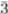Fabrication of Mesoporous Silica Shells on Solid Silica Spheres Using Anionic Surfactants and Their Potential Application in Controlling Drug Release
Abstract
:1. Introduction
2. Results and Discussion
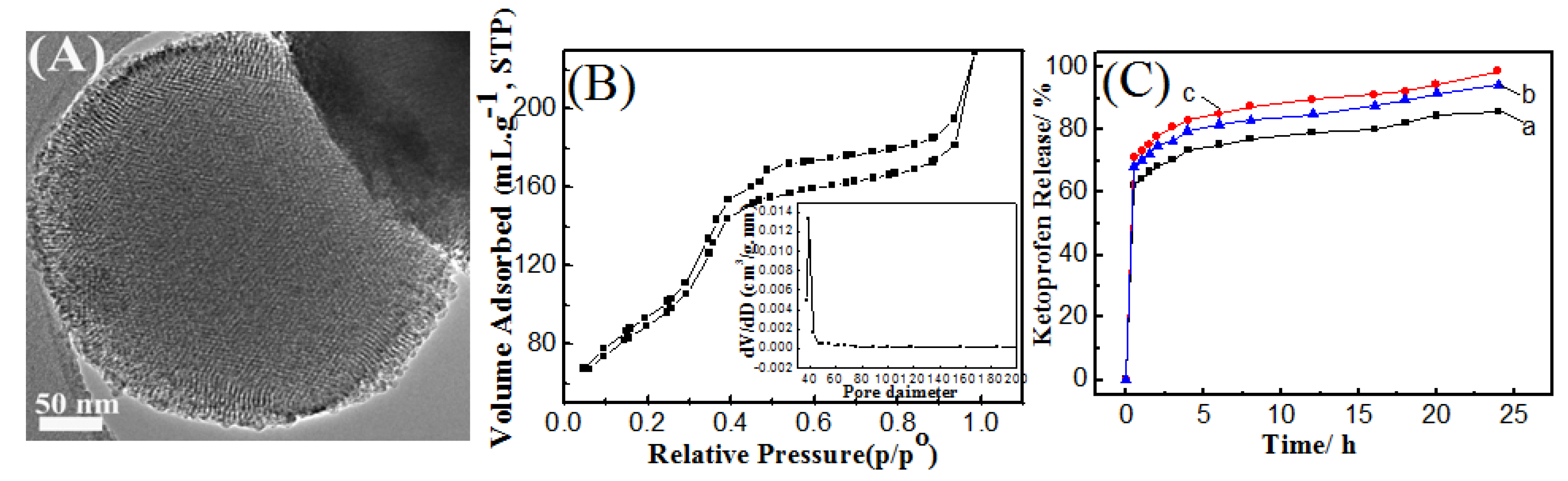
2.1. Effect of Synthesis Route
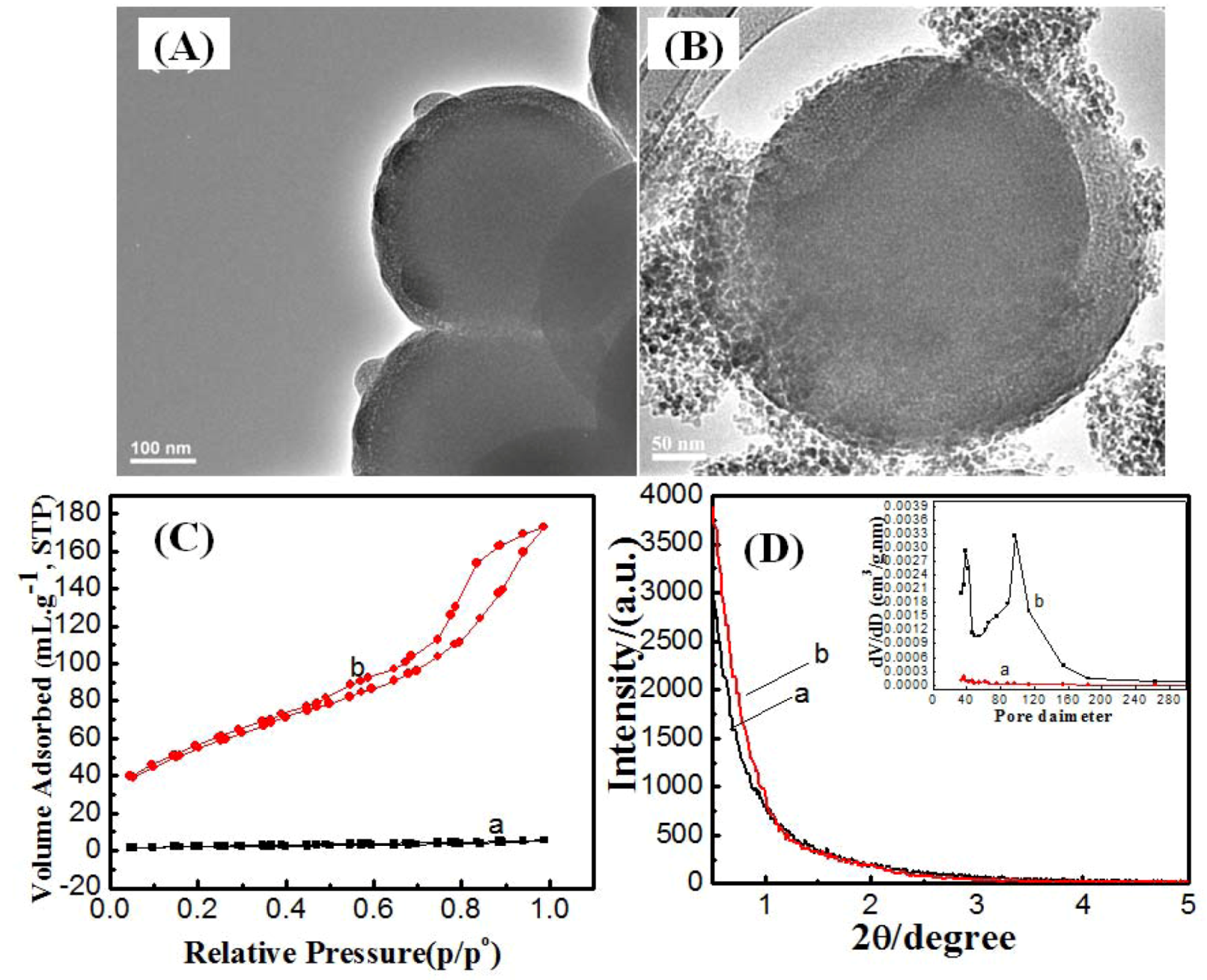
| Sample code | Synthesis route | Synthesis temperature/°C | Synthesis time | Surface area/cm2·g−1 | Pore volume/cc·g−1 | Shell thickness/nm | Pore size/nm | |
|---|---|---|---|---|---|---|---|---|
| TTEM | TDLS | |||||||
| SCMS-1 | One pot | 25 | 20 h | 7 | 0.008 | 34 | 38 | 3.6, 4, 6 |
| SCMS-2 | Two pot | 25 | 20 h | 199 | 0.207 | 41 | 43 | 3.6, 10 |
| SCMS-3 | Two pot | 50 | 20 h | 191 | 0.234 | 58 | 57 | 3.6, 10 |
| SCMS-4 | Two pot | 80 | 20 h | 286 | 0.243 | 65 | 71 | 3.6 |
| SCMS-5 | Two pot | 80 | 6 h | 324 | 0.261 | 50 | 53 | 3.6 |
| SCMS-6 | Two pot | 80 | 2 h | 226 | 0.184 | 42 | 46 | 3.6 |
| Hexa-an | - | 25 | 20 h | 332 | 0.354 | - | - | 3.6 |
2.2. Effect of Reaction Temperature
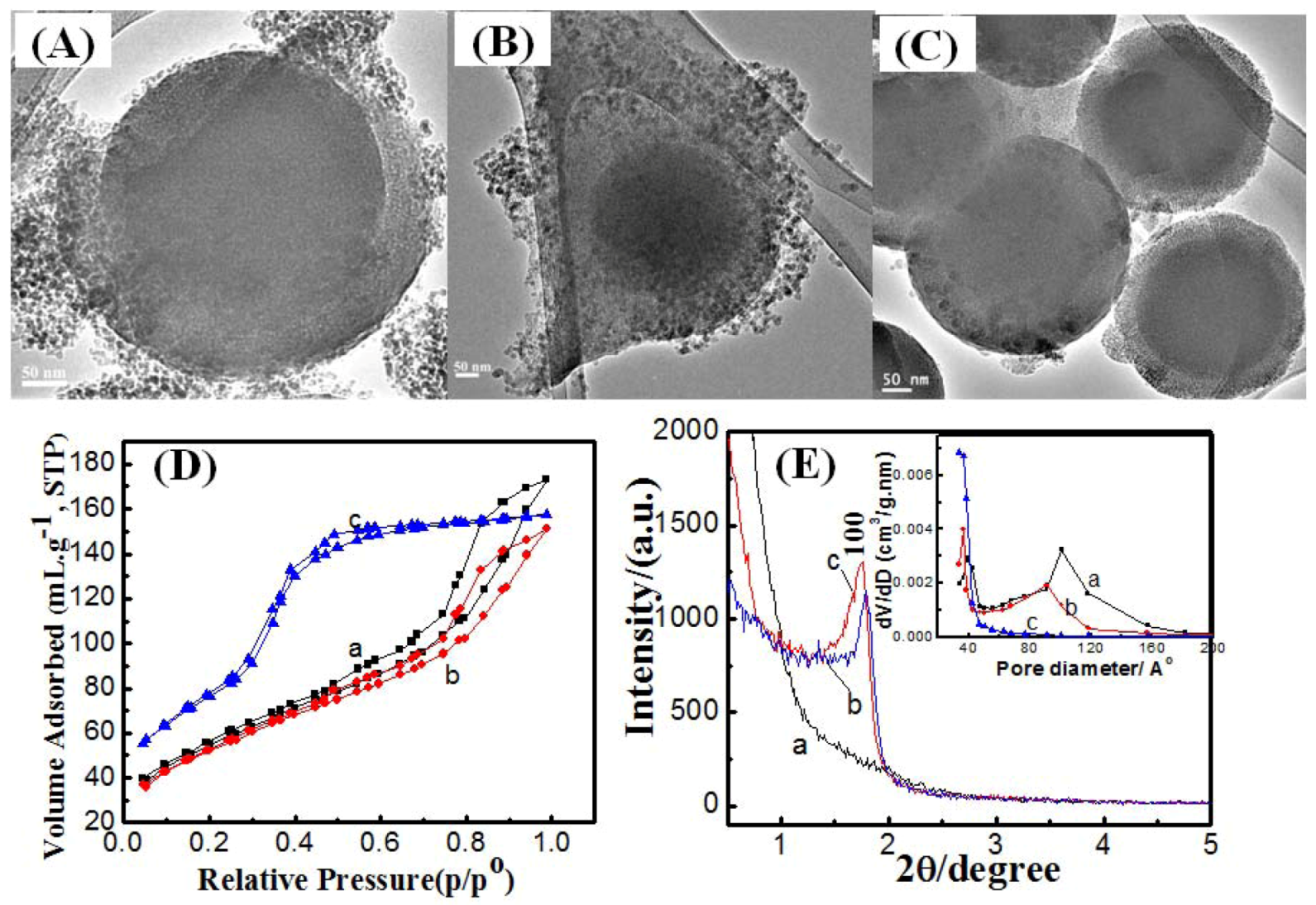
2.3. Effect of Reaction Time
3. Experimental
3.1. Chemicals
3.2. Synthesis
3.2.1. Synthesis of Solid Silica Cores
3.2.2. One-Pot Synthesis Route
3.2.3. Two-Pot Synthesis Route
3.2.4. In-Vitro Drug Storage Study
3.2.5. In Vitro Drug Release Study
3.3. Characterization
4. Conclusions
Acknowledgments
- Sample Availability: Small quantities, in micrograms, of the core-mesoporous shell silica spheres samples are available from the authors on request.
References
- Deng, Y.; Qi, D.; Deng, C.; Zhang, X.; Zhao, D. Superparamagnetic High-Magnetization Microspheres with an Fe3O4@SiO2 Core and Perpendicularly Aligned Mesoporous SiO2 Shell for Removal of Microcystins. J. Am. Chem. Soc. 2008, 130, 28–29. [Google Scholar] [CrossRef]
- Hah, H.J.; Um, J.I.; Han, S.H.; Koo, S.M. New synthetic route for preparing rattle-type silica particles with metal cores. Chem. Commun. 2004, 1012–1013. [Google Scholar]
- Zhao, W.; Chen, H.; Li, Y.; Li, L.; Lang, M.; Shi, J. Uniform Rattle-type Hollow Magnetic Mesoporous Spheres as Drug Delivery Carriers and their Sustained-Release Property. Adv. Funct. Mater. 2008, 18, 2780–2788. [Google Scholar] [CrossRef]
- Rosenholm, J.M.; Sahlgren, C.; Lindén, M. Towards multifunctional, targeted drug delivery systems using mesoporous silica nanoparticles—opportunities & challenges. Nanoscale 2010, 2, 1870–1883. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.E.; Lee, J.; Yu, J.H.; Kim, B.C.; An, K.; Hwang, Y.; Shin, C.-H.; Park, J.-G.; Kim, J.; et al. Magnetic Fluorescent Delivery Vehicle Using Uniform Mesoporous Silica Spheres Embedded with Monodisperse Magnetic and Semiconductor Nanocrystals. J. Am. Chem. Soc. 2006, 128, 688–689. [Google Scholar]
- Wu, H.; Liu, G.; Zhang, S.; Shi, J.; Zhang, L.; Chen, Y.; Chen, F.; Chen, H. Biocompatibility, MR imaging and targeted drug delivery of a rattle-type magnetic mesoporous silica nanosphere system conjugated with PEG and cancer-cell-specific ligands. J. Mater. Chem. 2011, 21, 3037–3045. [Google Scholar]
- Wu, H.; Zhang, S.; Zhang, J.; Liu, G.; Shi, J.; Zhang, L.; Cui, X.; Ruan, M.; He, Q.; Bu, W. Hollow-Core, Magnetic, and Mesoporous Double-Shell Nanostructure: In Situ Decomposition/ Reduction Synthesis, Bioimaging, and Drug-Delivery Properties. Adv. Funct. Mater. 2011, 21, 1850–1862. [Google Scholar] [CrossRef]
- Barmatova, M.V.; Ivanchikova, I.D.; Kholdeeva, O.A.; Shmakov, A.N.; Zaikovskii, V.I.; Mel’gunov, M.S. Magnetically separable titanium-silicate mesoporous materials with core-shell morphology: Synthesis, characterization and catalytic properties. J. Mater. Chem. 2009, 19, 7332–7339. [Google Scholar]
- Wang, C.; Tao, S.; Wei, W.; Meng, C.; Liu, F.; Han, M. Multifunctional mesoporous material for detection, adsorption and removal of Hg2+ in aqueous solution. J. Mater. Chem. 2010, 20, 4635–4641. [Google Scholar] [CrossRef]
- Kim, J.; Piao, Y.; Lee, N.; Park, Y.; Lee, I.-H.; Lee, J.-H.; Paik, S.R.; Hyeon, T. Magnetic Nanocomposite Spheres Decorated with NiO Nanoparticles for a Magnetically Recyclable Protein Separation System. Adv. Mater. 2010, 22, 57–60. [Google Scholar]
- Yoon, S.B.; Kim, J.-Y.; Kim, J.H.; Park, Y.J.; Yoon, K.R.; Park, S.-K.; Yu, J.-S. Synthesis of monodisperse spherical silica particles with solid core and mesoporous shell: Mesopore channels perpendicular to the surface. J. Mater. Chem. 2007, 17, 1758–1761. [Google Scholar]
- Büchel, G.; Unger, K.K.; Matsumoto, A.; Tsutsumi, K. A Novel Pathway for Synthesis of Submicrometer-Size Solid Core/Mesoporous Shell Silica Spheres. Adv. Mater. 1998, 10, 1036–1038. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, T.; Ge, J.; Yin, Y. Permeable Silica Shell through Surface-Protected Etching. Nano Lett. 2008, 8, 2867–2871. [Google Scholar] [CrossRef]
- Che, S.; Garcia-Bennett, A.E.; Yokoi, T.; Sakamoto, K.; Kunieda, H.; Terasaki, O.; Tatsumi, T. A novel anionic surfactant templating route for synthesizing mesoporous silica with unique structure. Nat. Mater. 2003, 2, 801–805. [Google Scholar] [CrossRef]
- Garcia-Bennett, A.E.; Miyasaka, K.; Terasaki, O.; Che, S. Structural Solution of Mesocaged Material AMS-8. Chem. Mater. 2004, 16, 3597–3605. [Google Scholar] [CrossRef]
- Gao, C.; Sakamoto, Y.; Sakamoto, K.; Terasaki, O.; Che, S. Synthesis and Characterization of Mesoporous Silica AMS-10 with Bicontinuous Cubic Pn
m Symmetry. Angew. Chem. Int. Ed. Engl. 2006, 45, 4295–4298. [Google Scholar] [CrossRef]
- Jin, H.; Liu, Z.; Ohsuna, T.; Terasaki, O.; Inoue, Y.; Sakamoto, K.; Nakanishi, T.; Ariga, K.; Che, S. Control of Morphology and Helicity of Chiral Mesoporous Silica. Adv. Mater. 2006, 18, 593–596. [Google Scholar]
- Yokoi, T.; Yoshitake, H.; Yamada, T.; Kubota, Y.; Tatsumi, T. Amino-functionalized mesoporous silica synthesized by an anionic surfactant templating route. J. Mater. Chem. 2006, 16, 1125–1135. [Google Scholar] [CrossRef]
- Mine, E.; Yamadaa, A.; Kobayashi, Y.; Konno, M.; Liz-Marzán, L.M. Direct coating of gold nanoparticles with silica by a seeded polymerization technique. J. Colloid. Interface Sci. 2003, 264, 385–390. [Google Scholar] [CrossRef]
- Wang, W.-Q.; Wang, J.-G.; Sun, P.-C.; Ding, D.-T.; Chen, T.-H. Effect of alcohol on morphology and mesostructure control of anionic-surfactant-templated mesoporous silica (AMS). J. Colloid. Interface Sci. 2009, 331, 156–162. [Google Scholar] [CrossRef]
- Niu, D.; Ma, Z.; Li, Y.; Shi, J. Synthesis of Core−Shell Structured Dual-Mesoporous Silica Spheres with Tunable Pore Size and Controllable Shell Thickness. J. Am. Chem. Soc. 2010, 132, 15144–15147. [Google Scholar] [CrossRef]
- Wang, J.; Li, F.; Zhou, H.; Sun, P.; Ding, D.; Chen, T. Silica Hollow Spheres with Ordered and Radially Oriented Amino-Functionalized Mesochannels. Chem. Mater. 2009, 21, 612–620. [Google Scholar] [CrossRef]
- Chen, Q.; Sakamoto, Y.; Terasaki, O.; Che, S. Synthesis of silica mesoporous crystals with controlled structure and morphology using gemini surfactant. Microporous Mesoporous Mater. 2007, 105, 24–33. [Google Scholar] [CrossRef]
- Lu, A.-H.; Zhao, D.; Wan, Y. Nanocasting: A Versatile Strategy for Creating Nanostructured Porous Materials; Royal Society of Chemistry: Cambridge, UK, 2009. [Google Scholar]
- Park, S.-J.; Kim, Y.-J.; Park, S.-J. Size-Dependent Shape Evolution of Silica Nanoparticles into Hollow Structures. Langmuir 2008, 24, 12134–12137. [Google Scholar] [CrossRef]
- Qu, F.; Zhu, G.; Huang, S.; Li, S.; Sun, J.; Zhang, D.; Qiu, S. Controlled release of Captopril by regulating the pore size and morphology of ordered mesoporous silica. Microporous Mesoporous. Mater. 2006, 92, 1–9. [Google Scholar] [CrossRef]
- Li, Z.-Z.; Wen, L.-X.; Shao, L.; Chen, J.-F. Fabrication of porous hollow silica nanoparticles and their applications in drug release control. J. Control. Release 2004, 98, 245–254. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Toni, A.M.; Khan, A.; Ibrahim, M.A.; Al-Hoshan, M.; Labis, J.P. Fabrication of Mesoporous Silica Shells on Solid Silica Spheres Using Anionic Surfactants and Their Potential Application in Controlling Drug Release. Molecules 2012, 17, 13199-13210. https://doi.org/10.3390/molecules171113199
El-Toni AM, Khan A, Ibrahim MA, Al-Hoshan M, Labis JP. Fabrication of Mesoporous Silica Shells on Solid Silica Spheres Using Anionic Surfactants and Their Potential Application in Controlling Drug Release. Molecules. 2012; 17(11):13199-13210. https://doi.org/10.3390/molecules171113199
Chicago/Turabian StyleEl-Toni, Ahmed Mohamed, Aslam Khan, Mohamed Abbas Ibrahim, Mansour Al-Hoshan, and Joselito Puzon Labis. 2012. "Fabrication of Mesoporous Silica Shells on Solid Silica Spheres Using Anionic Surfactants and Their Potential Application in Controlling Drug Release" Molecules 17, no. 11: 13199-13210. https://doi.org/10.3390/molecules171113199
APA StyleEl-Toni, A. M., Khan, A., Ibrahim, M. A., Al-Hoshan, M., & Labis, J. P. (2012). Fabrication of Mesoporous Silica Shells on Solid Silica Spheres Using Anionic Surfactants and Their Potential Application in Controlling Drug Release. Molecules, 17(11), 13199-13210. https://doi.org/10.3390/molecules171113199