Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Anti-bacterial activity
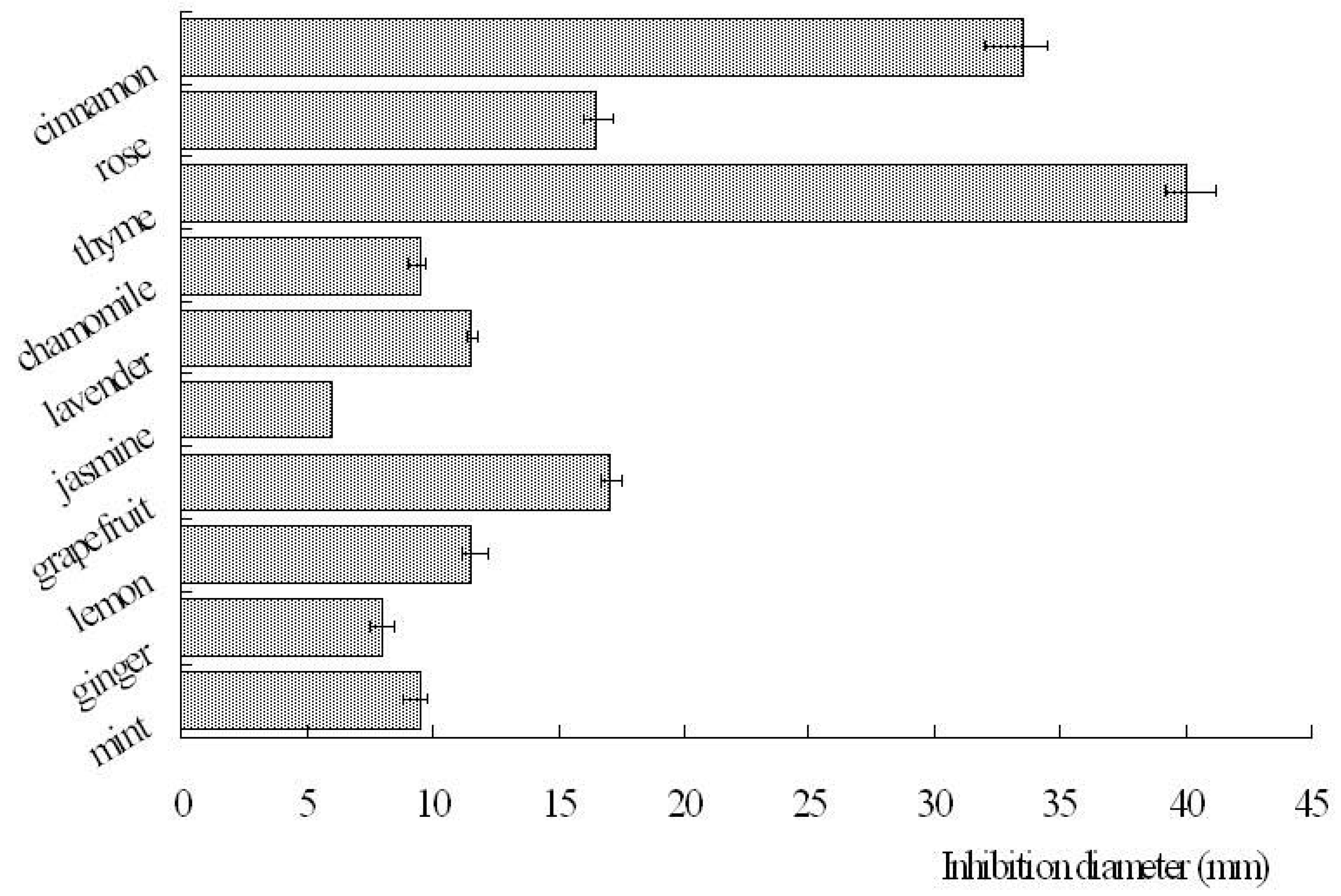
| cinnamon | rose | thyme | chamomile | lavender | jasmine | grapefruit | lemon | ginger | mint | |
| MIC | 0.016 | 0.031 | 0.016 | 0.125 | 0.125 | 0.500 | 0.250 | 0.250 | 0.250 | 0.250 |
| MBC | 0.016 | 0.031 | 0.016 | 0.125 | 0.125 | 0.500 | 0.250 | 0.250 | 0.250 | 0.250 |
2.2. Time-kill curves
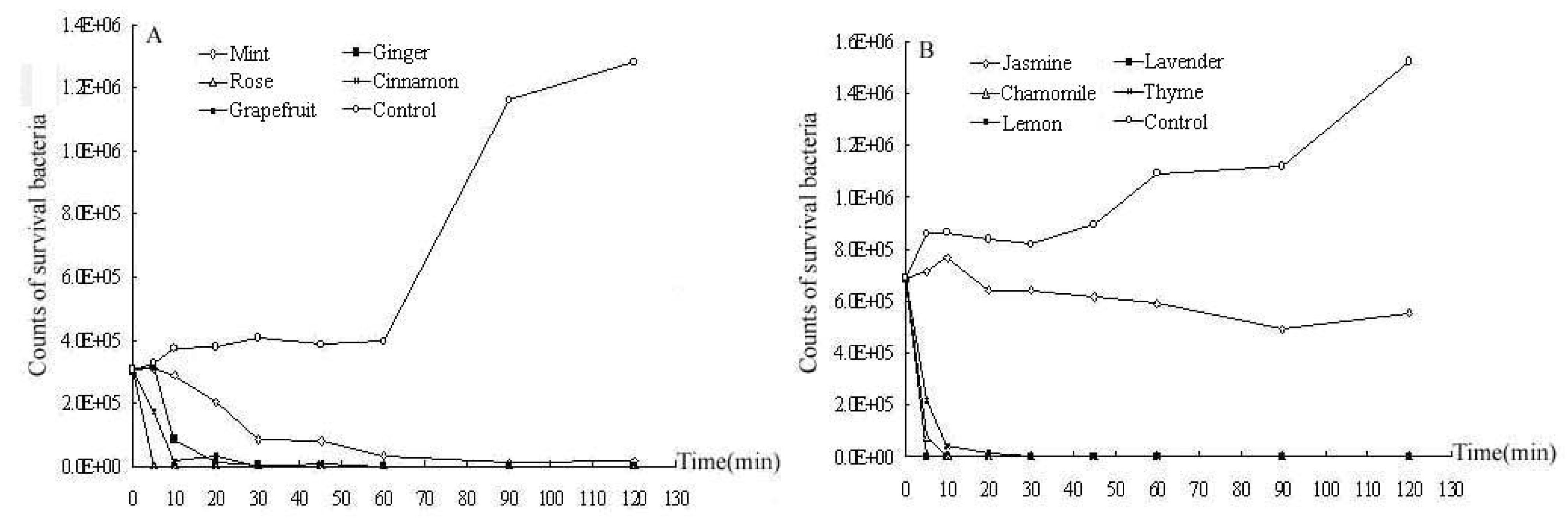
2.3. Cytotoxic activity towards cancer cells
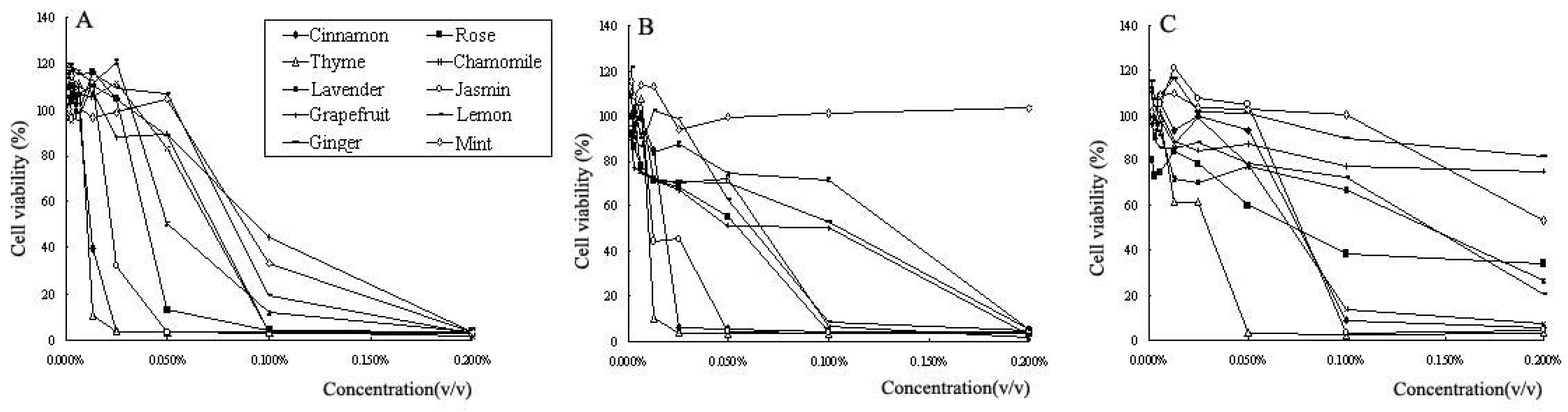
| cinnamon | rose | thyme | chamomile | lavender | jasmine | grapefruit | lemon | ginger | mint | |
| PC-3 | 0.012 | 0.040 | 0.010 | 0.071 | 0.050 | 0.022 | 0.094 | 0.083 | 0.077 | 0.088 |
| A549 | 0.017 | 0.055 | 0.011 | 0.067 | 0.133 | 0.012 | 0.100 | 0.061 | 0.107 | ------- |
| MCF-7 | 0.076 | 0.074 | 0.030 | 0.072 | 0.142 | 0.077 | ------- | 0.143 | ------- | ------- |
3. Experimental
3.1. Essential oils
3.2. Maintenance of Proprionibacterium acnes
3.3. Disc diffusion assay
3.4. Determinition of MICs and MBCs values
3.5. Time-kill dynamic curves
3.6. Maintenance of human cancer cell lines
3.7. Cytotoxicity assay
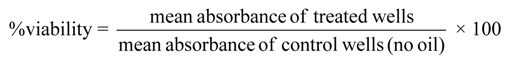
4. Conclusions
Acknowledgements
References and Notes
- Buckle, J. Use of aromatherapy as a complementary treatment for chronic pain. Altern Ther Health Med. 1999, 5, 42–51. [Google Scholar]
- Sylvestre, M.; Pichette, A.; Longtin, A.; Nagau, F.; Legault, J. Essential oil analysis and anticancer activity of leaf essential oil of Croton flavens L. from Guadeloupe. J. Ethnopharmacol. 2006, 103, 99–102. [Google Scholar] [CrossRef]
- Mimica-Dukic, N.; Bozin, B.; Sokovic, M.; Simin, N. Antimicrobial and antioxidant activities of Melissa officinalis L. (Lamiaceae) essential oil. J. Agric. Food. Chem. 2004, 52, 2485–2489. [Google Scholar] [CrossRef]
- Sylvestre, M.; Legault, J.; Dufour, D.; Pichette, A. Chemical composition and anticancer activity of leaf essential oil of Myrica gale L. Phytomedicine 2005, 12, 299–304. [Google Scholar] [CrossRef]
- Park, J.; Lee, J.; Jung, E.; Park, Y.; Kim, K.; Park, B.; Jung, K.; Park, E.; Kim, J.; Park, D. In vitro antibacterial and anti-inflammatory effects of honokiol and magnolol against Propionibacterium sp. Eur. J. Pharmacol. 2004, 496, 189–195. [Google Scholar] [CrossRef]
- Jappe, U. Pathological mechanisms of acne with special emphasis on Propionibacterium acnes and related therapy. Acta Derm. Venereo. 2003, 83, 241–248. [Google Scholar] [CrossRef]
- Poulin, Y. Practical approach to the hormonal treatment of acne. J. Cutan. Med. Surg. 2004, 4, 6–21. [Google Scholar]
- Tan, A.W.; Tan, H.H. Acne vulgaris: a review of antibiotic therapy. Expert Opin. Pharmacother. 2005, 6, 409–418. [Google Scholar]
- Leyden, J.J. Antibiotic resistance in the topical treatment of acne vulgaris. Cutis 2004, 73, 6–10. [Google Scholar]
- Yemisci, A.; Gorgulu, A.; Piskin, S. Effects and side-effects of spironolactone therapy in women with acne. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 163–166. [Google Scholar] [CrossRef]
- Lertsatitthanakorn, P.; Taweechaisupapong, S.; Aromdee, C.; Khunkitti, W. In vitro bioactivities of essential oils used for acne control. Int. J. Astrobiol. 2006, 16, 43–49. [Google Scholar]
- Efferth, T.; Fu, Y.J.; Zu, Y.G.; Schwarz, G.; Konkimall, V.S.; Wink, M. Molecular target-guided tumor therapy with natural products derived from traditional Chinese medicine. Curr. Med. Chem. 2007, 14, 2024–2032. [Google Scholar] [CrossRef]
- Lai, P.K.; Roy, J. Antimicrobial and chemopreventive properties of herbs and spices. Curr. Med. Chem. 2004, 11, 1451–1460. [Google Scholar] [CrossRef]
- Soković, M.D.; Vukojević, J.; Marin, P.D.; Brkić, D.D.; Vajs, V.; van Griensven, L.J. Chemical composition of essential oils of Thymus and Mentha species and their antifungal activities. Molecules 2009, 14, 238–249. [Google Scholar] [CrossRef]
- Sabulal, B.; Dan, M.; Kurup, R.; Pradeep, N.S.; Valsamma, R.K.; George, V. Caryophyllene-rich rhizome oil of Zingiber nimmonii from South India: Chemical characterization and antimicrobial activity. Phytochemistry 2006, 67, 2469–2473. [Google Scholar] [CrossRef]
- Ganzera, M.; Schneider, P.; Stuppner, H. Inhibitory effects of the essential oil of chamomile (Matricaria recutita L.) and its major constituents on human cytochrome P450 enzymes. Life Sci. 2006, 78, 856–861. [Google Scholar] [CrossRef]
- Shen, J.; Niijima, A.; Tanida, M.; Horii, Y.; Nakamura, T.; Nagai, K. Mechanism of changes induced in plasma glycerol by scent stimulation with grapefruit and lavender essential oils. Neurosci. Lett. 2007, 416, 241–246. [Google Scholar] [CrossRef]
- Hamdan, D.; El-Readi, M.Z.; Nibret, E.; Sporer, F.; Farrag, N.; El-Shazly, A.; Wink, M. Chemical composition of the essential oils of two Citrus species and their biological activities. Pharmazie 2010, 65, 141–147. [Google Scholar]
- Lauriola, M.M.; De Bitonto, A.; Sena, P. Allergic contact dermatitis due to cinnamon oil in galenic vaginal suppositories. Acta Derm. Venereol. 2010, 90, 187–188. [Google Scholar] [CrossRef]
- Wei, A.; Shibamoto, T. Antioxidant activities and volatile constituents of various essential oils. J. Agric. Food Chem. 2007, 55, 1737–1742. [Google Scholar] [CrossRef]
- Mourey, A.; Canillac, N. Anti-Listeria monocytogenes activity of essential oils components of conifers. Food Contr. 2002, 13, 289–292. [Google Scholar] [CrossRef]
- Oumzil, H.; Ghoulami, S.; Rhajaoui, M.; Ilidrissi, A.; Fkih-Tetouani, S.; Faid, M.; Benjouad, A. Antibacterial and antifungal activity of essential oils of Mentha suaveolens. Phytother. Res. 2002, 16, 727–731. [Google Scholar] [CrossRef]
- Legault, J.; Dahl, W.; Debiton, E.; Pichette, A.; Madelmont, J.C. Antitumor activity of balsam fir oil: production of reactive oxygen species induced by alpha-humulene as possible mechanism of action. Planta Med. 2003, 69, 402–407. [Google Scholar] [CrossRef]
- Wiseman, D.A.; Werner, S.R.; Crowell, P.L. Cell cycle arrest by the isoprenoids perillyl alcohol, geraniol, and farnesol is mediated by p21 (Cip1) and p27 (Kip1) in human pancreatic adenocarcinoma cells. J. Pharmacol. Exp. Ther. 2007, 320, 1163–1170. [Google Scholar]
- Gören, A.; Topçu, G.; Bilsel, G.; Bilsel, M.; Aydoğmuş, Z.; Pezzuto, J.M. The chemical constituents and biological activity of essential oil of Lavandula stoechas ssp. stoechas. Z. Naturforsch C. 2002, 57, 797–800. [Google Scholar]
- Hata, T.; Sakaguchi, I.; Mori, M.; Ikeda, N.; Kato, Y.; Minamino, M.; Watabe, K. Induction of apoptosis by Citrus paradisi essential oil in human leukemic (HL-60) cells. In Vivo 2003, 17, 553–559. [Google Scholar]
- Ait M'barek, L.; Ait Mouse, H.; Jaâfari, A.; Aboufatima, R.; Benharref, A.; Kamal, M.; Bénard, J.; El Abbadi, N.; Bensalah, M.; Gamouh, A.; Chait, A.; Dalal, A.; Zyad, A. Cytotoxic effect of essential oil of thyme (Thymus broussonettii) on the IGR-OV1 tumor cells resistant to chemotherapy. Braz. J. Med. Biol. Res. 2007, 40, 1537–1544. [Google Scholar] [CrossRef]
- Hernández, T.; Canales, M.; Avila, J.G.; García, A.M.; Martínez, A.; Caballero, J.; de Vivar, A.R.; Lira, R. Composition and antibacterial activity of essential oil of Lantana achyranthifolia Desf. (Verbenaceae). J. Ethnopharmacol. 2005, 96, 551–554. [Google Scholar] [CrossRef]
- Yu, J.Q.; Lei, J.C.; Yu, H.D.; Cai, X.; Zou, G.L. Chemical composition and antimicrobial activity of the essential oil of Scutellaria barbata. Phytochemistry. 2004, 65, 881–884. [Google Scholar] [CrossRef]
- Avila, J.G.; de Liverant, J.G.; Martínez, A.; Martínez, G.; Muñoz, J.L.; Arciniegas, A.; de Vivar, A.R. Mode of action of Buddleja cordata verbascoside against Staphylococcus aureus. J. Ethnopharmacol. 1999, 66, 75–78. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Meth. 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Tomaino, A.; Cimino, F.; Zimbalatti, V.; Venuti, V.; Sulfaro, V.; De Pasquale, A. Influence of heating on antioxidant activity and the chemical composition of some spice essential oils. Food Chem. 2005, 89, 549–554. [Google Scholar] [CrossRef]
- Falcone, P.M.; Mastromatteo, M.; Del Nobile, M.A.; Corbo, M.R.; Sinigaglia, M. Evaluating in vitro antimicrobial activity of thymol toward hygiene-indicating and pathogenic bacteria. J. Food Prot. 2007, 70, 425–431. [Google Scholar]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C.; Saija, A.; Mazzanti, G.; Bisignano, G. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef]
- Singh, G.; Maurya, S.; DeLampasona, M.P.; Catalan, C.A. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- Chami, N.; Bennis, S.; Chami, F.; Aboussekhra, A.; Remmal, A. Study of anticandidal activity of carvacrol and eugenol in vitro and in vivo. Oral Microbiol. Immunol. 2005, 20, 106–111. [Google Scholar] [CrossRef]
- Ghosh, R.; Nadiminty, N.; Fitzpatrick, J.E.; Alworth, W.L.; Slaga, T.J.; Kumar, A.P. Eugenol causes melanoma growth suppression through inhibition of E2F1 transcriptional activity. J. Biol. Chem. 2005, 280, 5812–5819. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2010 by the authors;
Share and Cite
Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells. Molecules 2010, 15, 3200-3210. https://doi.org/10.3390/molecules15053200
Zu Y, Yu H, Liang L, Fu Y, Efferth T, Liu X, Wu N. Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells. Molecules. 2010; 15(5):3200-3210. https://doi.org/10.3390/molecules15053200
Chicago/Turabian StyleZu, Yuangang, Huimin Yu, Lu Liang, Yujie Fu, Thomas Efferth, Xia Liu, and Nan Wu. 2010. "Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells" Molecules 15, no. 5: 3200-3210. https://doi.org/10.3390/molecules15053200
APA StyleZu, Y., Yu, H., Liang, L., Fu, Y., Efferth, T., Liu, X., & Wu, N. (2010). Activities of Ten Essential Oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 Cancer Cells. Molecules, 15(5), 3200-3210. https://doi.org/10.3390/molecules15053200




