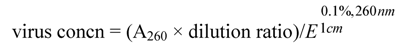Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives
Abstract
:1. Introduction
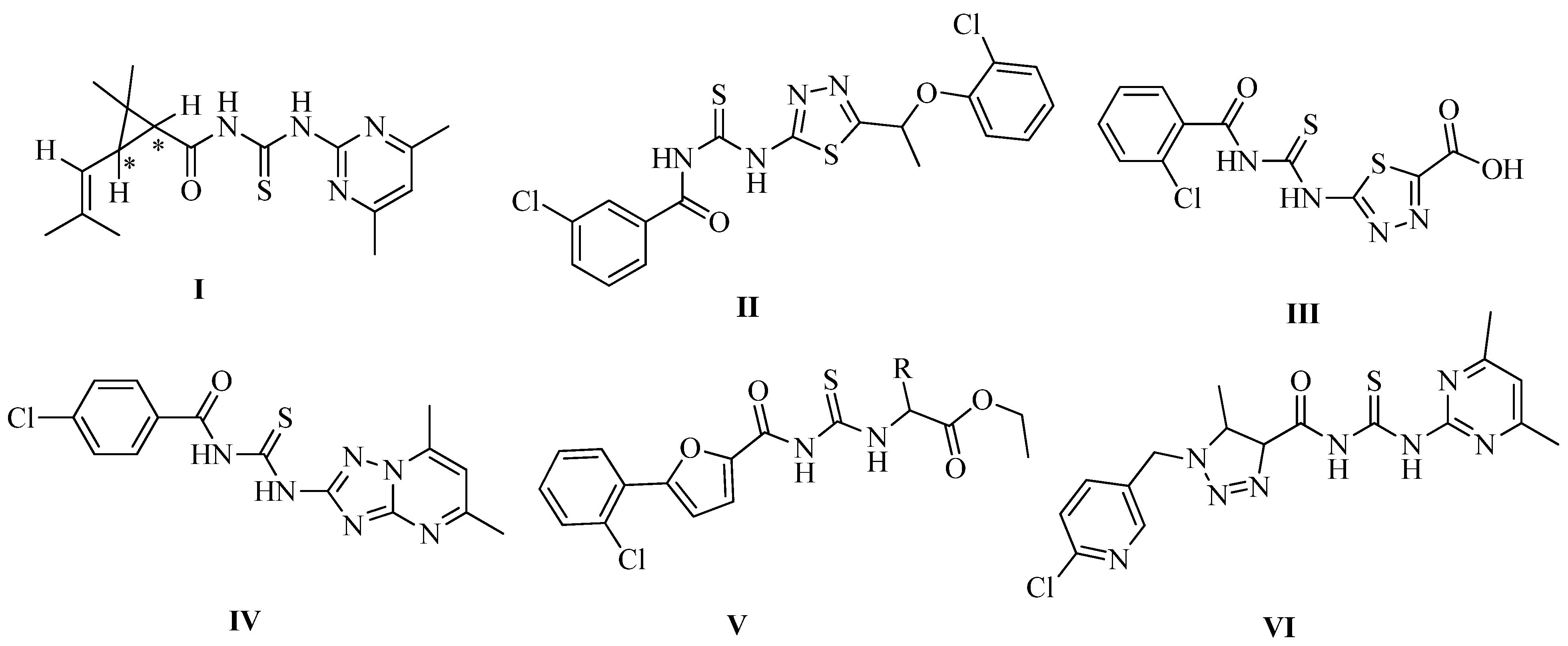
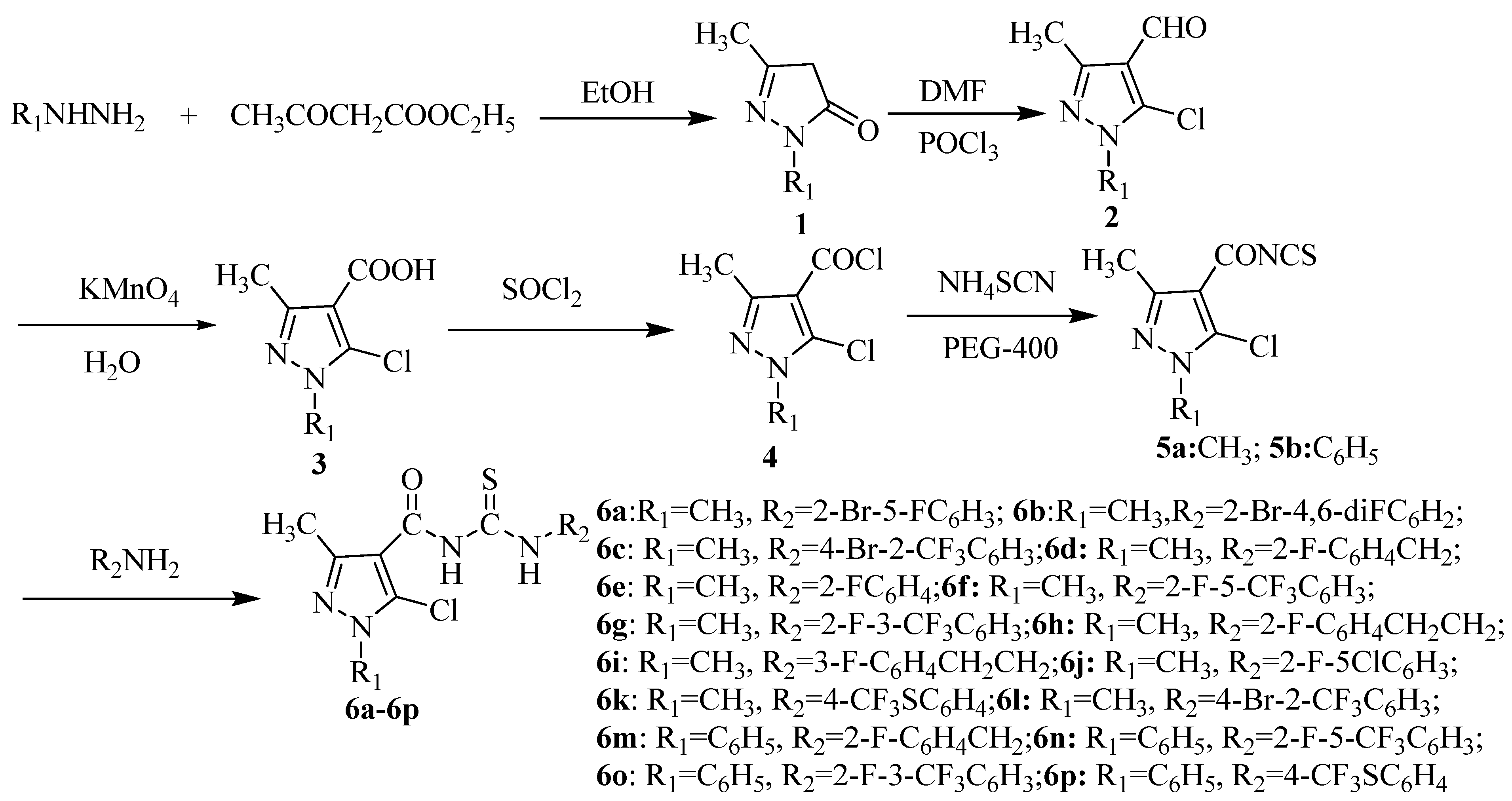
2. Results and Discussion
2.1. Chemistry
| Method | Agent | Reaction Temperature | Reaction Time | Yield |
|---|---|---|---|---|
| phase transfer catalysis | NH4SCN/PEG-400 | 25 °C | 3 h | 82.6% |
| conventional method | NH4SCN | 82 °C | 3 h | 51.2% |
2.2. Antifungal Activity Bioassay
| Compd. (100 µg/mL) | G. zeae | F. oxysporum | C. mandshurica |
|---|---|---|---|
| 6a | 2.84 | 1.46 | −5.79 |
| 6b | 24.7 | 50.3 | 48.7 |
| 6c | 8.24 | 6.92 | 1.98 |
| 6d | 12.8 | −3.21 | 8.16 |
| 6e | 9.57 | −1.15 | 8.47 |
| 6f | −0.57 | 4.39 | 3.42 |
| 6g | 17.6 | −1.46 | −3.68 |
| 6h | 48.6 | 57.9 | 59.7 |
| 6i | 19.9 | 4.09 | 6.57 |
| 6j | 4.55 | −3.80 | −6.31 |
| 6k | 15.4 | 11.8 | 7.91 |
| 6l | 22.9 | 0.86 | 4.80 |
| 6m | −1.14 | 6.73 | 3.42 |
| 6n | 5.85 | −0.29 | 5.08 |
| 6o | 7.18 | 5.19 | 11.9 |
| 6p | 3.46 | 9.22 | 4.24 |
| hymexazol | 69.7 | 67.1 | 69.5 |
3. Experimental
3.1. General
3.2. Preparation of the Intermediate Acyl Isothiocyanates 5
| Intermediate | R1 | Appearance | m.p./°C | Yield/% |
|---|---|---|---|---|
| 5a | CH3 | Yellow solid | 51~52 °C | 82.6% |
| 5b | C6H5 | White solid | 69~71 °C | 81.7% |
3.3. Preparation of Title Compounds 6a–p
3.4. Antifungal Bioassays
4. Conclusions
Acknowledgements
References and Notes
- Duan, L.P.; Xue, J.; Xu, L.L.; Zhang, H.B. Synthesis 1-acyl-3-(2'-aminophenyl) thioureas as anti-intestinal nematode prodrugs. Molecules 2010, 15, 6941–6947. [Google Scholar] [CrossRef]
- Zhong, Z.M.; Xing, R.; Liu, S.; Wang, L.; Cai, S.B.; Li, P.C. Synthesis of acyl thiourea derivatives of chitosan and their antimicrobial activities in vitro. Carbohydr. Res. 2008, 343, 566–570. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Wang, S.; Song, X.J.; Wang, Y.G. Synthesis and biological activity of N-5-(1-o-chlorophenoxyethy1)-1,3,4-thiodiazol-2-y1]-N-aryloxyacetyl thioureas. Chin. J. Pestic. Sci. 2005, 7, 282–284. [Google Scholar]
- Jurasek, A.; Safer, P.; Zvak, V. Synthesis and reactions of N-aryl-N'-(3-furoyl) thioureas. Chem. Pap. 1987, 41, 693–702. [Google Scholar]
- Nakamura, K.; Koike, K.; Sakamoto, M.; Nasumo, I. Pyrazole Derivatives. US 5607898, 1997. [Chem. Abstr. 1998, 125, 275866]. [Google Scholar]
- Dorfmeister, G.; Franke, H.; Geisler, J.; Hartfiel, U.; Bohner, J.; Rees, R. Substituted Pyrazole Derivatives and Their Use as Herbicides. US 5756424, 1998. [Chem. Abstr. 1998, 121, 230765]. [Google Scholar]
- Elbe, H.; Rieck, H.; Dunkel, R. Pyrazolyl Biphenyl Carboxamides. WO 02008195, 2002. [Chem. Abstr. 2002, 136, 151162]. [Google Scholar]
- Chen, H.S.; Li, Z.M.; Han, Y.F. Synthesis andfungicidal activity against Rhizoctonia solani of 2-alkyl(alkylthio)-5-pyrazoryl-1,3,4-oxadiazoles(thiodiazoles). J. Agric. Food Chem. 2002, 50, 3757–3760. [Google Scholar] [CrossRef]
- Xu, D.F.; Li, J.Z.; Guo, Y.L.; Xue, S.J. Synthesis and biological activity of N'-(substituted pyrimidin-2-yl)-N-chrysanthemoylthiourea derivatives. Chin. J. Org. Chem. 2005, 25, 1053–1056. [Google Scholar]
- Wang, Z.Y.; Gong, Y.X.; Chen, C.B.; Song, X.J.; Wang, Y.G. Synthesis and biological activity of N-[5-(1-o(p)-chlorophenoxy-ethyl)-1,3,4-thiodiazol-2-yl]-N'-aroyl thioureas. Chin. J. Org. Chem. 2005, 25, 1306–1310. [Google Scholar]
- Gong, Y.X.; Wang, Z.Y.; Zhang, Z.W.; Chen, C.B.; Wang, Y.G. Synthesis and biological activity of N-(2-carboxyl-1,3,4-thiadizol-5-yl)-N'-aroyl thioureas and aryloxyacetyl thioureas. Chin. J. Org. Chem. 2006, 26, 360–363. [Google Scholar]
- Wang, S.; Feng, G.R.; Liu, G.H.; Ye, W.F.; Wang, Y.G. Synthesis and biological activity of N-(1,2,4-triazolo-[1,5-a]pyrimidin-2-yl)-N'-aroyl thioureas. Chin. J. Org. Chem. 2006, 26, 1584–1586. [Google Scholar]
- Wang, Q.D.; Xue, S.J.; Shen, J.F.; Cai, Z.J. Synthesis and bioactivity of N-[5-(2-chlorophenyl)-2-furamidothiocarbonyl]-L-α-amino acid ethyl esters. Chin. J. Org. Chem. 2008, 28, 521–524. [Google Scholar]
- Yan, M.; Shi, D.Q. Synthesis and herbicidal activity of N-(4,6-disubstituted pyrimidin-2-yl)-N'-[1-(heteroaryl)methyl-5-methyl-1H-1,2,3-triazol-4-ylcarbonyl]thiourea. Chin. J. Org. Chem. 2008, 28, 736–740. [Google Scholar]
- Abd El-Wahab, A.H.F.; Al-Fifi, Z.I.A.; Bedair, A.H.; Ali, F.M.; Halawa, A.H.A.; El-Agrody, A.M. Synthesis, reactions and biological evaluation of some new naphtho[2,1-b]furan derivatives bearing a pyrazole nucleus. Molecules 2011, 16, 307–318. [Google Scholar] [CrossRef]
- Ouyang, G.P.; Cai, X.J.; Chen, Z.; Song, B.A.; Bhadury, P.S.; Yang, S.; Jin, L.H.; Xue, W.; Hu, D.Y.; Zeng, S. Synthesis and antiviral activities of pyrazole derivatives containing oxime ethers moiety. J. Agric Food Chem. 2008, 56, 10160–10167. [Google Scholar]
- Saad, H.A.; Osman, N.A.; Moustafa, A.H. Synthesis and analgesic activity of some new pyrazoles and triazoles bearing a 6,8-dibromo-2-methylquinazoline moiety. Molecules 2011, 16, 10187–10201. [Google Scholar] [CrossRef]
- Wei, T.B.; Zhang, Y.M.; Gao, L.M. An efficient synthesis of polymethylene-bis-aroyl thiourea derivatives under the condition of phase transfer catalysis. Synth. Commun. 2000, 30, 493–500. [Google Scholar] [CrossRef]
- Illi, V.O. Phase transfer catalyzed acylation. Tetrahedron Lett. 1979, 20, 2431–2432. [Google Scholar]
- Lv, Y.P.; Wang, X.Y.; Song, B.A.; Yang, S.; Yan, K.; Xu, G.F.; Bhadury, P.S.; Liu, F.; Jin, L.H.; Hu, D.Y. Synthesis, antiviral and antifungal bioactivity of 2-cyanoacrylate derivatives containing phosphonyl moieties. Molecules 2007, 12, 965–978. [Google Scholar] [CrossRef]
- Ouyang, G.; Zhang, P.; Xu, G.; Song, B.; Yang, S.; Jin, L.; Xue, W.; Hu, D.; Lu, P.; Chen, Z. Synthesis and antifungal bioactivities of 3-alkylquinazolin-4-one derivatives. Molecules 2006, 11, 383–392. [Google Scholar] [CrossRef]
- Gooding, G.V.Jr.; Hebert, T.T. A simple technique for purification of tobacco mosaic virus in large quantities. Phytopathology 1967, 57, 1285–1290. [Google Scholar]
- Gao, X.; Cai, X.; Yan, K.; Song, B.; Gao, L.; Chen, Z. Synthesis and antiviral bioactivities of 2-aryl- or 2-methyl-3-(substituted-Benzalamino)-4(3H)-quinazolinone derivatives. Molecules 2007, 12, 2621–2642. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the intermediates 1-5 and title compounds 6a-p are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wu, J.; Shi, Q.; Chen, Z.; He, M.; Jin, L.; Hu, D. Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives. Molecules 2012, 17, 5139-5150. https://doi.org/10.3390/molecules17055139
Wu J, Shi Q, Chen Z, He M, Jin L, Hu D. Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives. Molecules. 2012; 17(5):5139-5150. https://doi.org/10.3390/molecules17055139
Chicago/Turabian StyleWu, Jian, Qing Shi, Zhuo Chen, Ming He, Linhong Jin, and Deyu Hu. 2012. "Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives" Molecules 17, no. 5: 5139-5150. https://doi.org/10.3390/molecules17055139
APA StyleWu, J., Shi, Q., Chen, Z., He, M., Jin, L., & Hu, D. (2012). Synthesis and Bioactivity of Pyrazole Acyl Thiourea Derivatives. Molecules, 17(5), 5139-5150. https://doi.org/10.3390/molecules17055139