Synthesis, Characterization, Anti-Inflammatory and in Vitro Antimicrobial Activity of Some Novel Alkyl/Aryl Substituted Tertiary Alcohols
Abstract
:1. Introduction
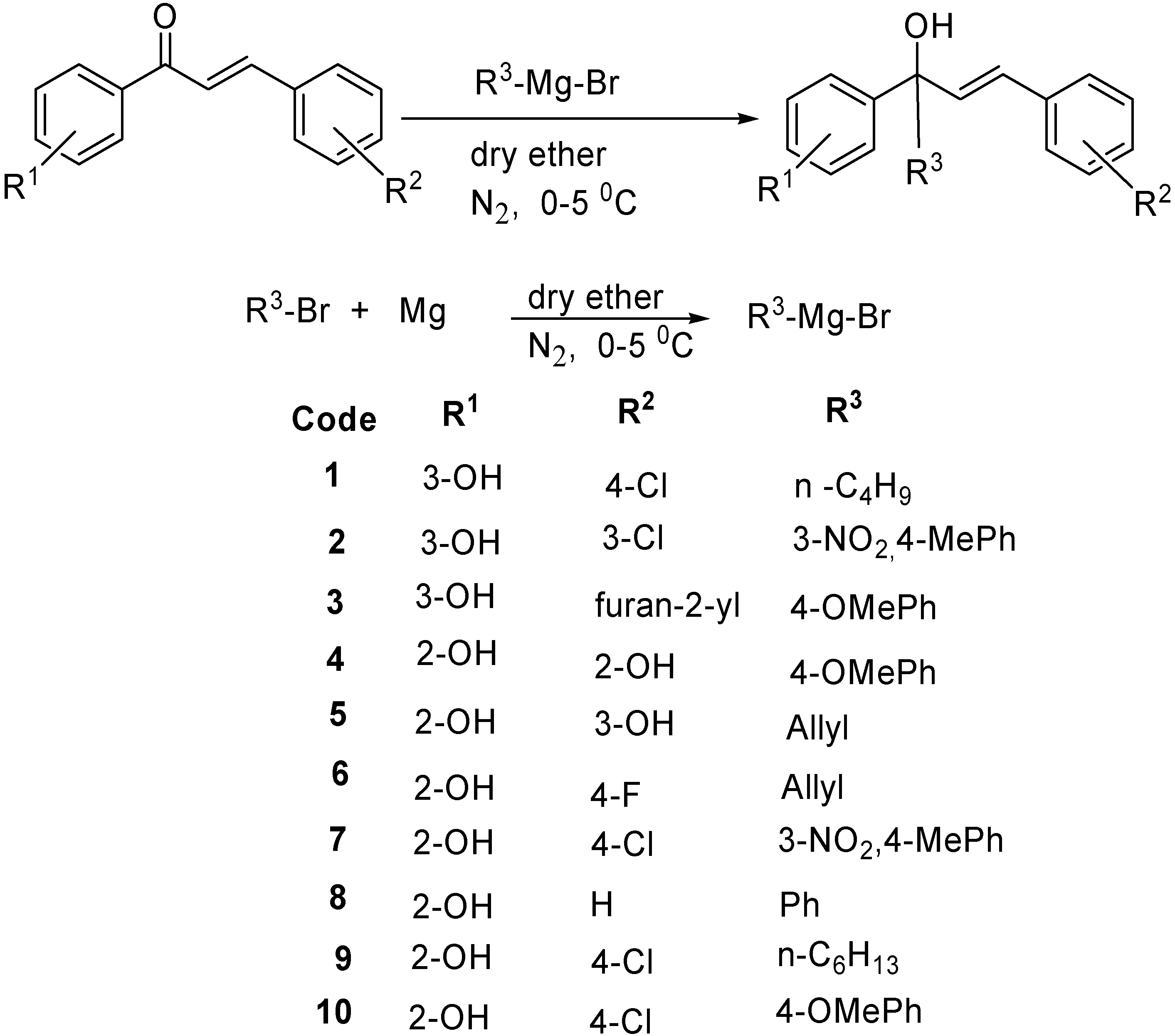
2. Results and Discussion
2.1. Antiinflammatory Activity
| Compounds | Edema volume ± S.E after 3 h | % Inhibition of inflammation a after 3 h | Potency b |
|---|---|---|---|
| 1 | 1.080 ± 0.030 * | 49.76 | 0.68 |
| 2 | 1.133 ± 0.049 * | 47.30 | 0.64 |
| 3 | 1.000 ± 0.025 * | 53.48 | 0.73 |
| 4 | 0.966 ± 0.021 * | 55.06 | 0.75 |
| 5 | 1.883 ± 0.074 | 12.41 | 0.17 |
| 6 | 1.833 ± 0.060 | 14.74 | 0.20 |
| 7 | 0.800 ± 0.036 * | 62.79 | 0.86 |
| 8 | 0.900 ± 0.051 * | 58.13 | 0.79 |
| 9 | 1.000 ± 0.025 * | 53.48 | 0.73 |
| 10 | 1.067 ± 0.033 * | 49.76 | 0.68 |
| Ibuprofen | 0.583 ± 0.060 * | 72.88 | 1 |
| Control | 2.150 ± 0.056 | - | - |
2.2. Antibacterial Activity
| Codes | P.m. | B.s. | E.c. | S a. | P. p. | P. a. | S. t. | M.l. | S. f. | K.p. |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 14 | 04 | 10 | 10 | 08 | 10 | 07 | 04 | 05 | 09 |
| 2 | 10 | - | 12 | 09 | 10 | 13 | 11 | 08 | 11 | 12 |
| 3 | 17 | 08 | 11 | - | 13 | 11 | 13 | 10 | 09 | 14 |
| 4 | 16 | 05 | 10 | 15 | 15 | - | 10 | - | 11 | 10 |
| 5 | 14 | - | 14 | 07 | 11 | 14 | 14 | 09 | 14 | 15 |
| 6 | 10 | - | 11 | 08 | 10 | 15 | 10 | 10 | 10 | 11 |
| 7 | 24 | 06 | 13 | 10 | 24 | 22 | 16 | 16 | 23 | 15 |
| 8 | 21 | 03 | 12 | 09 | 21 | 24 | 19 | - | 21 | 17 |
| 9 | 16 | 10 | 14 | 12 | 16 | 15 | 16 | 14 | 16 | 17 |
| 10 | 14 | 08 | 12 | - | 14 | 12 | 14 | 08 | 14 | 16 |
| Standard | 30 | 20 | 30 | 25 | 30 | 28 | 30 | 25 | 30 | 30 |
2.3. Antifungal Activity
| Codes | Aspergillus flavus | Aspergillus niger | Aspergillus pterus | ||
|---|---|---|---|---|---|
| 1 | 38 | 26 | 41 | ||
| 2 | 11 | 05 | 13 | ||
| 3 | 09 | - | 10 | ||
| 4 | 15 | 06 | 15 | ||
| 5 | 19 | 09 | 11 | ||
| 6 | 31 | 20 | 33 | ||
| 7 | 18 | - | 15 | ||
| 8 | 16 | 11 | 17 | ||
| 9 | 17 | - | 14 | ||
| 10 | 29 | 18 | 30 | ||
| Standard | 37 | 23 | 36 |
3. Experimental
3.1. Synthesis of Tertiary Alcohols 1-10
3.1.1. General Procedure
4. Conclusions
Acknowledgments
References and Notes
- Hong-Xi, X.; Song, F.L. Activity of plant flavonoids against antibiotic-resistant bacteria. Phytother. Res. 2001, 15, 39–43. [Google Scholar] [CrossRef]
- Patrick, G.L. An Introduction to Medicinal Chemistry; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Yasohara, Y.; Miyamoto, K.; Kizaki, N.; Hasegawa, J. A practical chemoenzymatic synthesis of a key intermediate of antifungal agents. Tetrahedron Lett. 2001, 42, 3331–3333. [Google Scholar] [CrossRef]
- Abdel-Rahman, H.M.; Al-Karamany, G.S.; El-Koussi, N.A.; Youssef, A.F.; Kiso, Y. HIV protease inhibitors: Peptidomimetic drugs and future perspectives. Curr. Med. Chem. 2002, 9, 1905–1922. [Google Scholar]
- Rodriguez-Barrios, F. HIV protease inhibition: Limited recent progress and advances in understanding current pitfalls. Curr. Top. Med. Chem. 2004, 4, 991–1007. [Google Scholar] [CrossRef]
- Xiongyu, W.; Per, O.; Jenny, K.E.; Johan, U.; Torsten, U.; Hans, W.; Bertil, S.; Anders, H.; Mars, L. Tow-carbon-clongated HIV-1 protease inhibitors with a tertiary alcohol containing transition-state mimic. J. Med. Chem. 2008, 51, 1053–1057. [Google Scholar] [CrossRef]
- Kotick, M.P.; Leland, D.L.; Polazzi, J.Q.; Schut, R.N. Analgesic narcotic antagonists. 1.8. beta. alkyl-, 8. beta. -acyl-, and 8. beta. (tertiary alcohol)dihydrocodeinones and -dihydromorphinones. J. Med. Chem. 1980, 23, 166–174. [Google Scholar] [CrossRef]
- Fiddler, G.I.; Lumley, P. Preliminary clinical studies with thromboxane synthetase inhibitors and thromboxane receptor blockers. Circulation 1990, 81, 1–69. [Google Scholar] [CrossRef]
- Cross, P.E.; Dickinson, R.P. Thromboxane synthetase inhibitors and antagonists. Annu. Rep. Med. Chem. 1987, 22, 95–105. [Google Scholar]
- Wang, Z.; Deng, Y.; Zang, X. The novel application of tertiary butyl alcohol in the preparation of hydrophobic drug-HP & CD complex. J. Pharm. Pharmacol. 2006, 58, 409–414. [Google Scholar]
- Alberto, M.; Anna, C.C.; Kelly, D.E.; Afranio, K. The first compound of a new class of potent anti-tuberculosis drugs. Chem. Future Microbiol. 2010, 5, 849–858. [Google Scholar] [CrossRef]
- Hans, R.H.; Gut, J.; Rosenthal, P.J.; Chibale, K. Comparison of the antiplasmodial and falcipain-2 inhibitory activity of β-amino alcohol thiolactone-chalcone and isatin-chalcone hybrids. Bioorg. Med. Chem. Lett. 2010, 20, 2234–2237. [Google Scholar]
- Coimbra, E.S.; Almeida, D.; Taveira, A.F.; de Costa, C.F.; de Almeida, A.C.; Reis, E.F.C.; da Silva, A.D. Synthesis and antileishmanial activity of lipidic amino alcohols. Chem. Biol. Drug Des. 2010, 75, 233–235. [Google Scholar] [CrossRef]
- Cordova, I.; Leon, L.G.; Leon, F.; Andres, S.L.; Luis, J.G.; Padron, J.M. Synthesis and antiproliferative activity of novel sugiol β-amino alcohol analogues. Eur. J. Med. Chem. 2006, 41, 1327–1332. [Google Scholar] [CrossRef]
- Costa, R.G.; Curto, M.J.; Furtado, O.R. Novel synthesis of haloaromatic tertiary alcohols using (η6-fluoroarene)tricarbonylchromium(0) complexes. Synth. Commun. 2000, 30, 1115–1137. [Google Scholar] [CrossRef]
- Merger, F.; Nestler, G. Preparation of Tertiary Alcohols. U.S. Patent 4,493,801, 1985. [Google Scholar]
- Özdemirhan, D.; Sezer, S.; Sönmez, Y. Enzyme-catalyzed resolution of aromatic ring fused cyclic tertiary alcohols. Tetrahedron: Asymmetry 2008, 19, 2717–2720. [Google Scholar] [CrossRef]
- Sakhthivel, K.; Notz, W.; Bui, T.; Barbas, C.F. Amino acid catalyzed direct asymmetric aldol reactions: A bioorganic approach to catalytic asymmetric carbon-carbon bond forming reactions. J. Am. Chem. Soc. 2001, 123, 5260–5267. [Google Scholar] [CrossRef]
- Bogevig, A.; Kumaragurubaran, N.; Jorgensen, K.A. Direct catalytic asymmetric aldol reactions of aldehydes. Chem. Commun. 2002, 6, 620–621. [Google Scholar]
- Phrmaceuticals, S.S. Synthesis of pharmaceutical intermediates aiming at construction of optically active tertiary alcohols as a key technology. 2005, 2005, 27–32. [Google Scholar]
- Ansari, F.L.; Nazir, S.; Noureen, H.; Mirza, B. Combinatorial synthesis and antibacterial evaluation of an indexed chalcone library. Chem. Biodivers. 2005, 2, 1656–1664. [Google Scholar] [CrossRef]
- Haugan, J.A. Total synthesis of C31-methyl ketone apocarotenoids 2: The first total synthesis of (3R)-triophaxanthin. Acta. Chem. Scand. 1997, 51, 1096–1103. [Google Scholar] [CrossRef]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenin-induced edema in hind paw of the rat as an assay for antiinflammatory drugs. Proc. Soc. Exp. Biol. Med. 1962, 111, 544–547. [Google Scholar]
- Okeke, M.I.; Iroegbu, C.U.; Eze, E.N.; Okoli, A.S.; Esimone, C.O. Evaluation of extracts of the root of landolphia owerrience for antibacterial activity. J. Ethnopharmacol. 2001, 78, 119–127. [Google Scholar] [CrossRef]
- Shastri, R.V.; Varudkar, J.S. Synthesis and antimicrobial of 3-propen 1,2-benzisoxazole derivatives. Indian J. Chem. Sec. 2009, 48B, 1156–1160. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Baseer, M.; Ansari, F.L.; Ashraf, Z.; SaeedulHaq, R. Synthesis, Characterization, Anti-Inflammatory and in Vitro Antimicrobial Activity of Some Novel Alkyl/Aryl Substituted Tertiary Alcohols. Molecules 2011, 16, 10337-10346. https://doi.org/10.3390/molecules161210337
Baseer M, Ansari FL, Ashraf Z, SaeedulHaq R. Synthesis, Characterization, Anti-Inflammatory and in Vitro Antimicrobial Activity of Some Novel Alkyl/Aryl Substituted Tertiary Alcohols. Molecules. 2011; 16(12):10337-10346. https://doi.org/10.3390/molecules161210337
Chicago/Turabian StyleBaseer, Muhammad, Farzana Latif Ansari, Zaman Ashraf, and Rafiuzzaman SaeedulHaq. 2011. "Synthesis, Characterization, Anti-Inflammatory and in Vitro Antimicrobial Activity of Some Novel Alkyl/Aryl Substituted Tertiary Alcohols" Molecules 16, no. 12: 10337-10346. https://doi.org/10.3390/molecules161210337
APA StyleBaseer, M., Ansari, F. L., Ashraf, Z., & SaeedulHaq, R. (2011). Synthesis, Characterization, Anti-Inflammatory and in Vitro Antimicrobial Activity of Some Novel Alkyl/Aryl Substituted Tertiary Alcohols. Molecules, 16(12), 10337-10346. https://doi.org/10.3390/molecules161210337




