Synthesis and Biological Evaluation of 2,5-Bis(alkylamino)-1,4-benzoquinones
Abstract
:1. Introduction
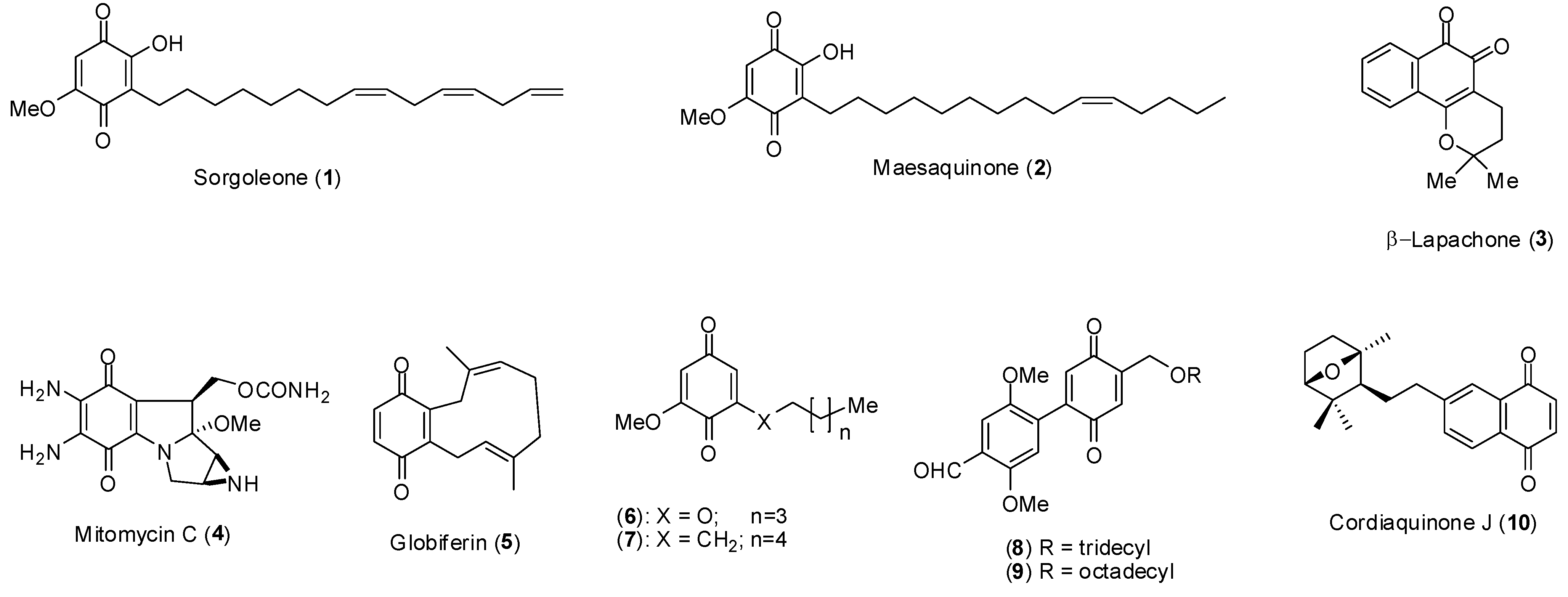
2. Results and Discussion
2.1. Synthesis of 2,5-diamino-p-benzoquinones
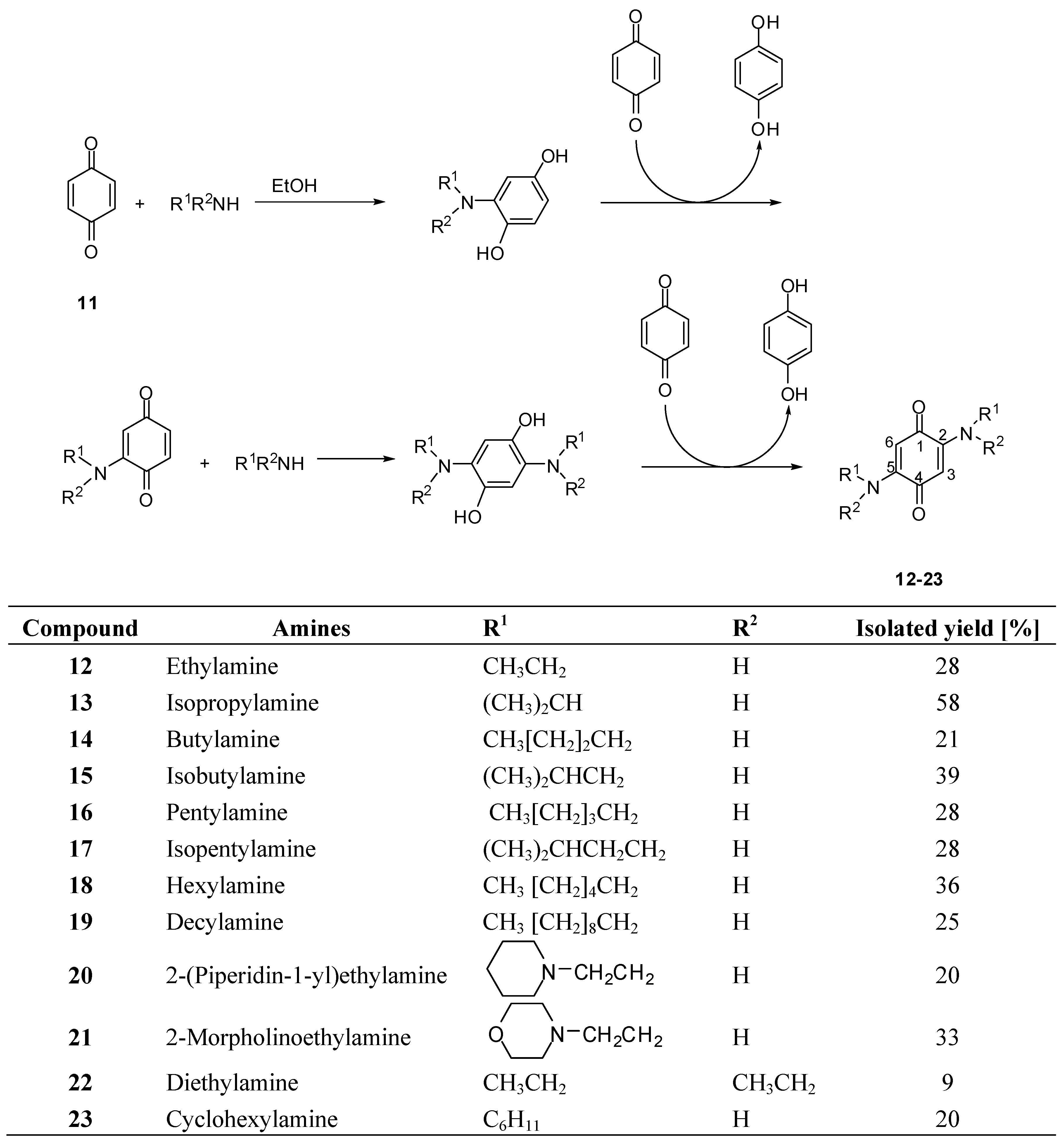
2.2. Phytotoxic activity
| Compound | Cucumis sativus | Sorghum bicolor | ||||||
|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 24 h | 48 h | |||||
| Radiclelength [cm]a | Inhibition [%] | Radiclelength [cm]a | Inhibition [%] | Radiclelenght [cm]a | Inhibition [%] | Radiclelength [cm]a | Inhibition [%] | |
| 12 | 1.40 def | 42 | 1.94 fg | 59 | 0.87 cd | 36 | 1.04 fg | 30 |
| 13 | 1.75 cde | 28 | 3.91 bcd | 18 | 1.90 ab | -40 | 3.50 abc | -135 |
| 14 | 2.17 abc | 10 | 4.87 a | -2 | 1.69 ab | -24 | 3.70 ab | -148 |
| 15 | 2.02 abc | 17 | 3.43 de | 28 | 1.14 bcd | 17 | 1.87 defg | -26 |
| 16 | 2.02 abc | 17 | 4.24 abcd | 11 | 1.52 abc | -12 | 1.91 defg | -28 |
| 17 | 1.89 bcd | 22 | 2.73 ef | 43 | 1.17 bcd | 14 | 2.25 bcdef | -51 |
| 18 | 1.25 ef | 48 | 1.25 g | 74 | 0.49 d | 64 | 0.51 g | 66 |
| 19 | 2.26 abc | 7 | 3.73 ef | 22 | 1.81 ab | -33 | 3.81 a | -156 |
| 20 | 1.85 cd | 24 | 2.33 f | 51 | 1.23 bcd | 10 | 2.07 cdef | -39 |
| 21 | 2.46 a | -2 | 4.41 abc | 7 | 2.09 a | -54 | 3.21 abcd | -115 |
| 22 | 1.16 f | 52 | 1.79 fg | 62 | 0.85 cd | 38 | 0.92 fg | 38 |
| 23 | 2.10 abc | 13 | 4.41 abc | 7 | 1.47 abc | -8 | 2.82 abcde | -89 |
| Control | 2.42 ab | 4.76 ab | 1.36 abc | 1.49 efg | ||||
| Compound | Ipomoea grandifolia | Brachiaria decumbens | ||||||
|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 24 h | 48 h | |||||
| Radiclelength [cm]a | Inhibition [%] | Radiclelength [cm]a | Inhibition [%] | Radiclelenght [cm]a | Inhibition [%] | Radiclelength [cm]a | Inhibition [%] | |
| 12 | 0.91 de | 48 | 0.91 d | 67 | 0.87 cd | 47 | 1.06 bcd | 43 |
| 13 | 1.19 cd | 32 | 2.47 ab | 10 | 1.82 a | -11 | 2.90 a | -56 |
| 14 | 1.41 abc | 20 | 2.74 a | 0 | 1.69 ab | -3 | 3.07 a | -65 |
| 15 | 1.43 abc | 19 | 2.39 ab | 13 | 1.48 abc | 10 | 2.35 ab | -26 |
| 16 | 1.48 abc | 16 | 2.41 ab | 12 | 1.10 bcd | 33 | 1.50 bcd | 19 |
| 17 | 1.23 cd | 30 | 1.49 bcd | 46 | 1.59 abc | 3 | 2.21 ab | -19 |
| 18 | 0.94 de | 47 | 0.94 d | 66 | 0.45 d | 73 | 0.45 d | 76 |
| 19 | 1.69 ab | 4 | 2.25 abc | 18 | 1.67 ab | -2 | 2.98 a | -60 |
| 20 | 1.22 cd | 31 | 1.57 bcd | 43 | 1.51 abc | 8 | 1.93 abc | -4 |
| 21 | 1.32 bcd | 26 | 2.40 ab | 12 | 1.80 ab | -10 | 2.82 a | -51 |
| 22 | 0.77 e | 56 | 1.35 cd | 51 | 0.65 d | 60 | 0.86 cd | 54 |
| 23 | 1.35 bc | 23 | 2.72 a | 1 | 1.73 ab | -5 | 3.02 a | -62 |
| Control | 1.76 a | 2.72 a | 1.64 ab | 1.86 abc | ||||
2.3. ATP assay
2.4. Anti-tumor activity
| Compound | Cellsa IC50b [µg/mL]; Confident interval | |||||
|---|---|---|---|---|---|---|
| HL-60 | SF-295 | HCT-8 | MDA-MB-435 | PBMC | ||
| 12 | 2.3(1.3-3.9) | 6.0 (4.6-7.9) | 5.2(2.4-11.2) | 5.6(3.8-8.4) | >25 | |
| 13 | >25 | >25 | >25 | >25 | Nd | |
| 14 | >25 | >25 | >25 | >25 | Nd | |
| 16 | >25 | >25 | >25 | >25 | Nd | |
| 17 | >25 | >25 | >25 | >25 | Nd | |
| 18 | >25 | >25 | >25 | >25 | Nd | |
| 19 | >25 | >25 | >25 | >25 | Nd | |
| 20 | 20.3(17.6-23.4) | 11.3(8.6-14.9) | 13.5(9.1-20.1) | 21.5(18.7-24.8) | 21,8 (15,6-30,1) | |
| 21 | >25 | >25 | >25 | >25 | Nd | |
| 22 | >25 | >25 | >25 | >25 | Nd | |
| 23 | >25 | >25 | >25 | >25 | Nd | |
| Doxorrubicin | 0.02(0.01-0.02) | 0.23(0.19-0.25) | 0.01(0.01-0.02) | 0.48(0.34-0.66) | 0,96 (0,51-1,71) | |
3. Experimental
3.1. General
3.2. Synthesis of compounds 12-16, 18-23, exemplified by the synthesis of 2,5-bis(isopentylamino)-1,4-benzoquinone (17)
3.3. Biological assays
3.3.1. Phytotoxic activity
3.3.2. Assay of radicle elongation on Petri dishes
3.3.3. Measurement of the ATP synthesis
3.4. Cytotoxicity screening
3.4.1. Cell lines and cell cultures
3.4.2. MTT assay
3.4.3. Alamar Blue assay
4. Conclusions
Acknowledgements
- Sample Availability: Contact the authors.
References and notes
- Oliveros-Bastidas, A.J.; Macías, F.A.; Fernández, C.C.; Marín, D.; Molinillo, J.M.G. Exudados dela raiz y su relevancia actual en las interacciones alelopaticas. Quim. Nova. 2009, 32, 198–213. [Google Scholar] [CrossRef]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef]
- Walker, T.S.; Bais, H.P.; Grotewold, E.; Vivanco, J.M. Root exudation and rhizosphere biology. Plant Physiol. 2003, 132, 44–51. [Google Scholar] [CrossRef]
- Bertin, C.; Yang, X.; Weston, L.A. The role of root exudates and allelochemicals in the rhizosphere. Plant Soil. 2003, 256, 67–83. [Google Scholar] [CrossRef]
- Macías, F.A.; Molinillo, J.M.G.; Varela, R.M.; Galindo, J.C.G. Allelopathy - A natural alternative for weed Control. Pest Manag. Sci. 2007, 63, 327–348. [Google Scholar] [CrossRef]
- Macías, F.A.; Galindo, J.C.G.; Molinillo, J.M.G.; Cutler, H.G. Allelopathy – Chemistry and Mode of Action of Allelochemicals; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Copping, L.G.; Duke, S.O. Natural products that have been used commercially as crop protection agents - A review. Pest Manag.Sci. 2007, 63, 524–554. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Teixeira, R.R.; Montanari, R.M. Phytotoxic Natural Products as Models for the development of crop protection agents. In Current Trends in Phytochemistry; Epifano, F., Ed.; Research Signpost: Kerala, India, 2008; pp. 21–59. [Google Scholar]
- Chang, M.; Netzly, D.H.; Butler, L.G.; Lynn, D.G. Chemical regulation of distance. Characterization of the first natural host germination stimulant for Striga asiatica. J. Am. Chem. Soc. 1986, 108, 7858–7860. [Google Scholar] [CrossRef]
- Einhellig, F.A.; Souza, I.F. Phytotoxicity of sorgoleone found in grain Sorghum root exudates. J. Chem. Ecol. 1992, 18, 1–11. [Google Scholar] [CrossRef]
- Gonzales, V.M.; Kazimir, J.; Nimbal, C.; Weston, L.A.; Cheniae, G.M. Inhibition of a photosystem II electron transfer reaction by the natural product sorgoleone. J. Agric. Food Chem. 1997, 45, 1415–1421. [Google Scholar] [CrossRef]
- Rasmussen, J.A.; Hejl, A.M.; Einhellig, F.A.; Thomas, J.A. Sorgoleone from root exudate inhibits mitochondrial functions. J. Chem. Ecol. 1992, 18, 197–207. [Google Scholar] [CrossRef]
- Czarnota, M.A.; Paul, R.N., Dayan; Nimbal, C.I.; Weston, L.A. Mode of action, localization, of production, chemical nature, and activity of sorgolene: A potent PSII inhibitor in Sorghum spp. root exudates. Weed Technol. 2001, 15, 813–825. [Google Scholar] [CrossRef]
- Streibig, J.C.; Dayan, F.E.; Rimando, A.M.; Duke, S.O. Joint action of natural and synthetic photosystem II inhibitors. Pest. Sci. 1999, 55, 137–146. [Google Scholar] [CrossRef]
- Hejl, A.M.; Koster, K.L. The allelochemical sorgoleone inhibits root H+-ATPase and water uptake. J. Chem. Ecol. 2004, 30, 2181–2191. [Google Scholar] [CrossRef]
- Kubo, I.; Chaudhuri, S.K. Structure of maesaquinone. Bioorg. Med. Chem. Lett. 1994, 4, 1131–1134. [Google Scholar] [CrossRef]
- Silva, M.N. da; Ferreira, V.F.; de Souza, M.C.B.V. Um panorama atual da química e da farmacologia de naftoquinonas, com ênfase na β-lapachona e derivados. Quim. Nova 2003, 26, 407–416. [Google Scholar] [CrossRef]
- Pan, S.S.; Andrews, P.A.; Glover, C.J. Reductive activation of mitomycin C and mitomycin C metabolites catalyzed by NADPH-cytochrome P-450 reductase and xanthine oxidase. J. Biol. Chem. 1984, 259, 959–966. [Google Scholar]
- Tomasz, M.; Palom, Y. The mitomycin bioreductive antitumor agents: cross-linking and alkylation of DNA as the molecular basis of their activity. Pharmacol. Ther. 1997, 76, 73–87. [Google Scholar] [CrossRef]
- Dettrakul, S.; Surerum, S.; Rajviroongit, S.; Kittakoop, P. Biomimetic transformation and biological activities of globiferin, a terpenoid benzoquinone from Cordia globifera. J. Nat. Prod. 2009, 72, 861–865. [Google Scholar] [CrossRef]
- Marinho-Filho, J.D.; Bezerra, D.P.; Araújo, A.J.; Montenegro, R.C.; Pessoa, C.; Diniz, J.C.; Viana, F.A.; Pessoa, O.D.; Silveira, E.R.; de Moraes, M.O.; Costa-Lotufo, L.V. Oxidative stress induction by (+)-cordiaquinone J triggers both mitochondria-dependent apoptosis and necrosis in leukemia cells. Chem. Biol. Interact. 2010, 183, 369–379. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Costa, A.V.; Veloso, D.P.; Lopes, J.L.C.; Hernandez-Terrones, M.G.; King-Diaz, B.; Lotina-Hennsen, B. Phytogrowth-inhibitory lactones derivatives of glaucolide B. Z Naturforsch. 2004, 59c, 803–810. [Google Scholar]
- Chaves, F.C.; Barbosa, L.C.A.; Demuner, A.J.; Silva, A.A. New helminthosporal analogues with plant-growth regulatory properties synthesized via oxyallyl cation. Z. Naturforsch. 2006, 61b, 1287–1294. [Google Scholar]
- Barbosa, L.C.A.; Demuner, A.J.; Maltha, C.R.A.; da Silva, P.S.; Silva, A.A. Sintese e avaliação da ativadade fitotóxica de novos análogos oxigenados do ácido helmintospórico. Quim. Nova 2003, 26, 655–660. [Google Scholar] [CrossRef]
- Demuner, A.J.; Barbosa, L.C.A.; Veloso, D.P.; Howarth, O.W. Synthesis and plant growth regulatory activity of 6α,7β-dihydroxyvouacapan-17β-oic acid derivatives. Aust. J. Chem. 1998, 51, 61–66. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Demuner, A.J.; Alvarenga, E.S.; Oliveira, A.; King-Diaz, B.; Lotina-Hennsen, B. Phytogrowth- and photosynthesis-inhibiting properties of nostoclide analogues. Pest Manag.Sci. 2006, 62, 214–222. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Demuner, A.J.; Maltha, C.R.A.; Teixeira, R.R.; Souza, K.A.P.; Bicalho, K.U. Phytogrowth activity of 3-(3-chlorobenzyl)-5-arylidenefuran-2(5H)-ones. Z. Naturforsch. 2009, 64b, 245–251. [Google Scholar]
- Barbosa, L.C.A.; Rocha, M.E.; Teixeira, R.R.; Maltha, C.R.A.; Forlani, G. Synthesis of 3-(4-bromobenzyl)-5-(aryl methylene)-5H-furan-2-ones and their activity as inhibitors of the photosynthetic electron transport chain. J. Agric. Food Chem. 2007, 55, 8562–8569. [Google Scholar] [CrossRef]
- Teixeira, R.R.; Barbosa, L.C.A.; Forlani, G.; Piló-Veloso, D.; Carneiro, J.W.M. Synthesis of photosynthesis-inhibiting nostoclide analogues. J. Agric. Food Chem. 2008, 56, 2321–2329. [Google Scholar] [CrossRef]
- Lima, L.S.; Barbosa, L.C.A.; Alvarenga, E.S.; Demuner, A.J.; Silva, A.A. Synthesis and phytotoxicity evaluation of substituted para-benzoquinones. Aust. J. Chem. 2003, 56, 625–630. [Google Scholar]
- Barbosa, L.C.A.; Alvarenga, E.S.; Demuner, A.J.; Virtuoso, L.S.; Silva, A.A. Synthesis of new phytogrowth-inhibitory substituted aryl-p-benzoquinones. Chem. Biod. 2006, 3, 553–567. [Google Scholar] [CrossRef]
- Bayen, S.; Barooah, N.; Sarma, R.J.; Sen, T.K.; Karmakar, A.; Baruah, J.B. Synthesis, structure and electrochemical properties of 2,5-bis(alkyl/arylamino)1,4-benzoquinones and 2-arylamino-1,4-naphthoquinones. Dye Pigment. 2007, 75, 770–775. [Google Scholar] [CrossRef]
- Hassan, S.S.M.; Iskander, M.L.; Nashed, N.E. Spectrophotometric determination of aliphatic primary and secondary amines by reaction with p-benzoquinone. Talanta 1985, 32, 301–305. [Google Scholar] [CrossRef]
- Tindale, C.R. Reactions of biogenic amines with quinones. Aust. J. Chem. 1984, 37, 611–617. [Google Scholar] [CrossRef]
- Nithianandam, V.S.; Erhan, S. Quinone-amine polymers: 18. A novel method for the synthesis of poly(alkyl aminoquinone)s. Polymer 1998, 39, 4095–4098. [Google Scholar] [CrossRef]
- Einhellig, F.A.; Schon, M.K.; Rasmussen, J.A. Synergistic effects of four cinnamic acid compounds on grain sorghum. J. Plant Growth Regul. 1982, 1, 251–258. [Google Scholar]
- Hennsen, B.L.; Achine, L.; Ruvalcaba, N.M.; Ortiz, A.; Hernández, J.; Farbán, N.; Martínez, M. A. 2,5-Diamino-p-benzoquinone derivatives as photosystem I electron acceptors: Synthesis and electrochemical and physicochemical properties. J. Agric. Food Chem. 1998, 46, 724–730. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Method. 1983, 16, 55–63. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Gogal, R.M.J.; Walsh, J.E. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes: An alternative to 3H-thymidine incorporation assay. J. Immunol. Method. 1994, 170, 211–224. [Google Scholar] [CrossRef]
- Zhi-Jun, Y.; Sriranganathan, N.; Vaught, T.; Arastu, S.K.; Ahmed, S.A. A dye-based lymphocyte proliferation assay that permits multiple immunological analyses: mRNA, cytogenetic, apoptosis, and immunophenotyping studies. J. Immunol. Method. 1997, 210, 25–39. [Google Scholar] [CrossRef]
- Chari, R.V.J. Targeted cancer therapy: conferring specificity to cytotoxic drugs. Acc. Chem. Res. 2008, 41, 98–107. [Google Scholar] [CrossRef]
- O′Brien, P.J. Molecular mechanisms of quinone cytotoxicity. Chem.-Biol. Interact. 1991, 80, 1–41. [Google Scholar] [CrossRef]
- Perrin, D.D.; Armarego, W.L.F. Purification of Laboratory Chemicals, 3rd ed; Pergamon: Oxford, UK, 1988. [Google Scholar]
- Brondani, D.J.; Bieber, L.W. Regioselective lithiation and alkylation of 2-methoxy-hydroquinone. Quim. Nova 1995, 18, 144–146. [Google Scholar]
- Zhou, Q.; Swager, T.M. Probing delocalization across alkyne-containing linkages: Synthesis and cyclic voltammetry of bridged phenylenediamines. J. Org. Chem. 1995, 60, 7096–7100. [Google Scholar] [CrossRef]
- Wellington, K.W.; Steenkamp, P.; Brady, D. Diamination by N-coupling using a commercial laccase. Bioorg. Med. Chem. 2010, 18, 1406–1414. [Google Scholar] [CrossRef]
- Garreau, Y. Oxidation products of gentisic acid in the presene of primary amines. Bull. Soc. Chim. Fr. 1950, 501–502. [Google Scholar]
- Garreau, Y. [Interaction of] phenols and sulfites. Ann. Chim. Applicata 1938, 10, 485–558. [Google Scholar]
- Torigoe, M. Cancerocidal substances. XIII. Effect of some quinones and related compounds upon the Yoshida sarcoma. Pharm. Bull. 1955, 3, 337–342. [Google Scholar]
- Makarova, A.N.; Berlin, A.Y. Reaction of ethylenimino-1,4-benzoquinones with amines. I. Reaction between ethylenimino-1,4-benzoquinones and secondary amines. Zh. Org. Khim. 1959, 29, 666–672. [Google Scholar]
- Cavalitto, C.J.; Soria, A.E.; Hoppe, J.O. Amino- and ammonium-alkylaminobenzoquinones as curarimimetic agents. J. Am. Chem. Soc. 1950, 72, 2661–2665. [Google Scholar] [CrossRef]
- Barbosa, L.C.A.; Pereira, U.A.; Teixeira, R.R.; Maltha, C.R.A.; Fernandes, S.A.; Forlani, G. Synthesis and phytotoxic activity of ozonides. J. Agric. Food Chem. 2008, 56, 9434–9440. [Google Scholar]
- Barbosa, L.C.A.; Maltha, C.R.A.; Cusati, R.C.; Teixeira, R.R.; Rodrigues, F.F.; Silva, A.A.; Drew, M.G.B.; Ismail, F.M.D. Synthesis and biological evaluation of new ozonides with improved plant growth regulatory activity. J. Agric. Food Chem. 2009, 57, 10107–10115. [Google Scholar]
- Gomes, F.P. Curso de Estatística Experimental, 13th ed; Nobel: Piracicaba, Brasil, 1990. [Google Scholar]
- Macías, M.L.; Rojas, I.S.; Matar, R.; Lotina-Hennsen, B. Effect of selected coumarins on spinach chloroplast photosynthesis. J. Agric. Food Chem. 1999, 47, 2137–2140. [Google Scholar] [CrossRef]
- Strain, H.H.; Coppe, B.T.; Svec, W.A. Analytical procedures for the isolation, identification, estimation and investigation of the chlorophylls. In Methods of Enzymology; San Pietro, A., Ed.; Academic Press: New York, NY, USA, 1971; p. 452. [Google Scholar]
- Demuner, A.J.; Barbosa, L.C.A.; Veiga, T.A.M.; Barreto, R.W.; King-Diaz, B.; Lotina-Hennsen, B. Phytotoxic constituents from Nimbya alternantherae. Bioch. Syst. Ecol. 2006, 34, 790–795. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Almeida Barbosa, L.C.; Alves Pereira, U.; Alvares Maltha, C.R.; Ricardo Teixeira, R.; Moreira Valente, V.M.; Oliveira Ferreira, J.R.; Costa-Lotufo, L.V.; Odorico Moraes, M.; Pessoa, C. Synthesis and Biological Evaluation of 2,5-Bis(alkylamino)-1,4-benzoquinones. Molecules 2010, 15, 5629-5643. https://doi.org/10.3390/molecules15085629
Almeida Barbosa LC, Alves Pereira U, Alvares Maltha CR, Ricardo Teixeira R, Moreira Valente VM, Oliveira Ferreira JR, Costa-Lotufo LV, Odorico Moraes M, Pessoa C. Synthesis and Biological Evaluation of 2,5-Bis(alkylamino)-1,4-benzoquinones. Molecules. 2010; 15(8):5629-5643. https://doi.org/10.3390/molecules15085629
Chicago/Turabian StyleAlmeida Barbosa, Luiz Cláudio, Ulisses Alves Pereira, Célia Regina Alvares Maltha, Róbson Ricardo Teixeira, Vânia Maria Moreira Valente, José Roberto Oliveira Ferreira, Letícia Veras Costa-Lotufo, Manoel Odorico Moraes, and Cláudia Pessoa. 2010. "Synthesis and Biological Evaluation of 2,5-Bis(alkylamino)-1,4-benzoquinones" Molecules 15, no. 8: 5629-5643. https://doi.org/10.3390/molecules15085629
APA StyleAlmeida Barbosa, L. C., Alves Pereira, U., Alvares Maltha, C. R., Ricardo Teixeira, R., Moreira Valente, V. M., Oliveira Ferreira, J. R., Costa-Lotufo, L. V., Odorico Moraes, M., & Pessoa, C. (2010). Synthesis and Biological Evaluation of 2,5-Bis(alkylamino)-1,4-benzoquinones. Molecules, 15(8), 5629-5643. https://doi.org/10.3390/molecules15085629




