Hepatoprotective Activity of Cichorium endivia L. Extract and Its Chemical Constituents
Abstract
:1. Introduction
2. Results and Discussion
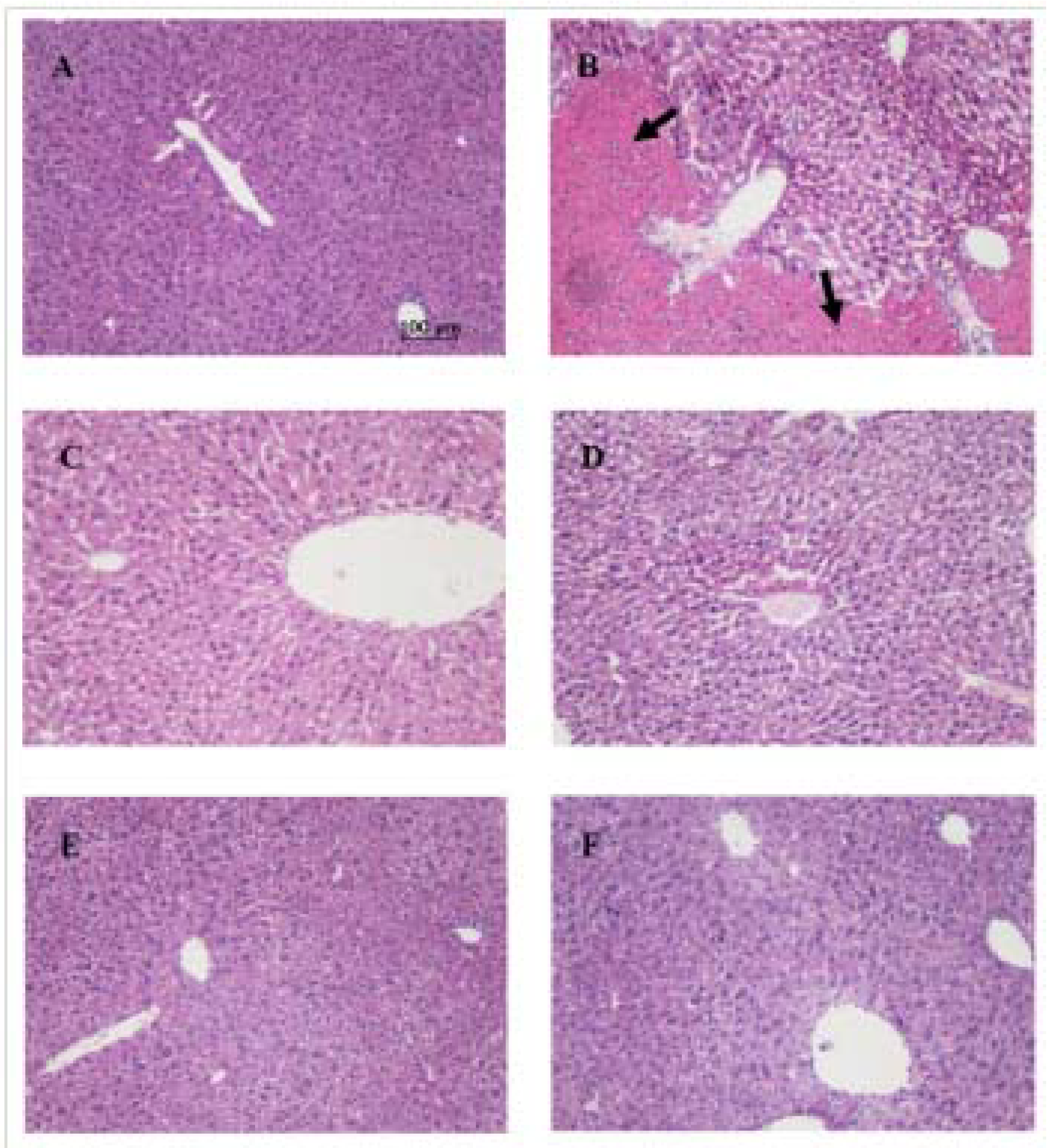
2.1. In Vivo Hepatoprotective Activity of CEE
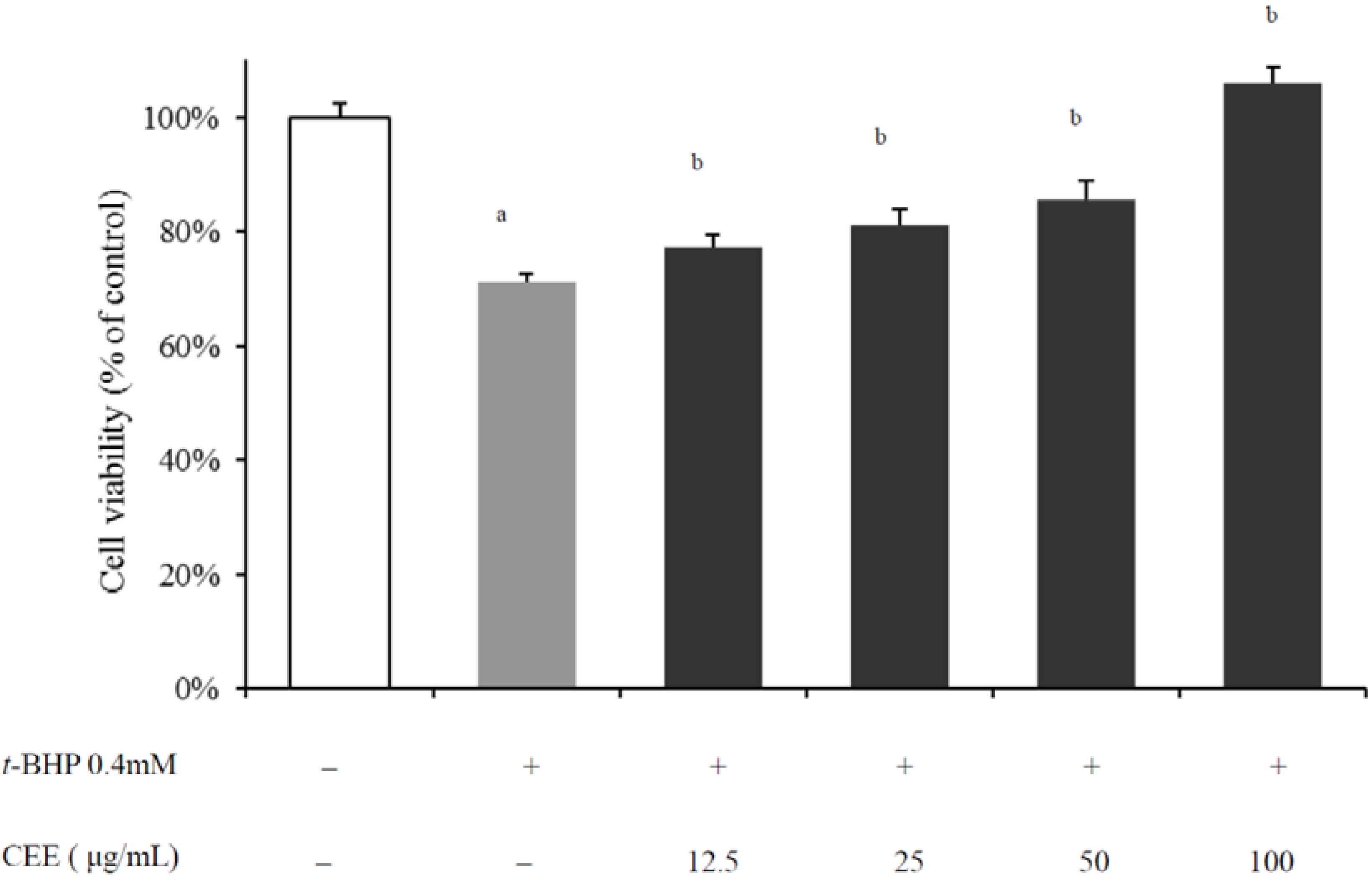
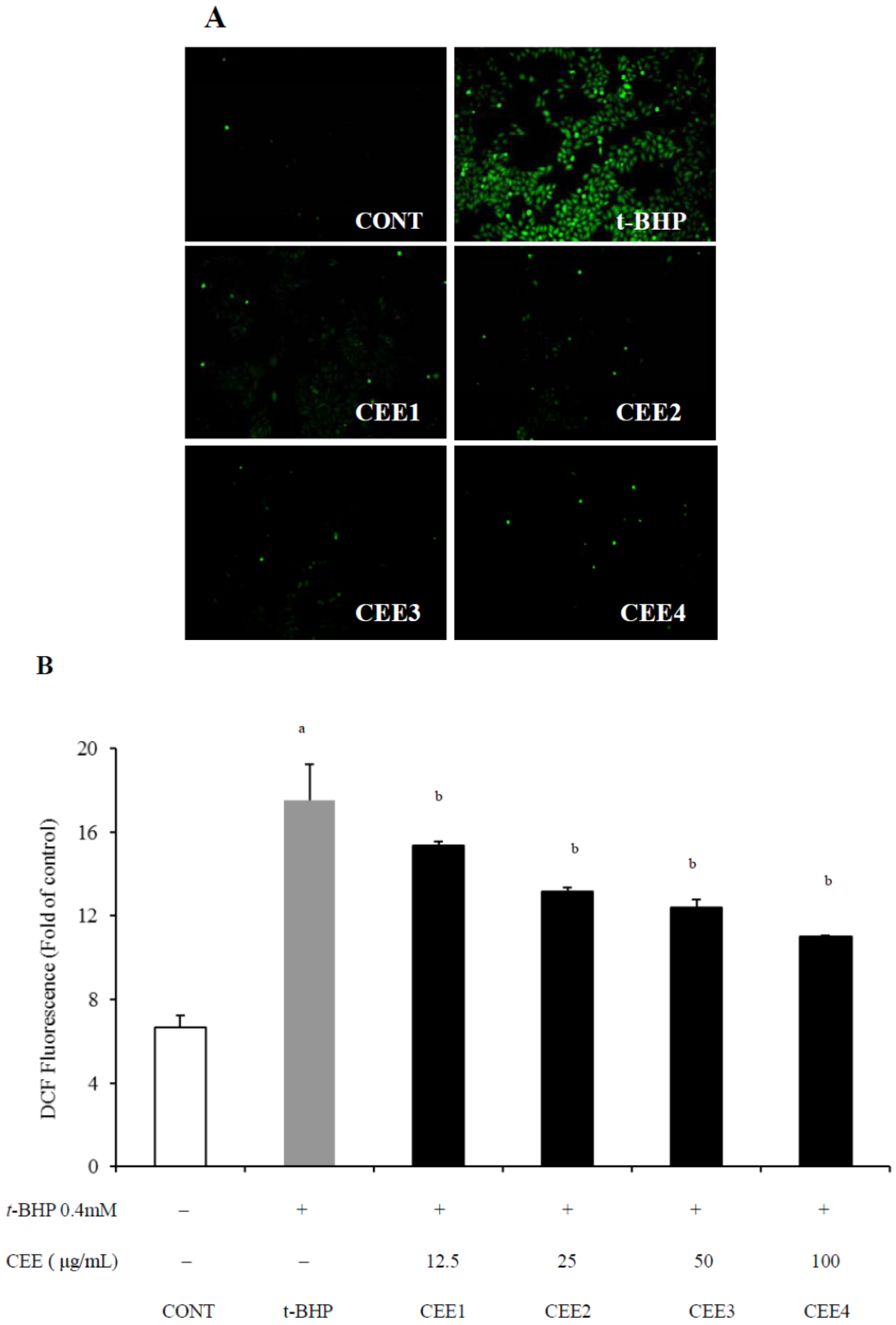
2.2. In Vivo Hepatoprotective Activity of CEE
| Group | Serum ALT(Karmen unit) | Serum AST(Karmen unit) | MDA(μmol/g protein) | SOD(U/mg protein) | GSH(μmol/g protein) | GST(U/mg protein) |
|---|---|---|---|---|---|---|
| Vehicle control | 11.88 ± 4.31 | 24.75 ± 4.56 | 0.81 ± 0.20 | 290.20 ± 10.88 | 8.25 ± 1.06 | 32.90 ± 1.36 |
| t-BHP treatment | 198.70 ± 30.72 b | 118.03 ± 11.58 b | 2.28 ± 0.29 b | 222.30 ± 17.41 b | 2.46 ± 0.32 b | 27.81 ± 2.78 a |
| 150 mg/kg DDB | 33.16 ± 14.15 d | 41.13 ± 14.45 d | 1.07 ± 0.27 d | 262.69 ± 15.36 d | 6.10 ± 0.39 d | 36.32 ± 4.14 d |
| 200 mg/kg CEE | 144.28 ± 17.17 c | 84.52 ± 10.94 d | 1.52 ± 0.16 d | 246.18 ± 5.86 c | 3.08 ± 0.57 c | 32.42 ± 5.03 |
| 400 mg/kg CEE | 75.85 ± 14.83 d | 51.67 ± 15.22 d | 1.22 ± 0.11 d | 253.09 ± 6.50 c | 4.05 ± 0.24 d | 35.64 ± 2.87 d |
| 800 mg/kg CEE | 33.10 ± 9.71 d | 38.76 ± 9.50 d | 1.03 ± 0.07 d | 271.47 ± 13.38 d | 4.79 ± 0.31 d | 36.14 ± 1.81 d |
2.3. Acute Oral Toxicity of CEE in Mice
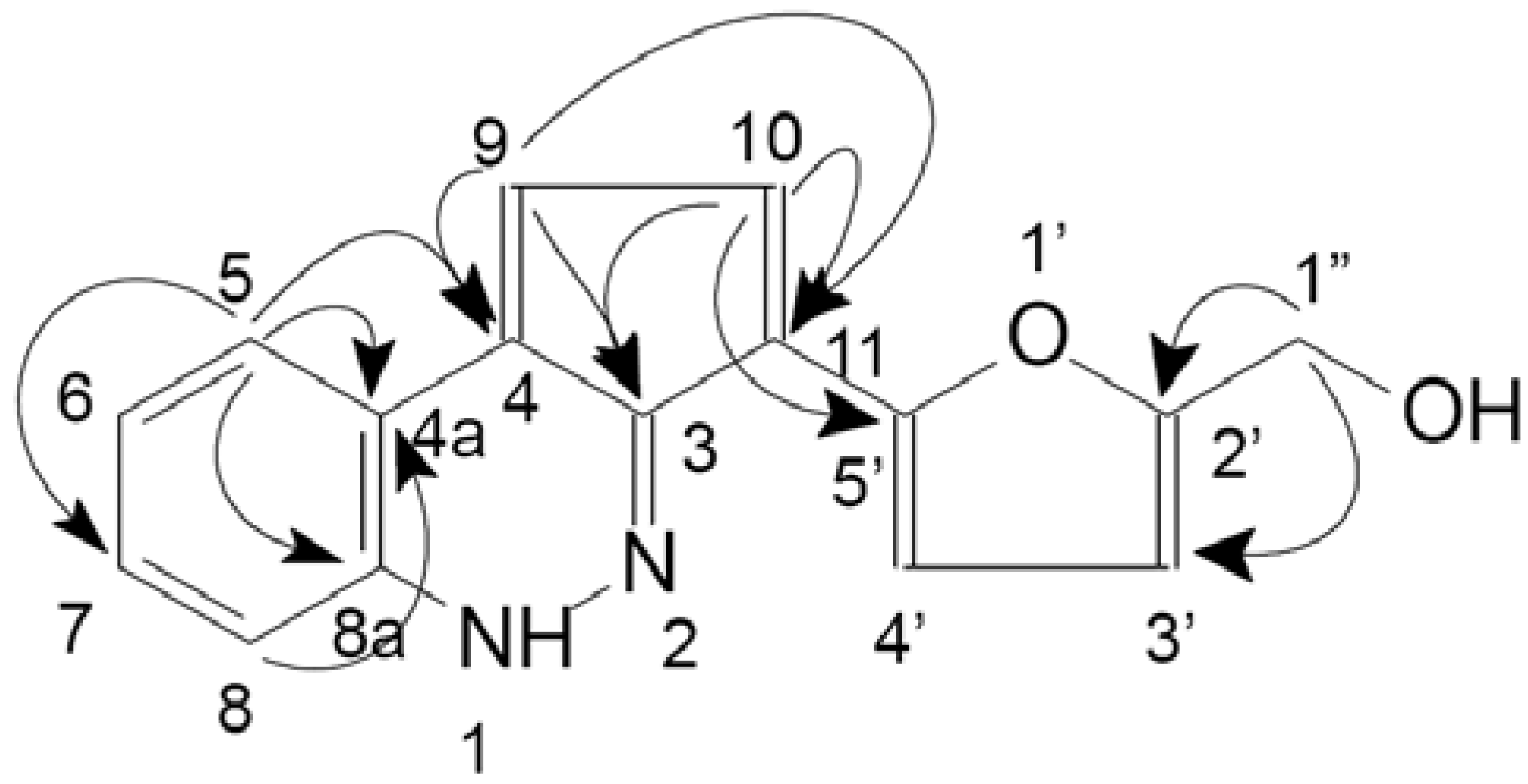
2.4. Structural Determination of Major Compounds from CEE
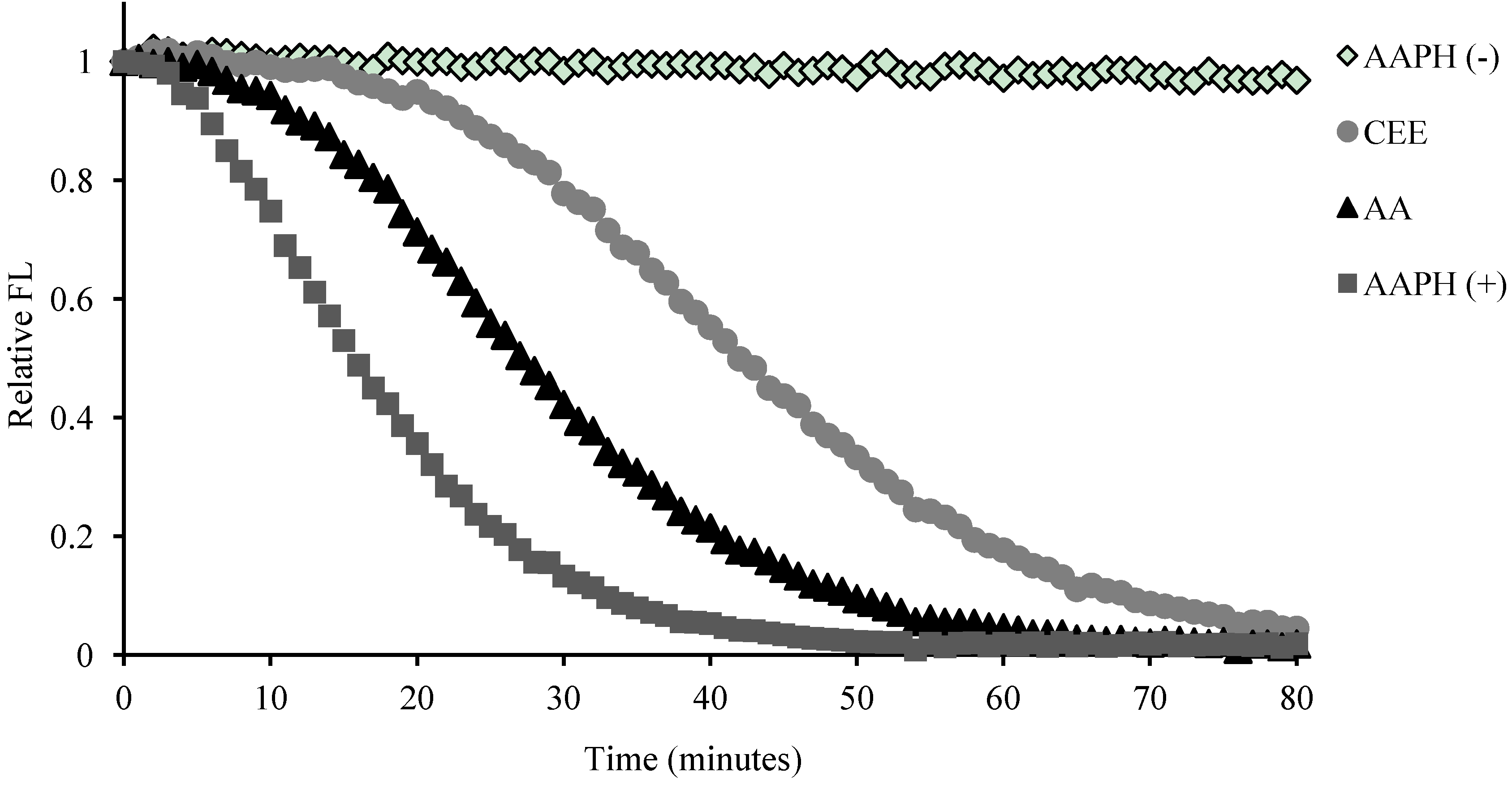
2.5. Antioxidant Activity of the CEE and Its Compounds
| Compound | 1 | 2 | 3 | 4 | 5 | AA |
|---|---|---|---|---|---|---|
| ORAC value (μmol TE) | 0.85 ± 0.01 | a | 10.20 ± 0.13 | 15.30 ± 0.19 | 1.12 ± 0.03 | 0.62 ± 0.01 |
3. Experimental
3.1. General
3.2. CEE Preparation
3.3. Cell Culture
3.4. Cell Viability Assay
3.5. Measurement of Intracellular ROS Level
3.6. Animals
3.7. Acute Liver Injury Induced by t-BHP in Mice
3.7.1. Hepatotoxicity Assessment
3.7.2. Lipid Peroxidation Assay
3.7.3. Antioxidant Enzyme Activity Assay
3.7.4. Pathological Histology
3.8. Acute oral Toxicity Test
3.9. Oxygen Radical Absorbance Capacity (ORAC) Assay
3.10. Statistical Analysis
3.11. Isolation and Identification of CEE
3.12. Characterization of 2-furanmethanol-(5'→11)-1,3-cyclopentadiene-[5,4-c]-1H-cinnoline (1)
4. Conclusions
Conflict of Interest
Acknowledgements
References and Notes
- Amat, N.; Upur, H.; Blazekovic, B. In vivo hepatoprotective activity of the aqueous extract of Artemisia absinthium L. against chemically and immunologically induced liver injuries in mice. J. Ethnopharmacol. 2010, 131, 478–484. [Google Scholar] [CrossRef]
- Jaeschke, H. Reactive oxygen and mechanisms of inflammatory liver injury: Present concepts. J. Gastroenterol. Hepatol. 2011, 26, 173–179. [Google Scholar] [CrossRef]
- Kaplowitz, N. Biochemical and cellular mechanisms of toxic liver injury. Semin. Liver. Dis. 2002, 22, 137–144. [Google Scholar] [CrossRef]
- Gowri Shankar, N.L.; Manavalan, R.; Venkappayya, D.; David Raj, C. Hepatoprotective and antioxidant effects of Commiphora berryi (Arn) Engl bark extract against CCl4-induced oxidative damage in rats. Food Chem. Toxicol. 2008, 46, 3182–3185. [Google Scholar] [CrossRef]
- Huang, B.; Ban, X.; He, J.; Tong, J.; Tian, J.; Wang, Y. Hepatoprotective and antioxidant activity of ethanolic extracts of edible lotus (Nelumbo nucifera Gaertn.) leaves. Food Chem. 2010, 120, 873–878. [Google Scholar] [CrossRef]
- Sunitha, S.; Nagaraj, M.; Varalakshmi, P. Hepatoprotective effect of lupeol and lupeol linoleate on tissue antioxidant defence system in cadmium-induced hepatotoxicity in rats. Fitoterapia 2001, 72, 516–523. [Google Scholar] [CrossRef]
- Llorach, R.; Martínez-Sánchez, A.; Tomás-Barberán, F.A.; Gil, M.I.; Ferreres, F. Characterisation of polyphenols and antioxidant properties of five lettuce varieties and escarole. Food Chem. 2008, 108, 1028–1038. [Google Scholar] [CrossRef]
- Chinese Phamacopoeia Commision. Chinese Pharmacopoeia; Chemical Industry Press: Beijing, China, 2005; p. 217.
- Ahmed, B.; Al-Howiriny, T.A.; Siddiqui, A.B. Antihepatotoxic activity of seeds of Cichorium intybus. J. Ethnopharmacol. 2003, 87, 237–240. [Google Scholar] [CrossRef]
- Zafar, R.; Mujahid Ali, S. Anti-hepatotoxic effects of root and root callus extracts of Cichorium intybus L. J. Ethnopharmacol. 1998, 63, 227–231. [Google Scholar] [CrossRef]
- Upur, H.; Amat, N.; Blazekovic, B.; Talip, A. Protective effect of Cichorium glandulosum root extract on carbon tetrachloride-induced and galactosamine-induced hepatotoxicity in mice. Food Chem. Toxicol. 2009, 47, 2022–2030. [Google Scholar] [CrossRef]
- Papetti, A.; Daglia, M.; Gazzani, G. Anti- and pro-oxidant water soluble activity of Cichorium genus vegetables and effect of thermal treatment. J. Agric. Food Chem. 2002, 50, 4696–4704. [Google Scholar] [CrossRef]
- Papetti, A.; Daglia, M.; Grisoli, P.; Dacarro, C.; Gregotti, C.; Gazzani, G. Anti-and pro-oxidant activity of Cichorium genus vegetables and effect of thermal treatment in biological systems. Food Chem. 2006, 97, 157–165. [Google Scholar] [CrossRef]
- Seto, M.; Miyase, T.; Umehara, K.; Ueno, A.; Hirano, Y.; Otani, N. Sesquiterpene lactones from Cichorium endivia L. and C. intybus L. and cytotoxic activity. Chem. Pharm. Bull. 1988, 36, 2423–2429. [Google Scholar] [CrossRef]
- Kisiel, W.; Michalska, K. Sesquiterpenoids and phenolics from roots of Cichorium endivia var. crispum. Fitoterapia 2006, 77, 354–357. [Google Scholar] [CrossRef]
- Papetti, A.; Daglia, M.; Aceti, C.; Sordelli, B.; Spini, V.; Carazzone, C.; Gazzani, G. Hydroxycinnamic acid derivatives occurring in Cichorium endivia vegetables. J. Pharm. Biomed. Anal. 2008, 48, 472–476. [Google Scholar] [CrossRef]
- Chen, C.J.; Qin, H.L.; Deng, A.J.; Wang, A.P. Antioxidant activity of extract from Cichorium endivia L. Food Drug 2011, 13, 93–96. [Google Scholar]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, N.J.; Kim, D.H. Hepatoprotective effect of ginsenoside Rb1 and compound K on tert-butyl hydroperoxide-induced liver injury. Liver Int. 2005, 25, 1069–1073. [Google Scholar] [CrossRef]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, D.H. Hepatoprotective effect of 20(S)-ginsenosides Rg3 and its metabolite 20(S)-ginsenoside Rh2 on tert-butyl hydroperoxide-induced liver injury. Biol. Pharm. Bull. 2005, 28, 1992–1994. [Google Scholar] [CrossRef]
- Liu, C.L.; Wang, J.M.; Chu, C.Y.; Cheng, M.T.; Tseng, T.H. In vivo protective effect of protocatechuic acid on tert-butyl hydroperoxide-induced rat hepatotoxicity. Food Chem. Toxicol. 2002, 40, 635–641. [Google Scholar] [CrossRef]
- Lee, K.J.; Choi, J.H.; Hwang, Y.P.; Chung, Y.C.; Jeong, H.G. Protective effect of caffeic acid phenethyl ester on tert-butyl hydroperoxide-induced oxidative hepatotoxicity and DNA damage. Food Chem. Toxicol. 2008, 46, 2445–2450. [Google Scholar] [CrossRef]
- Noh, J.R.; Gang, G.T.; Kim, Y.H.; Yang, K.J.; Hwang, J.H.; Lee, H.S. Antioxidant effects of the chestnut (Castanea crenata) inner shell extract in t-BHP-treated HepG2 cells, and CCl4- and high-fat diet-treated mice. Food Chem. Toxicol. 2010, 48, 3177–3183. [Google Scholar] [CrossRef]
- Ganie, S.A.; Haq, E.; Masood, A.; Hamid, A.; Zargar, M.A. Antioxidant and protective effect of ethyl acetate extract of podophyllum hexandrum rhizome on carbon tetrachloride induced rat liver injury. Evid. Based. Complement. Alternat. Med. 2011, 2011, 1–12. [Google Scholar]
- Chow, C.K. Nutritional influence on cellular antioxidant defense systems. Am. J. Clin. Nutr. 1979, 32, 1066–1081. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef]
- Davis, W., Jr.; Ronai, Z.; Tew, K.D. Cellular thiols and reactive oxygen species in drug-induced apoptosis. J. Pharmacol. Exp. Ther. 2001, 296, 1–6. [Google Scholar]
- Bandyopadhyay, U.; Das, D.; Banerjee, R.K. Reactive oxygen species: Oxidative damage and pathogenesis. Curr. Sci. 1999, 77, 658–666. [Google Scholar]
- Salvemini, D.; Riley, D.P.; Cuzzocrea, S. SOD mimetics are coming of age. Nat. Rev. Drug Discov. 2002, 1, 367–374. [Google Scholar] [CrossRef]
- Hayes, J.D.; Pulford, D.J. The glutathione S-transferase supergene family: Regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 445–600. [Google Scholar] [CrossRef]
- Halliwell, B.; Chirico, S. Lipid peroxidation: Its mechanism, measurement, and significance. Am. J. Clin. Nutr. 1993, 57, 715S–724S. [Google Scholar]
- Kennedy, G.L., Jr.; Ferenz, R.L.; Burgess, B.A. Estimation of acute oral toxicity in rats by determination of the approximate lethal dose rather than the LD50. J. Appl. Toxicol. 1986, 6, 145–148. [Google Scholar]
- Umehara, K.; Hattori, I.; Miyase, T.; Ueno, A.; Hara, S.C.K. Studies on the constituents of leaves of Citrus unshiu Marcov. Chem. Pharm. Bull. 1988, 36, 5004–5008. [Google Scholar] [CrossRef]
- Awaad, A.S.; Maitland, D.; Soliman, G. Hepatoprotective activity of Schouwia thebica webb. Bioorg. Med. Chem. Lett. 2006, 16, 4624–4628. [Google Scholar] [CrossRef]
- Wei, F.; Yan, W. Studies on the chemical constitutens of Vicia amoena Fisch. Acta Pharmacol. Sin. 1997, 32, 765–768. [Google Scholar]
- Rao, Y.J.; Reddy, C.R.; Gangadhar, N.; Krupadanam, G.L.D. Phytochemical investigation of the whole plant of Astragalus leucocephalus. Ind. J. Chem. 2009, 48, 1329–1332. [Google Scholar]
- Otsuka, H.; Takeuchi, M.; Inoshiri, S.; Sato, T.; Yamasaki, K. Phenolic compounds from Coix lachryma-jobi var. ma-yuen. Phytochemistry 1989, 28, 883–886. [Google Scholar] [CrossRef]
- Braham, H.; Mighri, Z.; Jannet, H.; Matthew, S.; Abreu, P. Antioxidant phenolic glycosides from Moricandia arvensis. J. Nat. Prod. 2005, 68, 517–522. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Bichem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Hendrickson, H.P.; Sahafayen, M.; Bell, M.A.; Kaufman, A.D.; Hadwiger, M.E.; Lunte, C.E. Relationship of flavonoid oxidation potential and effect on rat hepatic microsomal metabolism of benzene and phenol. J. Pharm. Biomed. Anal. 1994, 12, 335–341. [Google Scholar] [CrossRef]
- Oliveira, E.J.; Watson, D.G. In vitro glucuronidation of kaempferol and quercetin by human UGT-1A9 microsomes. FEBS Lett. 2000, 471, 1–6. [Google Scholar] [CrossRef]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as antioxidants: determination of radical-scavenging efficiencies. Methods Enzymol. 1990, 186, 343. [Google Scholar]
- Van Acker, S.; De Groot, M.; Van den Berg, D.J.; Tromp, M.; Donne-Op den Kelder, G.; Van der Vijgh, W.; Bast, A. A quantum chemical explanation of the antioxidantd activity of flavonoids. Chem. Res. Toxicol. 1996, 9, 1305–1312. [Google Scholar] [CrossRef]
- DuPont, M.S.; Mondin, Z.; Williamson, G.; Price, K. Effect of variety, processing, and storage on the flavonoid glycoside content and composition of lettuce and endive. J. Agric. Food Chem. 2000, 48, 3957–3964. [Google Scholar] [CrossRef]
- Song, E.K.; Kim, J.H.; Kim, J.S.; Cho, H.; Nan, J.X.; Sohn, D.H.; Ko, G.I.; Oh, H.; Kim, Y.C. Hepatoprotective phenolic constituents of Rhodiola sachalinensis on tacrine-induced cytotoxicity in HepG2 cells. Phytother. Res. 2003, 17, 563–565. [Google Scholar] [CrossRef]
- Oha, H.; Kimb, D.H.; Chob, J.H.; Kim, Y.C. Hepatoprotective and free radical scavenging activities of phenolic petrosins and flavonoids isolated from Equisetum arvense. J. Ethnopharmacol. 2004, 95, 421–424. [Google Scholar] [CrossRef]
- Matsushita, S.; Ibuki, F.; Aoki, A. Chemical reactivity of the nucleic acid bases. I. Antioxidative ability of the nucleic acids and their related substances on the oxidation of unsaturated fatty acids. Arch. Biochem. Biophys. 1963, 102, 446–451. [Google Scholar] [CrossRef]
- Bekhit, A. Fused cinnolines: Synthesis and biological activity. Boll. Chim. Farm. 2001, 140, 243. [Google Scholar]
- Lewgowd, W.; Stanczak, A. Cinnoline derivatives with biological activity. Arch. Pharm. 2007, 340, 65–80. [Google Scholar] [CrossRef]
- Cirrincione, G.; Almerico, A.M.; Barraja, P.; Diana, P.; Lauria, A.; Passannanti, A.; Musiu, C.; Pani, A.; Murtas, P.; Minnei, C. Derivatives of the new ring system indolo [1, 2-c] benzo [1, 2, 3] triazine with potent antitumor and antimicrobial activiy. J. Med. Chem. 1999, 42, 2561–2568. [Google Scholar] [CrossRef]
- Nargund, L.V.; Badiger, V.V.; Yarnal, S.M. Synthesis and antimicrobial and anti-inflammatory activities of substituted 2-mercapto-3-(N-aryl)pyrimido[5,4-c]cinnolin-4-(3H)-ones. J. Pharm. Sci. 1992, 81, 365–366. [Google Scholar] [CrossRef]
- Stefaska, B.; Arciemiuk, M.; Bontemps-Gracz, M.M.; Dzieduszycka, M.; Kupiec, A.; Martelli, S.; Borowski, E. Synthesis and biological evaluation of 2, 7-Dihydro-3H-dibenzo [de, h] cinnoline-3, 7-dione derivatives, a novel group of anticancer agents active on a multidrug resistant cell line. Bioorg. Med. Chem. 2003, 11, 561–572. [Google Scholar] [CrossRef]
- Duh, P.D.; Wang, B.S.; Liou, S.J.; Lin, C.J. Cytoprotective effects of pu-erh tea on hepatotoxicity in vitro and in vivo induced by tert-butyl-hydroperoxide. Food Chem. 2010, 119, 580–585. [Google Scholar] [CrossRef]
- Liu, C.L.; Wang, J.M.; Chu, C.Y.; Cheng, M.T.; Tseng, T.H. In vivo protective effect of protocatechuic acid on tert-butyl hydroperoxide-induced rat hepatotoxicity. Food Chem. Toxicol. 2002, 40, 635–641. [Google Scholar] [CrossRef]
- Huang, Y.N.; Zhao, Y.L.; Gao, X.L.; Zhao, Z.F.; Jing, Z.; Zeng, W.C.; Yang, R.; Peng, R.; Tong, T.; Wang, L.F.; et al. Intestinal alpha-glucosidase inhibitory activity and toxicological evaluation of Nymphaea stellata flowers extract. J. Ethnopharmacol. 2010, 131, 306–312. [Google Scholar] [CrossRef]
- Ribnicky, D.M.; Poulev, A.; O’Neal, J.; Wnorowski, G.; Malek, D.E.; Jager, R.; Raskin, I. Toxicological evaluation of the ethanolic extract of Artemisia dracunculus L. for use as a dietary supplement and in functional foods. Food Chem. Toxicol. 2004, 42, 585–598. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.; Prior, R. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the tested compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, C.-J.; Deng, A.-J.; Liu, C.; Shi, R.; Qin, H.-L.; Wang, A.-P. Hepatoprotective Activity of Cichorium endivia L. Extract and Its Chemical Constituents. Molecules 2011, 16, 9049-9066. https://doi.org/10.3390/molecules16119049
Chen C-J, Deng A-J, Liu C, Shi R, Qin H-L, Wang A-P. Hepatoprotective Activity of Cichorium endivia L. Extract and Its Chemical Constituents. Molecules. 2011; 16(11):9049-9066. https://doi.org/10.3390/molecules16119049
Chicago/Turabian StyleChen, Chao-Jie, An-Jun Deng, Chang Liu, Rui Shi, Hai-Lin Qin, and Ai-Ping Wang. 2011. "Hepatoprotective Activity of Cichorium endivia L. Extract and Its Chemical Constituents" Molecules 16, no. 11: 9049-9066. https://doi.org/10.3390/molecules16119049
APA StyleChen, C.-J., Deng, A.-J., Liu, C., Shi, R., Qin, H.-L., & Wang, A.-P. (2011). Hepatoprotective Activity of Cichorium endivia L. Extract and Its Chemical Constituents. Molecules, 16(11), 9049-9066. https://doi.org/10.3390/molecules16119049




