Synthesis, Acidity Constants and Tautomeric Structure of the Diazonium Coupling Products of 2-(Benzylsulfanyl)-7H-purin-6-one in Its Ground and Excited States
Abstract
:1. Introduction
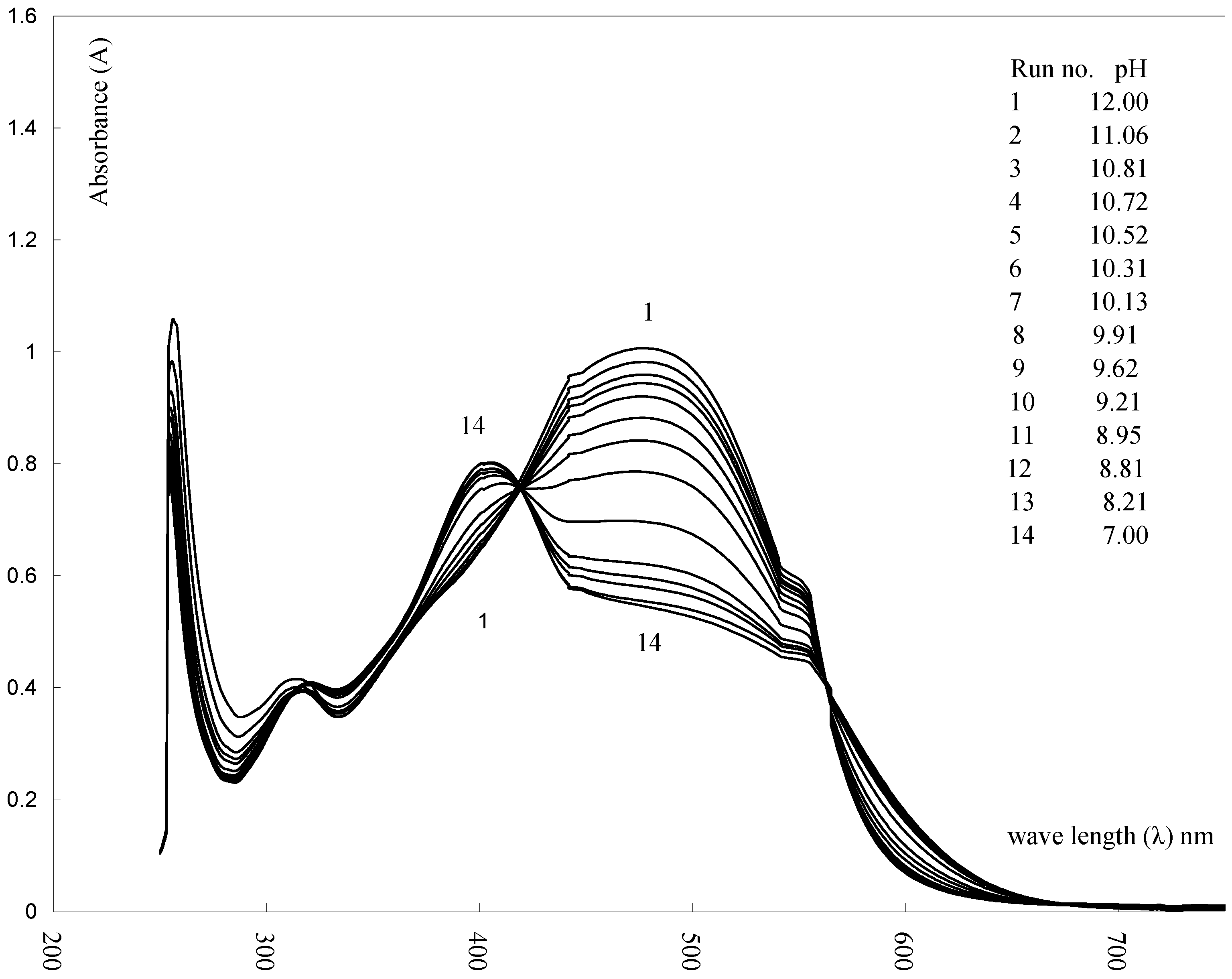
2. Results and Discussion
| Compd. no. | λmax nm (EtOH) | Compd. no. | λmax nm (EtOH) |
|---|---|---|---|
| 6a | 387 (4.17), 315 (4.10) | 6f | 388 (4.38), 288 (4.52) |
| 6b | 390 (4.42), 310 (4.35) | 6g | 396 (4.50), 317 (4.47) |
| 6c | 385 (4.21), 295(4.05) | 6h | 395 (4.65), 318 (4.51) |
| 6d + | 392 (4.26), 300 (4.45) | 6i | 405 (4.58), 310(4.33) |
| 6e | 394 (4.35), 298 (4.40) |
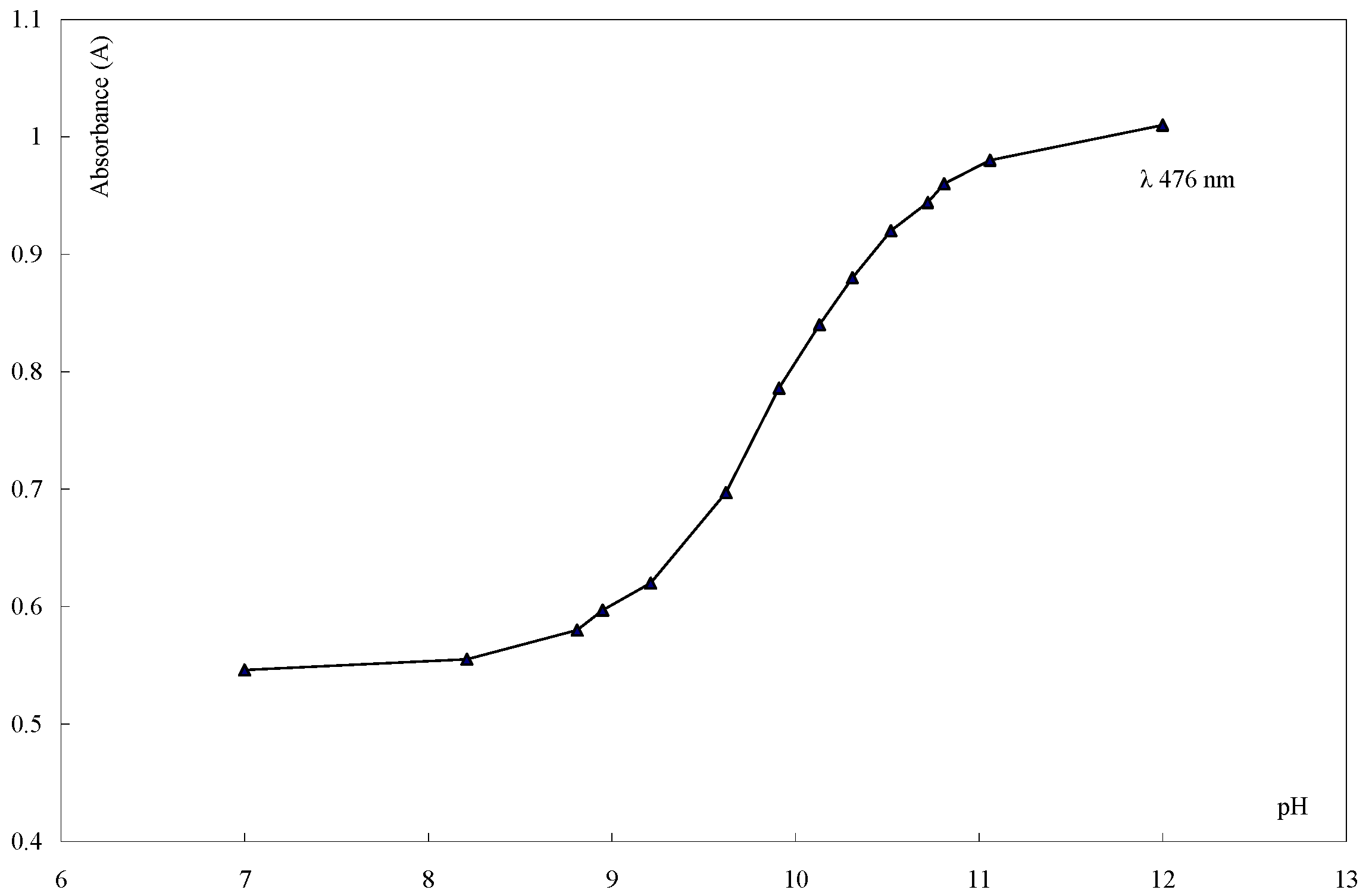
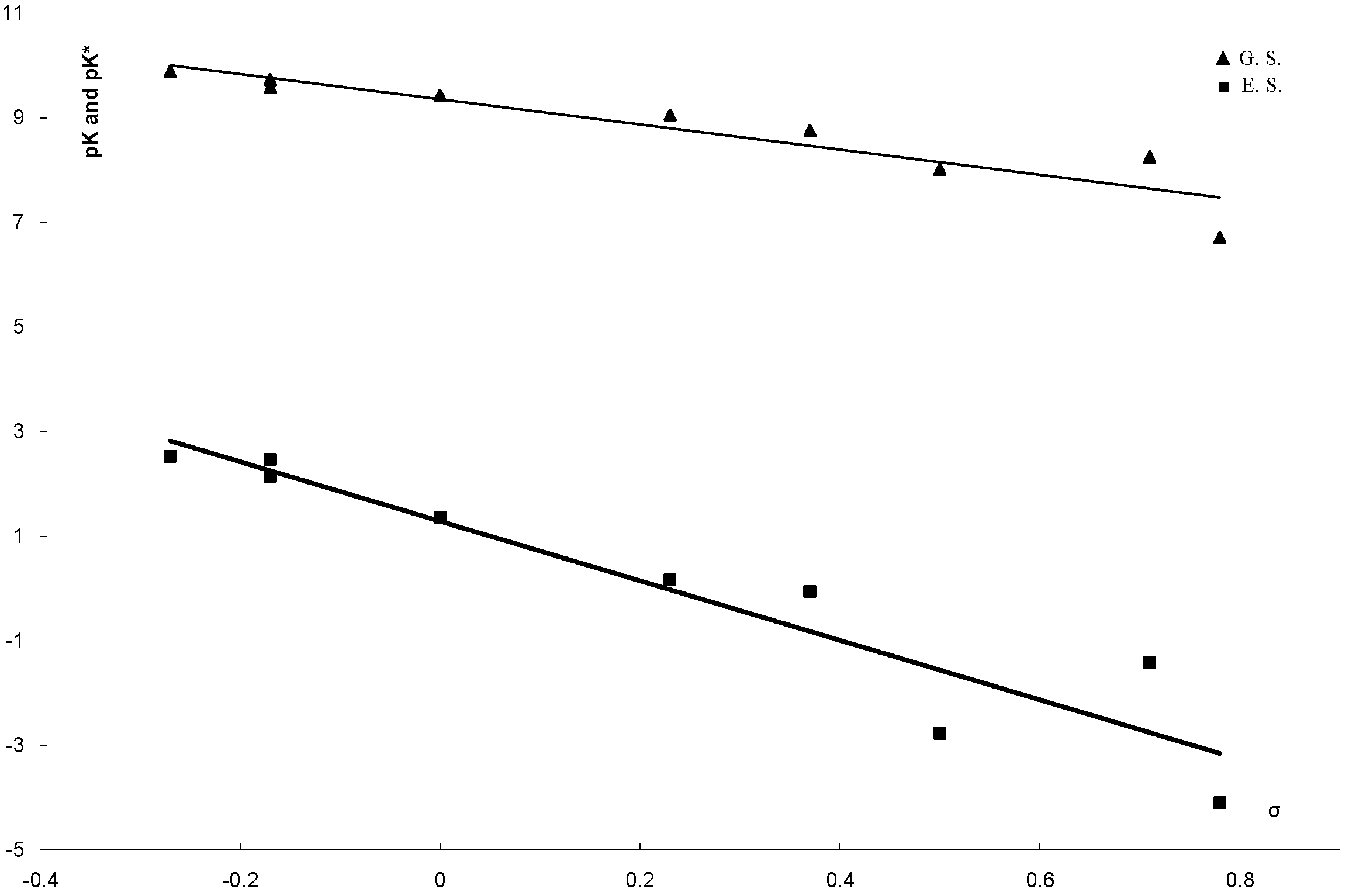
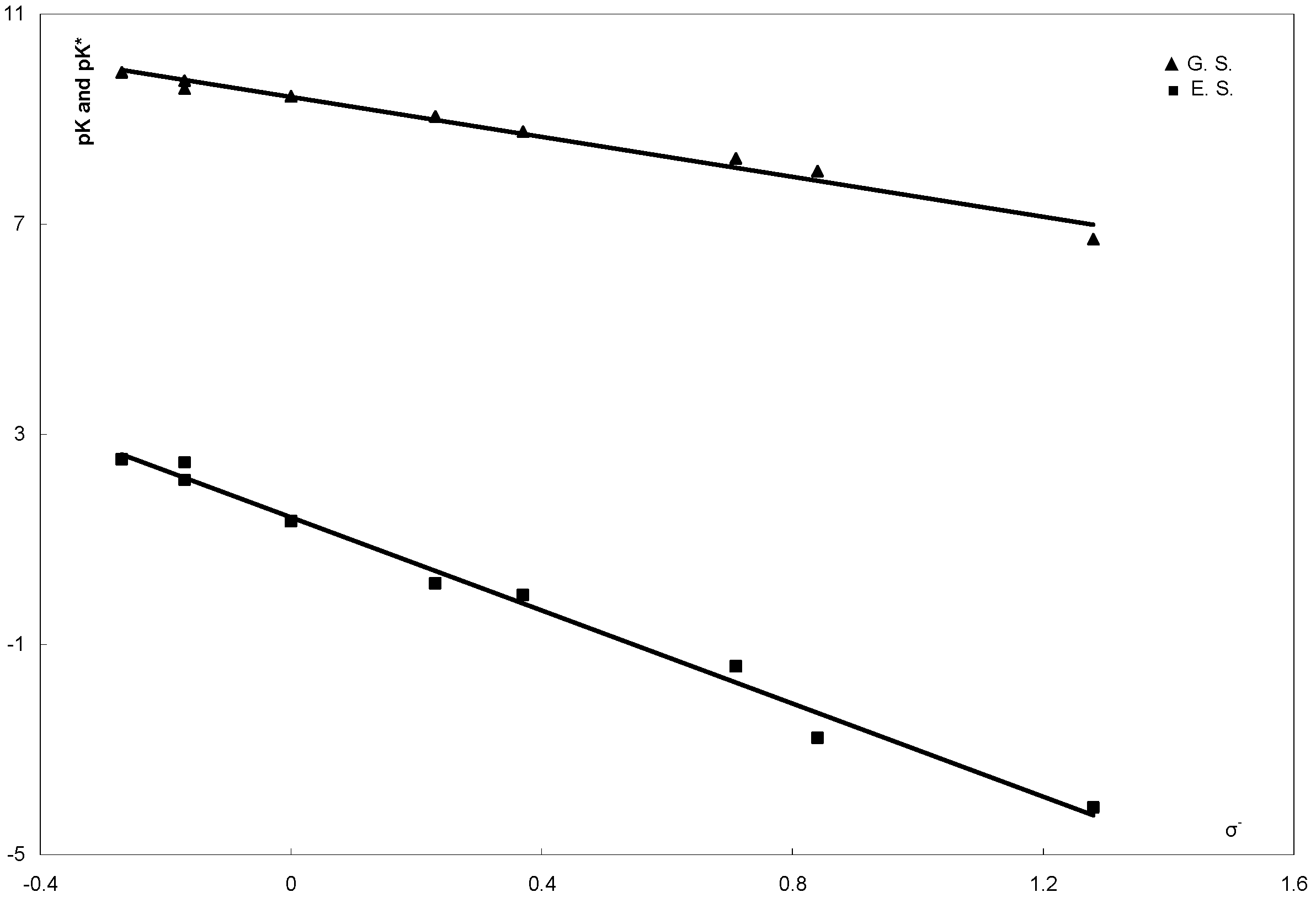
| Compd. No | σ | σ− | pK | λ max (a) | λ max (b) | ∆ν cm−1 | pK* |
|---|---|---|---|---|---|---|---|
| 6a | −0.27 | −0.27 | 9.89 | 405 | 476 | 3683 | 2.52 |
| 6b | −0.17 | −0.17 | 9.73 | 408 | 479 | 3633 | 2.46 |
| 6c | −0.17 | −0.17 | 9.58 | 400 | 470 | 3723 | 2.13 |
| 6d | 0 | 0 | 9.43 | 402 | 480 | 4042 | 1.35 |
| 6e | 0.23 | 0.23 | 9.05 | 397 | 482 | 4442 | 0.17 |
| 6f | 0.37 | 0.37 | 8.76 | 405 | 493 | 4407 | −0.05 |
| 6g | 0.71 | 0.71 | 8.25 | 404 | 502 | 4832 | −1.41 |
| 6h | 0.78 | 1.28 | 6.71 | 415 | 535 | 5405 | −4.10 |
| 6i | 0.5 | 0.84 | 8.01 | 400 | 510 | 5392 | −2.77 |
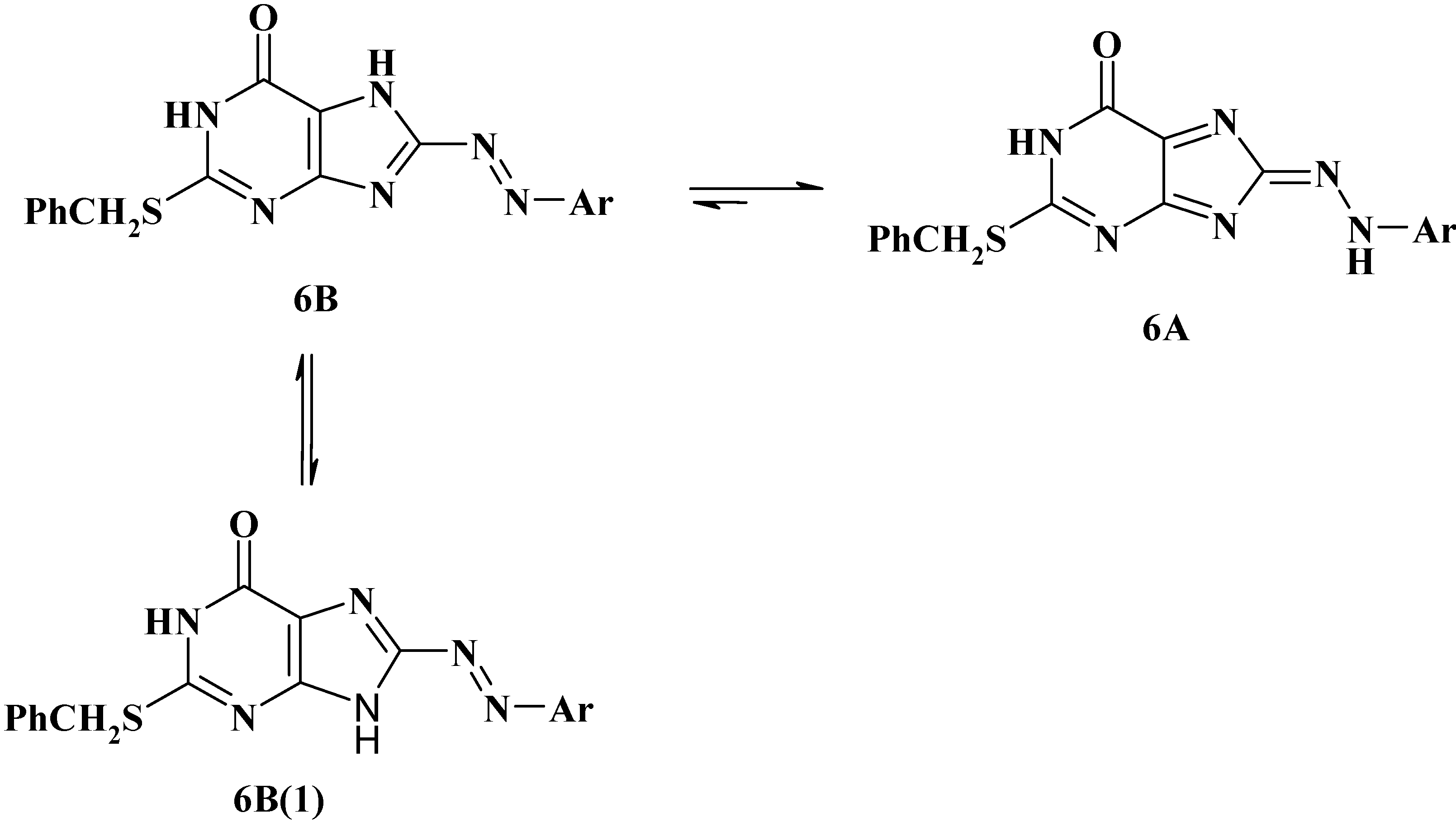
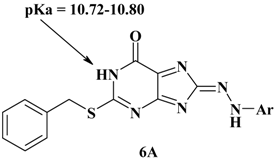
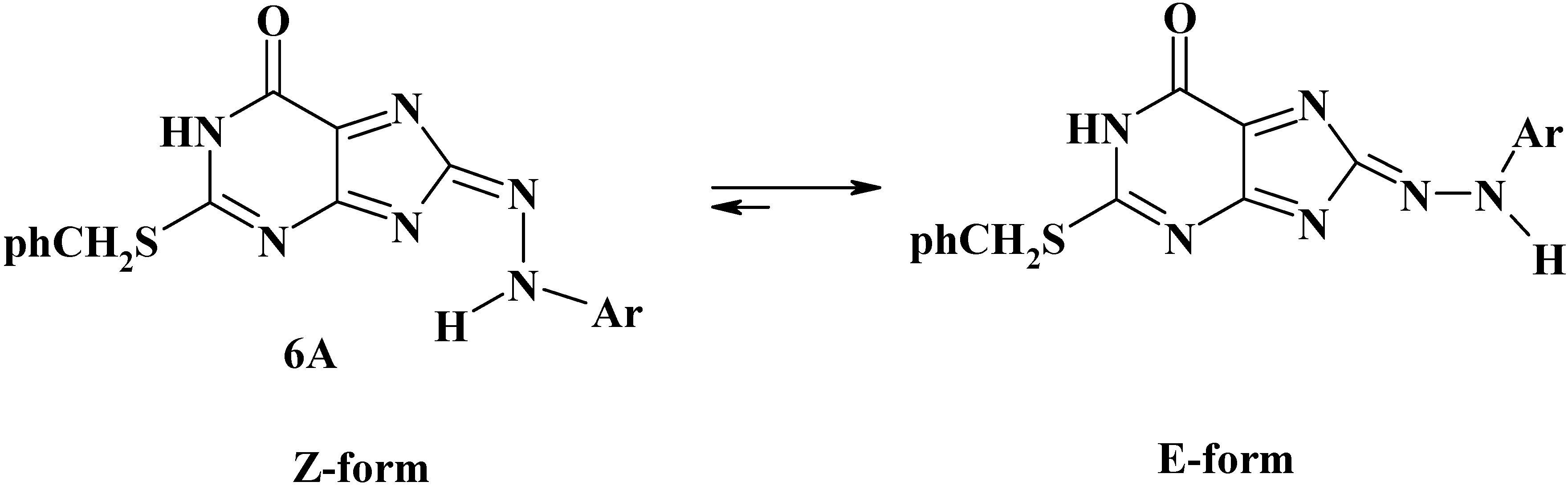
3. Experimental
3.1. General
3.2. General Procedure for Synthesis of 8-Arylhydrazono-2-benzylsulfanyl-1,8-dihydro-purin-6-ones 6a-i
3.3. pK Determination
4. Conclusions
References
- Rosemeyer, H. The chemodiversity of purine as a constituent of natural products. Chem. Biodivers. 2004, 3, 1361–1401. [Google Scholar]
- Legraverend, M.; Grierson, D.S. The purines: Potent and versatile small molecule inhibitors and modulators of key biological targets. Bioorg. Med. Chem. 2006, 14, 3987–4006. [Google Scholar] [CrossRef]
- Schmidt, A.P.; Lara, D.R.; Souza, D.O. Proposal of a guanine-based purinergic system in the mammalian central nervous system. Pharmacol. Ther. 2007, 116, 401–416. [Google Scholar] [CrossRef]
- Legraverend, M. Recent advances in the synthesis of purine derivatives and their precursors. Tetrahedron 2008, 64, 8585–8603. [Google Scholar] [CrossRef]
- Alberts, B.; Bray, D.; Lewis, J.; Raff, M.; Roberts, K.; Watson, J.D. Molecular Biology of the Cell, 3rd ed; Garland Publishing: New York, NY, USA, 1994; p. 736. [Google Scholar]
- Gao, Z.; Duhl, D.M.; Harrison, S.D. Chapter 20. Beyond kinases: Purine-binding enzymes as cancer targets. Ann. Rep. Med. Chem. 2003, 38, 193–202. [Google Scholar] [CrossRef]
- Elion, G.B. The purine path to chemotherapy. Science 1989, 244, 41–47. [Google Scholar]
- Karran, P. Thiopurines, DNA damage, DNA repair and therapy-related cancer. Br. Med. Bull. 2006, 79-80, 153–170. [Google Scholar] [CrossRef]
- Moylan, C.R.; McNelis, B.J.; Nathan, L.C.; Marques, M.A.; Hermstad, E.L.; Brichler, B.A. Challenging the auxiliary donor effect on molecular hyperpolarizability in thiophene-containing nonlinear chromophores: X-ray crystallographic and optical measurements on two new isomeric chromophores. J. Org. Chem. 2004, 69, 8239–8243. [Google Scholar]
- Trofimov, B.A.; Schmidt, E.Y.; Mikhaleva, A.I.; Vasiltsov, A.M.; Zaitsev, A.B.; Smolyanina, A.S.; Senotrusova, E.Y.; Afonin, A.V.; Ushakov, I.A.; Petrushenko, K.B.; Kazheva, I.N.; Dyachenko, O.A.; Smirnov, V.V.; Schmidt, A.F.; Markova, M.V.; Morozova, L.V. 2-Arylazo-1-vinylpyrroles: A novel promising family of reactive dyes. Eur. J. Org. Chem. 2006, 2006, 4021–4033. [Google Scholar]
- Hao, J.; Han, M.J.; Guo, K.; Zao, Y.; Qiu, L.; Shen, Y. A novel NLO azothiophene-based chromophore: Synthesis, characterization, thermal stability and optical nonlinearity. Mater. Lett. 2008, 62, 973–976. [Google Scholar]
- He, M.; Zhou, Y.; Liu, R.; Dai, J.; Cui, Y.; Zhang, T. Novel nonlinearity-transparency-thermal stability trade-off of thiazolylazopyrimidine chromophores for nonlinear optical application. Dyes Pigm. 2009, 80, 6–10. [Google Scholar] [CrossRef]
- Zhao, X. Synthesis, characterization and structure dependence of thermochromism of polythiophene derivatives. J. Mater. Sci. 2005, 40, 3423–3428. [Google Scholar] [CrossRef]
- Zhao, X.; Hu, X.; Gan, L.H. Photoluminescent behavior of poly(3-hexylthiophene) derivatives with a high azobenzene content in the side chains. Polym. Adv. Technol. 2005, 16, 370–377. [Google Scholar] [CrossRef]
- Sharma, G.D.; Choudhary, V.S.; Roy, M.S. Effect of annealing on the optical, electrical, and photovoltaic properties of bulk hetero-junction device based on PPAT:TY blend. Sol. Energ. Mater. Sol. Cell. 2007, 91, 275–284. [Google Scholar] [CrossRef]
- Corrêa, D.S.; de Boni, L.; Gonçalves, V.C.; Balogh, D.T.; Mendonça, C.R. Excited state absorption in conjugated polymers: Photoinduced transparency. Polymer 2007, 48, 5303–5307. [Google Scholar] [CrossRef]
- Dinçalp, H.; Toker, F.; Durucasu, I.; Avcibasi, N.; Icli, S. New thiophene-based azo ligands containing azo methine groups in the main chain for the determination of copper(II) ions. Dyes Pigm. 2007, 75, 11–24. [Google Scholar] [CrossRef]
- Matharu, A.S.; Jeeva, S.; Huddleston, P.R.; Ramanujam, P.S. Synthesis and optical storage properties of a thiophene-based holographic recording medium. J. Mater. Chem. 2007, 17, 4477–4482. [Google Scholar] [CrossRef]
- Towns, A.D. Developments in azo disperse dyes derived from heterocyclic diazo components. Dyes Pigm. 1999, 42, 3–28. [Google Scholar] [CrossRef]
- Gupta, S.; Sivasubramanian, A.; Rodrigues, L.M.; Esteves, A.P.; Hrdina, R.; Oliveira-Compos, A.M.F. Synthesis of dyes derived from 1-aryl-5-amino-4-cyanopyrazoles. Dyes Pigm. 2007, 75, 82–87. [Google Scholar] [CrossRef]
- Tsai, P.C.; Wang, I.J. A facile synthesis of some new pyrazolo[1,5-a]pyrimidine heterocyclic disazo dyes and an evaluation of their solvatochromic behaviour. Dyes Pigm. 2007, 74, 578–584. [Google Scholar] [CrossRef]
- Maradiya, H.R.; Patel, V.S. Synthesis and dyeing performance of some novel heterocyclic azo disperse dyes. J. Braz. Chem. Soc. 2001, 12, 710–714. [Google Scholar]
- Razus, A.C.; Birzan, L.; Surugiu, N.M.; Corbu, A.C.; Chiraleu, E. Syntheses of azulen-1-yl-benzothiazol-2-yl diazenes. Dyes Pigm. 2007, 74, 26–33. [Google Scholar] [CrossRef]
- Singh, K.; Singh, S.; Taylor, J.A. Monoazo disperse dyes-part 1: Synthesis, spectroscopic studies and technical evaluation of monoazo disperse dyes derived from 2-aminothiazoles original. Dyes Pigm. 2002, 54, 189–200. [Google Scholar] [CrossRef]
- Naik, S.J.; Halkar, U.P. Synthesis and application of novel 4,5,6,7-tetrahydrobenzothiazole based azo disperse dyes. ARKIVOC 2005, xiii, 141–149. [Google Scholar]
- Habibi, M.H.; Hassanzadeh, A.; Isfahani, A.Z. Spectroscopic studies of Solophenyl red 3BL polyazo dye tautomerism in different solvents using UV–visible, 1H NMR and steady-state fluorescence techniques. Dyes Pigm. 2006, 69, 93–101. [Google Scholar] [CrossRef]
- Garg, H.G.; Praksh, C. Potential antidiabetics. 7. N1-(β-hydroxybenzylmethyl)-3-methyl-4-arylhydrazono-2-pyrazolin-5-ones and N1-(β-hydroxybenzylmethyl)-3-methyl-4-arylazo-5- methyl- or -phenylpyrazoles. J. Med. Chem. 1971, 14, 175–176. [Google Scholar]
- Garg, H.G.; Praksh, C. Potential antidiabetics. 11. Preparation of 4-arylazo-3,5-disubstituted-(2H)-1,2,6-thiadiazine 1,1-dioxides. J. Med. Chem. 1972, 15, 435–436. [Google Scholar] [CrossRef]
- Parekh, N.; Maheria, K.; Patel, P.; Rathod, M. Study on antibacterial activity for multidrug resistance stain by using phenyl pyrazolones substituted 3-amino 1H-pyrazolon (3,4-b) quinoline derivative in vitro condition. Int. J. Pharm Tech Res. 2011, 3, 540–548. [Google Scholar]
- Fusaka, T.; Ujikawa, O.; Kajiwara, T.; Tanaka, Y. Preparation of 1H-1,2,3-triazole-1-carboxamides as herbicides. PCT Int. Appl. WO. 97 11075, Mar 27, 1997. Chem. Abstr. 1997, 126, 293354p. [Google Scholar]
- Terada, A.; Wachi, K.; Myazawa, H.; Lizuka, Y.; Hasegawa, K.; Tabata, K. Preparation of imidazopyrazole derivatives as anti-inflammatory, antiulcer, and antiallergy agents. Jpn. Kokai Tokkyo Koho JP 07278148, Oct 24, 1995. Chem. Abstr. 1996, 124, 87009k. [Google Scholar]
- Kinnaman, K.E.; Pon, B.T.; Hanson, W.L.; Waits, V.B. Activity of anticancer compounds against trypanosome cruzi-infected mice. Am. J. Trop. Med. Hyg. 1998, 58, 804-806. Chem. Abstr. 1998, 129, 225339z. [Google Scholar]
- Naka, T.; Inada, Y. Preparation of heterocyclic compounds as angiotensin II antagonists. Faming Zhuanli Shenqing Gongkai Shuomingshu CN. 1079966, Dec 29, 1993. Chem. Abstr. 1995, 123, 169626c. [Google Scholar]
- Robins, R.K. The purines and related ring-systems. In Heterocyclic Compounds; Elderfield, R.C., Ed.; John Wiley and Sons, Inc.: New York, NY, USA, 1967; Volume 8, p. 277, Chapter 3. [Google Scholar]
- Davis, J.R.; Jadhav, A.L.; Fareed, J. Substituted azopurines. 1. Synthesis of 8,8'-dioxo-6,6'-azopurine. J. Med. Chem. 1974, 17, 639–642. [Google Scholar] [CrossRef]
- Taylor, E.; Cheng, C. Notes-purine chemistry. VII. An aimproved synthesis of hypoxanthine. J. Org. Chem. 1960, 25, 148–149. [Google Scholar] [CrossRef]
- Shawalia, A.S.; Mosselhi, A.N.M.; Husseina, A.M. Reaction of pyridothiouracil with hydrazonoyl halides and the antimicrobial activity of the products, pyrido[2,3-d][1,2,4]triazolo-[4,3-a]pyrimidin-5(1H)-one derivatives. J. Sulfur Chem. 2006, 27, 329–339. [Google Scholar] [CrossRef]
- Nakashima, K. The coloration reaction mechanism of 6-aminouracil derivatives with ehrlich reagent and its application to the colorimetric determination method. II. Yakugaku Zasshi 1977, 97, 906-910. Chem. Abstr. 1977, 87, 206576x. [Google Scholar]
- Gibson, C.L.; La Rosa, S.; Ohta, K.; Boyle, H.P.; Leurquin, F.; Alexandra, L.A.; Suckling, C.J. The synthesis of 7-deazaguanines as potential inhibitors of guanosine triphosphate cyclohydrolase I. Tetrahedron 2004, 60, 943–959. [Google Scholar] [CrossRef]
- Gibson, C.L.; La Rosa, S.; Suckling, C.J. A prototype solid phase synthesis of pteridines and related heterocyclic compounds. Org. Biomol. Chem. 2003, 1, 1909–1918. [Google Scholar] [CrossRef]
- Baker, B.R.; Wood, W.F. Irreversible enzyme inhibitors. CXXIII. Candidate irreversible inhibitors of guanine deaminase and xanthine oxidase derived from 9-phenylguanine substituted with a terminal sulfonyl fluoride. J. Med. Chem. 1968, 11, 650–652. [Google Scholar] [CrossRef]
- Mosselhi, M.A.; Pfleiderer, W. Purines. XIV. Synthesis and Properties of 8-Nitroxanthine and its N-Methyl Derivatives. J. Heterocycl. Chem. 1993, 30, 1221–1228. [Google Scholar] [CrossRef]
- Mueller, C.E.; Sauer, R.; Geis, U.; Frobenius, W.; Talik, P.; Pawlowski, M. Aza-Analogs of 8-Styrylxanthines as A2A-Adenosine Receptor Antagonists. Arch. Pharm. 1997, 330, 181–189. [Google Scholar] [CrossRef]
- Mueller, C.E.; Shi, D.; Manning, M., Jr.; Daly, J.W. Synthesis of paraxanthine analogs (1,7-disubstituted xanthines) and other xanthines unsubstituted at the 3-position: Structure-activity relationships at adenosine receptors. J. Med. Chem. 1993, 36, 3341–3349. [Google Scholar] [CrossRef]
- Mueller, C.E. General synthesis and properties of 1-monosubstituted xanthines. Synthesis 1993, 125–128. [Google Scholar] [CrossRef]
- John, C.D. The Hammett Equation; Cambridge University Press: New York, NY, USA, 1973. [Google Scholar]
- Shawali, A.S.; Darwish, E.S.S.; Altlbawy, F.M.A. Synthesis of (4-amino-5-phenyl-1,2,4-triazol-3-yl)thiohydrazonates and spectrophotometric study of their cyclization products in ground and excited states. Asian J. Spectrosc. 2007, 11, 115–125. [Google Scholar]
- Shawali, A.S.; Zeid, I.F.; Abdelkader, M.H.; Elsherbini, A.A.; Altlbawy, F.M.A. Synthesis, acidity constants and tautomeric structure of 7-arylhydrazono[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines in ground and excited states. J. Chin. Chem. Soc. 2001, 48, 65–72. [Google Scholar]
- Shawali, A.S.; Darwish, E.S.S.; Altlbawy, F.M.A. Site selectivity in diazonium coupling of ethyl (3-phenyl-7H-[1,2,3]-triazolo[3,4-b][1,3,4] thiadiazin-6-yl)acetate and tautomeric structure of the coupling products in the ground and excited states. Asian J. Spectrosc. 2008, 12, 113–120. [Google Scholar]
- Shawali, A.S.; Abdelkader, M.H.; Altalbawy, F.M.A. Synthesis and tautomeric structure of novel 3,7-bis(arylazo)-2,6-diphenyl-1H-imidazo-[1,2-b]pyrazoles in ground and excited states. Tetrahedron 2002, 58, 2875–2880. [Google Scholar] [CrossRef]
- Shawali, A.S.; Mosselhi, A.M.; Altalbawy, F.M.A.; Farghaly, T.A.; Tawfik, N.M. Synthesis and tautomeric structure of 3,7-bis(arylazo)-6-methyl-2-phenyl-1H-imidazo[1,2-b]pyrazoles in ground and excited states. Tetrahedron 2008, 64, 5524–5530. [Google Scholar]
- Sample Availability: Samples are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Darwish, E.S.; Mosselhi, M.A.; Altalbawy, F.M.; Saad, H.A. Synthesis, Acidity Constants and Tautomeric Structure of the Diazonium Coupling Products of 2-(Benzylsulfanyl)-7H-purin-6-one in Its Ground and Excited States. Molecules 2011, 16, 8788-8802. https://doi.org/10.3390/molecules16108788
Darwish ES, Mosselhi MA, Altalbawy FM, Saad HA. Synthesis, Acidity Constants and Tautomeric Structure of the Diazonium Coupling Products of 2-(Benzylsulfanyl)-7H-purin-6-one in Its Ground and Excited States. Molecules. 2011; 16(10):8788-8802. https://doi.org/10.3390/molecules16108788
Chicago/Turabian StyleDarwish, Elham S., Mosselhi A. Mosselhi, Farag M. Altalbawy, and Hosam A. Saad. 2011. "Synthesis, Acidity Constants and Tautomeric Structure of the Diazonium Coupling Products of 2-(Benzylsulfanyl)-7H-purin-6-one in Its Ground and Excited States" Molecules 16, no. 10: 8788-8802. https://doi.org/10.3390/molecules16108788
APA StyleDarwish, E. S., Mosselhi, M. A., Altalbawy, F. M., & Saad, H. A. (2011). Synthesis, Acidity Constants and Tautomeric Structure of the Diazonium Coupling Products of 2-(Benzylsulfanyl)-7H-purin-6-one in Its Ground and Excited States. Molecules, 16(10), 8788-8802. https://doi.org/10.3390/molecules16108788







