Synthesis and Effect on Human HepG2 Cells of 1,2-bis- (2-Methylallyl)disulfane
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structural characterization of 1,2-bis(2-methylallyl)disulfane
2.1.1. GC-MS analysis results
| No. | Peaks detected in GC-MS ( m/z) | Possible groups | Accurate m/z value for the proposed groups |
|---|---|---|---|
| 1 | 39-43 | 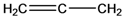 | 40 |
| 2 | 41-43 |  | 41 |
| 3 | 55 |  | 55 |
| 4 | 57 | Not characterized | |
| 5 | 71 | Not characterized | |
| 6 | 81 | Not characterized | |
| 7 | 86-88 | 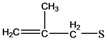 | 87 |
| 8 | 113 | Not characterized | |
| 9 | 118-121 | 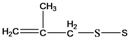 | 119 |
| 10 | 133 | 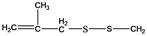 | 133 |
| 11 | 174 | 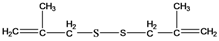 | 174 |
2.1.2. 1H-NMR analysis of 1,2-bis(2-methylallyl)disulfane
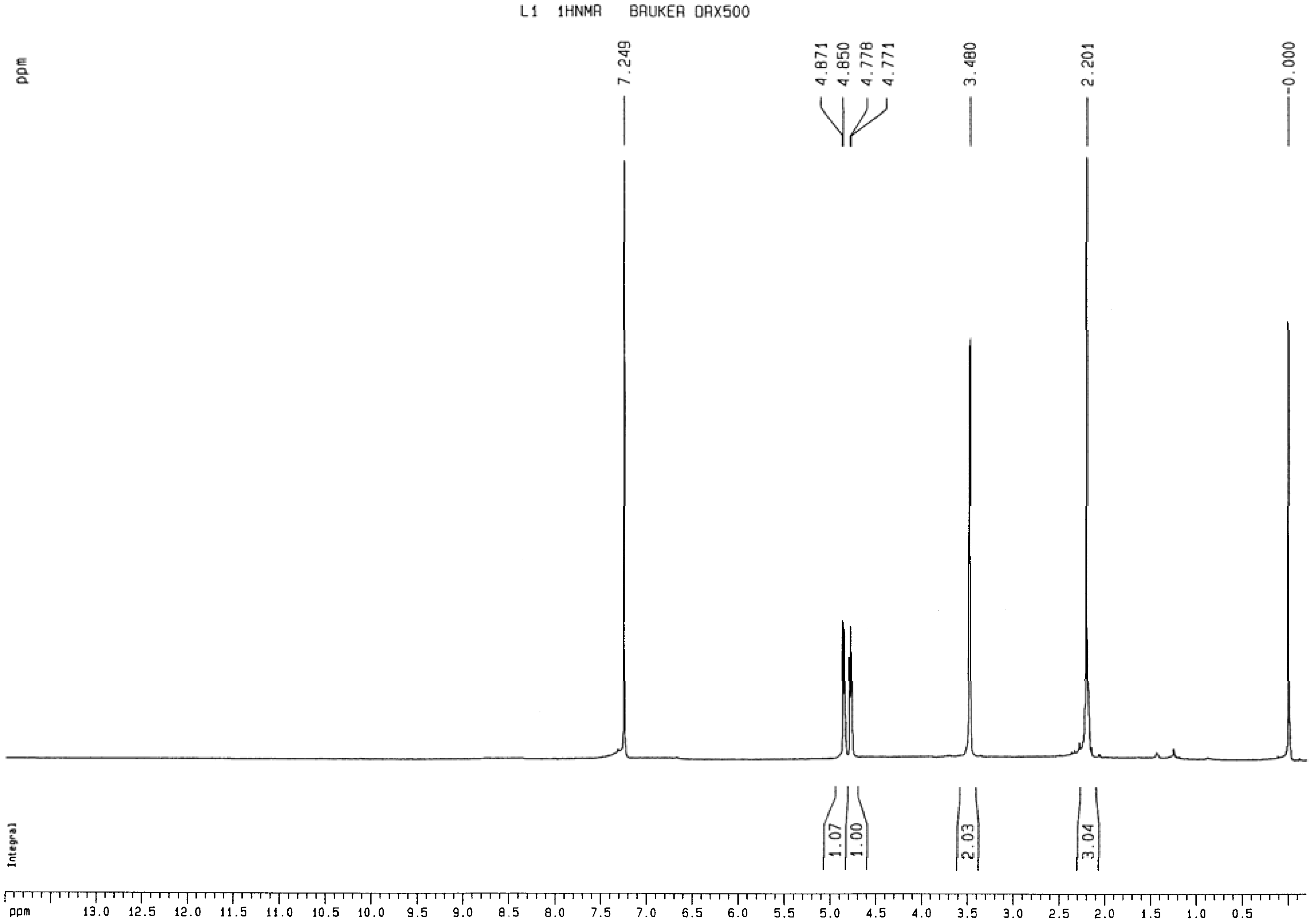
2.1.3. Elemental analysis of 1,2-bis(2-methylallyl)disulfane
| Elemental | C | H | S |
|---|---|---|---|
| Found (%) | 54.93 | 8.01 | 36.01 |
| Calculated (%) | 55.12 | 8.09 | 36.79 |
2.2. The effect of 1,2-bis(2-methylallyl)disulfane on human HepG2 cells
2.2.1. Cell activity
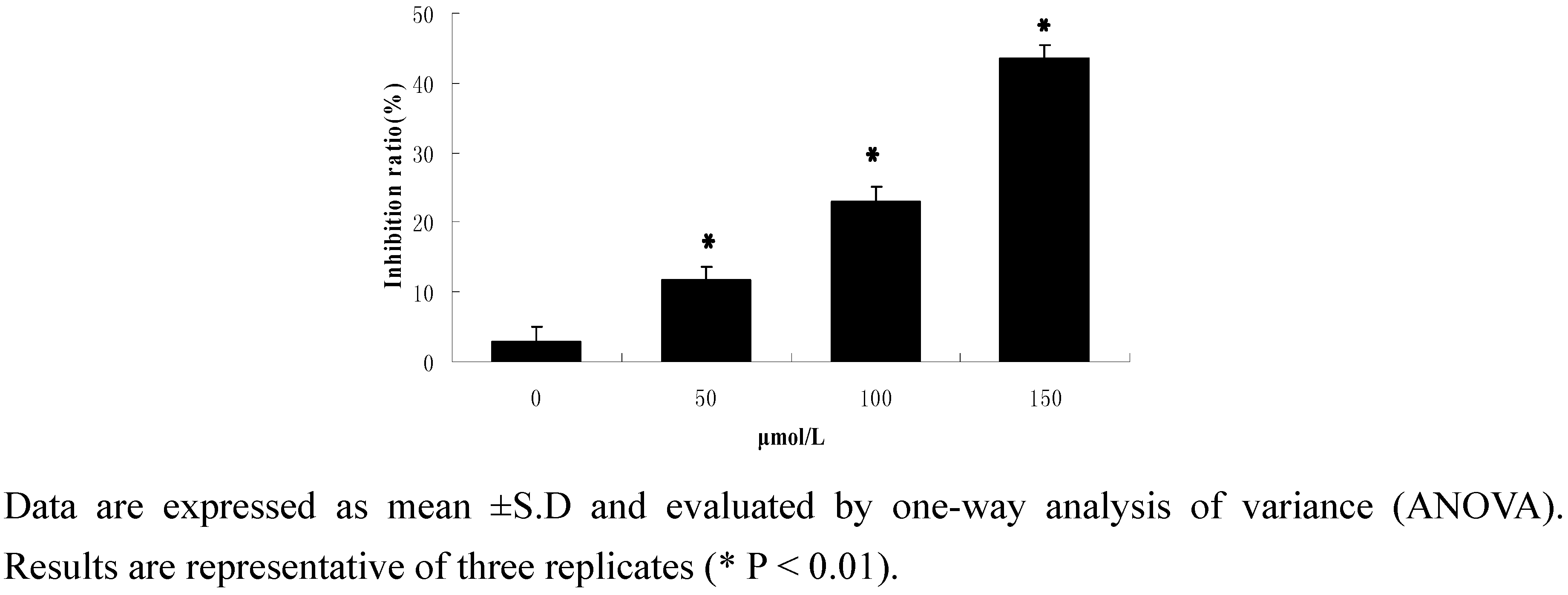
2.2.2. Flow-cytometric analysis of apoptosis
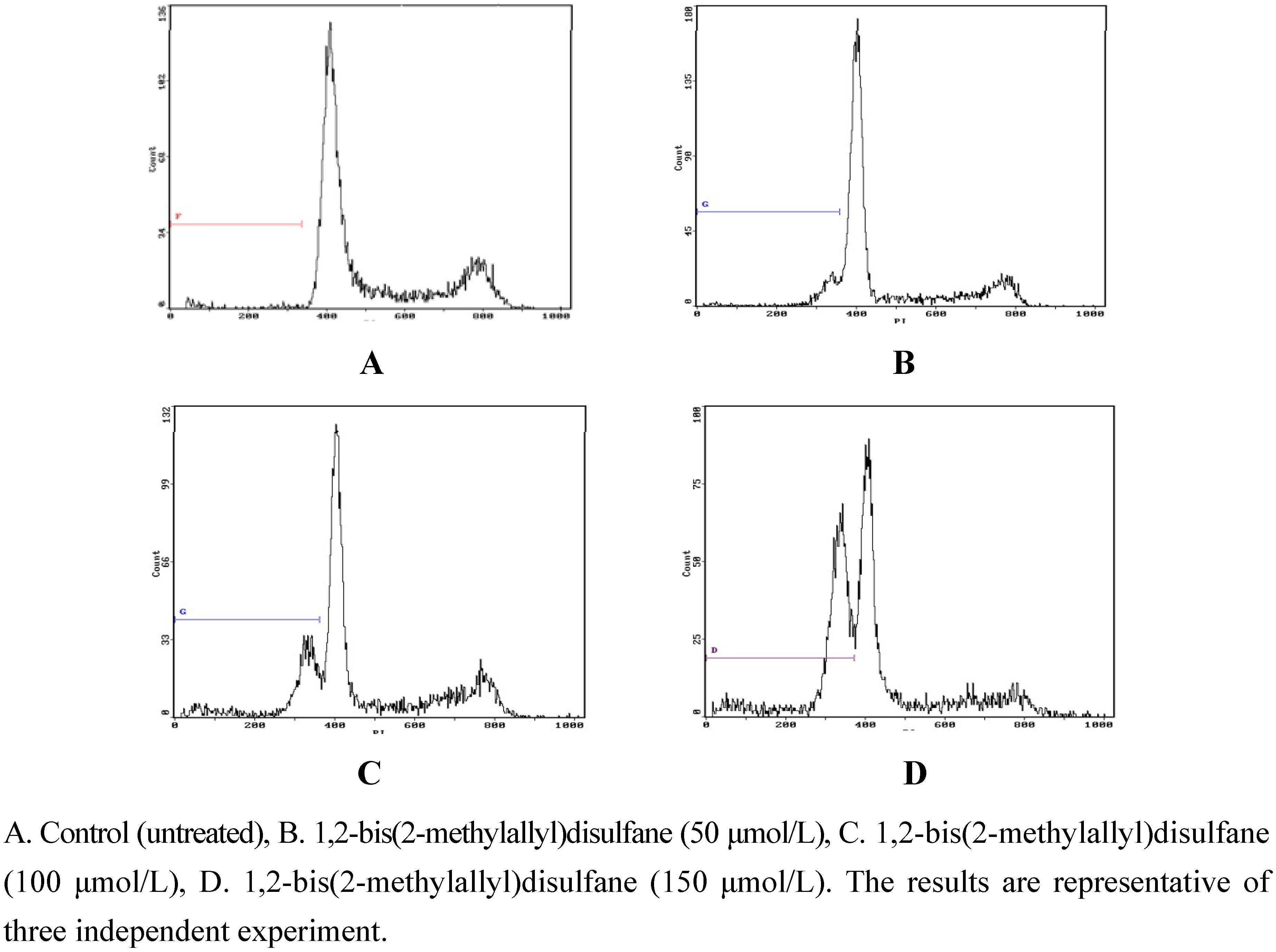
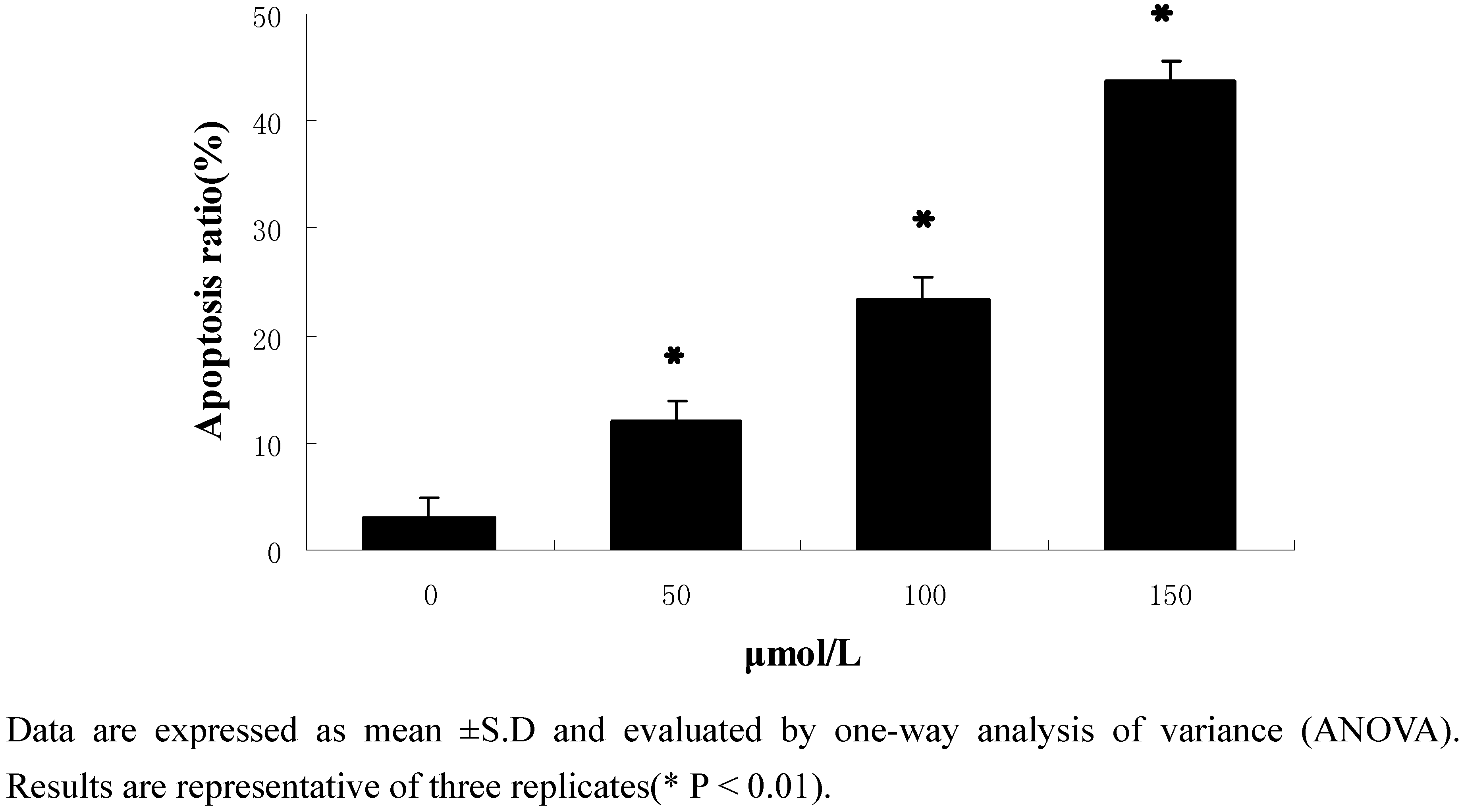
2.2.3. Protein expression
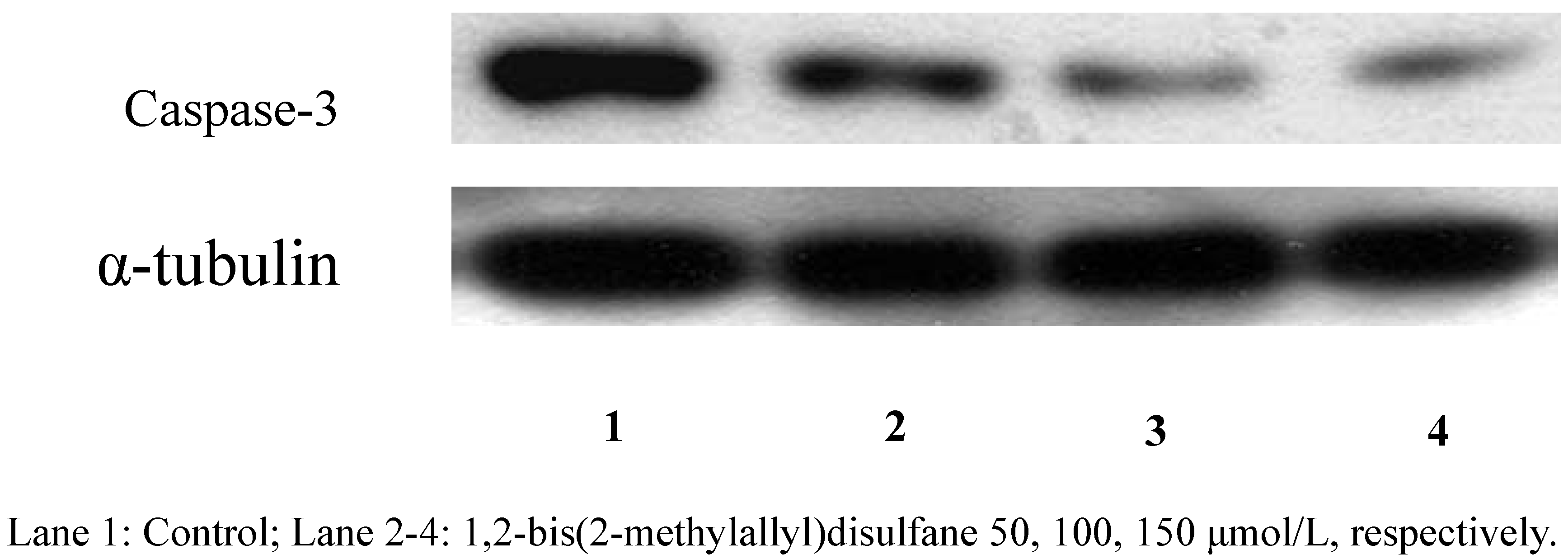
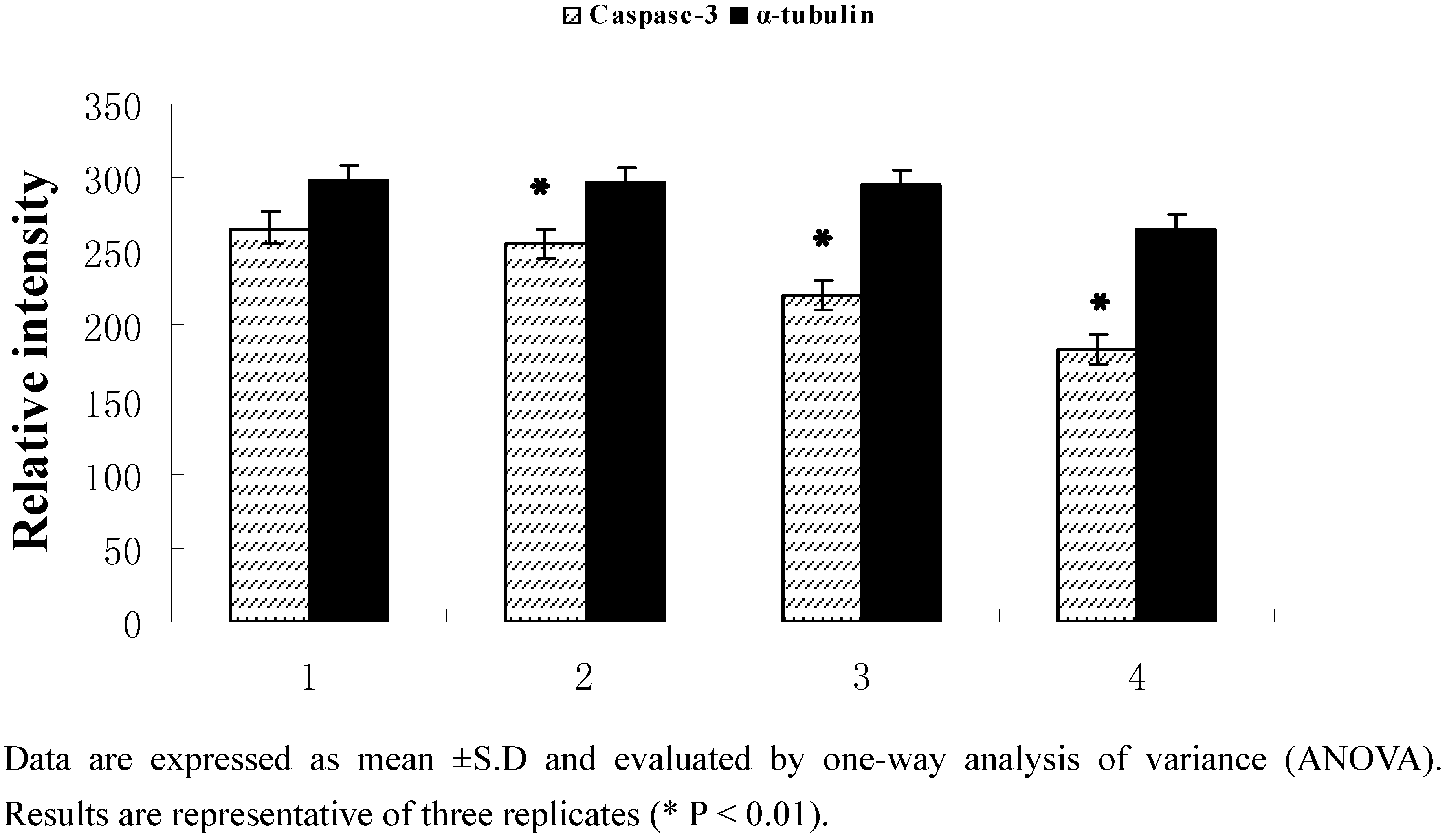
3. Experimental
3.1. General
3.2. Synthesis of 1,2-bis(2-methylallyl)disulfane

3.3. Cell culture
3.4. Cell viability assay
3.5. Flow cytometry analysis
3.6. Western-blotting
3.7. Statistics
4. Conclusions
References
- Sundaram, S.G.; Milner, J.A. Diallyl disulfide induces apoptosis of human colon tumor cells. Carcinogenesis 1996, 17, 669–673. [Google Scholar] [CrossRef]
- Kwon, K.B.; Yoo, S.J.; Ryu, D.G.; Yang, J.Y.; Rho, H.W.; Kim, J.S. Induction ofapoptosis by diallyl disulfide through activation of Caspase-3 in human leukemia HL-60 cells. Biochem. Pharmacol. 2002, 63, 41–47. [Google Scholar]
- Nakagawa, H.; Tsuta, K.; Kiuchi, K.; Senzaki, H.; Tanaka, K.; Hioki, K. Growth inhibitory effects of diallyl disfulfide on human breast cancer cell lines. Carcinogenesis 2001, 22, 891–897. [Google Scholar] [CrossRef]
- Wen, J.; Zhang, Y.W.; Chen, X.Q.; Shen, L.B.; Li, G.C.; Xu, M. Enhancement of diallyl disulfide-induced apoptosis by inhibitors of MAPKs in human HepG2 hepatoma cells. Biochem. Pharmacol. 2004, 68, 323–331. [Google Scholar]
- Druesne-Pecollo, N.; Pagniez, A.; Thomas, M.; Cherbuy, C.; Duee, P.H.; Martel, P.; Chaumontet, C. Diallyl Disulfide Increases CDKN1A Promoter-Associated Histone Acetylation in Human Colon Tumor Cell Lines. Agric. Food Chem. 2006, 54, 7503–7507. [Google Scholar]
- Bottone, F.G., Jr.; Baek, S.J.; Nixon, J.B.; Eling, T.E. Diallyl disulfide (DADS) induces the antitumorigenic NSAID-activated gene (NAG-1) by a p53 dependent mechanism in human colorectal HCT 116 cells. J. Nutr. 2002, 132, 773–778. [Google Scholar]
- Hong, Y.S.; Ham, Y.A.; Choi, J.H.; Kim, J. Effects of ally sulfur compounds and garlic extract on expressions of Bcl22, Bax, and p53 in nonsmall cell lung cancer cell lines. Exp. Mol. Med. 2000, 32, 127–134. [Google Scholar]
- Tsai, C.W.; Chen, H.W.; Yang, J.J.; Sheen, L.Y.; Lii, C.K. Diallyl Disulfide and Diallyl Trisulfide Up-Regulate the Expression of the π Class of Glutathione S-Transferase via an AP-1-Dependent Pathway. Agric. Food Chem. 2007, 55, 1019–1026. [Google Scholar] [CrossRef]
- Nabekura, T.; Kamiyama, S.; Kitagawa, S. Effects of dietary chemopre2 ventive phytochemicals on P-glycoprotein function. Biochem. Biophys. Res. Commun. 2005, 327, 866–870. [Google Scholar] [CrossRef]
- Xiao, D.; Choi, S.; Johnson, D.E. Diallyl trisulfide - induced apoptosis in human prostate cancer cells is mediated by activation of c-Jun N-terminal kinase and extracellular-signal regulated kinase mediated phosphoryl ation of Bcl-2. Oncogene 2004, 23, 5594–5606. [Google Scholar] [CrossRef]
- Srivastava, A.; Akoh, C.C.; Fischer, J.; Krewer, G. Effect of Anthocyanin Fractions from Selected Cultivars of Georgia-Grown Blueberries on Apoptosis and Phase II Enzymes. Agric. Food Chem. 2007, 55, 3180–3185. [Google Scholar] [CrossRef]
- Zhang, E.J.; Ng, K.M.; Luo, K.Q. Extraction and Purification of Isoflavones from Soybeans and Characterization of Their Estrogenic Activities. Agric. Food Chem. 2007, 55, 6940–6950. [Google Scholar] [CrossRef]
- Toshikazu, T.; Daisaku, S.; Yoshimasa, M.; Nobuo, Y.; Nobuhiro, K. Aromatic Hydrocarbon-Catalyzed Direct Reaction of Sulfur and Sodium in a Heterogeneous System: Selective and Facile Synthesis of Sodium Monosulfide and Disulfide. Inorg. Chem. 2003, 42, 3712–3714. [Google Scholar] [CrossRef]
- Yoon, H.; Liu, R.H. Effect of Selected Phytochemicals and Apple Extracts on NF-κB Activation in Human Breast Cancer MCF-7 Cells. Agric. Food Chem. 2007, 55, 3167–3173. [Google Scholar] [CrossRef]
- Dorant, E.; Van den Brandi, P.A.; Goldbohm, R.A.; Hermus, R.J.; Sturmans, F. Garlic and its significance for the prevention of cancer in humans. Br. J. Cancer 1993, 67, 424–429. [Google Scholar] [CrossRef]
- Kern, M.; Pahlke, G.; Balavenkatraman, K.K.; Bohmer, F.D.; Marko, D. Apple Polyphenols Affect Protein Kinase C Activity and the Onset of Apoptosis in Human Colon Carcinoma Cells. Agric. Food Chem. 2007, 55, 4999–5006. [Google Scholar]
- Gedye, R.; Smith, F.; Westaway, K.; Ali, H.; Baldisera, L.; Laberge, L.; Rousell, J. The use of microwave ovens for rapid organic synthesis. Tetrahedron Lett. 1986, 27, 279–282. [Google Scholar]
- Hansen, M.B.; Nielsen, S.E.; Berg, K. Re-examination and further development of a precise and rapid dye method for measuring cell grewth/cell kil1. Immunol. Meth. 1989, 119, 203–210. [Google Scholar] [CrossRef]
- Gerard, M.; Michael, F. Organoleptic characteristics of flavor materials. Perfumer Flavorist 1992, l7, 41–42. [Google Scholar]
- Roman, K.; Jan, V. Sulfur-containing volatiles arising by thermal degradation of alliin and deoxyalliin. J. Agric. Food Chem. 1997, 45, 3580–3585. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2010 by the authors;
Share and Cite
Ji, C.; Ren, F.; Dai, J.; Xu, M. Synthesis and Effect on Human HepG2 Cells of 1,2-bis- (2-Methylallyl)disulfane. Molecules 2010, 15, 3634-3642. https://doi.org/10.3390/molecules15053634
Ji C, Ren F, Dai J, Xu M. Synthesis and Effect on Human HepG2 Cells of 1,2-bis- (2-Methylallyl)disulfane. Molecules. 2010; 15(5):3634-3642. https://doi.org/10.3390/molecules15053634
Chicago/Turabian StyleJi, Chunxiao, Fenglian Ren, Jun Dai, and Ming Xu. 2010. "Synthesis and Effect on Human HepG2 Cells of 1,2-bis- (2-Methylallyl)disulfane" Molecules 15, no. 5: 3634-3642. https://doi.org/10.3390/molecules15053634
APA StyleJi, C., Ren, F., Dai, J., & Xu, M. (2010). Synthesis and Effect on Human HepG2 Cells of 1,2-bis- (2-Methylallyl)disulfane. Molecules, 15(5), 3634-3642. https://doi.org/10.3390/molecules15053634




