Preparation and Evaluation of Berberine Alginate Beads for Stomach-Specific Delivery
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation and Evaluation of Floating Berberine Beads

2.1.1. Diameter
| Ratios (SA: berberine) | 1:0 | 1:2 | 1:4 | 1:6 | 1:8 | 1:10 |
|---|---|---|---|---|---|---|
| Mean Diameter (mm) | 0.85 ± 0.02 | 1.24 ± 0.01 | 1.42 ± 0.01 | 1.69 ± 0.01 | 1.86 ± 0.01 | 2.07 ± 0.01 |
| Drug Loading (%) | 0 | 62.1 | 75.5 | 81.4 | 85.4 | 88.7 |
| Entrapped Efficiency (%) | 0 | 94.6 | 94.4 | 94.7 | 91.3 | 95.1 |
2.1.2. Drug Loading and Drug Entrapment Efficiency
2.1.3. Floating Ability
| No. | Ratio (SA:octodecanol:bererine) | Octodecanol (%) | Lag time (h) | Lasting floating time (h) |
|---|---|---|---|---|
| F1 | 1:1:2 | 25% | 0 | >24 |
| F2 | 1:0.5:2 | 14.29% | 0 | >24 |
| F3 | 1:0.2:2 | 6.25% | 0 | >24 |
| F4 | 1:0.1:2 | 3.23% | 0.1 * | 15.3 |
| F5 | 1:0:2 | 0 | sank immediately and then suspended in the central of vessels after 1 h | |
2.2. In vitro Dissolution Studies
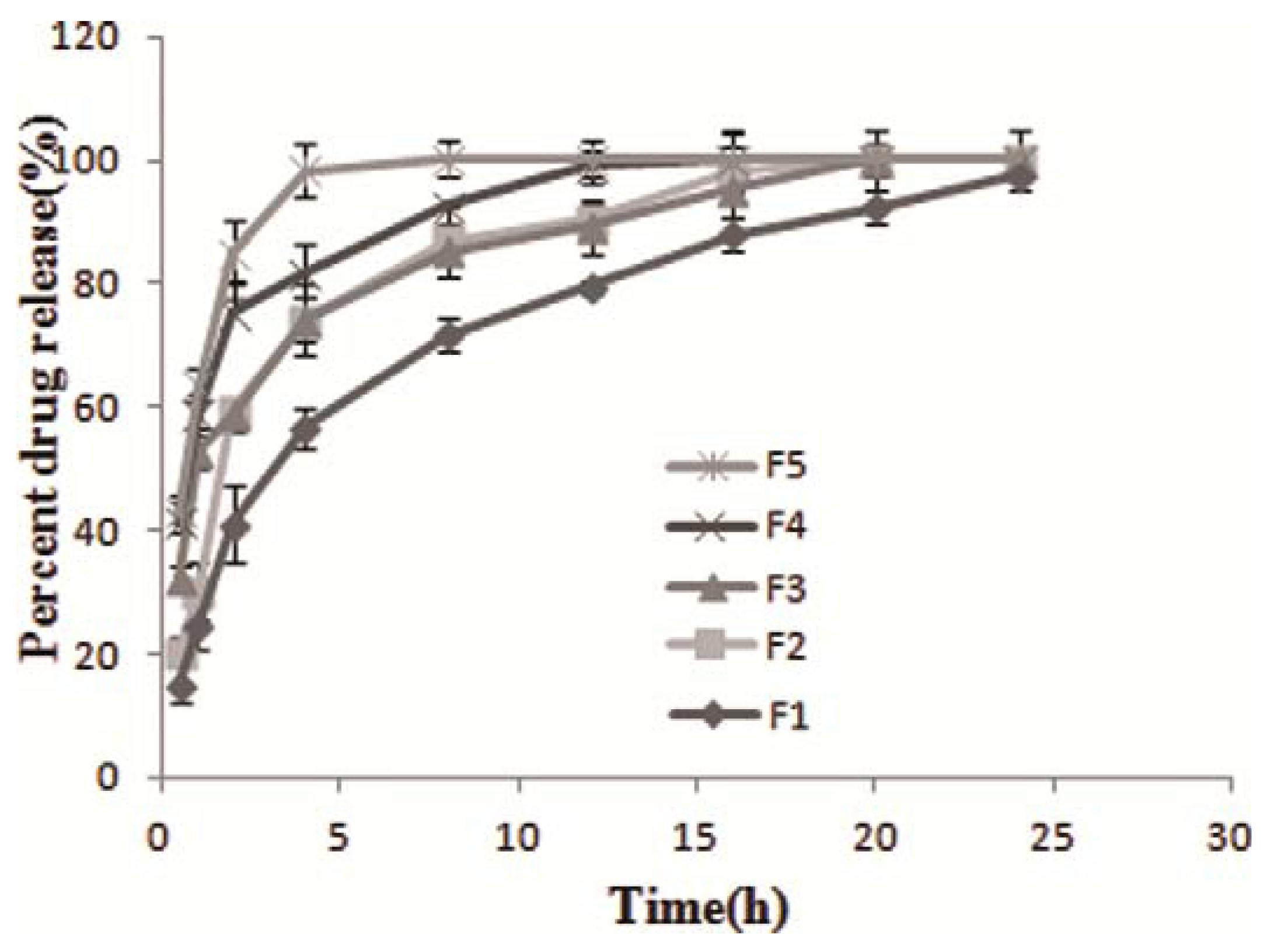
| No | Model | Results | r2 |
|---|---|---|---|
| 1 | Zero order | Qt = 5.1061t | 0.3365 |
| 2 | Fist order | Qt = 21.707Lnt + 26.995 | 0.9973 |
| 3 | Higuich | Qt = 21.595t1/2 | 0.9362 |
| 4 | Hixson-Crowell | (100 − Qt)1/3 = −0.1208t + 4.2502 | 0.9860 |
| 5 | Korsemeyer-Peppas | LnQt = 0.3837Lnt + 2.8229 | 0.9968 |
2.3. In Vivo Gastric Residence Time Study in Rat
| Formula | Remaining bead (12 h) | Remaining bead (24 h) |
|---|---|---|
| berberine alginate beads 1 | 30 | 30 |
| berberine alginate beads 2 | 30 | 29 |
| berberine alginate beads 3 | 27 | 22 |
| free alginate beads | 22 | 15 |
| sustained-release micro-pellets | 12 | 6 |
3. Experimental
3.1. Materials
3.2. Preparation and Evaluation of Floating Berberine Beads
3.2.1. Determination of Mean Diameter
3.2.2. Determination of Drug Loading and Drug Entrapment Efficiency
3.2.3. Determination of Floating Time
3.3. In Vitro Dissolution Study
3.4. In Vitro Gastric Residence Time Study in Rat
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Conflicts of Interest
References and Notes
- Goodman, K.J.; Cockburn, M. The role of epidemiology in understanding thehealth effects of Helicobacter pylori. Epidemiology 2001, 12, 266–271. [Google Scholar] [CrossRef]
- de Sousa Falcão, H.; Leite, J.A.; Barbosa-Filho, J.M.; de Athayde-Filho, P.F.; de Oliveira Chaves, M.C.; Moura, M.D.; Ferreira, A.L.; de Almeida, A.B.; Souza-Brito, A.R.; de Fátima Formiga Melo Diniz, M.; et al. Gastric and Duodenal Antiulcer Activity of Alkaloids: A Review. Molecules 2008, 13, 3198–3223. [Google Scholar]
- Konturek, P.C.; Konturek, S.J.; Brzozowski, T. Helicobacter pylori infection in gastric cancerogenesis. J. Physiol. Pharmacol. 2009, 60, 3–21. [Google Scholar]
- Schreiber, S.; Konradt, M.; Groll, C.; Scheid, P.; Hanauer, G.; Werling, H.O.; Josenhans, C.; Suerbaum, S. The spatial orientation of Helicobacter pylori in the gastric mucus. Proc. Natl. Acad. Sci. USA 2004, 101, 5024–5029. [Google Scholar]
- Bardonnet, P.L.; Faivre, V.; Pugh, W.J.; Piffaretti, J.C.; Falson, F. Gastroretentive dosageforms: Overview and special case of Helicobacter pylori. J. Control. Release 2006, 111, 1–18. [Google Scholar] [CrossRef]
- Davis, S.S.; Stockwell, A.F.; Taylor, M.J.; Hardy, J.G.; Whalley, D.R.; Wilson, C.G.; Bechgaard, H.; Christensen, F.N. The effect of density on the gastric emptying of single- and multiple-unit dosage forms. Pharm. Res. 1986, 3, 208–213. [Google Scholar] [CrossRef]
- Chang, C.H.; Huang, W.Y.; Lai, C.H.; Hsu, Y.M.; Yao, Y.H.; Chen, T.Y.; Wu, J.Y.; Peng, S.F.; Lin, Y.H. Development of novel nanoparticles shelled with heparin for Berberine chloride delivery to treat Helicobacter pylori. Acta Biomater. 2011, 7, 593–603. [Google Scholar] [CrossRef]
- Eftaiha, A.F.; Qinna, N.; Rashid, I.S.; Al Remawi, M.M.; Al Shami, M.R.; Arafat, T.A.; Badwan, A.A. Bioadhesive Controlled Metronidazole Release Matrix Based on Chitosan and Xanthan Gum. Mar. Drugs. 2010, 8, 1716–1730. [Google Scholar] [CrossRef]
- Hwang, S.J.; Park, H. Gastro retentive drug-delivery systems. Crit. Rev. Ther. Drug Carrier Syst. 1998, 15, 234–284. [Google Scholar]
- Kharia, A.A.; Hiremath, S.N.; Singhai, A.K.; Omray, L.K.; Jain, S.K. Design and optimization of floating drug delivery system of acyclovir. Indian J. Pharm. Sci. 2010, 72, 599–606. [Google Scholar] [CrossRef]
- Sathish, D.; Himabindu, S.; Kumar, Y.S.; Shayeda; Rao, Y.M. Floating drug delivery systems for prolonging gastric residence time: A review. Curr. Drug Deliv. 2011, 8, 494–510. [Google Scholar] [CrossRef]
- D'Ayala, G.G.; Malinconico, M.; Laurienzo, P. Marine derived polysaccharides for biomedical applications: chemical modification approaches. Molecules 2008, 13, 2069–2106. [Google Scholar] [CrossRef]
- Murata, Y.; Jinno, D.; Liu, D.; Isobe, T.; Kofuji, K.; Kawashima, S. The drug release profile from calcium-induced alginate gel beads coated with an alginate hydrolysate. Molecules 2007, 12, 2559–2566. [Google Scholar] [CrossRef]
- Whitehead, L.; Collett, J.H.; Fell, J.T. Amoxycillin release from a floating dosage form based on alginates. Int. J. Pharm. 2000, 210, 45–49. [Google Scholar] [CrossRef]
- Dmmrcp, H.L.S.; Atkinson, M.M.F. Comparison of a dimethicone/antacid (Asilone gel) with an alginate/antacid (Gaviscon liquid) in the management of reflux oesophagitis. J. Roy. Soc. Med. 1990, 83, 554–556. [Google Scholar]
- Poynard, T.; Vernisse, B.; Agostini, H. Randomized, multicentre comparison of sodium alginate and cisapride in the symptomatic treatment of uncomplicated gastro-oesophageal reflux. Aliment. Pharmacol. Ther. 1998, 12, 159–165. [Google Scholar] [CrossRef]
- Shiraishi, S.; Imai, T.; Otagiri, M. Controlled-release preparation of indomethacin using calcium alginate gel. Biol. Pharm. Bull. 1993, 16, 1164–1168. [Google Scholar] [CrossRef]
- Murata, Y.; Kofuji, K.; Kawashima, S. Preparation of floating alginate beads for drug delivery to gastric mucosa. J. Biomater. Sci. Polym. Ed. 2003, 14, 581–588. [Google Scholar] [CrossRef]
- Stops, F.; Fell, J.T.; Collett, J.H.; Martini, L.G. Floating dosage forms to prolong gastro-retention-The characterisation of calcium alginate beads. Int. J. Pharm. 2008, 350, 301–311. [Google Scholar] [CrossRef]
- Imanshahidi, M.; Hosseinzadeh, H. Pharmacological and therapeutic effects of Berberis vulgaris and its active constituent, berberine. Phytother. Res. 2008, 22, 999–1012. [Google Scholar] [CrossRef]
- Kuo, C.L.; Chi, C.W.; Liu, T.Y. The anti-inflammatory potential of Berberine chloride in vitro and in vivo. Cancer Lett. 2004, 203, 127–137. [Google Scholar] [CrossRef]
- Ji, H.F.; Shen, L. Berberine: A potential multipotent natural product to combat Alzheimer's disease. Molecules 2011, 16, 6732–6740. [Google Scholar] [CrossRef]
- Chung, J.G.; Hsia, T.C.; Kuo, H.M.; Li, Y.C.; Lee, Y.M.; Lin, S.S.; Hung, C.F. Inhibitory actions of luteolin on the growth and arylamine N-acetyltransferase activity in strains of Helicobacter pylori from ulcer patients. Toxicol. In Vitro 2001, 15, 191–198. [Google Scholar] [CrossRef]
- Vani, M.; Meena, A.; Savio, F.G.; Priya, M. Design and evaluation of gastro retentive floating beads of ranitidine hydrochloride. Int. J. Pharm. Biomed. Sci. 2010, 1, 1–4. [Google Scholar]
- Stops, F.; Fell, J.T; Collett, J.H.; Martini, L.G.; Sharma, H.L.; Smith, A.M. The use of citric acid to prolong the in vivo gastro-retention of a floating dosage form in the fasted state. Int. J. Pharm. 2006, 308, 8–13. [Google Scholar] [CrossRef]
- Klokk, T.I.; Melvik, J.E. Controlling the size of alginate gel beads by use of a high electrostatic potential. J. Microencapsul. 2002, 19, 415–424. [Google Scholar] [CrossRef]
- Patel, Y.; Sher, P.; Pawar, A. The effect of drug concentration and curing time on processing and properties of calcium alginate beads containing metronidazole by response surface methodology. AAPS PharmSciTech 2006, 7, E24–E30. [Google Scholar] [CrossRef]
- Wei, Z.; Hongtao, S.; Qian, Z. Preparation of gastric floating sustained-release capsules of Tripterygium wilfordii by using multiparticulate time-controlled release technology. Zhongguo Zhong Yao Za Zhi 2009, 34, 2867–2871. [Google Scholar]
- Raval, J.A.; Patel, J.K.; Li, N.H.; Patel, M.M. Ranitidine hydrochloride floating matrix tablets based on low density powder: Effects of formulation and processing parameters on drug release. Asian J. Pharm. Sci. 2007, 2, 146–158. [Google Scholar]
- Singhvi, G.; Singh, M. Review: In-vitro drug release characterization models. Int. J. Pharmaceut. Sci. Res. 2011, 77–84. [Google Scholar]
- Sample Availability: Contact the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Z.-H.; Sun, Y.-S.; Pang, H.; Munyendo, W.L.L.; Lv, H.-X.; Zhu, S.-L. Preparation and Evaluation of Berberine Alginate Beads for Stomach-Specific Delivery. Molecules 2011, 16, 10347-10356. https://doi.org/10.3390/molecules161210347
Zhang Z-H, Sun Y-S, Pang H, Munyendo WLL, Lv H-X, Zhu S-L. Preparation and Evaluation of Berberine Alginate Beads for Stomach-Specific Delivery. Molecules. 2011; 16(12):10347-10356. https://doi.org/10.3390/molecules161210347
Chicago/Turabian StyleZhang, Zhen-Hai, Yong-Shun Sun, Hui Pang, Were L.L. Munyendo, Hui-Xia Lv, and Sheng-Liang Zhu. 2011. "Preparation and Evaluation of Berberine Alginate Beads for Stomach-Specific Delivery" Molecules 16, no. 12: 10347-10356. https://doi.org/10.3390/molecules161210347
APA StyleZhang, Z.-H., Sun, Y.-S., Pang, H., Munyendo, W. L. L., Lv, H.-X., & Zhu, S.-L. (2011). Preparation and Evaluation of Berberine Alginate Beads for Stomach-Specific Delivery. Molecules, 16(12), 10347-10356. https://doi.org/10.3390/molecules161210347





