Two New Triterpene Glycosides with Monomethyl Malonate Groups from the Rhizome of Cimifuga foetida L.
Abstract
:1. Introduction
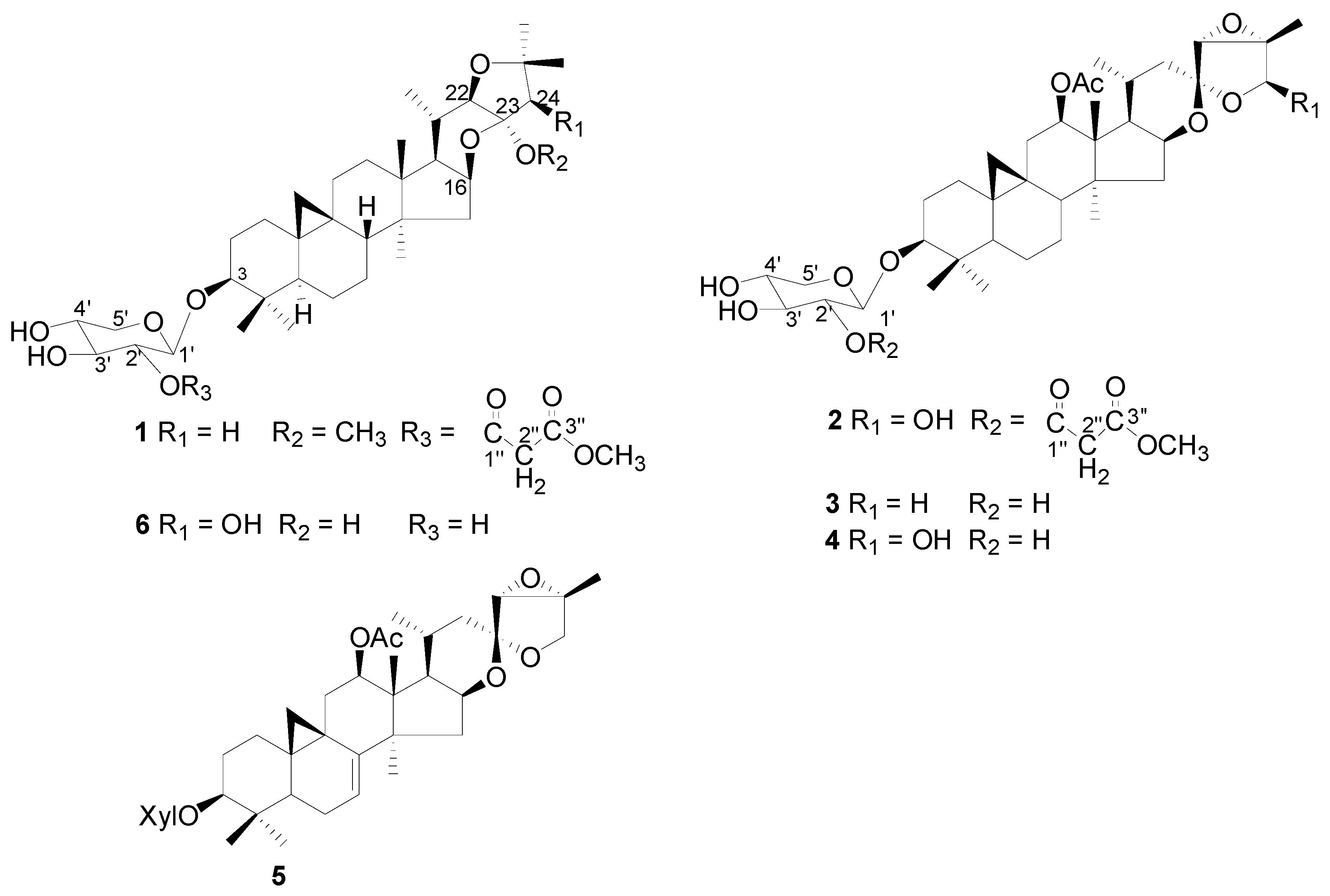
2. Results and Discussion
| No | 1 a | 2 b | No | 1 a | 2 b | ||||
|---|---|---|---|---|---|---|---|---|---|
| 13C | 1H | 13C | 1H | 13C | 1H | 13C | 1H | ||
| 1 | 32.0 t | 1.20 m; 1.52 m | 31.8 t | 1.15 m; 1.52 m | 20 | 34.3 d | 2.15 m | 26.0 d | 1.79 m |
| 2 | 29.8 t | 1.85 m; 2.20 m | 29.7 t | 1.88 m; 2.28 m | 21 | 17.5 q | 1.15 d 6.4 | 21.0 q | 0.94 d 6.3 |
| 3 | 88.5 d | 3.39 d 3.5 | 88.2 d | 3.31 dd 4.5, 11.5 | 22 | 86.3 d | 3.67 m | 37.6 t | 1.65 m; 2.20 m |
| 4 | 41.0 s | / | 40.9 s | / | 23 | 109.7 s | / | 105.8 s | / |
| 5 | 47.3 d | 1.33 m | 46.9 d | 1.26 m | 24 | 30.0 t | 1.54 m | 63.5 d | 3.90 s |
| 6 | 20.9 t | 0.73 m; 1.31 m | 20.1 t | 0.65 m; 1.45 m | 25 | 83.4 s | / | 65.6 s | / |
| 7 | 26.1 t | 1.61 m | 25.7 t | 0.95 m; 1.30 m | 26 | 27.1 q | 1.51 s | 98.4 d | 5.70 s |
| 8 | 47.5 d | 1.49 m | 45.8 d | 1.52 m | 27 | 24.6 q | 1.61 s | 13.1 q | 1.74 s |
| 9 | 19.7 s | / | 20.4 s | / | 28 | 19.7 q | 0.83 s | 19.5 q | 0.75 s |
| 10 | 26.4 s | / | 26.7 s | / | 29 | 25.6 q | 1.30 s | 25.6 q | 1.08 s |
| 11 | 26.5 t | 1.79 m | 36.7 t | 1.16 m; 2.68 dd 8.3, 15.9 | 30 | 15.2 q | 0.97 s | 15.1 q | 0.91 s |
| 12 | 33.4 d | 1.50 m | 77.1 d | 5.05 dd 3.7, 8.5 | 1' | 104.2 d | 4.83 d 7.4 | 104.1 d | 4.79 d 7.8 |
| 13 | 46.9 s | / | 48.7 s | / | 2' | 76.8 d | 5.54 t 6.9 | 76.7 d | 5.50 t 8.4 |
| 14 | 45.3 s | / | 47.8 s | / | 3' | 76.6 d | 4.15 m | 76.1 d | 4.11 m |
| 15 | 42.8 t | 1.58 m; 1.87 m | 43.6 t | 1.89 dd 8.1, 12.6; 1.71 m | 4' | 71.3 d | 4.18 m | 71.3 d | 4.15 m |
| 16 | 72.5 d | 4.40 dd 6.0, 12.5 | 73.0 d | 4.58 dd 7.1, 14.2 | 5' | 67.1 t | 3.64 m; 4.29 dd 3.5, 8.7 | 67.1 t | 3.60 m; 4.25 dd 7.1, 11.9 |
| 17 | 51.6 d | 1.50 m | 56.4 d | 1.77 m | 1'' | 166.5 s | / | 166.5 s | / |
| 18 | 20.6 q | 1.14 s | 13.6 q | 1.33 s | 2'' | 42.0 t | 3.78 s | 42.0 t | 3.74 s |
| 19 | 30.2 t | 0.13 d 2.9; 0.39 d 2.9 | 29.6 t | 0.17 d 4.0; 0.50 d 4.0 | 3'' | 167.2 s | / | 167.2 s | / |


3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Acid Hydrolysis of Compounds 1, 2
4. Conclusions
Acknowledgements
References
- Lieberman, S.J. A review of the effectiveness of Cimicifuga racemosa (black cohosh) for the symptoms of menopause. Women’s Health 1998, 7, 525–529. [Google Scholar] [CrossRef]
- Mckenna, D.J.; Jones, K.; Humphrey, S.; Hughes, K. Black cohosh: Efficacy, safety, and use in clinical and preclinical applications. Altern. Ther. 2001, 7, 93–100. [Google Scholar]
- Liske, E.; Wustenberg, P. Therapy of climacteric complaints with Cimicifuga racemosa: herbal medicine with clinically proven evidence. Menopause 1998, 5, 250–256. [Google Scholar]
- Lin, Y.P.; Qiu, M.H.; Li, Z.R. Studies on the chemical constituents and biologic activities of Cimicifuga. Nat. Prod. Res. Dev. 2002, 14, 58–76. [Google Scholar]
- Qiu, M.H.; Kim, J.H.; Lee, H.K.; Min, B.S. Anticomplement activity of cycloartane glycosides from the rhizome of Cimicifuga foetida. Phytother. Res. 2006, 20, 945–948. [Google Scholar] [CrossRef]
- Sun, L.R.; Yan, J.; Pei, S.J.; Qiu, M.H. A new cycloartane triterpenoid from the rhizome of Cimicifuga foetida collected in Dali. Acta Bot. Yunnan 2005, 27, 331–336. [Google Scholar]
- Sun, L.R.; Qing, C.; Zhang, Y.L.; Jia, S.Y.; Li, Z.R.; Pei, S.J.; Qiu, M.H.; Michael, L.G.; Qiu, S.X. Cimicifoetisides A and B, two cytotoxic cycloartane triterpenoid glycosides from the rhizomes of Cimicifuga foetida, inhibit proliferation of cancer cells. Beilstein J. Org. Chem. 2007, 3, 3. [Google Scholar]
- Sun, L.R.; Yan, J.; Lu, L.; Pei, S.J.; Li, Z.R.; Zhou, L.; Zhang, X.M.; Qiu, M.H. Cimicifine A: A novel triterpene alkaloid from the rhizomes of Cimicifuga foetida. Helv. Chim. Acta 2007, 7, 1313–1318. [Google Scholar]
- Sun, L.R.; Yan, J.; Nian, Y.; Zhou, L.; Zhang, H.J.; Qiu, M.H. New triterpene diglycosides from the rhizome of Cimicifuga foetida. Molecules 2008, 13, 1712–1721. [Google Scholar] [CrossRef]
- Lu, L.; Chen, J.C.; Nian, Y.; Sun, Y.; Qiu, M.H. Trinor-cycloartane glycosides from the rhizomes of Cimicifuga foetida. Molecules 2009, 14, 1578–1584. [Google Scholar] [CrossRef]
- Nian, Y.; Zhang, Y.L.; Chen, J.C.; Lu, L.; Qiu, M.H.; Qing, C. Cytotoxic chemical constituents from the roots of Cimicifuga fetida. J. Nat. Prod. 2010, 73, 93–98. [Google Scholar] [CrossRef]
- Chen, S.N.; Li, W.K.; Fabricant, D.S.; Santarsiero, B.D.; Mesecar, A.; Fitzloff, J.F.; Fong, H.H.S.; Farnsworth, N.R. Isolation, structure elucidation, and absolute configuration of 26-deoxyactein from Cimicifuga racemosa and clarification of nomenclature associated with 27-deoxyactein. J. Nat. Prod. 2002, 65, 601–605. [Google Scholar] [CrossRef]
- Li, C.J.; Li, Y.H.; Chen, S.F.; Xiao, P.G. Triterpenoids from Cimicifuga foetida L. Acta Pharm. Sin. 1994, 29, 449–453. [Google Scholar]
- Kusano, A.; Takahira, M.; Shibano, M.; Miyase, T.; Kusano, G. Studies on the constituents of Cimicifuga species. XXVI. Twelve new cyclolanostanol glycosides from the underground parts of Cimicifuga simplex Wormsk. Chem. Pharm. Bull. 1999, 47, 511–516. [Google Scholar] [CrossRef]
- Kusan, A.; Yakahira, M.; Shibano, M.; Miyase, T.; Okuyama, T.; Kusano, G. Structures of two new cyclolanostanol xylosides: cimiacerosides A and B. Heterocycles 1998, 48, 1003–1014. [Google Scholar] [CrossRef]
- Kusano, A.; Shibano, M.; Kusano, G. Studies on the constituents of Cimicifuga species. XXVII. Malonyl cyclolanostanol glycosides from the underground parts of Cimicifuga simplex Wormsk. Chem. Pharm. Bull. 1999, 47, 1175–1179. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, D.H.; Si, J.Y.; Tu, G.Z.; An, D.G. Two new cyclolanostanol xylosides from the aerial parts of Cimicifuga dahurica. J. Nat. Prod. 2002, 65, 1486–1488. [Google Scholar] [CrossRef]
- Li, J.X.; Yu, Z.Y. Cimicifugae rhizoma: from origins, bioactive constituents to clinical outcomes. Curr. Med. Chem. 2006, 13, 2927–2951. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–6 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, L.-R.; Yan, J.; Zhou, L.; Li, Z.-R.; Qiu, M.-H. Two New Triterpene Glycosides with Monomethyl Malonate Groups from the Rhizome of Cimifuga foetida L. Molecules 2011, 16, 5701-5708. https://doi.org/10.3390/molecules16075701
Sun L-R, Yan J, Zhou L, Li Z-R, Qiu M-H. Two New Triterpene Glycosides with Monomethyl Malonate Groups from the Rhizome of Cimifuga foetida L. Molecules. 2011; 16(7):5701-5708. https://doi.org/10.3390/molecules16075701
Chicago/Turabian StyleSun, Li-Rong, Jian Yan, Lin Zhou, Zhong-Rong Li, and Ming-Hua Qiu. 2011. "Two New Triterpene Glycosides with Monomethyl Malonate Groups from the Rhizome of Cimifuga foetida L." Molecules 16, no. 7: 5701-5708. https://doi.org/10.3390/molecules16075701
APA StyleSun, L.-R., Yan, J., Zhou, L., Li, Z.-R., & Qiu, M.-H. (2011). Two New Triterpene Glycosides with Monomethyl Malonate Groups from the Rhizome of Cimifuga foetida L. Molecules, 16(7), 5701-5708. https://doi.org/10.3390/molecules16075701





