An Efficient and Chemoselective Procedure for Acylal Synthesis
Abstract
:1. Introduction
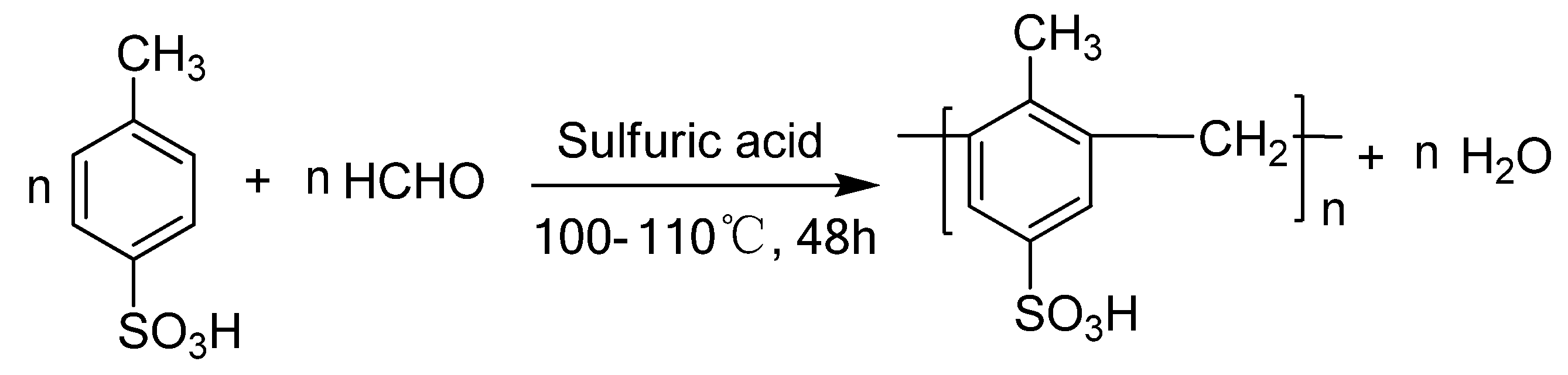
2. Results and Discussion
2.1. Characterization of the novel catalyst
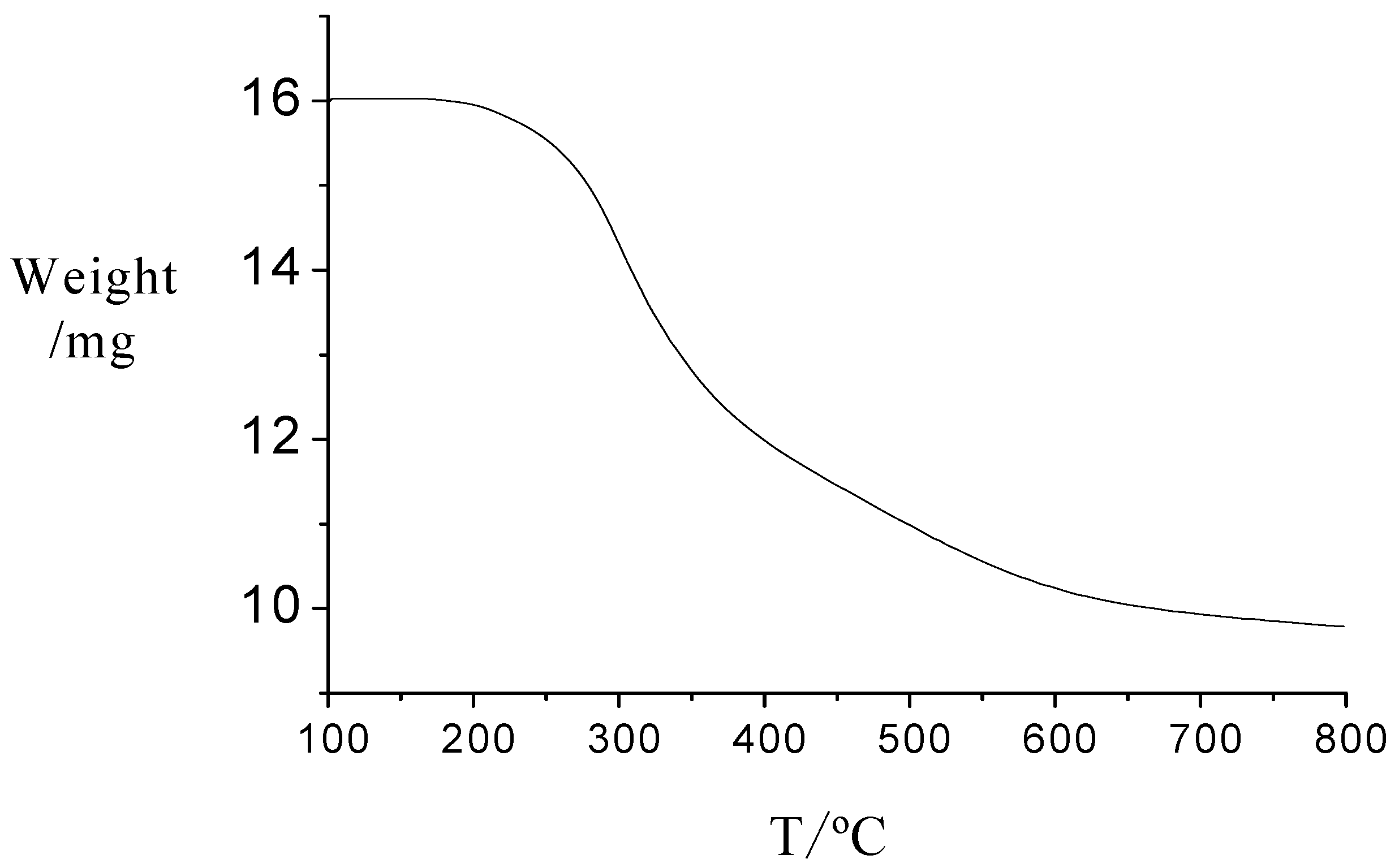
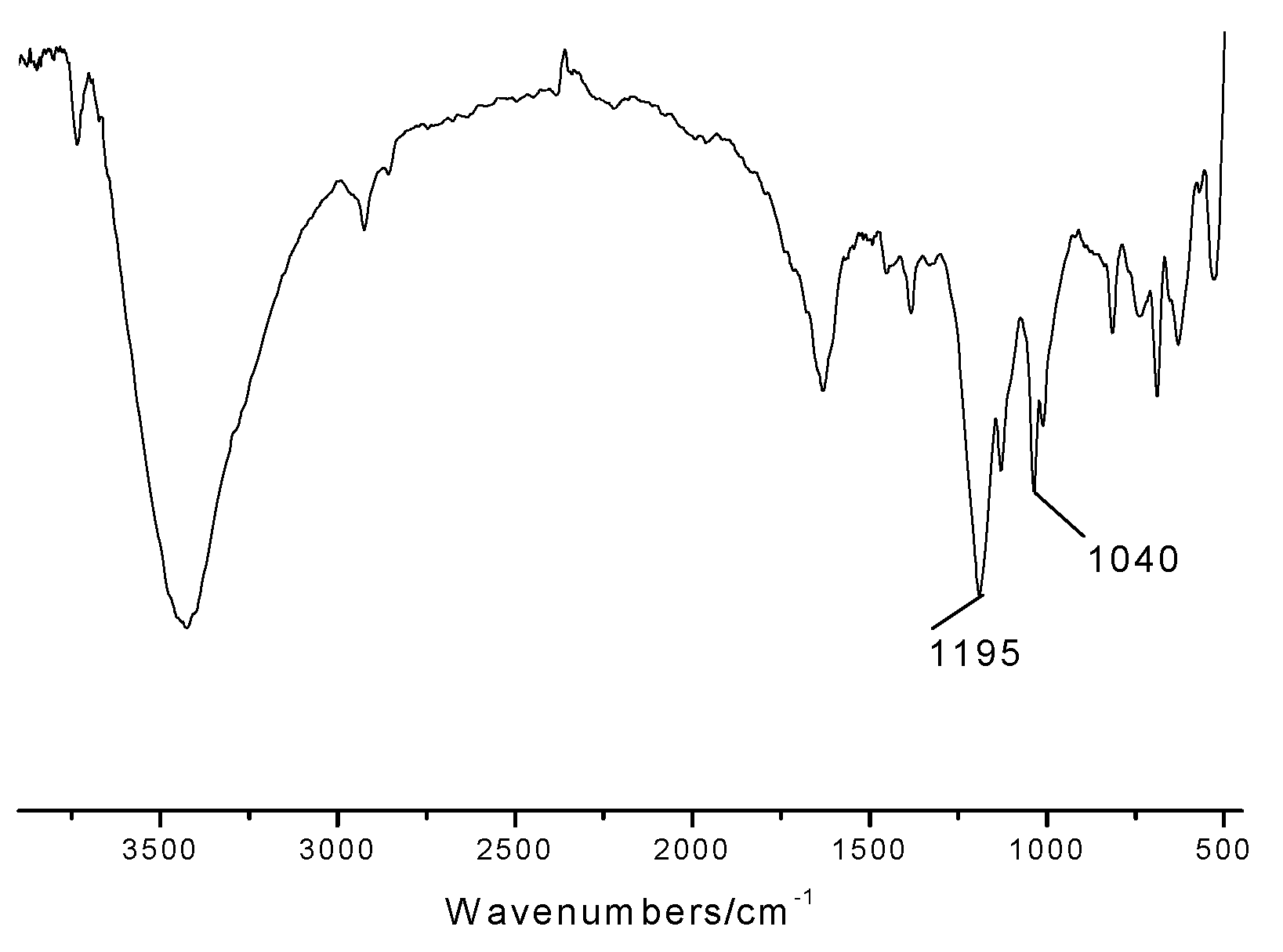
2.2. Catalytic procedure for the synthesis of acylals
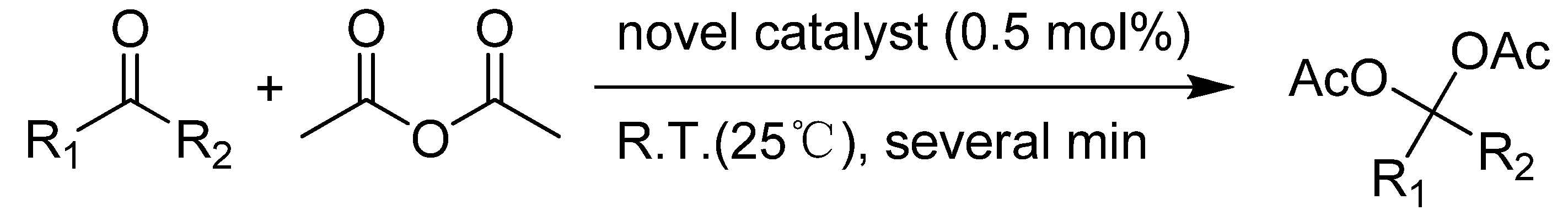
| Entry | Substrate | Product | Reaction time/min | Yield/% a, b |
|---|---|---|---|---|
| 1 |  | 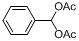 | 3 | 98.9 |
| 2 | 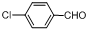 |  | 4 | 97.0 |
| 3 | 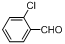 | 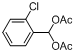 | 6 | 94.0 |
| 4 | 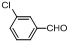 |  | 5 | 95.0 |
| 5 | 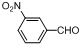 | 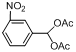 | 2 | 99.4 |
| 6 | 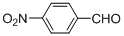 | 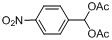 | 3 | 99.2 |
| 7 | 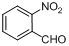 | 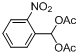 | 4 | 99.0 |
| 8 | 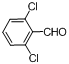 | 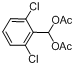 | 15 | 98.0 |
| 9 | 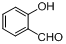 | 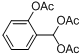 | 6 | 94.0 |
| 10 |  | 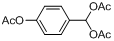 | 5 | 95.0 |
| 11 | 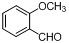 | 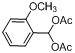 | 6 | 94.0 |
| 12 | 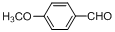 | 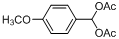 | 5 | 95.0 |
| 13 | 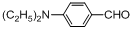 | 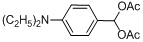 | 35 | 45.0 |
| 14 | 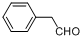 | 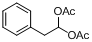 | 10 | 90.0 |
| 15 | 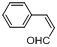 | 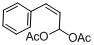 | 12 | 91.0 |
| 16 | 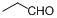 | 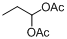 | 16 | 92.0 |
| 17 | 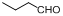 | 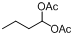 | 18 | 90.0 |
| 18 |  | 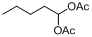 | 23 | 90.0 |
2.3. Comparative study on the catalytic activities of the novel catalyst and other catalysts
| Entry | Catalyst | Catalyst loading/mol% | Reaction time/min | Yield/% a, b |
|---|---|---|---|---|
| 1 | H2SO4 | 1 | 5 | 88.5 |
| 2 | PTSA | 1 | 5 | 87.4 |
| 3 | FeCl3 | 1 | 5 | 84.3 |
| 4 | NH2SO3H | 1 | 14 | 88.4 |
| 5 | Bi(OTf)3 | 1 | 15 | 90.3 |
| 6 | ZrCl4 | 1 | 8 | 85.4 |
| 7 | Iodine | 1 | 25 | 80.5 |
| 8 | NBS | 1 | 13 | 83.2 |
| 9 | Cu(OTf)3 | 1 | 10 | 85.6 |
| 10 | CAN | 1 | 15 | 87.8 |
| 11 | HY | 70 mg | 30 | 82.3 |
| 12 | SO42-/TiO2 | 80 mg | 15 | 90.3 |
| 13 | Novel catalyst | 25 mg | 3 | 98.9 |
2.4. The reuse of the catalyst
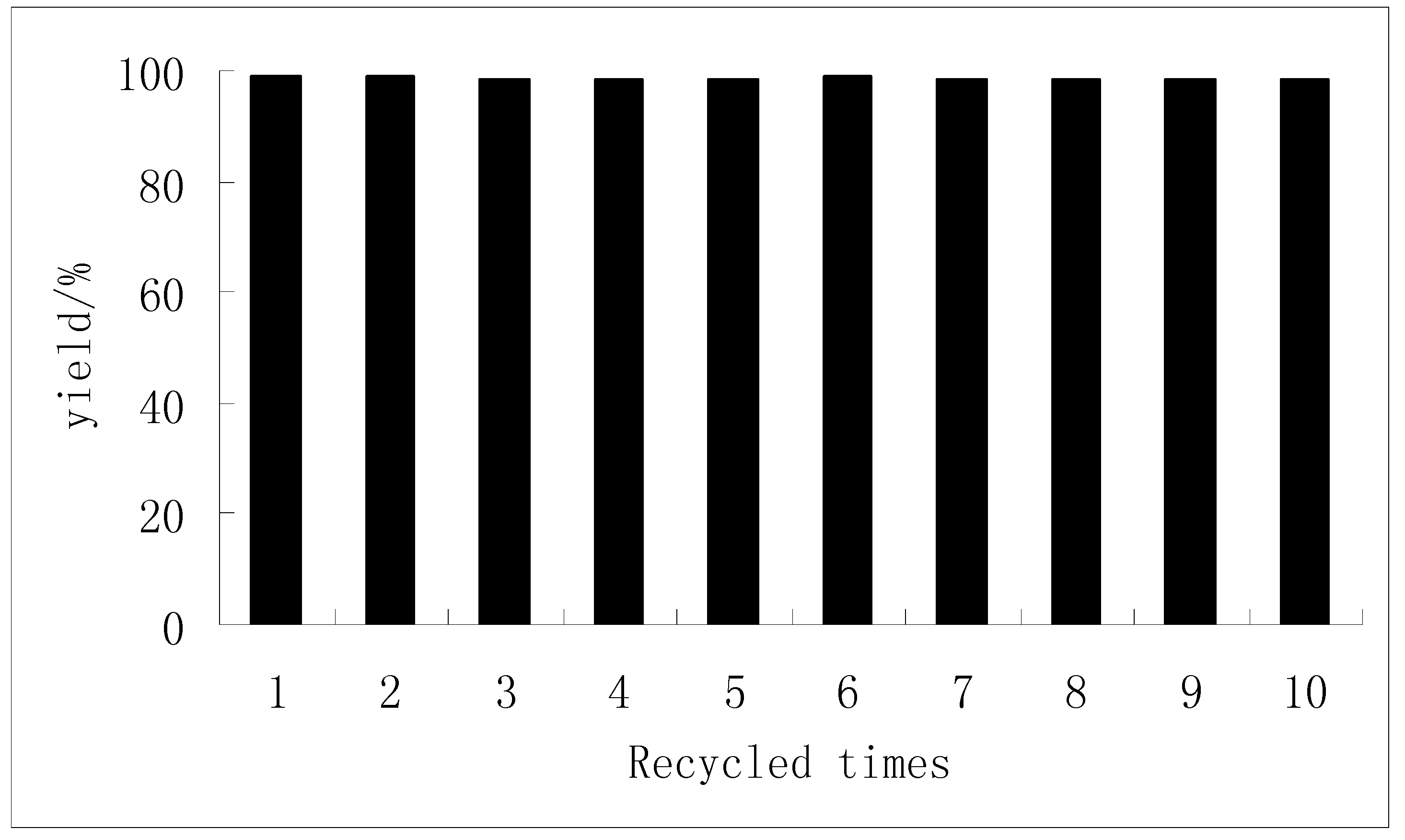
2.5. The chemoselectivity of the catalyst

3. Experimental
3.1. General
3.2. Synthesis of the catalyst
3.3. Preparation of acylals
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from the authors.
References and Notes
- Greene, T.W.; Wuts, P.G.M. Protective Groups in Organic Synthesis, 3rd ed; John Wiley and Sons: New York, NY, USA, 1999. [Google Scholar]
- Frick, J.G.; Harper, R.J. Acetals as Crosslinking Reagents for Cotton. J. Appl. Polym. Sci. 1984, 1433–1447. [Google Scholar]
- Shelke, K.; Sapkal, S.; Kategaonkar, A.; Shingate, B.; Shingare, M.S. An efficient and green procedure for the preparation of acylals from aldehydes catalyzed by alum [KAl(SO4)212H2O]. South Afr. J. Chem. 2009, 62, 109–112. [Google Scholar]
- VanHeerden, F.R.; Huyser, J.J.; Bardley, D.; Williams, G.; Holzapfel, C.W. Palladium-catalysed substitution reactions of geminal allylic diacetates. Tetrahedron Lett. 1998, 39, 5281–5283. [Google Scholar] [CrossRef]
- Andberg, M.; Sydness, L.K. The chemistry of acylals. Part II. Formation of nitriles by treatment of acylals with trimethylsilyl azide in the presence of a Lewis acid. Tetrahedron Lett. 1998, 39, 6361–6364. [Google Scholar] [CrossRef]
- Yadav, J.S.; Subba Reddy, V.B.; Shrihari, P. Scandium triflate catalyzed allylation of acetals and gem-diacetates: a facile synthesis of homoallyl ethers and acetates. Synlett 2001, 673–675. [Google Scholar]
- Michie, J.K.; Miller, J.A. Phosphorus trichloride as a catalyst in the preparation of 1,1-diacetates from aldehydes. Synthesis 1981, 824. [Google Scholar]
- Daka, N.; Borah, R.; Kalita, D.J.; Sarma, J.C. Synthesis of 1,1-Diacetates from Aldehydes using Trimethylchlorosilane and Sodium Iodide as Catalyst. J. Chem. Res. 1998, (S), 94–95. [Google Scholar]
- Deka, N.; Kalita, D.J.; Borah, R.; Sarma, J.C. Iodine as Acetylation Catalyst in the Preparation of 1,1-Diacetates from Aldehydes. J. Org. Chem. 1997, 62, 1563–1564. [Google Scholar] [CrossRef]
- Kochhar, K.S.; Bal, B.S.; Deshpande, R.P.; Rajadhyaksha, S.N.; Pinnick, H.W. Protecting groups in organic synthesis. Part 8. Conversion of aldehydes into geminal diacetates. J. Org. Chem. 1983, 48, 1765–1567. [Google Scholar]
- Li, Y.-Q. A rapid and convenient synthesis of 1,1-diacetates from aldehydes and acetic anhydride catalyzed by PVC-FeCl3 catalyst. Synth. Commun. 2000, 30, 3913–3917. [Google Scholar] [CrossRef]
- Karimi, B.; Seradj, H.; Ebrahimian, R.G. Mild and efficient conversion of aldehydes to 1,1-diacetates catalyzed with N-bromosuccinimide (NBS). Synlett 2000, 623–624. [Google Scholar]
- Firouzabadi, H.; Iranpoor, N.; Amani, K. Heteropoly acids as heterogeneous catalysts for thioacetalization and transthioacetalization reactions. Synthesis 2002, 59–62. [Google Scholar]
- Lawesson, S.O.; Berglund, C.; Gronwall, S. Peroxy compounds. VIII. Preparation of acylals and related compounds by Cu salt catalyzed reaction of tert-butyl perbenzoate with simple ethers and sulfides. Acta Chem. Scand. 1961, 15, 249–259. [Google Scholar] [CrossRef]
- Roy, S.C.; Banerjee, B. A mild and efficient method for the chemoselective synthesis of acylals from aldehydes and their deprotection catalyzed by ceric ammonium nitrate. Synlett 2002, 1677–1678. [Google Scholar]
- Smitha, G.; Reddy, C.S. A facile and efficient ZrCl4 catalyzed conversion of aldehydes to geminal-diacetates and dipivalates and their cleavage. Tetrahedron 2003, 59, 9571–9576. [Google Scholar] [CrossRef]
- Aggen, D.H.; Arnold, J.N.; Hayes, P.D.; Smoter, N.J.; Mohan, R.S. Bismuth compounds in organic synthesis. Bismuth nitrate catalyzed chemoselective synthesis of acylals from aromatic aldehydes. Tetrahedron 2004, 60, 3675–3679. [Google Scholar]
- Nagy, N.M.; Jakab, M.A.; Konya, J.; Antus, S. Convenient preparation of 1,1-diacetates from aromatic aldehydes catalyzed by zinc-montmorillonite. Appl. Clay Sci. 2002, 21, 213–216. [Google Scholar] [CrossRef]
- Li, Y.Q.; Cheng, L.H. A rapid and convenient synthesis of acylals from aldehydes and acetic anhydrides catalyzed by SnCl4/SiO2. Chin. Chem. Lett. 2001, 12, 565–568. [Google Scholar]
- Jin, T.; Feng, G.; Yang, M.; Li, T. An efficient and practical procedure for synthesis of 1,1-diacetates from aldehydes catalyzed by zirconium sulfate tetrahydrate-silica gel. Synth. Commun. 2004, 34, 1645–1651. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Xu, Y.N.; Wang, Z.Z.; Dai, L.Y. A new approach to the synthesis of pentaerythritol diacetals (diketals) catalyzed by SO3H-functionalized ionic liquids. Chin. Chem. Lett. 2010, 21, 524–528. [Google Scholar] [CrossRef]
- Niknam, K.; Saberi, D.; Sefat, M.N. Silica-bonded S-sulfonic acid as a recyclable catalyst for chemoselective synthesis of 1,1-diacetates. Tetrahedron Lett. 2009, 50, 4058–4062. [Google Scholar] [CrossRef]
- Khan, A.T.; Choudhury, L.H.; Ghosh, S. Silica-supported perchloric acid (HClO4-SiO2): A highly efficient and reusable catalyst for geminal diacylation of aldehydes under solvent-free conditions. J. Mol. Catal. A: Chem. 2006, 255, 230–235. [Google Scholar] [CrossRef]
- Wang, M.; Song, Z.; Gong, H.; Jiang, H. Synthesis of 1,1-diacetates using a new combined catalytic system, copper p-toluenesulfonate/HOAc. Synth. Commun. 2008, 38, 961–966. [Google Scholar] [CrossRef]
- Rabindran, J.B.; Pandurangan, A. Synthesis of geminal diacetates (acylals) using heterogeneous H3PW12O40 supported MCM-41 molecular sieves. Catal. Commun. 2008, 9, 577–583. [Google Scholar] [CrossRef]
- Lermontov, S.A.; Yurkova, L.L. Solid superacids as catalysts for the synthesis of acylals from aldehydes and acetic anhydride. Russ. Chem. Bull. 2008, 57, 2561–2563. [Google Scholar] [CrossRef]
- Margelefsky, E.L.; Anissa, B.; Zeidan, R.K.; Dufaud, V.; Davis, M.E. Nanoscale Organization of Thiol and Arylsulfonic Acid on Silica Leads to a Highly Active and Selective Bifunctional, Heterogeneous Catalyst. J. Am. Chem. Soc. 2008, 130, 13442–13449. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fan, D.-H.; Wang, H.; Mao, X.-X.; Shen, Y.-M. An Efficient and Chemoselective Procedure for Acylal Synthesis. Molecules 2010, 15, 6493-6501. https://doi.org/10.3390/molecules15096493
Fan D-H, Wang H, Mao X-X, Shen Y-M. An Efficient and Chemoselective Procedure for Acylal Synthesis. Molecules. 2010; 15(9):6493-6501. https://doi.org/10.3390/molecules15096493
Chicago/Turabian StyleFan, Da-He, Hui Wang, Xing-Xing Mao, and Yong-Miao Shen. 2010. "An Efficient and Chemoselective Procedure for Acylal Synthesis" Molecules 15, no. 9: 6493-6501. https://doi.org/10.3390/molecules15096493
APA StyleFan, D.-H., Wang, H., Mao, X.-X., & Shen, Y.-M. (2010). An Efficient and Chemoselective Procedure for Acylal Synthesis. Molecules, 15(9), 6493-6501. https://doi.org/10.3390/molecules15096493




