Pellitorine, a Potential Anti-Cancer Lead Compound against HL60 and MCT-7 Cell Lines and Microbial Transformation of Piperine from Piper Nigrum
Abstract
:1. Introduction
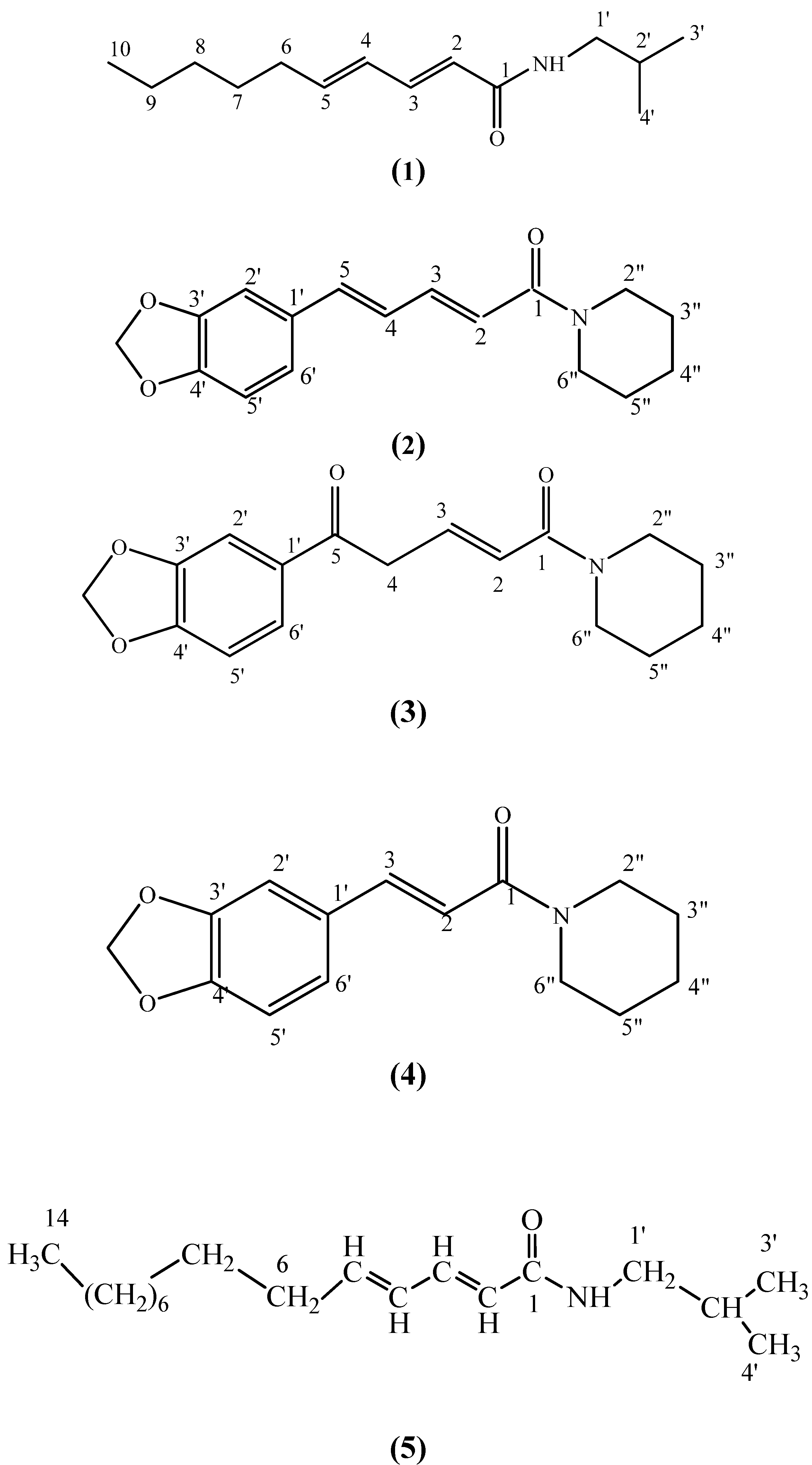
2. Results and Discussion
| Position | 1H NMR, δ ppm | 13C NMR, δ ppm | HMBC |
|---|---|---|---|
| 1 | 164.95 | ||
| 2 | 6.26 (1H, d, J=13.7 Hz) | 120.43 | 164.95 (C-1) (2J), 142.59(C-3) (2J), 46.96(C-4) (3J) |
| 3 | 6.82 (1H, m) | 142.59 | 164.95 (C-1) (3J), 120.43 (C-2) (2J), 46.96 (C-4) (2J) |
| 4 | 3.89 (2H, m) | 46.96 | 120.43 (C-2) (3J), 142.59(C-3) (2J), 169.12(C-5) (2J), 131.05 (C-1’) (3J) |
| 5 | 169.12 | ||
| 1’ | 131.05 | ||
| 2’ | 6.69 (1H, s) | 105.36 | 131.05 (C-1’) (2J), 122.12 (C-6’) (3J) |
| 3’ & 4’ | - | 147.55 | |
| 5’ | 6.56 (1H, d, J=8.2 Hz) | 108.10 | 131.05 (C-1’) (3J), 122.12 (C-6’) (2J) |
| 6’ | zhzhzzzz | 122.12 | 108.10 (C-5’ )(2J) |
| OCH2O | 6.61 (1H, d, J=2.7 Hz) | 100.88 | 147.55 (C-3’) (3J), 147.55 (C-4’) (3J) |
| 2” | 5.86 (2H, s) | 43.06 | 25.54 (C-2”) (2J), 25.54 (C-3”) (3J) |
| 3” & 5” | 3.40 (2H, m) | 25.54 | 43.06 (C-1”) (2J), (3J) |
| 4” | 1.57 (4H, m) | 26.56 | 25.54 (C-3” ) (2J), 25.54 (C-2”) (3J) |
| 6” | 1.57 (2H,m) | 46.36 | 26.56 (C-4”) (2J) |
| 3.40 (2H, m) |
3. Experimental
3.1. Plant material
3.2. General
3.3. Extraction and isolation
3.4. Biotransformation
3.4.1. Screening procedures
3.4.2. Preparative biotransformation
3.4.3. Extraction and purification of biotransformation products
3.5. Cytotoxicity assay
4. Conclusions
Acknowledgements
References and Notes
- Scott, I.M.; Puniani, E.; Jensen, H.; Livesey, J.F.; Poveda, L.; Vindas, P.S.; Durst, T.; Arnason, J.T. Analysis of Piperaceae germplasm by HPLC and LCMS: a method for isolating and identifying unsaturated amides from Piper spp extracts. J. Agric. Food Chem. 2005, 53, 1907–1913. [Google Scholar]
- Miyakado, M.; Nakayama, I.; Yoshioka, H. The Piperaceae amides I: structure of pipercide, a new insecticidal amide form Piper nigrum L. Agric. Bio. Chem. 1979, 43, 1609–1611. [Google Scholar] [CrossRef]
- Rosario, S.L.; Silva, A.J.; Parente, J.P. Alkamides from Cissampelos glaberrima. Planta Medica. 1996, 62, 376–377. [Google Scholar] [CrossRef]
- Schobert, R.; Siegfried, S.; Gordon, G.J. Three-component synthesis of (E)-α,β-unsaturated amides of the piperine family. J. Chem. Soc., Perk Trans 1. 2001, 2393–2397. [Google Scholar]
- Greger, H.; Grenz, M.; Bohlmann, F. Amides from Achillea species and Leucocyclus formosus. Phytochemistry 1981, 11, 2578–2581. [Google Scholar]
- Parmar, V.S.; Jain, S.C.; Gupta, S.; Talwar, S.; Rajwanshi, V.K.; Kumar, R.; Azim, A.; Malhotra, S.; Kumar, N.; Jain, R.; Sharma, N.K.; Tyagi, O.D.; Lawrie, S.J.; Ernugton, W.; Howarch, O.W.; Olsen, C.E.; Singh, S.K.; Wengel, J. Polyphenols and alkaloids from Piper species. Phytochemistry 1998, 49, 1069–1078. [Google Scholar]
- MacKeen, M.M.; Ali, A.M.; El-Shakarwy, S.H.; Mnap, M.Y.; Salleh, K.M.; Lajis, N.H.; Kawazu, K. Antimicrobial and cytotoxic properties of some Malaysian traditional vegetables. Int. J. Pharmacol. 1997, 35, 174–178. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–5 are available from authors.
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ee, G.C.L.; Lim, C.M.; Rahmani, M.; Shaari, K.; Bong, C.F.J. Pellitorine, a Potential Anti-Cancer Lead Compound against HL60 and MCT-7 Cell Lines and Microbial Transformation of Piperine from Piper Nigrum. Molecules 2010, 15, 2398-2404. https://doi.org/10.3390/molecules15042398
Ee GCL, Lim CM, Rahmani M, Shaari K, Bong CFJ. Pellitorine, a Potential Anti-Cancer Lead Compound against HL60 and MCT-7 Cell Lines and Microbial Transformation of Piperine from Piper Nigrum. Molecules. 2010; 15(4):2398-2404. https://doi.org/10.3390/molecules15042398
Chicago/Turabian StyleEe, Gwendoline Cheng Lian, Chyi Meei Lim, Mawardi Rahmani, Khozirah Shaari, and Choon Fah Joseph Bong. 2010. "Pellitorine, a Potential Anti-Cancer Lead Compound against HL60 and MCT-7 Cell Lines and Microbial Transformation of Piperine from Piper Nigrum" Molecules 15, no. 4: 2398-2404. https://doi.org/10.3390/molecules15042398
APA StyleEe, G. C. L., Lim, C. M., Rahmani, M., Shaari, K., & Bong, C. F. J. (2010). Pellitorine, a Potential Anti-Cancer Lead Compound against HL60 and MCT-7 Cell Lines and Microbial Transformation of Piperine from Piper Nigrum. Molecules, 15(4), 2398-2404. https://doi.org/10.3390/molecules15042398




