 47, 1209–1220. [Google Scholar] [CrossRef]
- Vallejo, F.; Tomás-Barberán, F.A.; Ferreres, F. Characterisation of flavonols in broccoli (Brassica oleracea L. var. italica) by liquid chromatography–UV diode–array detection-electrospray ionisation mass spectrometry. J. Chromatogr. A 2004, 1054, 181–193. [Google Scholar] [CrossRef]
- Ferreres, F.; Sousa, C.; Valentão, P.; Pereira, J.A.; Seabra, R.M.; Andrade, P.B. Tronchuda cabbage flavonoids uptake by Pieris brassicae. Phytochemistry 2007, 68, 361–367. [Google Scholar]
- Ferreres, F.; Valentão, P.; Pereira, J.A.; Bento, A.; Noites, A.; Seabra, R.M.; Andrade, P.B. HPLC-DAD-MS/MS-ESI screening of phenolic compounds in Pieris brassicae L. reared on Brassica rapa var. rapa L. J. Agric. Food Chem. 2008, 56, 844–853. [Google Scholar] [CrossRef]
- Ferreres, F.; Fernandes, F.; Pereira, D.M.; Pereira, J.A.; Valentão, P.; Andrade, P.B. Phenolics metabolism in insects: Pieris brassicae-Brassica oleracea var. costata ecological duo. J. Agric. Food Chem. 2009, 57, 9035–9043. [Google Scholar] [CrossRef]
- Pereira, D.M.; Noites, A.; Valentão, P.; Ferreres, F.; Pereira, J.A.; Vale-Silva, L.; Pinto, E.; Andrade, P.B. Targeted metabolite analysis and biological activity of Pieris brassicae fed with Brassica rapa var. rapa. J. Agric. Food Chem. 2009, 57, 483–489. [Google Scholar]
- Sousa, C.; Pereira, D.M.; Valentão, P.; Ferreres, F.; Pereira, J.A.; Seabra, R.M.; Andrade, P.B. Pieris brassicae inhibits xanthine oxidase. J. Agric. Food Chem. 2009, 57, 2288–2294. [Google Scholar]
- Fernandez-Panchon, M.S.; Villano, D.; Troncoso, A.M.; Garcia-Parrilla, M.C. Antioxidant activity of phenolic compounds: from in vitro results to in vivo evidence. Crit. Rev. Food Sci. Nutr. 2008, 48, 649–671. [Google Scholar] [CrossRef]
- Lima, C.F.; Carvalho, F.; Fernandes, E.; Bastos, M.L.; Santos-Gomes, P.C.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Evaluation of toxic/protective effects of the essential oil of Salvia officinalis on freshly isolated rat hepatocytes. Toxicol. In Vitro 2004, 18, 457–465. [Google Scholar] [CrossRef]
- Sousa, C.; Pontes, H.; Carmo, H.; Dinis-Oliveira, R.J.; Valentão, P.; Andrade, P.B.; Remião, F.; Bastos, M.L.; Carvalho, F. Water extracts of Brassica oleracea var. costata potentiate paraquat toxicity to rat hepatocytes in vitro. Toxicol. In Vitro 2009, 23, 1131–1138. [Google Scholar] [CrossRef]
- Talorete, T.P.N.; Bouaziz, M.; Sayadi, S.; Isoda, H. Influence of medium type and serum on MTT reduction by flavonoids in the absence of cells. Cytotechnology 2006, 52, 189–198. [Google Scholar]
- Murray, J.I.; Whitfield, M.L.; Trinklein, N.D.; Myers, R.M.; Brown, P.O.; Botstein, D. Diverse and specific gene expression responses to stresses in cultured human cells. Mol. Biol. Cell 2004, 15, 2361–2374. [Google Scholar] [CrossRef]
- Ündeğer, Ü.; Başaran, A.; Degen, G.H.; Başaran, N. Antioxidant activities of major thyme ingredients and lack of (oxidative) DNA damage in V79 Chinese hamster lung fibroblast cells at low levels of carvacrol and thymol. Food Chem. Toxicol. 2009, 47, 2037–2043. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Kang, J.-S.; Park, J.H.Y.; Lee, Y.-J.; Choi, J.-S.; Kang, Y.-H. Polyphenolic flavonoids differ in their antiapoptotic efficacy in hydrogen peroxide-treated human vascular endothelial cells. J. Nutr. 2003, 133, 985–991. [Google Scholar]
- Nakayama, T. Suppression of hydroperoxide-induced cytotoxicity by polyphenols. Cancer Res. 1994, 54, 1991S–1993S. [Google Scholar]
- Piao, M.J.; Kang, K.A.; Zhang, R.; Ko, D.O.; Wang, Z.H.; You, H.J.; Kim, H.S.; Kim, J.S.; Kang, S.S.; Hyun, J.W. Hyperoside prevents oxidative damage induced by hydrogen peroxide in lung fibroblast cells via an antioxidant effect. Biochim. Biophys. Acta 2008, 1780, 1448–1457. [Google Scholar] [CrossRef]
- Zou, Y.P.; Lu, Y.H.; Wei, D.Z. Protective effects of a flavonoid-rich extract of Hypericum perforatum L. against hydrogen peroxide-induced apoptosis in PC12 cells. Phytother. Res. 2010, 24, S6–S10. [Google Scholar] [CrossRef]
- Halliwell, B. Reactive oxygen species and the central nervous system. J. Neurochem. 1992, 59, 1609–1623. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar]
- Skibola, C.F.; Smith, M.T. Potential health impacts of excessive flavonoid intake. Free Radic. Biol. Med. 2000, 29, 375–383. [Google Scholar] [CrossRef]
- Liu, R.H.; Finley, J. Potential cell culture models for antioxidant research. J. Agric. Food Chem. 2005, 53, 4311–4314. [Google Scholar] [CrossRef]
- Aherne, S.A.; O’Brien, N.M. Lack of effect of the flavonoids, myricetin, quercetin, and rutin, on repair of H2O2-induced DNA single-strand breaks in Caco-2, Hep G2, and V79 cells. Nutr. Cancer 2000, 38, 106–115. [Google Scholar] [CrossRef]
- Qu, W.; Fan, L.; Kim, Y.C.; Ishikawa, S.; Iguchi-Ariga, S.M.; Pu, X.P.; Ariga, H. Kaempferol derivatives prevent oxidative stress-induced cell death in a DJ-1-dependent manner. J. Pharmacol. Sci. 2009, 110, 191–200. [Google Scholar] [CrossRef]
- Myhrstad, M.C.; Carlsen, H.; Nordström, O.; Blomhoff, R.; Moskaug, J.Ø. Flavonoids increase the intracellular glutathione level by transactivation of the gamma-glutamylcysteine synthetase catalytical subunit promoter. Free Radic. Biol. Med. 2002, 32, 386–393. [Google Scholar] [CrossRef]
- Jornot, L.; Petersen, H.; Junod, A.F. Hydrogen peroxide-induced DNA damage is independent of nuclear calcium but dependent on redox-active ions. Biochem. J. 1998, 335, 85–94. [Google Scholar]
- Carmo, H.; Brulport, M.; Hermes, M.; Oesch, F.; de Boer, D.; Remião, F.; Carvalho, F.; Schön, M.R.; Krebsfaenger, N.; Doehmer, J.; et al. CYP2D6 increases toxicity of the designer drug 4-methylthioamphetamine (4-MTA). Toxicology 2007, 229, 236–244. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fernandes, F.; Sousa, C.; Ferreres, F.; Valentão, P.; Remião, F.; Pereira, J.A.; Andrade, P.B. Kale Extract Increases Glutathione Levels in V79 Cells, but Does not Protect Them against Acute Toxicity Induced by Hydrogen Peroxide. Molecules 2012, 17, 5269-5288. https://doi.org/10.3390/molecules17055269
Fernandes F, Sousa C, Ferreres F, Valentão P, Remião F, Pereira JA, Andrade PB. Kale Extract Increases Glutathione Levels in V79 Cells, but Does not Protect Them against Acute Toxicity Induced by Hydrogen Peroxide. Molecules. 2012; 17(5):5269-5288. https://doi.org/10.3390/molecules17055269
Chicago/Turabian StyleFernandes, Fátima, Carla Sousa, Federico Ferreres, Patrícia Valentão, Fernando Remião, José A. Pereira, and Paula B. Andrade. 2012. "Kale Extract Increases Glutathione Levels in V79 Cells, but Does not Protect Them against Acute Toxicity Induced by Hydrogen Peroxide" Molecules 17, no. 5: 5269-5288. https://doi.org/10.3390/molecules17055269
APA StyleFernandes, F., Sousa, C., Ferreres, F., Valentão, P., Remião, F., Pereira, J. A., & Andrade, P. B. (2012). Kale Extract Increases Glutathione Levels in V79 Cells, but Does not Protect Them against Acute Toxicity Induced by Hydrogen Peroxide. Molecules, 17(5), 5269-5288. https://doi.org/10.3390/molecules17055269








