Structure of Salvioccidentalin, a Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia occidentalis
Abstract
:1. Introduction
2. Results and Discussion
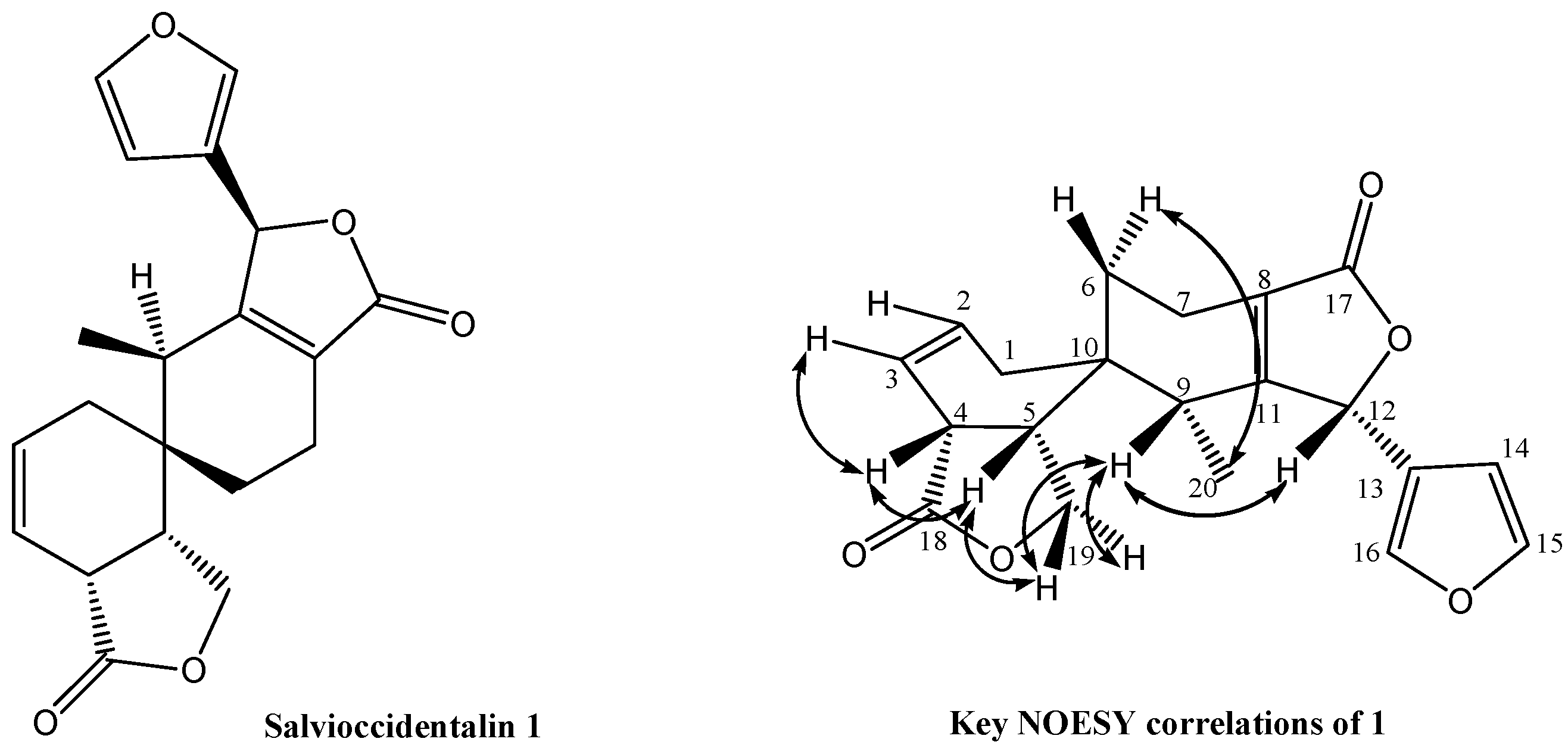
| Position | δH (mult, J in Hz) | COSY (H-H) | NOESY (H-H) | δC (DEPT) | HMBC (H→C) |
|---|---|---|---|---|---|
| 1 Heq | 1.92 (br dd, J = 18.0, 4.0) | H-1ax, H-2 | 2, 3, 5, 6, 9, 10 | ||
| 1 Hax | 2.10 (br dddd, J = 18.0, 2.5, 2.5, 2.5) | H-1eq, H-3, H-4 | H-19A | 31.2 (CH2) | |
| 2 | 5.93 (dddd, J = 10.5, 6.0, 2.5, 2.5) | H-1eq, H-3 | H-1eq, H-3 | 127.8 (CH) | 4, 10 |
| 3 | 5.69 (br d, J = 10.5) | H-1eq, H-2, H-4 | H-2, H-4 | 119.9 (CH) | 1, 4, 5 |
| 4 | 3.14 (m) | H-1eq, H-3, H-5 | H-3, H-5, H-6eq | 40.8 (CH) | 3, 18, 19 |
| 5 | 2.75 (ddd, J = 11.0, 8.5, 8.5) | H-4, H-19A, H-19B | H-4, H-19B | 37.4 (CH) | 1, 3, 4, 19 |
| 6 Hax | 1.60 (Hax, ddd, J = 14.5, 12.0, 6.5) | H-6eq, H-7ax, H-7eq | H-6eq, H-20 | 24.4 (CH2) | 7, 8, 9 |
| 6 Heq | 1.85 (Heq, dd, J = 14.5, 6.5) | H-6ax, H-7ax | H-6ax, H-4 | ||
| 7 Heq | 2.47 (Heq, dd, J = 19.0, 6.5) | H-7ax , H-6ax | H-7ax | 17.5 (CH2) | 6, 8, 10, 11 |
| 7 Hax | 2.25 (Hax, m) | H-7eq, H-6ax, H-6eq | H-7eq | ||
| 8 | 126.2 (C) | ||||
| 9 | 2.01 (q, J = 7.5) | H-20 | H-12, H19A, H19B, H-20 | 35.9 (CH) | 5, 6, 8, 10, 11, 12, 20 |
| 10 | 34.3 (C) | ||||
| 11 | 163.9 (C) | ||||
| 12 | 5.68 (br s) | H-7ax, H-7eq | H-9, H-16 | 77.6 (CH) | 8, 13, 14, 16 |
| 13 | 120.8 (C) | ||||
| 14 | 6.23 (dd, J = 1.5, 0.5) | H-15 | H-15 | 108.3 (CH) | 13, 15, 16 |
| 15 | 7.45 (dd, J = 1.5, 1.5) | H-14, H-16 | H-14 | 144.5 (CH) | 13, 14 |
| 16 | 7.58 (br s) | H-15 | H-12 | 141.3 (CH) | 13, 15 |
| 17 | 172.1 (C) | ||||
| 18 | 175.9 (C) | ||||
| 19A H pro-S | 4.05 (dd, J = 11.0, 8.5) | H-5, H-19B | H-1ax, H-9, H-19B | 67.9 (CH2) | 4, 5, 10, 18 |
| 19B H pro-R | 4.21 (dd, J = 8.5, 8.5) | H-5, H-19A | H-5, H-9, H-19A | ||
| 20 | 0.74 (d, J = 7.5 Hz) | H-9 | H-6ax, H-9 | 14.3 (CH3) | 9, 10, 11 |


3. Experimental
3.1. General
3.2. Extraction and Isolation
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Lawrence, B.M. Chemical Components of Labiatae Oils and Their Exploitation. In Advances in Labiate Sciences, 1st; Harley, R.M., Reynolds, T., Eds.; Royal Botanic Gardens: Kew, UK, 1992; pp. 399–436. [Google Scholar]
- Kabouche, A.; Kabouche, Z. Bioactive Diterpenoids of Salvia Species. Stud. Nat. Prod. Chem. 2008, 35, 753–833. [Google Scholar] [CrossRef]
- Kamatou, G.P.P.; Makunga, N.P.; Ramogola, W.P.N.; Viljoen, A.M. South African Salvia Species: A Review of Biological Activities and Phytochemistry. J. Ethnopharmacol. 2008, 119, 664–672. [Google Scholar] [CrossRef]
- Piozzi, F.; Bruno, M.; Rosselli, S.; Maggio, A. Structure and Biological Activity of the Furan-Diterpenoids from the Genera Leonotis and Leonurus. Heterocycles 2007, 74, 31–52. [Google Scholar] [CrossRef]
- Rodríguez-Hahn, L.; Cárdenas, J. Comparative Chemotaxonomy in Labiatae. Curr. Top. Phytochem. 1999, 2, 91–102. [Google Scholar]
- Rodríguez-Hahn, L.; Esquivel, B.; Cárdenas, J. Neo-clerodane Diterpenoids from American Salvia Species. Recent Adv. Phytochem. 1995, 29, 311–332. [Google Scholar]
- Rodríguez-Hahn, L.; Esquivel, B.; Cárdenas, J. Clerodane Diterpenes in Labiatae. Prog. Chem. Org. Nat. Prod. 1994, 63, 107–196. [Google Scholar]
- Esquivel, B. Rearranged Clerodane and Abietane Derived Diterpenoids from American Salvia Species. Nat. Prod. Commun. 2008, 3, 989–1002. [Google Scholar]
- Epling, C. A Revision of Salvia Subgenus Calosphace. In Repertorium Specierum Novarum regni Vegetabilis; Verlag des Repertoriums: Berlin, Germany, 1939; pp. 1–15. [Google Scholar]
- Aoyagi, Y.; Yamazaki, A.; Nakatsugawa, C.; Fukaya, H.; Takeya, K.; Kawauchi, S.; Izumi, H. A Novel Diterpenoid with an Unprecedented Rearranged Neoclerodane Skeleton from Salvia leucantha Cav. Org. Lett. 2008, 10, 4429–4432. [Google Scholar] [CrossRef]
- Narukawa, Y.; Hatano, K.; Takeda, T. A Novel Diterpenoid with a Rearranged Neoclerodane Skeleton from Salvia leucantha CAV. J. Nat. Med. 2006, 60, 206–209. [Google Scholar] [CrossRef]
- Esquivel, B.; Cárdenas, J.; Toscano, A.; Soriano-García, M.; Rodríguez-Hahn, L. Structure of Salvigenolide, a Novel Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia fulgens. Tetrahedron 1985, 41, 3213–3217. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compound 1 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jaime-Vasconcelos, M.Á.; Frontana-Uribe, B.A.; Morales-Serna, J.A.; Salmón, M.; Cárdenas, J. Structure of Salvioccidentalin, a Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia occidentalis. Molecules 2011, 16, 9109-9115. https://doi.org/10.3390/molecules16119109
Jaime-Vasconcelos MÁ, Frontana-Uribe BA, Morales-Serna JA, Salmón M, Cárdenas J. Structure of Salvioccidentalin, a Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia occidentalis. Molecules. 2011; 16(11):9109-9115. https://doi.org/10.3390/molecules16119109
Chicago/Turabian StyleJaime-Vasconcelos, Miguel Ángel, Bernardo Antonio Frontana-Uribe, José Antonio Morales-Serna, Manuel Salmón, and Jorge Cárdenas. 2011. "Structure of Salvioccidentalin, a Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia occidentalis" Molecules 16, no. 11: 9109-9115. https://doi.org/10.3390/molecules16119109
APA StyleJaime-Vasconcelos, M. Á., Frontana-Uribe, B. A., Morales-Serna, J. A., Salmón, M., & Cárdenas, J. (2011). Structure of Salvioccidentalin, a Diterpenoid with a Rearranged neo-Clerodane Skeleton from Salvia occidentalis. Molecules, 16(11), 9109-9115. https://doi.org/10.3390/molecules16119109





