Neuroprotective Effects of Herbal Ethanol Extracts from Gynostemma pentaphyllum in the 6-Hydroxydopamine-Lesioned Rat Model of Parkinson's Disease
Abstract
:1. Introduction
2. Results
2.1. TH-immunopositive neuronal cell survival in 6-OHDA-lesioned rats by GP-EX administration
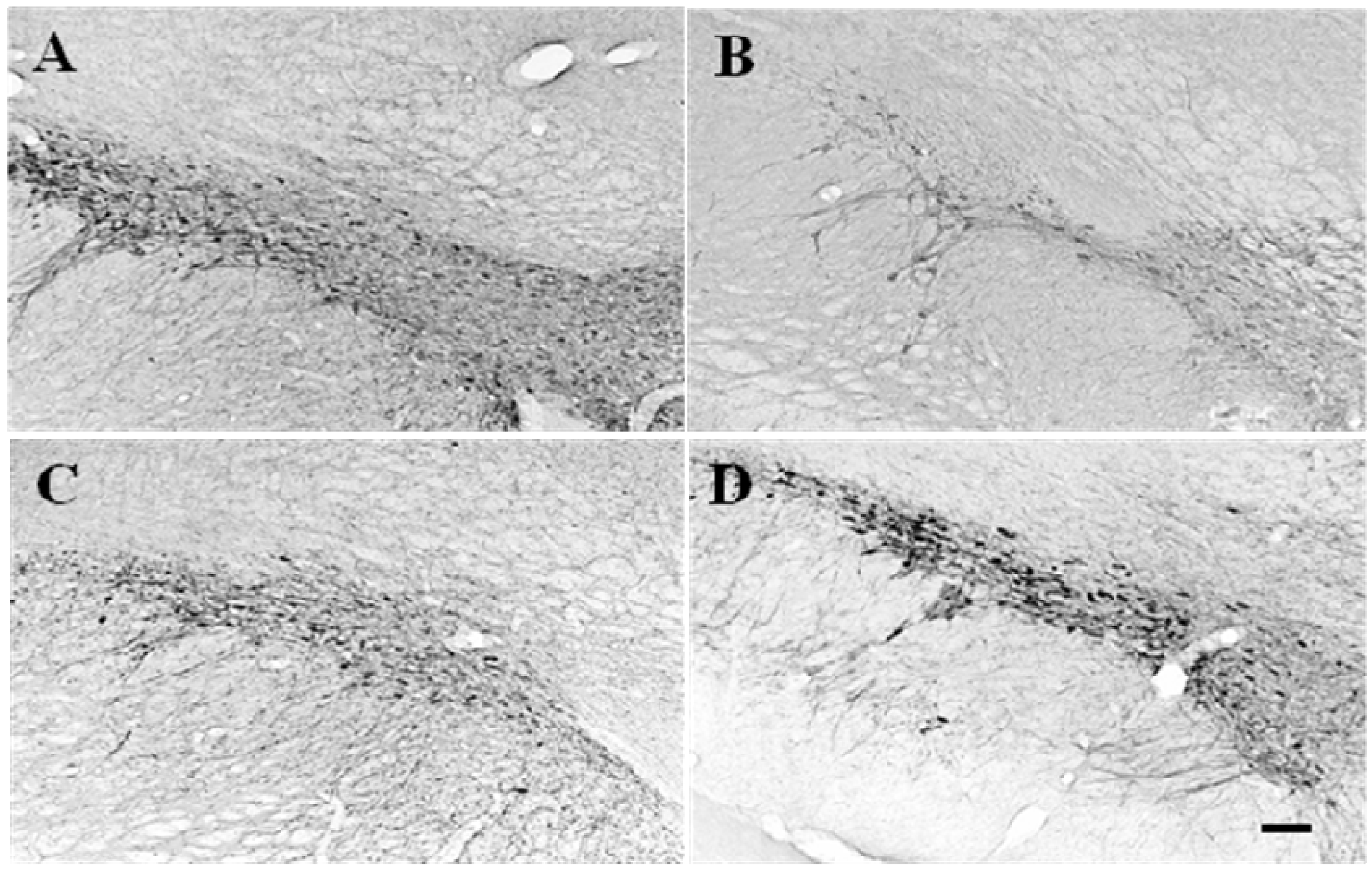
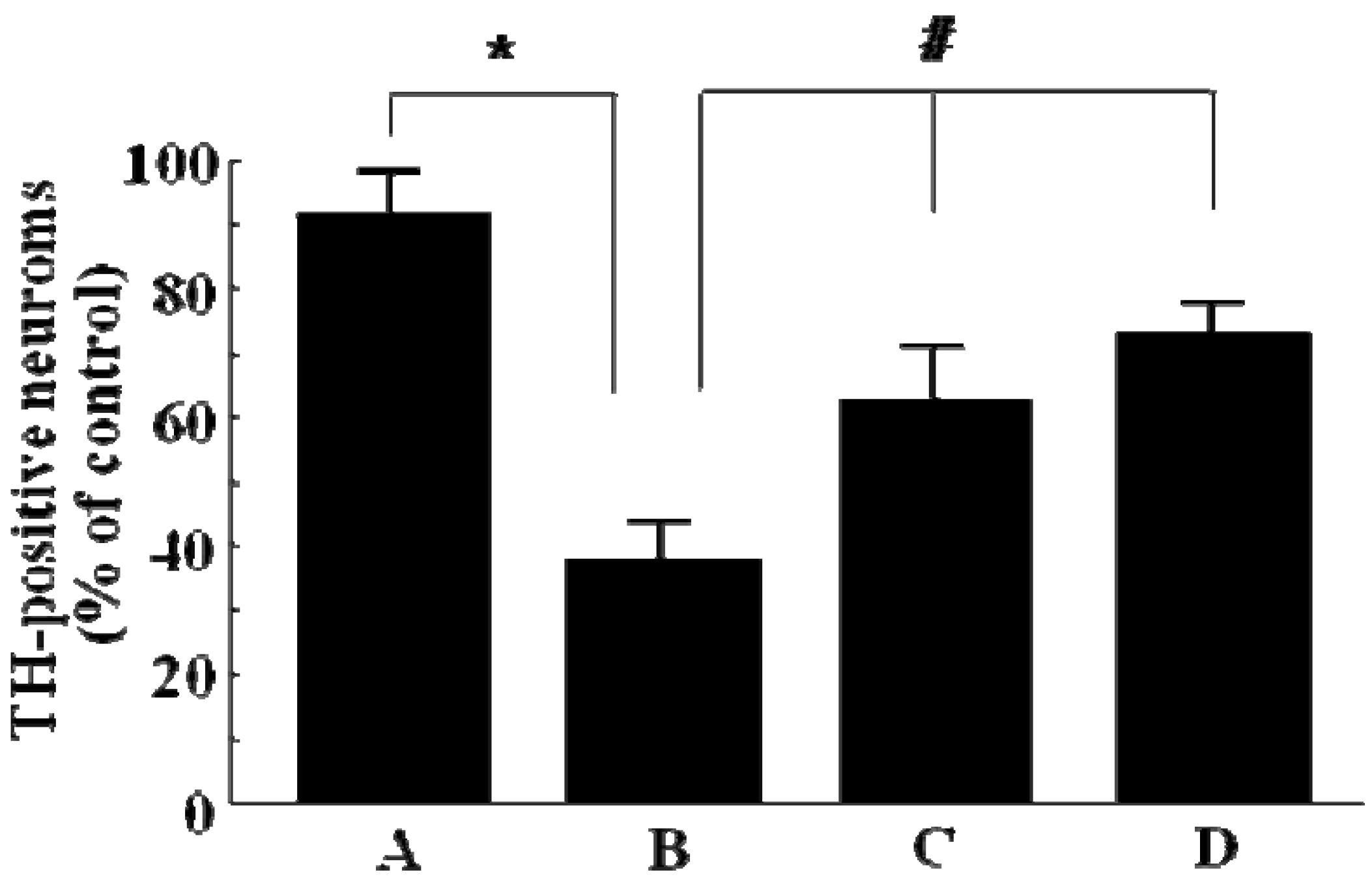
2.2. The levels of dopamine, DOPAC, HVA and norepinephrine in striatum of 6-OHDA-lesioned rats by GP-EX administration
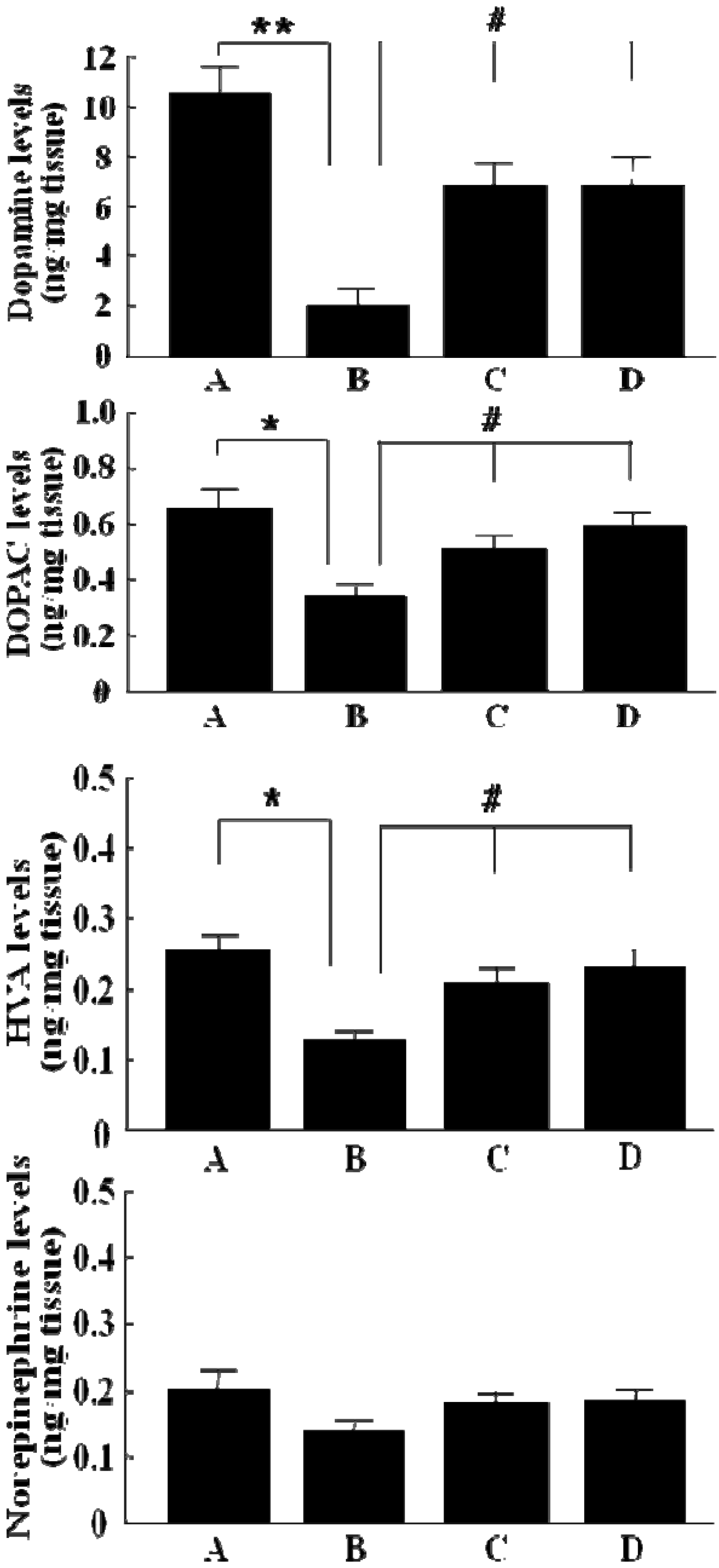
3. Discussion
4. Experimental
4.1. Materials
4.2. Preparation and treatment of GP-EX
4.3. Unilateral 6-OHDA lesion
4.4. TH immunohistochemistry
4.5. Determination of dopamine, DOPAC, HVA and norepinephrine
4.6. Separation of gypenosides from GP-EX
4.7. Statistical analysis
5. Conclusions
Acknowledgements
References
- Neil, A.; David, B. Parkinson’s disease. Medicine 2008, 36, 630–635. [Google Scholar] [CrossRef]
- Hornykiewicz, O. Imbalance of Brain monoamines and clinical disorders. Prog. Brain Res. 1982, 55, 419–429. [Google Scholar] [CrossRef]
- Marsden, C.D. Parkinson's disease. J. Neurol. Neurosurg. Psychiatry 1994, 57, 672–681. [Google Scholar] [CrossRef]
- Jankovic, J. Motor fluctuations and dyskinesias in Parkinson's disease: clinical manifestations. Mov. Disord. 2005, 20, S11–S16. [Google Scholar] [CrossRef]
- Perese, D.A.; Ulman, J.; Viola, J.; Ewing, S.E.; Bankiewicz, K.S. A 6-hydroxydopamine-induced selective parkinsonian rat model. Brain Res. 1989, 494, 285–293. [Google Scholar] [CrossRef]
- Schober, A. Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell Tissue Res. 2004, 318, 215–224. [Google Scholar] [CrossRef]
- Slater, T.F. Free-radical mechanisms in tissue injury. Biochem. J. 1984, 15, 1–15. [Google Scholar]
- Fahn, S.; Cohen, G. The oxidant stress hypothesis in Parkinson's disease: Evidence supporting it. Ann. Neurol. 1992, 32, 804–812. [Google Scholar] [CrossRef]
- Cadet, J.L.; Brannock, C. Free radicals and the pathobiology of brain dopamine systems. Neurochem. Int. 1998, 32, 117–131. [Google Scholar]
- Maharaj, H. L-DOPA administration enhances 6-hydroxydopamine generation. Brain Res. 2005, 1063, 180–186. [Google Scholar]
- Razmovski-Naumovski, V.; Huang, T.H-.W.; Tran, V.H.; Li, G.Q.; Duke, C.C.; Roufogalis, B.D. Chemistry and pharmacology of Gynostemma Pentaphyllum. Phytochem. Rev. 2005, 4, 197–219. [Google Scholar] [CrossRef]
- Choi, H.S.; Lim, S.A.; Park, M.S.; Hwang, B.Y.; Lee, C.K.; Kim, S.H.; Lim, S.C.; Lee, M.K. Ameliorating effects of the ethanol extracts from Gynostemma Pentaphyllum on electric footshock stress. Kor. J. Pharmacogn. 2008, 39, 341–346. [Google Scholar]
- Im, S.A.; Choi, H.S.; Hwang, B.Y.; Lee, M.K.; Lee, C.K. Augmentation of Immune Responses by oral administration of Gynostemma Pentaphyllum ethanol extract. Kor. J. Pharmacogn. 2009, 40, 35–40. [Google Scholar]
- Nagai, M.; Izawa, K.; Nagumo, S.; Sakurai, N.; Inoue, T. Two glycosides of a novel dammarane alcohol from Gynostemma Pentaphyllum. Chem. Pharm. Bull. 1981, 29, 779–783. [Google Scholar] [CrossRef]
- Takemoto, T.; Arihara, S.; Yoshikawa, K.; Kawasaki, J.; Nakajima, T.; Okuhira, M. Studies on the constituents of Cucurbitaceae plants. XI. On the saponin constituents of Gynostemma pentaphyllum MAKINO. (7). Yakugaku Zasshi 1984, 104, 1043–1049. [Google Scholar]
- Yoshikawa, K.; Arimitsu, M.; Kuki, K.; Takemoto, T.; Arihara, S. Studies on the constituents of Cucurbitaceae plants. XVIII. On the saponin constituents of Gynostemma pentaphyllum MAKINO. (13). Yakugaku Zasshi 1987, 107, 361–366. [Google Scholar]
- Ren, T.; Sagar, S.M. Induction of c-fos immunostaining in the rat brain after the systemic administration of nicotine. Brain Res. Bull. 1992, 29, 589–597. [Google Scholar] [CrossRef]
- Attawish, A.; Chivapat, S.; Phadungpat, S.; Bansiddhi, J.; Techadamrongsin, Y.; Mitrijit, O.; Chaorai, B.; Chavalittumrong, P. Chronic toxicity of Gynostemma pentaphyllum. Fitoterapia 2004, 75, 539–551. [Google Scholar] [CrossRef]
- Lin, J.M.; Lin, C.C.; Chiu, H.F.; Yang, J.J.; Lee, S.G. Evaluation of the anti-inflammatory and liver-protective effects of Anoectochilus formosanus, Ganoderma lucidum and Gynostemma pentaphyllum in rats. Am. J. Chin. Med. 1993, 21, 59–69. [Google Scholar] [CrossRef]
- Huang, W.C.; Kuo, M.L.; Li, M.L.; Yang, R.C.; Liou, C.J.; Shen, J.J. The extract of Gynostemma pentaphyllum enhanced the production of antibodies and cytokines in mice. Yakugaku Zasshi 2007, 127, 889–896. [Google Scholar] [CrossRef]
- Wang, Z.J.; Luo, D.H. Antioxidant activities of different fractions of polysaccharide purified from Gynostemma pentaphyllum Makino. Carbohyd. Polym. 2007, 68, 54–58. [Google Scholar] [CrossRef]
- Shang, L.; Liu, J.; Zhu, Q.; Zhao, L.; Feng, Y.; Wang, X.; Cao, W.; Xin, H. Gypenosides protect primary cultures of rat cortical cells against oxidative neurotoxicity. Brain Res. 2006, 1102, 163–174. [Google Scholar]
- Curtius, H.C.; Wolfensberger, M.; Steinmann, B.; Redweik, U.; Siegfried, J. Mass fragmentography of dopamine and 6-hydroxydopamine. Application to the determination of dopamine in human brain biopsies from the caudate nucleus. J. Chromatogr. 1974, 99, 529–540. [Google Scholar] [CrossRef]
- Severson, J.A.; Marcusson, J.; Winblad, B.; Finch, C.E. Age-correlated loss of dopaminergic binding sites in human basal ganglia. J. Neurochem. 1982, 39, 1623–1631. [Google Scholar] [CrossRef]
- Linert, W.; Herlinger, E.; Jameson, R.F.; Kienzl, E.; Jellinger, K.; Youdim, M.B. Dopamine, 6-hydroxydopamine, iron, and dioxygen—their mutual interactions and possible implication in the development of Parkinson's disease. Biochim. Biophys. Acta 1996, 1316, 160–168. [Google Scholar]
- Napolitano, A.; Pezzella, A.; Prota, G. New reaction pathways of dopamine under oxidative stress conditions: nonenzymatic iron-assisted conversion to norepinephrine and the neurotoxins 6-hydroxydopamine and 6,7-dihydroxytetrahydroisoquinoline. Chem. Res. Toxicol. 1999, 12, 1090–1097. [Google Scholar] [CrossRef]
- Basma, A.N.; Morris, E.J.; Nicklas, W.J.; Geller, H.M. L-dopa cytotoxicity to PC12 cells in culture is via its autoxidation. J. Neurochem. 1995, 64, 825–832. [Google Scholar]
- Cheng, N.N.; Maeda, T.; Kume, T.; Kaneko, S.; Kochiyama, H.; Akaike, A.; Goshima, Y.; Misu, Y. Differential neurotoxicity induced by L-DOPA and dopamine in cultured striatal neurons. Brain Res. 1996, 743, 278–283. [Google Scholar] [CrossRef]
- Yacoubian, T.A.; Standaert, D.C. Targets for neuroprotection in Parkinson’s disease. Biochim. Biophys. Acta 2009, 1792, 676–687. [Google Scholar]
- Myllylä, V.V.; Sotaniemi, K.A.; Vuorinen, J.A.; Heinonen, E.H. Selegiline as initial treatment in de novo parkinsonian patients. Neurology 1992, 42, 339–343. [Google Scholar]
- Przedborski, S.; Levivier, M.; Jiang, H.; Ferreira, M.; Jackson-Lewis, V.; Donaldson, D.; Togasaki, D.M. Dose-dependent lesions of the dopaminergic nigrostriatal pathway induced by intrastriatal injection of 6-hydroxydopamine. Neuroscience 1995, 67, 631–647. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 2nd ed; Academic Press: Tokyo, Australia, 1986. [Google Scholar]
- Hudson, J.L.; van Horne, C.G.; Strömberg, I.; Brock, S.; Clayton, J.; Masserano, J.; Hoffer, B.J.; Gerhardt, G.A. Correlation of apomorphine- and amphetamine-induced turning with nigrostriatal dopamine content in unilateral 6-hydroxydopamine lesioned rats. Brain Res. 1993, 626, 167–174. [Google Scholar] [CrossRef]
- Tariq, M.; Khan, H.A.; Al Moutaery, K.; Al Deeb, S. Protective effect of quinacrine on striatal dopamine levels in 6-OHDA and MPTP models of Parkinsonism in rodents. Brain Res. Bull. 2001, 54, 77–82. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds (gynosaponin TN-1, gynosaponin TN-2, gypenoside XLV and gypenoside LXXIV) are available from the authors.
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Choi, H.S.; Park, M.S.; Kim, S.H.; Hwang, B.Y.; Lee, C.K.; Lee, M.K. Neuroprotective Effects of Herbal Ethanol Extracts from Gynostemma pentaphyllum in the 6-Hydroxydopamine-Lesioned Rat Model of Parkinson's Disease. Molecules 2010, 15, 2814-2824. https://doi.org/10.3390/molecules15042814
Choi HS, Park MS, Kim SH, Hwang BY, Lee CK, Lee MK. Neuroprotective Effects of Herbal Ethanol Extracts from Gynostemma pentaphyllum in the 6-Hydroxydopamine-Lesioned Rat Model of Parkinson's Disease. Molecules. 2010; 15(4):2814-2824. https://doi.org/10.3390/molecules15042814
Chicago/Turabian StyleChoi, Hyun Sook, Mi Sook Park, Seung Hwan Kim, Bang Yeon Hwang, Chong Kil Lee, and Myung Koo Lee. 2010. "Neuroprotective Effects of Herbal Ethanol Extracts from Gynostemma pentaphyllum in the 6-Hydroxydopamine-Lesioned Rat Model of Parkinson's Disease" Molecules 15, no. 4: 2814-2824. https://doi.org/10.3390/molecules15042814
APA StyleChoi, H. S., Park, M. S., Kim, S. H., Hwang, B. Y., Lee, C. K., & Lee, M. K. (2010). Neuroprotective Effects of Herbal Ethanol Extracts from Gynostemma pentaphyllum in the 6-Hydroxydopamine-Lesioned Rat Model of Parkinson's Disease. Molecules, 15(4), 2814-2824. https://doi.org/10.3390/molecules15042814



