Screening of Natural Antioxidants from Traditional Chinese Medicinal Plants Associated with Treatment of Rheumatic Disease
Abstract
:1. Introduction
2. Results and Discussion
2.1. Antioxidant capacities of 50 selected medicinal plants
2.2. Total phenolic content of 50 selected medicinal plants
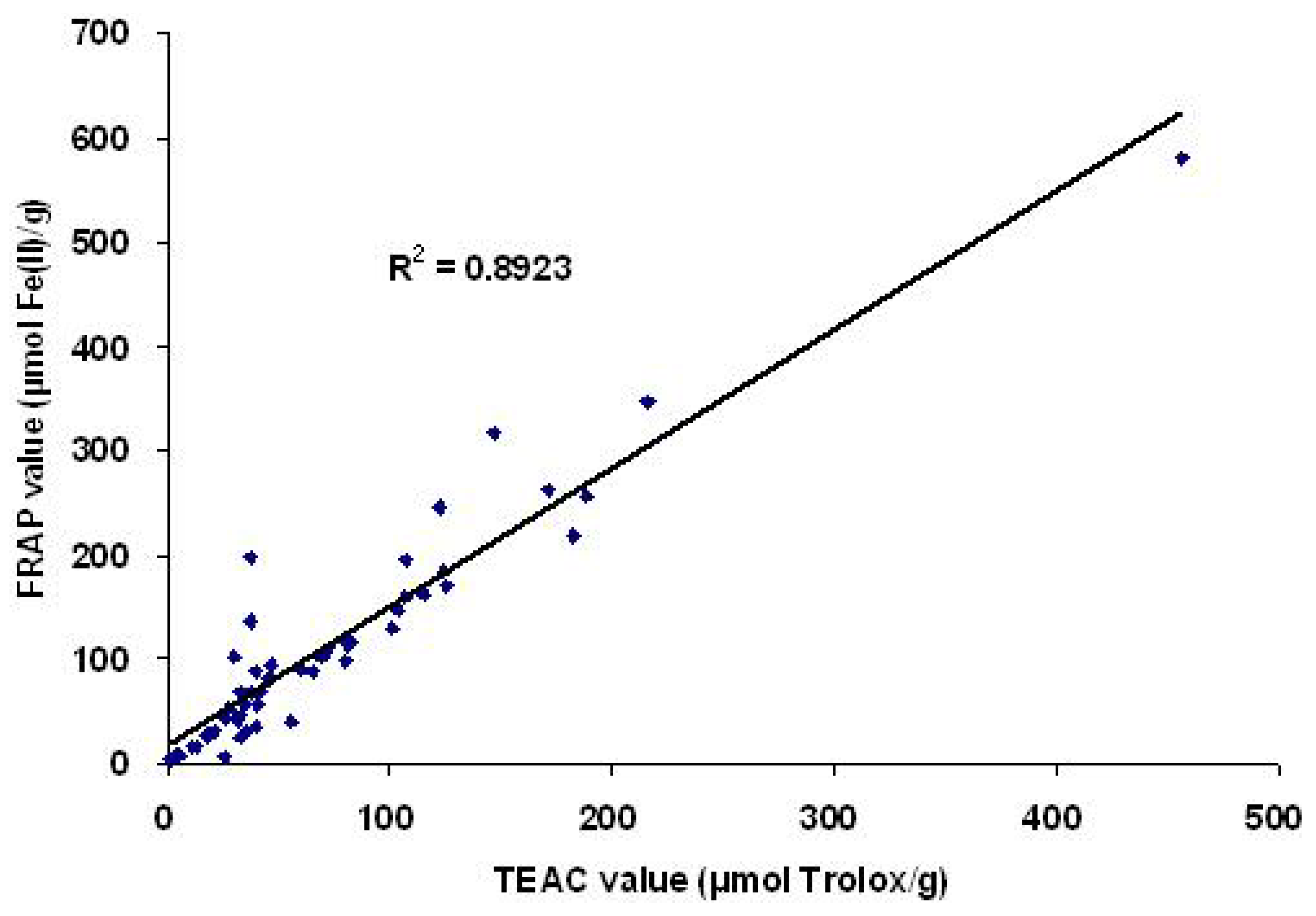
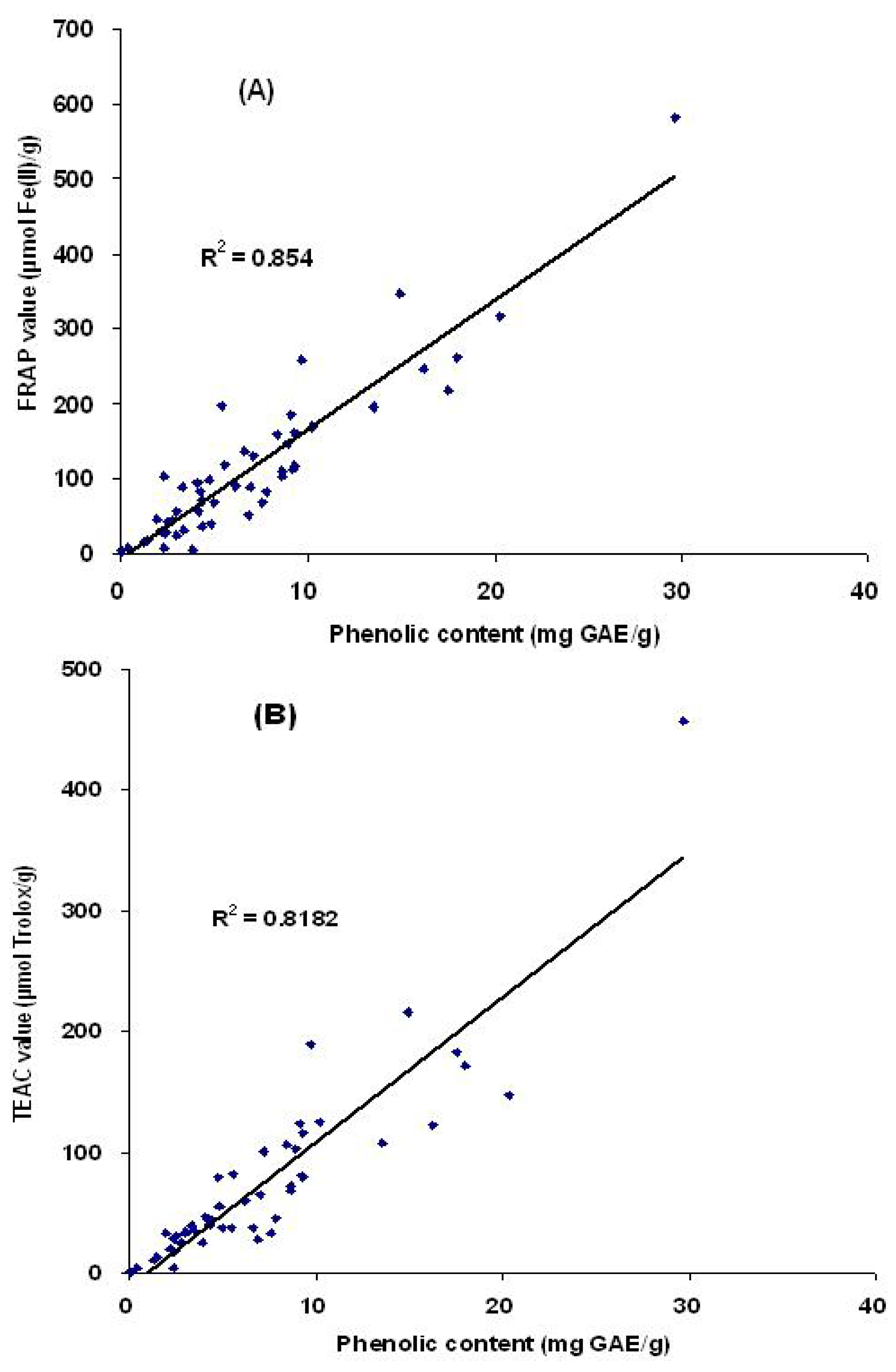
2.3. Correlation between antioxidant capacities and total phenolic content
3. Experimental
3.1. Chemicals and plant materials
3.2. Sample preparation
3.3. Ferric-reducing antioxidant power (FRAP) assay
3.4. Trolox equivalent antioxidant capacity (TEAC) assay
3.5. Determination of total phenolic content
3.6. Statistical analysis
4. Conclusions
Acknowledgements
References and Notes
- Azizova, O.A. Role of free radical processes in the development of atherosclerosis. Biol. Membrany. 2002, 19, 451–471. [Google Scholar]
- Quintero, M.; Brennan, P.A.; Thomas, G.J.; Moncada, S. Nitric oxide is a factor in the stabilization of hypoxia-inducible factor-1 alpha in cancer: Role of free radical formation. Cancer Res. 2006, 66, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Nagler, R.; Reznick, A.; Shafir, Y.; Shehadeh, N. Free radical-related effects and antioxidants in saliva and serum of adolescents with type 1 diabetes mellitus. Free Radic. Res. 2006, 40, S156. [Google Scholar]
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative diseases and oxidative stress. Nat. Rev. Drug Discov. 2004, 3, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Wickens, A.P. Ageing and the free radical theory. Resp. Physiol. 2001, 128, 379–391. [Google Scholar] [CrossRef]
- Stamler, J.S.; Hausladen, A. Oxidative modifications in nitrosative stress. Nat. Struct. Biol. 1998, 5, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, J.; Arner, E.S. Reactive oxygen species, antioxidants and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 31, 1287–1312. [Google Scholar] [CrossRef]
- Duthie, G.G.; Duthie, S.J.; Kyle, J.A.M. Plant polyphenols in cancer and heart disease: Implications as nutritional antioxidants. Nutr. Res. Rev. 2000, 13, 79–106. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Li, D.; Zhang, Y.; Gan, R.Y.; Song, F.L.; Chen, F. Antioxidant properties of Chinese medicinal plants. In Reactive Oxygen Species and Antioxidants in Higher Plants; Gupta, S.D., Ed.; Sciences Publishers: Enfield, CT, USA, 2010; Chapter 15; pp. 331–362. [Google Scholar]
- Wong, C.C.; Li, H.B.; Cheng, K.W.; Chen, F. A systematic survey of antioxidant activity of 30 Chinese medicinal plants using the ferric reducing antioxidant power assay. Food Chem. 2006, 97, 705–711. [Google Scholar] [CrossRef]
- Liu, H.Y.; Qiu, N.X.; Ding, H.H.; Yao, R.Q. Polyphenols contents and antioxidant capacity of 68 Chinese herbals suitable for medical or food uses. Food Res. Int. 2008, 41, 363–370. [Google Scholar] [CrossRef]
- Cai, Y.Z.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.L.; Plumb, G.W.; Bennett, R.N.; Bao, Y.P. Antioxidant activities of extracts from five anti-viral medicinal plants. J. Ethnopharmacol. 2005, 96, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Wong, C.C.; Cheng, K.W.; Chen, F. Antioxidant properties in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT-Food Sci. Technol. 2008, 41, 385–390. [Google Scholar] [CrossRef]
- Zhu, Y.Z.; Huang, S.H.; Tan, B.K.H.; Sun, J.; Whiteman, M.; Zhu, Y.C. Antioxidants in Chinese herbal medicines: A biochemical perspective. Nat. Prod. Rep. 2004, 21, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.; Banbury, L.K.; Leach, D.N. Antioxidant activity of 45 Chinese herbs and the relationship with their TCM characteristics. Evid. Bas. Complem. Alternat. Med. 2008, 5, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Dragland, S.; Senoo, H.; Wake, K.; Holte, K.; Blomhoff, R. Several culinary and medicinal herbs are important sources of dietary antioxidants. J. Nutr. 2003, 133, 1286–1290. [Google Scholar] [CrossRef] [PubMed]
- Delfino, R.J.; Staimer, N.; Tjoa, T.; Polidori, A.; Arhami, M.; Gillen, D.L.; Kleinman, M.T.; Vaziri, N.D.; Longhurst, J.; Zaldivar, F.; Siouta, S.C. Circulating biomarkers of inflammation, antioxidant activity and platelet activation are associated with primary combustion aerosols in subjects with coronary artery disease. Environ. Health Persp. 2008, 116, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Schinella, G.R.; Tournier, H.A.; Prieto, J.M.; Mordujovich de Buschiazzo, P.; Ríos, J.L. Antioxidant activity of anti-inflammatory plant extracts. Life Sci. 2002, 70, 1023–1033. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, V.; Milos, M.; Kulisic, T.; Jukic, M. Screening of 70 medicinal plant extracts for antioxidant capacity and total phenols. Food Chem. 2006, 94, 550–557. [Google Scholar] [CrossRef]
- Li, H.B.; Cheng, K.W.; Wong, C.C.; Fan, K.W.; Chen, F.; Jiang, Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method Enzymol. 1999, 299, 152–178. [Google Scholar]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef] [PubMed]
- Tawaha, K.; Alali, F.Q.; Gharaibeh, M.; Mohammad, M.; El-Elimat, T. Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chem. 2007, 104, 1372–1378. [Google Scholar] [CrossRef]
- Hinneburg, I.; Dorman, H.J.D.; Hiltunen, R. Antioxidant activities of extracts from selected culinary herbs and spices. Food Chem. 2006, 97, 122–129. [Google Scholar] [CrossRef]
- Kardosova, A.; Machova, E. Antioxidant activity of medicinal plant polysaccharides. Fitoterapia 2006, 77, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
Sample Availability: Plant samples are available from the authors. |
| Scientific Name | FRAP Assay (µmol Fe(II)/g) | TEAC Assay (μmol Trolox/g) | Phenolic Contents (mg GAE/g) |
|---|---|---|---|
| Acanthopanax gracilistylus W. W. Smith | 170.19 ± 3.96 | 125.08 ± 7.32 | 10.23 ± 0.20 |
| Agadtacge rygisa O. Kuntze. | 95.19 ± 4.53 | 46.31 ± 2.39 | 4.10 ± 0.07 |
| Akebia trifoliata (Thunb.) Koidz. | 102.18 ± 4.32 | 29.15 ± 1.29 | 2.38 ± 0.11 |
| Alisma orientale (Sam.) Juz. | 5.54 ± 0.74 | 25.69 ± 2.11 | 3.90 ± 0.16 |
| Alpinia galanga (L.) Willd. | 82.21 ± 2.92 | 45.45 ± 0.82 | 4.25 ± 0.10 |
| Alpinia katsumadai Hayat | 42.89 ± 0.95 | 31.51 ± 0.99 | 2.52 ± 0.08 |
| Amomun kravanh Pierre ex Gagnep | 43.54 ± 2.73 | 25.50 ± 0.88 | 2.77 ± 0.14 |
| Amomum tsao-ko Crevostet Lemarie | 130.16 ± 2.85 | 100.61 ± 1.71 | 7.15 ± 0.17 |
| Amomum villosum Lour | 117.57 ± 1.43 | 80.16 ± 0.97 | 9.29 ± 0.13 |
| Angelica biserrata Yuan et Shan | 68.99 ± 3.26 | 32.67 ± 2.03 | 7.63 ± 0.58 |
| Artemisia capillaris Thunb. | 158.87 ± 7.50 | 106.55 ± 3.63 | 8.38 ± 0.20 |
| Atractylodes lancea (Thunb.) DC. | 28.15 ± 0.25 | 17.61 ± 2.56 | 2.41 ± 0.41 |
| Benincasa hispida (Thunb.) Cogn. | 57.28 ± 4.34 | 40.81 ± 0.42 | 4.21 ± 0.21 |
| Capsella bursapastoris (L.) Medic. | 69.99 ± 7.85 | 41.37 ± 4.01 | 4.35 ± 0.09 |
| Chaenomeles speciosa (Sweet) Nakai | 195.15 ± 2.78 | 107.61 ± 1.09 | 13.58 ± 0.13 |
| Clematis chinensis Osbeck | 82.27 ± 4.41 | 45.04 ± 0.70 | 7.85 ± 0.03 |
| Coix lacryma-jobi L. | 7.75 ± 0.15 | 4.69 ± 0.16 | 2.34 ± 0.44 |
| Cynanchum paniculatum (Bge.) Kitag. | 35.93 ± 0.62 | 39.45 ± 3.81 | 4.35 ± 0.10 |
| Dianthus superbus L. | 68.57 ± 2.00 | 37.12 ± 3.94 | 5.00 ± 0.07 |
| Dioscorea collettii Hook. F. | 16.05 ± 0.11 | 10.82 ± 0.28 | 1.31 ± 0.03 |
| Dioscorea nipponica Makino. | 39.64 ± 0.51 | 54.86 ± 3.27 | 4.82 ± 0.22 |
| Drosera burmannii Vahl | 99.80 ± 3.70 | 79.76 ± 4.18 | 4.76 ± 0.15 |
| Erythrina variegata L. | 185.91 ± 0.68 | 124.31 ± 5.69 | 9.12 ± 0.23 |
| Eupatorium fortunei Turcz. | 111.80 ± 9.46 | 71.93 ± 0.55 | 8.65 ± 0.10 |
| Gentiana macrophylla Pall. | 52.29 ± 1.69 | 27.55 ± 1.05 | 6.89 ± 0.48 |
| Geranium wilfordii Maxim. | 347.33 ± 7.99 | 215.98 ± 4.10 | 14.98 ± 0.64 |
| Homalomena occulta (Lour.) Schott | 32.30 ± 0.47 | 35.57 ± 0.61 | 3.40 ± 0.12 |
| Juncus effusus L. | 56.69 ± 7.33 | 34.77 ± 0.94 | 3.00 ± 0.18 |
| Kochia scparia (L.) Schrad | 103.22 ± 2.28 | 68.83 ± 1.69 | 8.63 ± 0.12 |
| Liquidambar formosana Hance | 118.44 ± 2.44 | 81.88 ± 11.11 | 5.58 ± 0.07 |
| Loranthus parasiticus (L.) Merr. | 580.02 ± 31.32 | 457.00 ± 6.41 | 29.67 ± 0.99 |
| Lysima chiachristinae Hance | 88.85 ± 1.44 | 65.30 ± 0.78 | 6.99 ± 0.05 |
| Magnolia officinalis Rehd. et Wils. | 257.45 ± 9.28 | 188.70 ± 12.01 | 9.68 ± 0.22 |
| Malva verticillata L. | 30.67 ± 2.17 | 20.32 ± 0.73 | 2.18 ± 0.07 |
| Morus alba L. | 46.96 ± 3.00 | 33.03 ± 2.38 | 1.96 ± 0.33 |
| Pinus tabulaeformis Carr. | 17.72 ± 0.25 | 13.30 ± 0.26 | 1.46 ± 0.01 |
| Piper kadsura (Choisy) Ohwi | 147.41 ± 3.64 | 103.41 ± 8.11 | 8.94 ± 0.16 |
| Plantago asiatica L. | 88.06 ± 13.26 | 39.94 ± 1.41 | 3.34 ± 0.43 |
| Plantago major L. | 137.23 ± 7.07 | 37.77 ± 0.85 | 6.62 ± 0.18 |
| Polygonum aviculare L. | 263.19 ± 4.73 | 171.65 ± 10.78 | 18.00 ± 0.25 |
| Polyporus umbellatus (Pers) Fr. | 7.93 ± 1.59 | 4.18 ± 0.09 | 0.38 ± 0.05 |
| Poria cocos (Schw.) Wolf. | 3.88 ± 0.15 | 1.31 ± 0.11 | 0.10 ± 0.01 |
| Pyrola calliantha H. Andr. | 160.96 ± 5.25 | 115.77 ± 10.03 | 9.31 ± 0.72 |
| Pyrrosia sheaeri (Bak.) Ching | 316.72 ± 4.82 | 147.26 ± 4.87 | 20.29 ± 0.17 |
| Siegesbeckia orientalis L. | 91.25 ± 4.39 | 60.23 ± 9.49 | 6.18 ± 0.14 |
| Sinomenium acutum Rehd.et Wils. | 245.94 ± 9.25 | 122.24 ± 1.74 | 16.21 ± 0.09 |
| Trachelospermum jasminoides Lem. | 113.22 ± 3.29 | 81.21 ± 6.10 | 9.19 ± 0.14 |
| Tripterygium wilfordii Hook. F. | 217.94 ± 2.49 | 181.98 ± 2.87 | 17.51 ± 0.30 |
| Vigna umbellata Ohwi et Ohashi | 24.74 ± 0.80 | 32.82 ± 1.62 | 3.01 ± 0.08 |
| Zanthoxylum nitidum (Roxb.) DC. | 198.25 ± 14.13 | 37.45 ± 1.95 | 5.44 ± 0.16 |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gan, R.-Y.; Kuang, L.; Xu, X.-R.; Zhang, Y.; Xia, E.-Q.; Song, F.-L.; Li, H.-B. Screening of Natural Antioxidants from Traditional Chinese Medicinal Plants Associated with Treatment of Rheumatic Disease. Molecules 2010, 15, 5988-5997. https://doi.org/10.3390/molecules15095988
Gan R-Y, Kuang L, Xu X-R, Zhang Y, Xia E-Q, Song F-L, Li H-B. Screening of Natural Antioxidants from Traditional Chinese Medicinal Plants Associated with Treatment of Rheumatic Disease. Molecules. 2010; 15(9):5988-5997. https://doi.org/10.3390/molecules15095988
Chicago/Turabian StyleGan, Ren-You, Lei Kuang, Xiang-Rong Xu, Yuan Zhang, En-Qin Xia, Feng-Lin Song, and Hua-Bin Li. 2010. "Screening of Natural Antioxidants from Traditional Chinese Medicinal Plants Associated with Treatment of Rheumatic Disease" Molecules 15, no. 9: 5988-5997. https://doi.org/10.3390/molecules15095988
APA StyleGan, R.-Y., Kuang, L., Xu, X.-R., Zhang, Y., Xia, E.-Q., Song, F.-L., & Li, H.-B. (2010). Screening of Natural Antioxidants from Traditional Chinese Medicinal Plants Associated with Treatment of Rheumatic Disease. Molecules, 15(9), 5988-5997. https://doi.org/10.3390/molecules15095988






