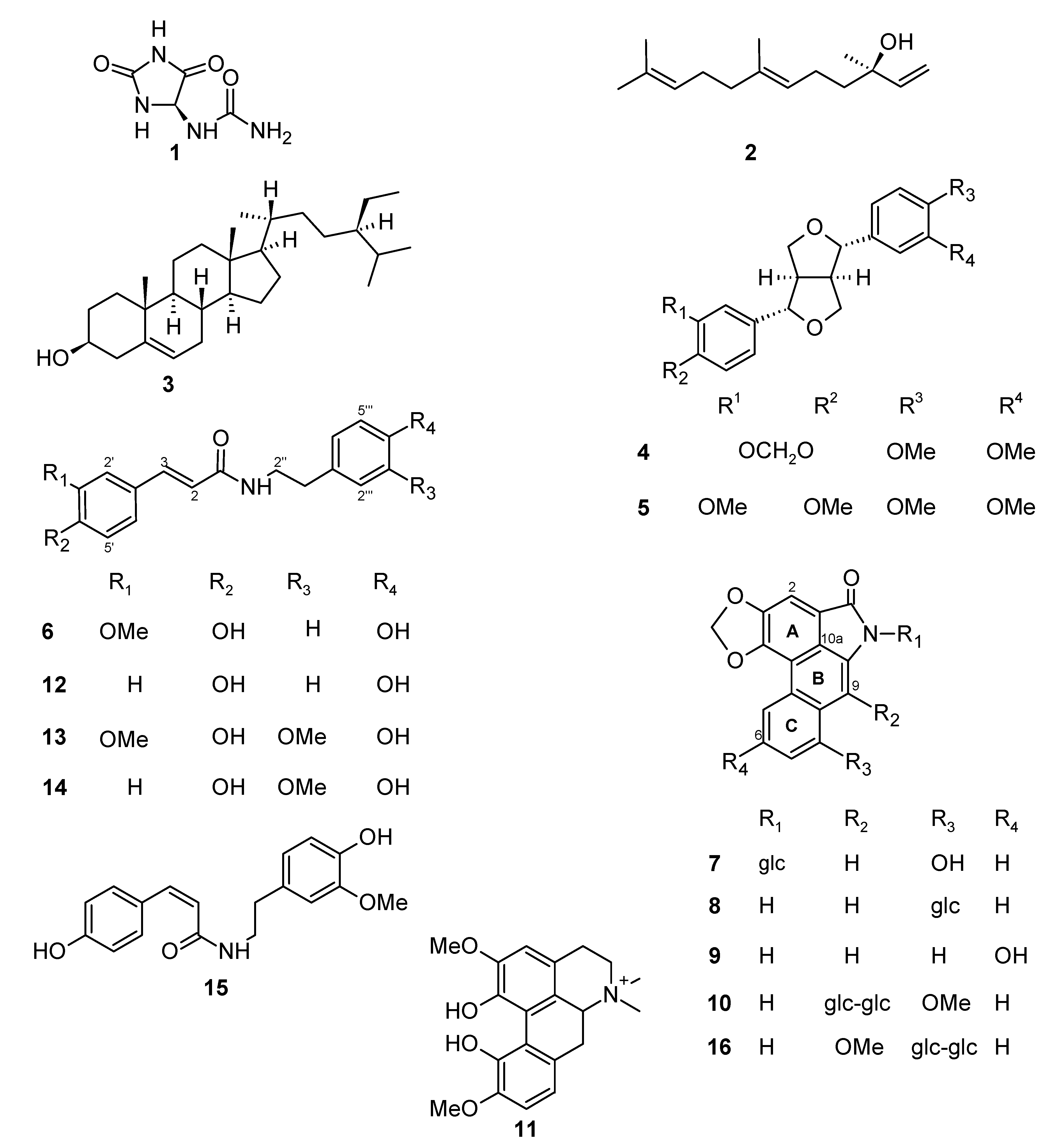
Aristolactams and Alkamides of Aristolochia gigantea
Abstract
:1. Introduction
2. Results and Discussion
| Position | δH | δC | Position | δH | δC |
|---|---|---|---|---|---|
| 1 | 119.1 | OCH2O | 6.46 s | 103.0 | |
| 2 | 7.65 s | 105.7 | OCH3 | 3.94 s | 56.0 |
| 3 | 148.2 | 1′ | 5.08 d (6.5) | 103.0 | |
| 4 | 146.9 | 2′ | 3.92 dd (8.5, 6.5) | 81.0 | |
| 4a | 109.0 | 3′ | 3.54 t (8.5) | 75.8 | |
| 4b | 127.9 | 4′ | 3.47 t (8.5) | 69.4 | |
| 5 | 8.26 dd (8.5, 1.0) | 118.4 | 5′ | 3.18 m | 76.9 |
| 6 | 7.56 dd (8.0, 8.5) | 126.1 | 6′α, 6′β | 3.8 − 3.6 m | 60.5 |
| 7 | 7.23 dd (8.0, 1.0) | 110.8 | 1″ | 4.65 d (7.5) | 102.4 |
| 8 | 157.2 | 2″ | 3.06 dd (7.5, 8.5) | 74.1 | |
| 8a | 120.0 | 3″ | 3.10 t (8.5) | 76.1 | |
| 9 | 132.6 | 4″ | 3.17 t (8.5) | 69.3 | |
| 10 | b | 5″ | 2.99 ddd (8.5, 4.7, 2.5) | 76.4 | |
| 10a | 124.4 | 6″α, 6″β | 3.35 m | 60.3 | |
| 3.8 − 3.6 m | |||||
| CO | 167.4 | NH | 10.18 s |
| Position | 14 δH | 15 δH |
|---|---|---|
| 2 | 6.38 d (15.5) | 5.75 d (13.0) |
| 3 | 7.28 d (15.5) | 6.48 d (13.0) |
| 2′, 6′ | 7.36 d (8.5) | 7.56 d (8.5) |
| 3′, 5′ | 6.76 d (8.5) | 6.68 d (8.5) |
| 2′ | 3.34 m b | 3.34 m b |
| 3′ | 2.62 t (5.5) | 2.62 t (5.5) |
| 2′′′ | 6.75 d (2.0) | 6.74 d (2.0) |
| 5′′′ | 6.66 d (8.0) | 6.66 d (8.0) |
| 6′′′ | 6.59 dd (8.0, 2.0) | 6.58 dd (8.0, 2.0) |
| OCH3 | 3.72 s | 3.71 s |
| NH | 8.00 t (5.5) | 7.98 t (5.5) |
3. Experimental
3.1. General
3.2. Plant material
3.3. Extraction and isolation of the chemical constituents
3.4. Spectral data
 −17 ° (CHCl3, c 0.2) [lit. −12.5 ° (CHCl3, c 0.02)] [13]. 1H-NMR (CDCl3) δ 5.15 (1H, dd, J 17.5, 1.5 Hz, H-1α), 5.00 (1H, dd, J 10.5, 1.5 Hz, H-1β), 5.86 (1H, dd, J 17.5, 10.5 Hz, H-2), 1.54–1.51 (2H, m, H-4), 2.00–1.92 (6H, m, H-5, H-8, H-9), 5.08–5.01 (2H, m, H-6, H-10), 1.60 (3H, s, H-12), 1.22 (3H, s, H-15), 1.54 (6H, s, H-13, H-14). 13C-NMR data were consistent with those previously reported [14].
−17 ° (CHCl3, c 0.2) [lit. −12.5 ° (CHCl3, c 0.02)] [13]. 1H-NMR (CDCl3) δ 5.15 (1H, dd, J 17.5, 1.5 Hz, H-1α), 5.00 (1H, dd, J 10.5, 1.5 Hz, H-1β), 5.86 (1H, dd, J 17.5, 10.5 Hz, H-2), 1.54–1.51 (2H, m, H-4), 2.00–1.92 (6H, m, H-5, H-8, H-9), 5.08–5.01 (2H, m, H-6, H-10), 1.60 (3H, s, H-12), 1.22 (3H, s, H-15), 1.54 (6H, s, H-13, H-14). 13C-NMR data were consistent with those previously reported [14]. −15.2 ° (CHCl3, c 0.2) [lit. −26.1 ° (CHCl3, c 0.1)] [15]. 13C- NMR data were consistent with those previously reported [16].
−15.2 ° (CHCl3, c 0.2) [lit. −26.1 ° (CHCl3, c 0.1)] [15]. 13C- NMR data were consistent with those previously reported [16]. +51.4 ° (CHCl3, c 0.21) [lit. +58.0 ° (CHCl3, c 0.03)] [17]. 1H- NMR (CDCl3) δ 6.83 (1H, d, J 2.0 Hz, H-2), 6.78 (1H, d, J 2.0 Hz, H-2′), 6.71 (1H, d, J 8.0 Hz, H-5), 6.77 (1H, d, J 8.0 Hz, H-5′), 6.81 (1H, dd, J 2.0, 8.0 Hz, H-6), 6.74 (1H, dd, J 2.0, 8.0 Hz, H-6′), 4.67 (2H, d, J 5.5 Hz, H-7β, H-7′β), 3.02 (2H, m, H-8α, H-8′α), 4.18 (2H, dd, J 7.0, 14.0 Hz, H-9β, H-9′β), 3.82 (2H, m, H-9α, H-9′α), 3.83 (3H, s, OCH3), 3.80 (3H, s, OCH3), 5.85 (2H, s, OCH2O).
+51.4 ° (CHCl3, c 0.21) [lit. +58.0 ° (CHCl3, c 0.03)] [17]. 1H- NMR (CDCl3) δ 6.83 (1H, d, J 2.0 Hz, H-2), 6.78 (1H, d, J 2.0 Hz, H-2′), 6.71 (1H, d, J 8.0 Hz, H-5), 6.77 (1H, d, J 8.0 Hz, H-5′), 6.81 (1H, dd, J 2.0, 8.0 Hz, H-6), 6.74 (1H, dd, J 2.0, 8.0 Hz, H-6′), 4.67 (2H, d, J 5.5 Hz, H-7β, H-7′β), 3.02 (2H, m, H-8α, H-8′α), 4.18 (2H, dd, J 7.0, 14.0 Hz, H-9β, H-9′β), 3.82 (2H, m, H-9α, H-9′α), 3.83 (3H, s, OCH3), 3.80 (3H, s, OCH3), 5.85 (2H, s, OCH2O). +17.5 ° (CHCl3, c 0.12) [lit. +61 ° (CHCl3, c 0.4)] [17]. 1H-NMR and 13C-NMR data were consistent with those previously reported [17].
+17.5 ° (CHCl3, c 0.12) [lit. +61 ° (CHCl3, c 0.4)] [17]. 1H-NMR and 13C-NMR data were consistent with those previously reported [17].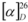 −7.9 ° (MeOH, c 0.1) [lit. −9.9 ° (MeOH, c 0.07)] [19]. 1H-NMR data were consistent with those previously reported [19].
−7.9 ° (MeOH, c 0.1) [lit. −9.9 ° (MeOH, c 0.07)] [19]. 1H-NMR data were consistent with those previously reported [19].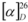 −8.2 ° (MeOH, c 0.1) [lit. −10.5 ° (MeOH, c 0.2)] [20]. 1H-NMR data were consistent with those previously reported [20].
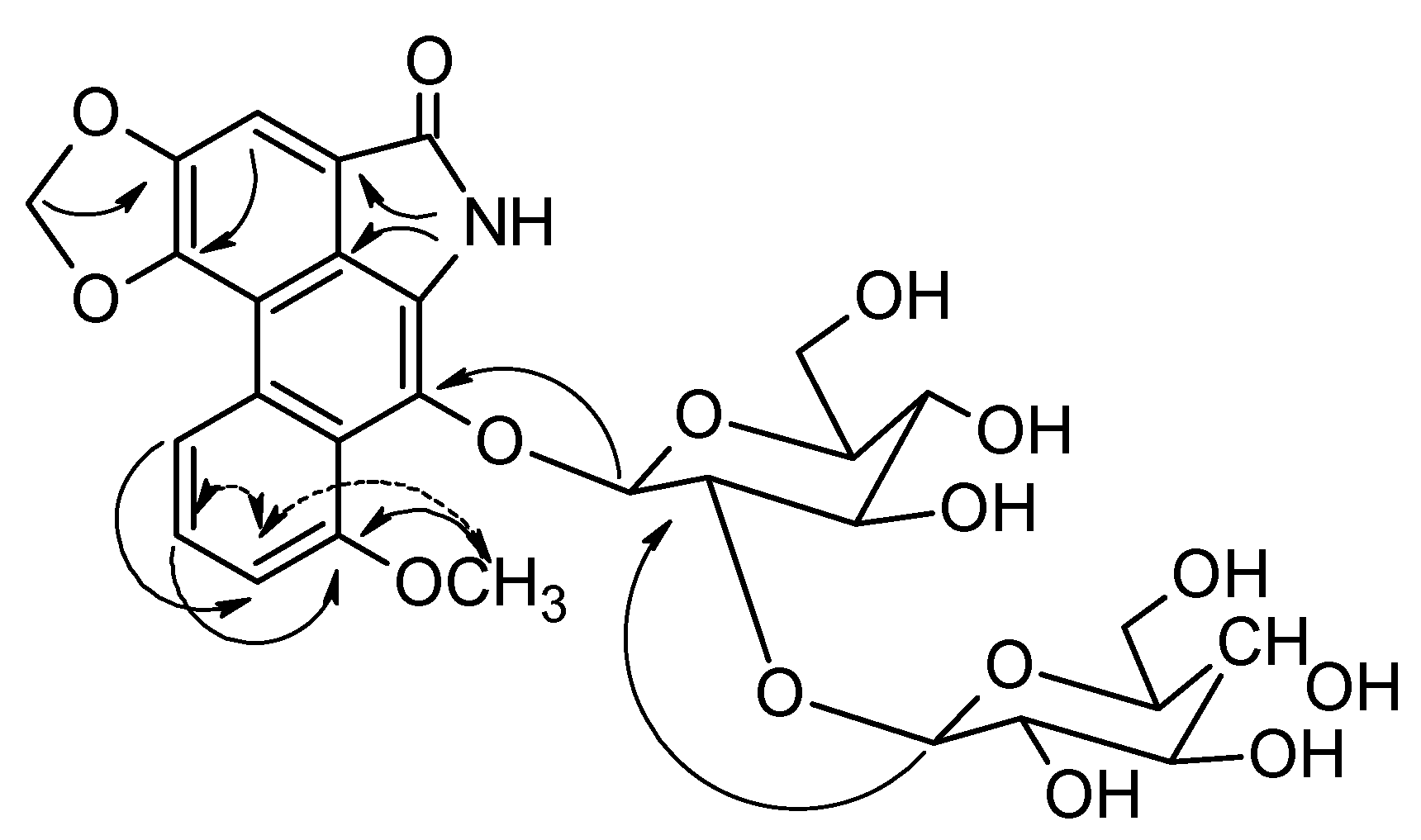
−8.2 ° (MeOH, c 0.1) [lit. −10.5 ° (MeOH, c 0.2)] [20]. 1H-NMR data were consistent with those previously reported [20].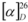 −3.9 ° (c 0.5, MeOH). 1H-NMR (CDCl3) and 13C-NMR (CDCl3) spectra see Table 1; ESI-HR-TOF-MS (probe), 4000 V, m/z (rel. int.): 632. 1614 [M − H]− (100) (calculated for C29H31O15N − H = 632.1615); ESI-MS/MS (probe) 4,500 V from ions at m/z 632.1 (100), m/z (rel. int.): 308.1 [M − glc-glc]− (92).
−3.9 ° (c 0.5, MeOH). 1H-NMR (CDCl3) and 13C-NMR (CDCl3) spectra see Table 1; ESI-HR-TOF-MS (probe), 4000 V, m/z (rel. int.): 632. 1614 [M − H]− (100) (calculated for C29H31O15N − H = 632.1615); ESI-MS/MS (probe) 4,500 V from ions at m/z 632.1 (100), m/z (rel. int.): 308.1 [M − glc-glc]− (92). +164.4 ° (MeOH, c 0.03) [lit. +150.0 ° (MeOH, c 0.1)] [23]. 1H-NMR (DMSO-d6) δ 6.49 (1H, s, H-3), 2.7–2.8 (2H, m, H-4α, H-4β), 3.7–3.6 (2H, m, H-5α, H-5β), 4.34 (1H, br d, J 13.0, H-6a), 2.59 (1H, t, J 13.0, H-7α), 3.10 (1H, br d, J 13.0, H-7β), 6.35 (1H, br d, J 8.0, H-8), 6.59 (1H, d, J 8.0, H-9), 3.65 (3H, s, OCH3-10), 3.68 (3H, s, OCH3-2), 2.88 (3H, s, N-CH3), 3.29 (3H, s, N-CH3). 13C-NMR (DMSO-d6) δ 152.1 (C-1), 151.2 (C-2), 108.8 (C-3), 111.6 (C-3a), 23.2 (C-4), 60.5 (C-5), 69.2 (C-6a), 30.4 (C-7), 125.1 (C-7a), 112.4 (C-8), 109.9 (C-9), 150.3 (C-10), 152.5 (C-11), 122.6 (C-11a), 123.1 (C-1a), 120.0 (C-1b), 55.2, 55.7 (OCH3), 42.5, 52.6 (N-CH3).
+164.4 ° (MeOH, c 0.03) [lit. +150.0 ° (MeOH, c 0.1)] [23]. 1H-NMR (DMSO-d6) δ 6.49 (1H, s, H-3), 2.7–2.8 (2H, m, H-4α, H-4β), 3.7–3.6 (2H, m, H-5α, H-5β), 4.34 (1H, br d, J 13.0, H-6a), 2.59 (1H, t, J 13.0, H-7α), 3.10 (1H, br d, J 13.0, H-7β), 6.35 (1H, br d, J 8.0, H-8), 6.59 (1H, d, J 8.0, H-9), 3.65 (3H, s, OCH3-10), 3.68 (3H, s, OCH3-2), 2.88 (3H, s, N-CH3), 3.29 (3H, s, N-CH3). 13C-NMR (DMSO-d6) δ 152.1 (C-1), 151.2 (C-2), 108.8 (C-3), 111.6 (C-3a), 23.2 (C-4), 60.5 (C-5), 69.2 (C-6a), 30.4 (C-7), 125.1 (C-7a), 112.4 (C-8), 109.9 (C-9), 150.3 (C-10), 152.5 (C-11), 122.6 (C-11a), 123.1 (C-1a), 120.0 (C-1b), 55.2, 55.7 (OCH3), 42.5, 52.6 (N-CH3).4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from the authors.
References and Notes
- Lopes, L.M.X.; Nascimento, I.R.; Silva, T. Phytochemistry of the Aristolochiaceae Family. In Research Advances in Phytochemistry; Mohan, R.M.M., Ed.; Global Research Network: Kerala, India, 2001; Volume 2, pp. 19–108. [Google Scholar]
- Li, Y.; Liu, Z.; Guo, X.; Shu, J.; Chen, Z.; Li, L. Aristolochic Acid I-induced DNA Damage and Cell Cycle Arrest in Renal Tubular Epithelial Cells in Vitro. Arch. Toxicol. 2006, 80, 524–532. [Google Scholar] [CrossRef]
- Li, J.; Zhang, L.; Jiang, Z.; Shu, B.; Li, F.; Bao, Q.; Zhang, L. Toxicities of Aristolochic Acid I and Aristololactam I in Cultured Renal Epithelial Cells. Toxicol. In Vitro 2010, 24, 1092–1097. [Google Scholar] [CrossRef]
- Zhang, J.; Xiao, Y.; Feng, J.; Wu, S.L.; Xue, X.; Zhang, X.; Liang, X. Selectively Preparative Purification of Aristolochic Acids and Aristololactams from Aristolochia Plants. J. Pharm. Biomed. Anal. 2010, 52, 446–451. [Google Scholar] [CrossRef]
- Choi, Y.L.; Kim, J. K.; Choi, S.-U.; Min, Y.-K.; Bae, M.-A.; Kim, B.T.; Heo, J.-N. Synthesis of Aristolactam Analogues and Evaluation of Their Antitumor Activity. Bioorg. Med. Chem. Lett. 2009, 19, 3036–3040. [Google Scholar]
- Kumar, V.; Prasad, A.K.; Parmar, V.S. Naturally Occurring Aristolactams, Aristolochic Acids and Dioxoaporphines and Their Biological Activities. Nat. Prod. Rep. 2003, 20, 565–583. [Google Scholar] [CrossRef]
- Lopes, L.M.X.; Humpfer, E. 8-Benzylberbine and N-Oxide Alkaloids from Aristolochia gigantea. Phytochemistry 1997, 45, 431–435. [Google Scholar]
- Francisco, C.S.; Messiano, G.B.; Lopes, L.M.X.; Tininis, A.G.; de Oliveira, J.E.; Capellari, L., Jr. Classification of Aristolochia Species Based on GC-MS and Chemometric Analyses of Essential Oils. Phytochemistry 2008, 69, 168–175. [Google Scholar]
- Leitão, G.G.; Lopes, D.; Menezes, F.D.S.; Kaplan, M.A.C.; Craveiro, A.A.; Alencar, J.W. Essential Oils from Brazilian Aristolochia. J. Essent. Oil Res. 1991, 3, 403–408. [Google Scholar] [CrossRef]
- Cortes, D.; Dadoun, H.; Paiva, R.L.R.; de Oliveira, A.B. Nouveaux Alcaloïdes Bis-benzylisoquinoleiques Isoles des Feuilles de Aristolochia gigantea. J. Nat. Prod. 1987, 50, 910–914. [Google Scholar] [CrossRef]
- Lopes, L.M.X. 8-Benzylberbine Alkaloids from Aristolochia gigantea. Phytochemistry 1992, 31, 4005–4009. [Google Scholar] [CrossRef]
- Sang, S.; Lao, A.; Wang, H.; Chen, Z.; Uzawa, J.; Fujimoto, Y. A Phenylpropanoid Glycoside from Vaccaria segetalis. Phytochemistry 1998, 48, 569–571. [Google Scholar]
- Cane, D.E.; Ha, H.-J.; McIlwaine, D.B.; Pascoe, K.O. The Synthesis of (3R)-Nerolidol. Tetrahedron Lett. 1990, 31, 7553–7554. [Google Scholar] [CrossRef]
- Suarez, L.E.C.; Menichini, F.; Monache, F.D. Tetranortriterpenoids and Dihydrocinnamic Acid Derivatives from Hortia colombiana. J. Braz. Chem. Soc. 2002, 13, 339–344. [Google Scholar] [CrossRef]
- Nakabayashi, R.; Kusano, M.; Kobayashi, M.; Tohge, T.; Yonekura-Sakakibara, K.; Kogure, N.; Yamazaki, M.; Kitajima, M.; Saito, K.; Takayama, H. Metabolomics-oriented Isolation and Structure Elucidation of 37 Compounds Including Two Anthocyanins from Arabidopsis thaliana. Phytochemistry 2009, 70, 1017–1029. [Google Scholar]
- Nes, W.D.; Norton, R.A.; Benson, M. Carbon-13 NMR Studies on Sitosterol Biosynthesized from [13C] Mevalonates. Phytochemistry 1992, 31, 805–811. [Google Scholar]
- Latip, J.; Hartley, T.G.; Waterman, P.G. Lignans and Coumarins Metabolites from Melico pehayesii. Phytochemistry 1999, 51, 107–110. [Google Scholar]
- Chang, Y.-C.; Chen, C.-Y.; Chang, F.-R.; Wub, Y.-C. Alkaloids from Lindera glauca. J. Chin. Chem. Soc. 2001, 48, 811–815. [Google Scholar]
- Leu, Y.-L.; Chan, Y.-Y.; Hsu, M.-Y.; Chen, I.-S.; Wu, T.-S. The Constituents of the Stem and Roots of Aristolochia foveolata. J. Chin. Chem. Soc. 1998, 45, 539–541. [Google Scholar]
- Zhang, Y.T.; Jiang, J.Q. Alkaloids from Aristolochia manshuriensis (Aristolochiaceae). Helv. Chim. Acta 2006, 89, 2665–2670. [Google Scholar] [CrossRef]
- Hegde, V.R.; Borges, S.; Patel, M.; Das, P.R.; Wu, B.; Gullo, V.P.; Chan, T.-M. New Potential Antitumor Compounds from the Plant Aristolochia manshuriensis as Inhibitors of the CDK2 Enzyme. Bioorg. Med. Chem. Lett. 2010, 20, 1344–1346. [Google Scholar] [CrossRef]
- Priestap, H.A. Seven Aristolactams from Aristolochia argentina. Phytochemistry 1985, 24, 849–852. [Google Scholar]
- Chen, J.-H.; Du, Z.-Z.; Shen, Y.-M.; Yang, Y.-P. Aporphine Alkaloids from Clematis parviloba and their Antifungal Activity. Arch. Pharm. Res. 2009, 32, 3–5. [Google Scholar] [CrossRef]
- Cutillo, F.; D’Abrosca, B.; DellaGreca, M.; Di Marino, C.; Golino, A.; Previtera, L.; Zarrelli, A. Cinnamic Acid Amides from Chenopodium album: Effects on Seeds Germination and Plant Growth. Phytochemistry 2003, 64, 1381–1387. [Google Scholar]
- Lin, W.-H.; Fu, H.-Z.; Hano, Y.; Nomura, T. Alkaloids from the Roots of Aristolochia Triangularis (I). J. Chin. Pharm. Sci. 1997, 6, 8–13. [Google Scholar]
- Pedersen, H.A.; Steffensen, S.K.; Christophersen, C. Cinnamoylphenethylamine 1H-NMR Chemical Shifts: A Concise Reference for Ubiquitous Compounds. Nat. Prod. Commun. 2010, 5, 1259–1262. [Google Scholar]
- Navickiene, H.M.D.; Lopes, L.M.X. Alkamides and Phenethyl Derivatives from Aristolochia gehrtii. J. Braz. Chem. Soc. 2001, 12, 467–472. [Google Scholar]
- Ferreira, D.T.; Alvares, P.S.M.; Houghton, P.J.; Braz-Filho, R. Constituintes Químicos das Raízes de Pyrostegia Venusta e Considerações Sobre a sua Importância Medicinal. Quim. Nova 2000, 23, 42–46. [Google Scholar] [CrossRef]
- Lee, M.-Y.; Lee, N.-H.; Jung, D.; Lee, J.-A.; Seo, C.-S.; Lee, H.; Kim, J.-H.; Shin, H.-K. Protective Effects of Allantoin Against Ovalbumin (OVA)-Induced Lung Inflammation in a Murine Model of Asthma. Int. Immunopharmacol. 2010, 10, 474–480. [Google Scholar] [CrossRef]
- Simas, N.K.; Lima, E.C.; Conceição, S.R.; Kuster, R.M.; Oliveira, A.M. Produtos Naturais para o Controle da Transmissão da Dengue: Atividade Larvicida de Myroxylon balsamum (óleo vermelho) e de Terpenóides e Fenilpropanóides. Quim. Nova 2004, 27, 46–49. [Google Scholar]
- Shen, Y.; Li, C.G.; Zhou, S.F.; Pang, E.C.K.; Story, D.F.; Xue, C.C.L. Chemistry and Bioactivity of Flos Magnoliae, a Chinese Herb for Rhinitis and Sinusitis. Curr. Med. Chem. 2008, 15, 1616–1627. [Google Scholar] [CrossRef]
- Tringali, C.; Spatafora, C.; Calì, V.; Simmonds, M.S.J. Antifeedant Constituents from Fagara macrophylla. Fitoterapia 2001, 72, 538–543. [Google Scholar] [CrossRef]
- Yokozawa, T.; Satoh, A.; Cho, E.J.; Kashiwada, Y.; Ikeshiro, Y. Protective Role of Coptidis Rhizoma Alkaloids Against Peroxynitrite-induced Damage to Renal Tubular Epithelial Cells. J. Pharm.Pharmacol. 2005, 57, 367–374. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Holzbach, J.C.; Lopes, L.M.X. Aristolactams and Alkamides of Aristolochia gigantea. Molecules 2010, 15, 9462-9472. https://doi.org/10.3390/molecules15129462
Holzbach JC, Lopes LMX. Aristolactams and Alkamides of Aristolochia gigantea. Molecules. 2010; 15(12):9462-9472. https://doi.org/10.3390/molecules15129462
Chicago/Turabian StyleHolzbach, Juliana C., and Lucia M. X. Lopes. 2010. "Aristolactams and Alkamides of Aristolochia gigantea" Molecules 15, no. 12: 9462-9472. https://doi.org/10.3390/molecules15129462
APA StyleHolzbach, J. C., & Lopes, L. M. X. (2010). Aristolactams and Alkamides of Aristolochia gigantea. Molecules, 15(12), 9462-9472. https://doi.org/10.3390/molecules15129462




