Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes
Abstract
:1. Introduction
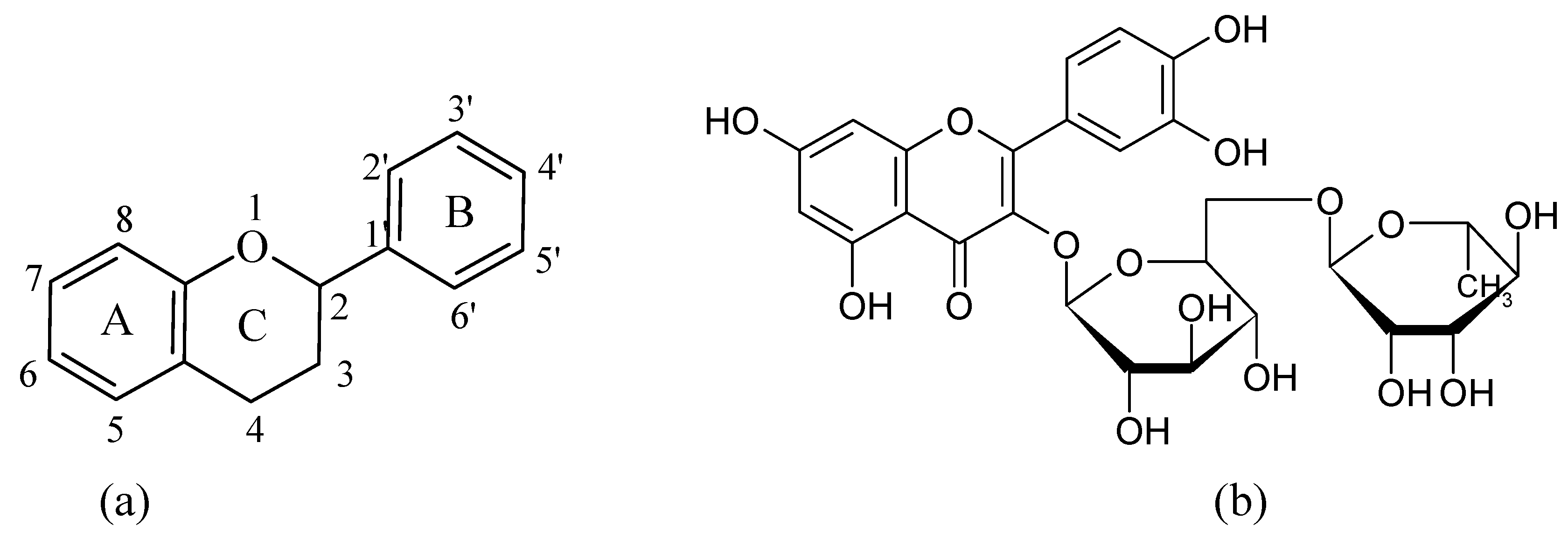
2. Results and Discussion
2.1. Characterization of the complexes
| Compound | ν(OH) | ν(C=O) | ν(C-O-C) | ν(V=O) | ν3(SO42-) | ν4(SO42-) |
|---|---|---|---|---|---|---|
| Rutin | 3,429 | 1,656 (s)* | 1,296 (s) | |||
| 1 | 3,401 | 1,631 (s) | 1,296 (s) | 983 (m)** | 1,023 (s) | 609 (m) |
| 2 | 3,422 | 1,622 (s) | 1,294 (s) | 972 (m) |
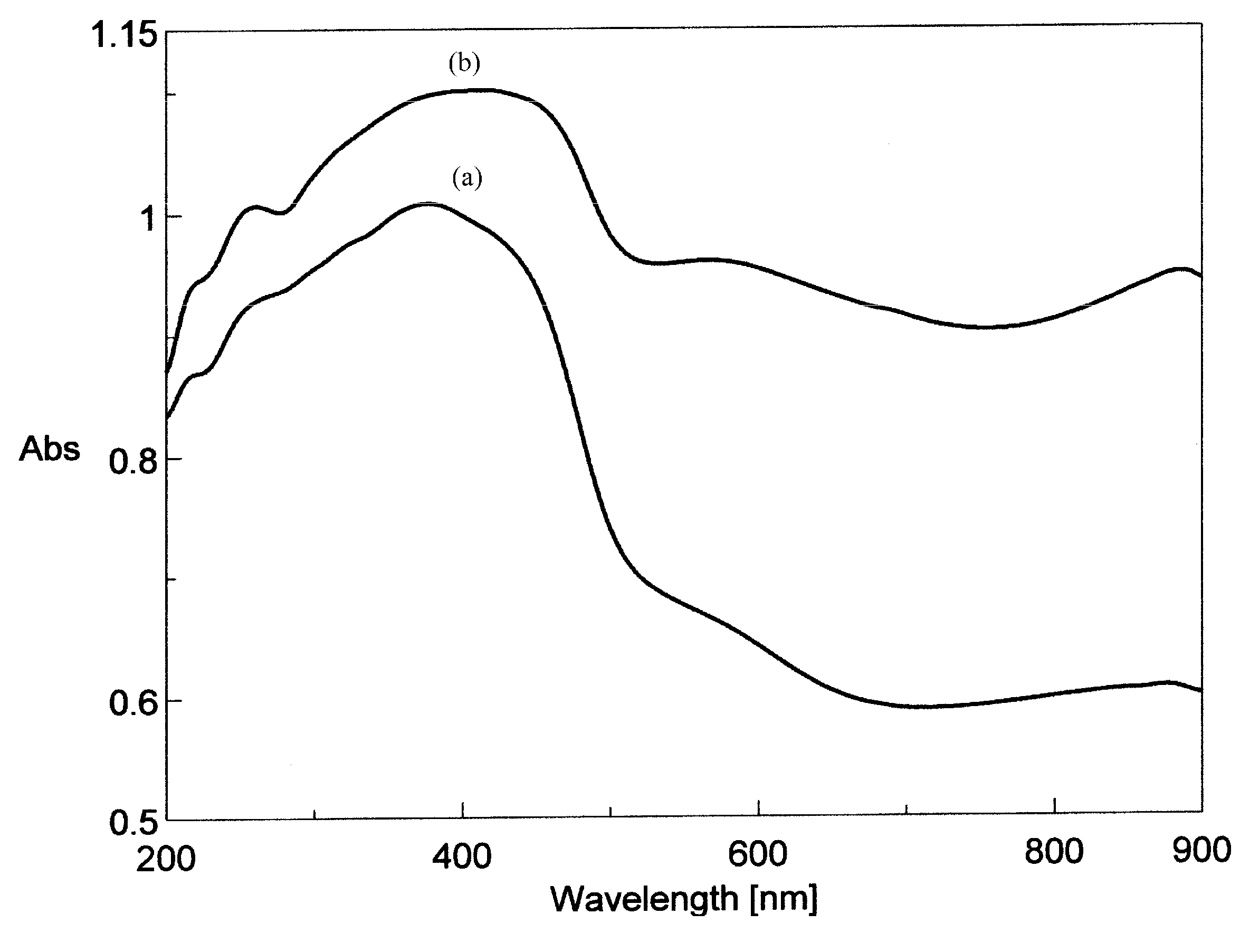
| Compound | λ (nm) | ||||
|---|---|---|---|---|---|
| Rutin | 260 | 360 | - | - | - |
| (1) | 261 | 377 | 420 (sh) | 579 (sh)* | 879 |
| (2) | 260 | 382 | 417 | 566 | 886 |
| Complex | Step | Thermal effect | Temperature range/°C | Δmexp/% | Δmcalcd/% | ||||
|---|---|---|---|---|---|---|---|---|---|
| [VO(rutin)(H2O)2](SO4)0.5 2H2O | 1 | Endothermic | 78–140 | 9.04 | 9.04 | ||||
| 2 | Exothermic | 140–388 | 40.52 | 40.66 | |||||
| 3 | Exothermic | 388–812 | 33.94 | 33.88 | |||||
| 4 | Exothermic | 812–900 | 4.98 | 5.02 | |||||
| [VO(Rut)2] 4H2O | 1 | Endothermic | 55–94 | 5.38 | 5.31 | ||||
| 2 | Exothermic | 94–362 | 47.68 | 47.75 | |||||
| 3 | Exothermic | 362–900 | 40.87 | 40.83 | |||||

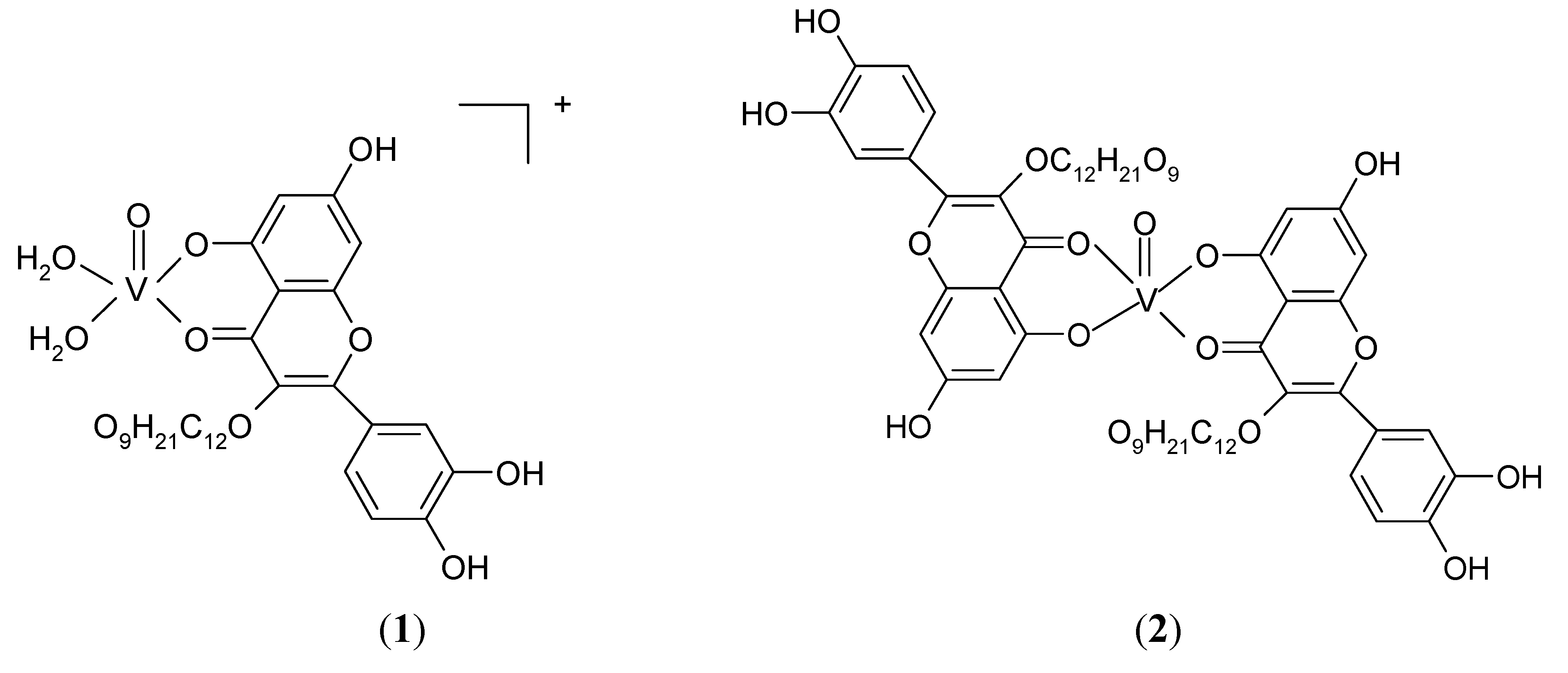
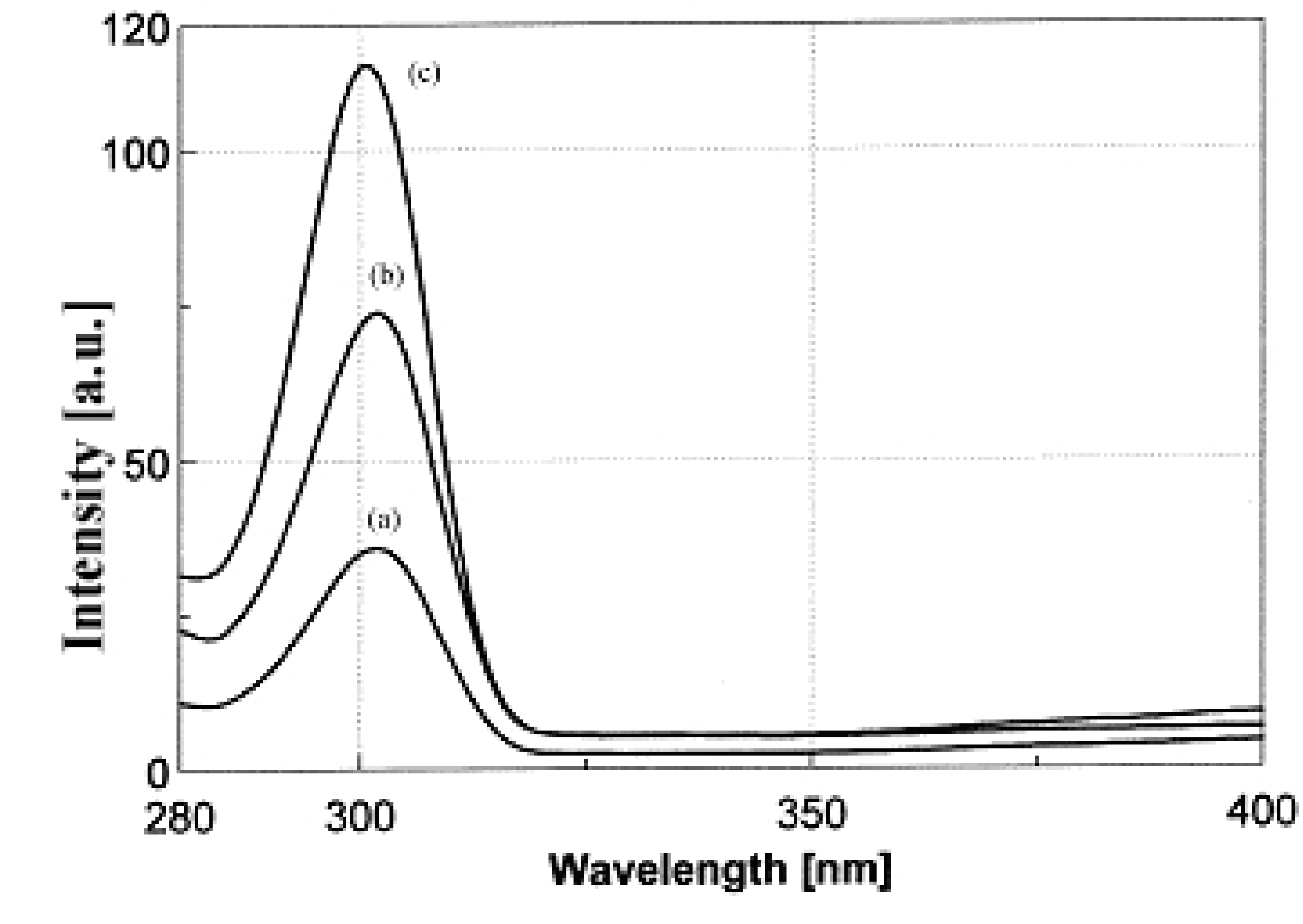
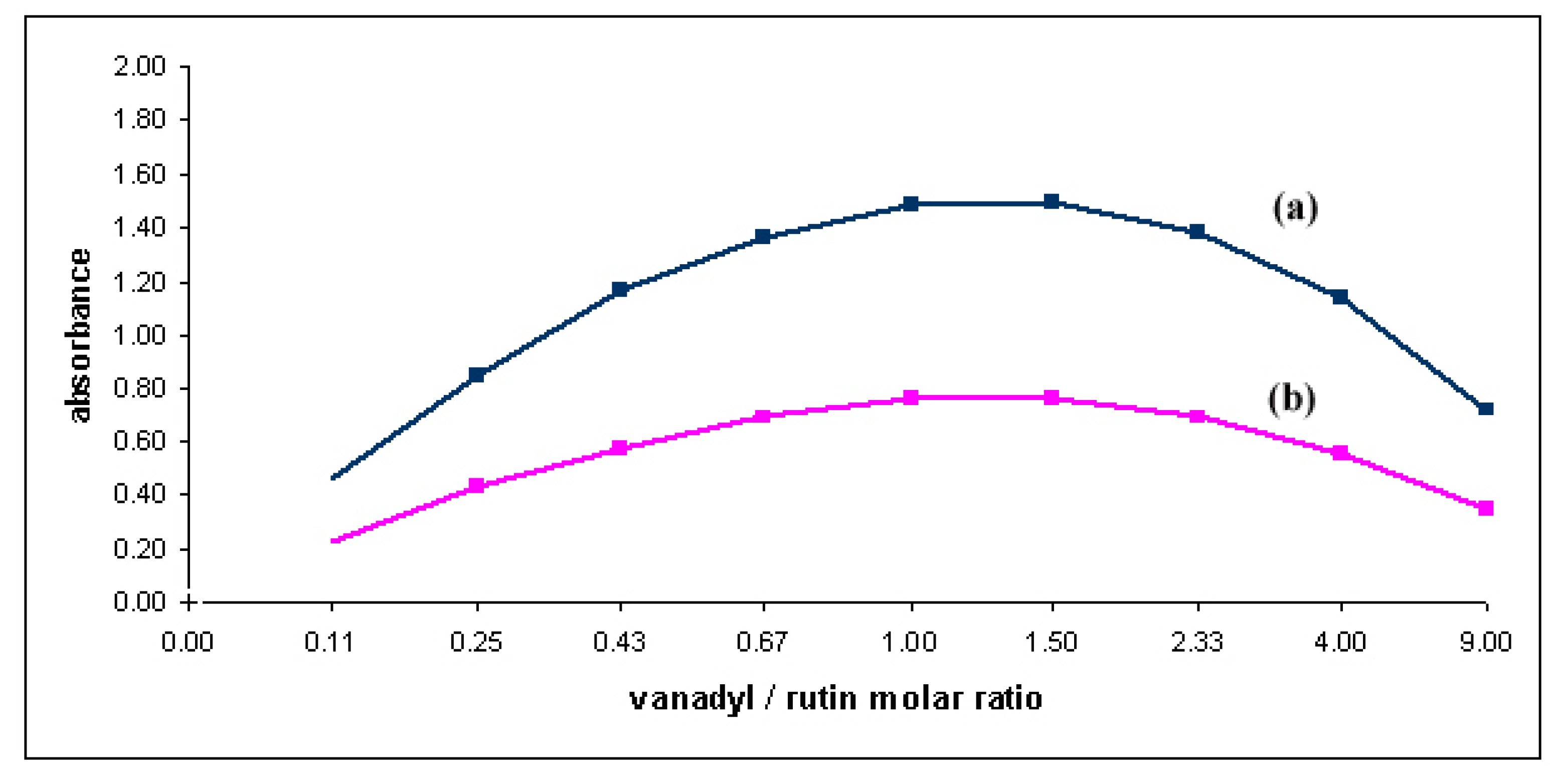
2.2. Complex formation and stability studies
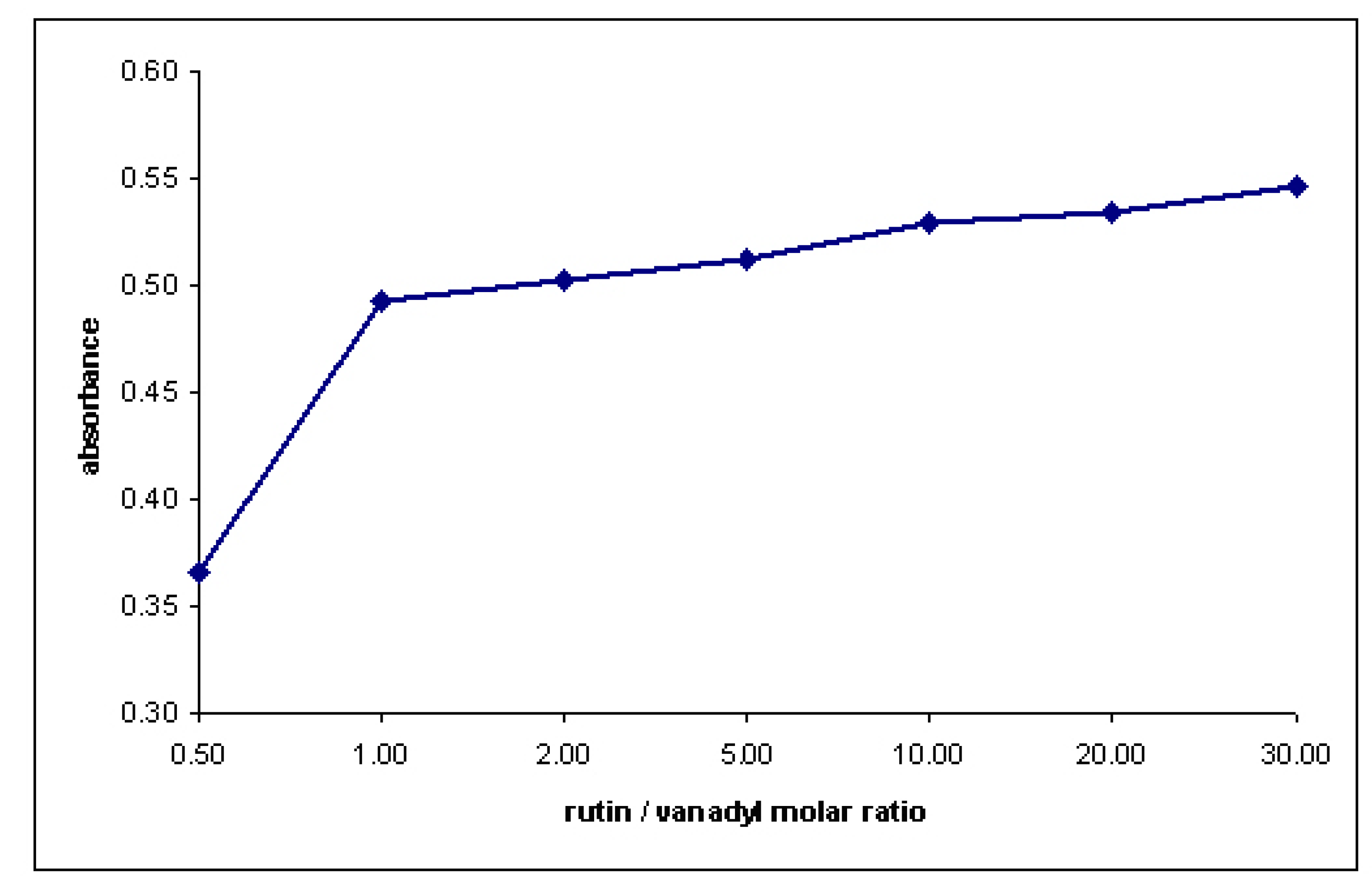


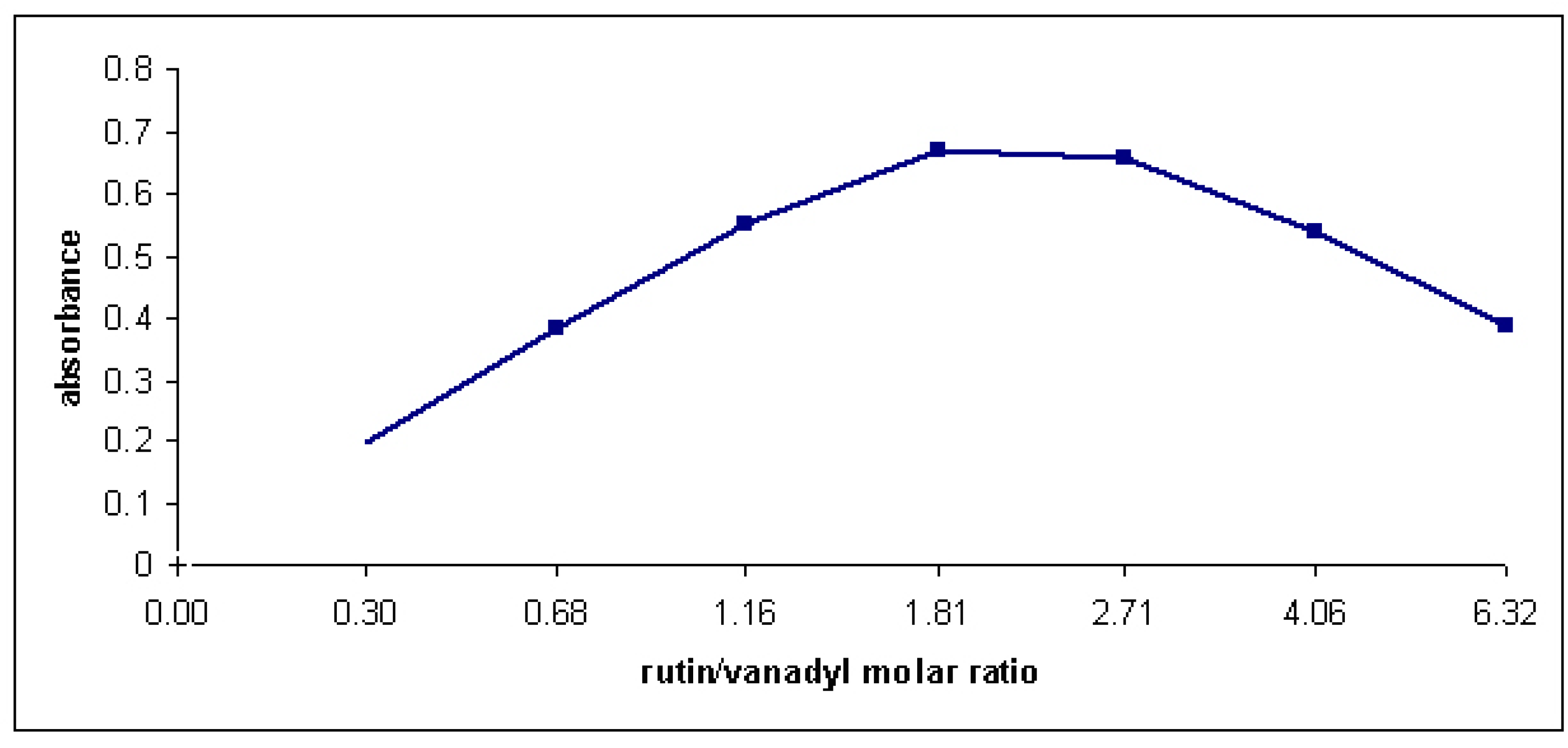
3. Experimental
3.1. General
3.2. Synthesis of the complex 1
3.3. Synthesis of the complex 2
4. Conclusions
Acknowledgements
- Samples Availability: Samples of the compounds 1 and 2 are available from the author V. Uivarosi.
References and Notes
- Cushnie, T.; Lamb, A. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Havsteen, B.H. The biochemistry and medical significance of flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.C.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A.M. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113, 71–88. [Google Scholar]
- Manach, C.; Regerat, F; Texier, O.; Agullo, G.; Demigne, C.; Remesy, C. Bioavailability, metabolism and physiological impact of 4-oxo-flavonoids. Nutr. Res. 1996, 16, 517–544. [Google Scholar]
- Stavric, B. Antimutagens and anticarcinogens in food. Food Chem. Toxicol. 1994, 32, 79–89. [Google Scholar]
- Reznichenko, L.; Amit, T.; Zheng, H.; Avramovich-Tirosh, Y.; Youdim, M.B.; Weinreb, O.; Mandel, S. Reduction of iron-regulated amyloid precursor protein and beta-amyloid peptide by (-)-epigallocatechin-3-gallate in cell cultures: Implications for iron chelation in Alzheimer's disease. J. Neurochem. 2006, 97, 527–536. [Google Scholar] [CrossRef]
- Cook, N.C.; Samman, S. Flavonoids-Chemistry, metabolism, cardioprotective effects, and dietary sources. J. Nutr. Biochem. 1996, 2, 66–76. [Google Scholar]
- Teixeira, S.; Siquet, C.; Aloes, C.; Boal, I.; Marques, M.P.; Borges, F.; Lima, J.L.F.C.; Reis, S. Structure-property studies on the antioxidant activity of flavonoids present in diet. Free Radic. Biol. Med. 2005, 39, 1099–1108. [Google Scholar] [CrossRef]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Van Acker, S.A.B.E.; van Den Berg, D.-J.; Tromp, M.N.J.L.; Griffioen, D.H.; van Bennekom, W.P.; van der Vijgh, W.J.F.; Bast, A. Structural aspects of antioxidant activity of flavonoids. Free Radic. Biol. Med. 1996, 20, 331–342. [Google Scholar] [CrossRef]
- Pereira, R.M.S.; Andrades, N.E.D.; Paulino, N.; Bast, A.; Sawaya, A.C.H.F.; Eberlin, M.N.; Marcucci, M.C.; Favero, G.M.; Novak, E.M.; Bydlowski, S.P. Synthesis and characterization of metal complex containing naringin and Cu, and its antioxidant, antimicrobial, antiiinflammatory and tumor cell cytotoxicity. Molecules 2007, 12, 1352–1366. [Google Scholar] [CrossRef]
- Afanas’ev, I.B.; Dorozhko, A.I.; Brodskii, A.V.; Kostyuk, V.A.; Potapovich, A.I. Chelating and free radical scavenging mechanisms of inhibitory action of rutin and quercetin in lipid peroxidation. Biochem. Pharmacol. 1989, 38, 1763–1769. [Google Scholar]
- Grazul, M.; Budzisz, E. Biological activity of metal ions complexes of chromones, coumarins and flavones. Coord. Chem. Rev. 2009, 253, 2588–2598. [Google Scholar] [CrossRef]
- Ong, K.C.; Khoo, H.E. Insulinomimetic effects of myricetin on lipogenesis and glucose transport in rat adipocytes but not glucose transporter translocation. Biochem. Pharmacol. 1996, 51, 423–429. [Google Scholar]
- Vessal, M.; Hemmati, M.; Vasei, M. Antidiabetic effects of quercetin in streptozotocin-induced diabetic rats. Comp. Biochem. Physiol. 2003, C135, 357–364. [Google Scholar]
- Cazarolli, L.H.; Zanatta, L.; Jorge, A.P.; de Sousa, E.; Horst, H.; Woehl, V.M.; Pizzolatti, M.G.; Szpoganicz, B.; Silva, F.R.M.B. Follow-up studies on glycosylated flavonoids and their complexes with vanadium: Their anti-hyperglycemic potential role in diabetes. Chemico-Biol. Interact. 2006, 163, 177–191. [Google Scholar]
- Shukla, R.; Barve, V.; Padhye, S.; Bhonde, R. Reduction of oxidative stress induced vanadium toxicity by complexing with a flavonoid, quercetin: A pragmatic therapeutic approach for diabetes. BioMetals 2006, 19, 685–693. [Google Scholar] [CrossRef]
- Shukla, R.; Barve, V.; Padhye, S.; Bhonde, R. Synthesis, structural properties and insulin-enhancing potential of bis(quercetinato)oxovanadium(IV) conjugate. Bioorg. Med. Chem. Lett. 2004, 14, 4961–4965. [Google Scholar] [CrossRef]
- Ferrer, E.G.; Salinas, M.V.; Correa, M.J.; Naso, L.; Barrio, D.A..; Etcheverry, S.B.; Lezama, L.; Rojo, T.; Williams, P.A. Synthesis, characterization, antitumoral and osteogenic activities of quercetin vanadyl(IV) complexes. J. Biol. Inorg. Chem. 2006, 11, 791–801. [Google Scholar]
- Etcheverry, S.B.; Ferrer, E.G.; Naso, L.; Rivadeneira, J.; Salinas, V.; Williams, P.A.M. Antioxidant effects of the VO(IV) hesperidin complex and its role in cancer chemoprevention. J. Biol. Inorg. Chem. 2008, 13, 435–447. [Google Scholar] [CrossRef]
- La Casa, C.; Villegas, I.; Alarcon de la Lastra, C.; Motilva, V.; Martin Calero, M.J. Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol induced gastric lesions. J. Ethnopharmacol. 2000, 71, 45–53. [Google Scholar] [CrossRef]
- Guardia, T.; Rotelli, A.E.; Juarez, A.O.; Pelzer, L.E. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Farmaco 2001, 56, 683–687. [Google Scholar]
- Han, Y. Rutin has therapeutic effect on septic arthritis caused by Candida albicans. Int.Immunopharmacol. 2009, 9, 207–211. [Google Scholar] [CrossRef]
- Korkmaz, A.; Kolankaya, D. Protective effect on rutin on ischemia/reperfusion induce damage in rat kidney. J. Surg. Res. 2009. [Google Scholar] [CrossRef]
- Perez Guerrero, C.; Martin, M.J.; Marhuenda, E. Prevention by rutin of gastric lesions induced by ethanol in rats: role of endogenous prostaglandins. Gen. Pharmacol. 1994, 25, 575–580. [Google Scholar] [CrossRef]
- Undeger, U.; Aydin, S.; Basaran, A.A.; Basaran, N. The modulation effects on quercetin and rutin on the mitomycin C induced DNA damage. Toxicol. Lett. 2004, 151, 143–149. [Google Scholar]
- Nassiri-Asl, M.; Shariati-Rad, S.; Zamansoltani, F. Anticonvulsive effects of intracerebroventricular administration of rutin in rats. Progr. Neuro Psychopharmacol. Biol. Psychiatr. 2008, 32, 989–993. [Google Scholar] [CrossRef]
- Kostyuk, V.A.; Potapovich, A.I.; Kostyuk, T.V.; Cherian, M.G. Metal complexes of dietary flavonoids: evaluation of radical scavenger properties and protective activity against oxidative stress in vivo. Cell. Mol. Biol. 2007, 53, 62–69. [Google Scholar]
- Afanas'ev, I.B.; Ostrachovich, E.A.; Korkina, L.G. Effect of rutin and its copper complex on superoxide formation and lipid peroxidation in rat liver microsomes. FEBS Lett. 1998, 425, 256–258. [Google Scholar] [CrossRef]
- Afanas’ev, I.B.; Ostrakhovitch, E.A.; Mikhal’chik, E.V.; Ibragimova, G.A.; Korkina, L.G. Enhancement of antioxidant and anti-inflammatory activities of bioflavonoid rutin by complexation with transition metals. Biochem. Pharmacol. 2001, 61, 677–684. [Google Scholar]
- Kaushal, G.P.; Sekhon, B.S.; Bhatia, I.S. Spectrophotometric determination of quercetin with VO2+. Microchim. Acta 1979, 71, 365–370. [Google Scholar] [CrossRef]
- Hathaway, B.J. Oxyanions. In Comprehensive Coordination Chemistry; Wilkinson, G., Gillard, R.D., McCleverty, J.A., Eds.; Pergamon Press: Oxford, UK, 1987; pp. 413–433. [Google Scholar]
- Lever, A.B.P. Inorganic Electronic Spectroscopy; Elsevier: Amsterdam, The Netherlands, 1986; pp. 385–392. [Google Scholar]
- Malesev, D.; Kuntic, V. Investigation of metal–flavonoid chelates and determination of flavonoids via metal–flavonoid complexing reactions. J. Serb. Chem. Soc. 2007, 72, 921–939. [Google Scholar] [CrossRef]
- Job, P. Recherches sur la formation de complexes mineraux en solution, et sur leur stabilite(in Paris). Ann. Chim. 1928, 9, 113–203. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Uivarosi, V.; Barbuceanu, S.F.; Aldea, V.; Arama, C.-C.; Badea, M.; Olar, R.; Marinescu, D. Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes. Molecules 2010, 15, 1578-1589. https://doi.org/10.3390/molecules15031578
Uivarosi V, Barbuceanu SF, Aldea V, Arama C-C, Badea M, Olar R, Marinescu D. Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes. Molecules. 2010; 15(3):1578-1589. https://doi.org/10.3390/molecules15031578
Chicago/Turabian StyleUivarosi, Valentina, Stefania Felicia Barbuceanu, Victoria Aldea, Corina-Cristina Arama, Mihaela Badea, Rodica Olar, and Dana Marinescu. 2010. "Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes" Molecules 15, no. 3: 1578-1589. https://doi.org/10.3390/molecules15031578
APA StyleUivarosi, V., Barbuceanu, S. F., Aldea, V., Arama, C.-C., Badea, M., Olar, R., & Marinescu, D. (2010). Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes. Molecules, 15(3), 1578-1589. https://doi.org/10.3390/molecules15031578








