Antifungal Activity of the Essential Oil of Illicium verum Fruit and Its Main Component trans-Anethole
Abstract
:1. Introduction
2. Results and Discussion
2.1. Essential oil analysis
| Compounda | RIb | RA (%)c |
|---|---|---|
| Camphene | 950 | 0.2 |
| β-Myrcene | 991 | 0.1 |
| α-Phellandrene | 1003 | 0.1 |
| δ-3-Carene | 1016 | 0.3 |
| p-Cymene | 1029 | 0.1 |
| Limonene | 1033 | 0.4 |
| trans-Ocimene | 1040 | 0.1 |
| γ-Terpinene | 1060 | 0.1 |
| Terpinolene | 1100 | 0.1 |
| Linalool | 1114 | 0.3 |
| Terpinen-4-ol | 1177 | 0.2 |
| Isobornyl thiocyanoacetate | 1203 | 0.4 |
| γ-Terpineol | 1198 | 0.4 |
| cis-Anethole | 1254 | 0.7 |
| trans-Anethole | 1387 | 89.5 |
| β-Elemene | 1389 | 0.1 |
| Cyperene | 1398 | 0.2 |
| β-Caryophyllene | 1418 | 0.1 |
| α-Caryophyllene | 1450 | 0.1 |
| (+)-9-Epiledene | 1507 | 0.1 |
| Cubebene | 1672 | 0.1 |
| 2-(1-cyclopentenyl)-Furan | 1694 | 0.9 |
| Total identified | 94.6 | |
| Monoterpene hydrocarbons | 1.5 | |
| Oxygenated monoterpenes | 1.3 | |
| Sesquiterpene hydrocarbons | 0.4 |
2.2. Antifungal activity by direct contact assay
| Fungus | Fruit oil IC50 (mg/mL) | trans-Anethole IC50 (mg/mL) |
|---|---|---|
| Alternaria solani | 0.09 | 0.11 |
| Bipolaris maydis | 0.07 | 0.06 |
| Botryodiplodia theobromae | 0.11 | 0.09 |
| Fusarium graminearum | 0.08 | 0.10 |
| Fusarium oxysporum f. sp. cucumerinum | 0.16 | 0.17 |
| Fusarium oxysporum f. sp. lycopersici | 0.14 | 0.14 |
| Fusarium oxysporumf. sp. vasinfectum | 0.25 | 0.20 |
| Magnaporthe oryzae | 0.22 | 0.25 |
| Pythium aphanidermatum | 0.09 | 0.07 |
| Rhizoctonia cerealis | 0.10 | 0.11 |
| Rhizoctonia solani | 0.08 | 0.07 |
2.3. Antifungal activity by vapor contact assay
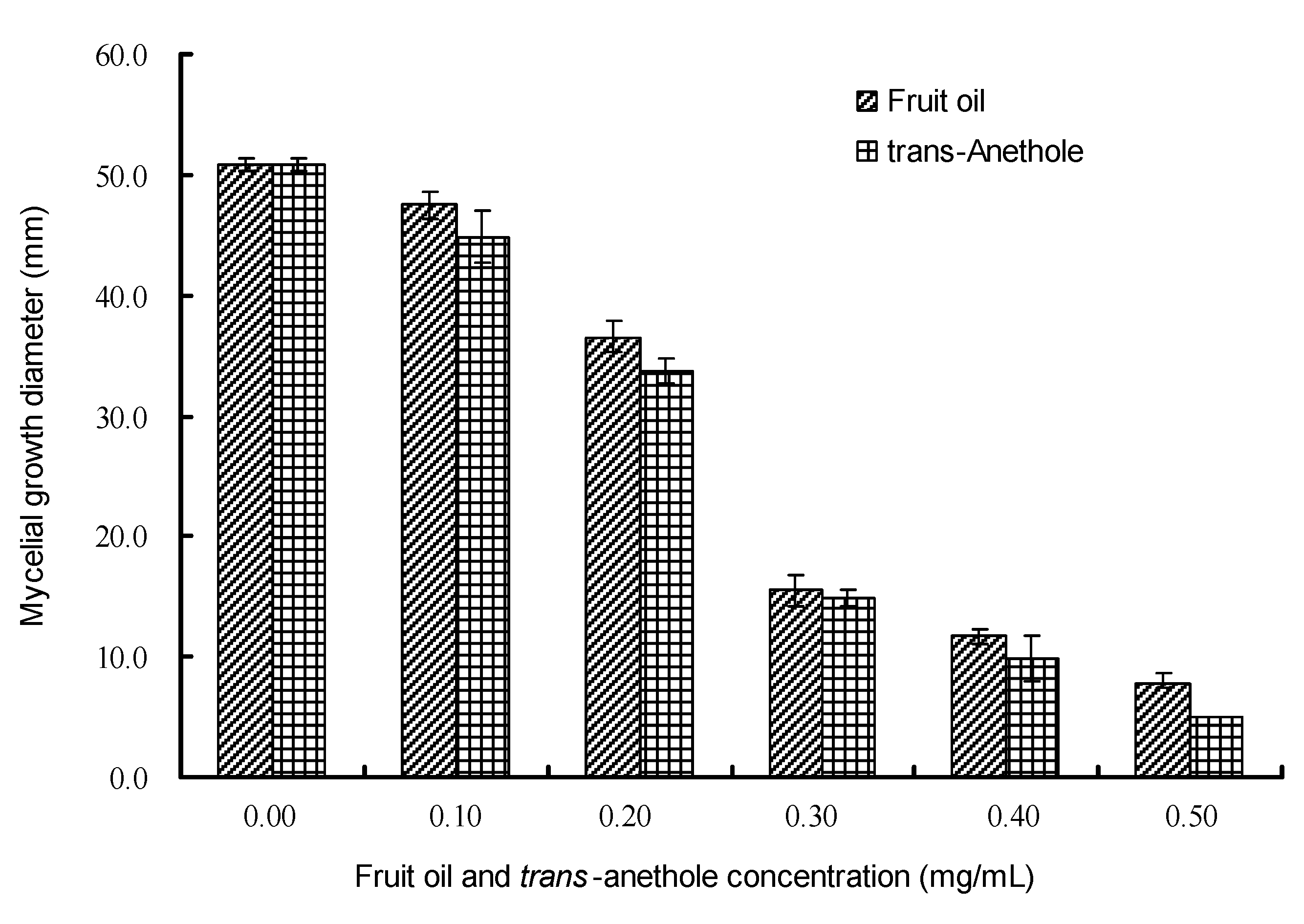
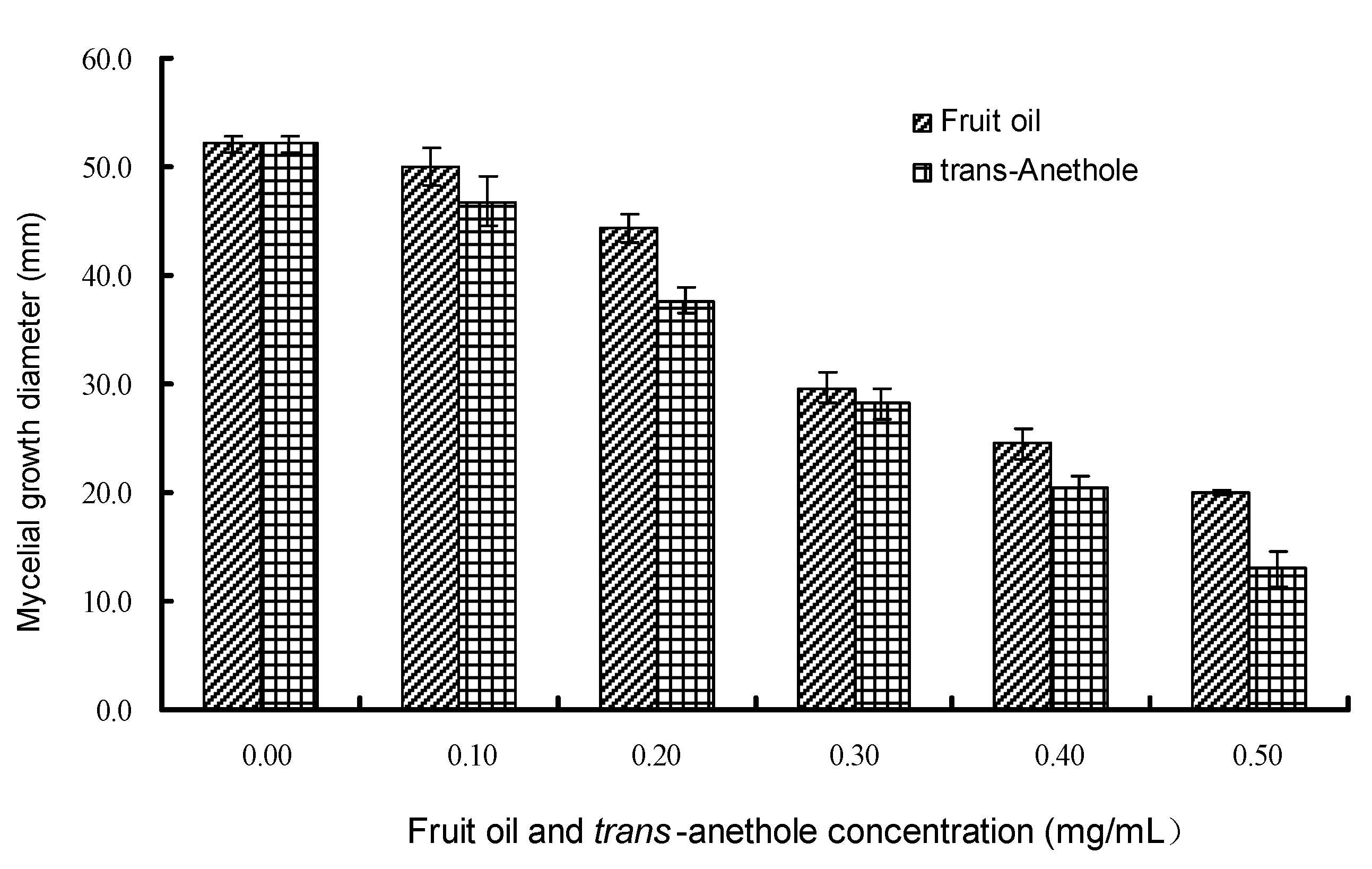
2.4. Inhibitory activity on spore germination
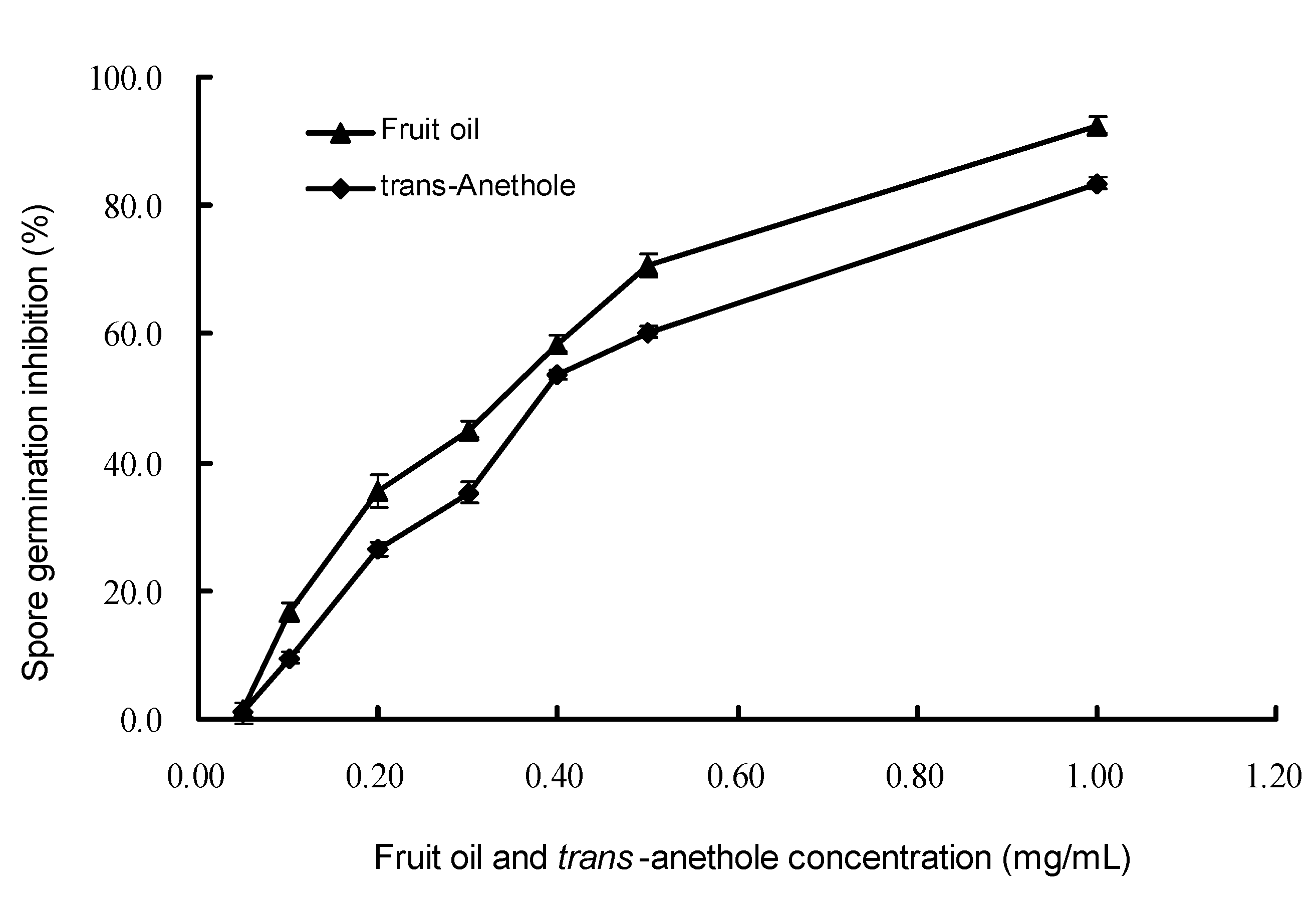
3. Experimental
3.1. Plant material
3.2. Preparation of the essential oil
3.3. Essential oil analysis
3.4. Test fungi
3.5. Evaluation of the antifungal activity
3.5.1. Determination of antifungal activity by direct contact assay
3.5.2. Determination of antifungal activity by vapor contact assay
3.5.3. Determination of inhibitory activity on spore germination
4. Conclusions
Acknowledgements
- Samples Availability: Samples of the compound and essential oil are available from the authors.
References
- Pedras, M.S.C. Prospects for controlling plant fungal diseases: alternatives based on chemical ecology and biotechnology. Can. J. Chem. 2004, 82, 1329–1335. [Google Scholar] [CrossRef]
- Fletcher, J.; Bender, C.; Budowle, B.; Cobb, W.T.; Gold, S.E.; Ishimaru, C.A.; Luster, D.; Melcher, U.; Murch, R.; Scherm, H.; Seem, R.C.; Sherwood, J.L.; Sobral, B.W.; Tolin, S.A. Plant pathogen forensics: capabilities, needs, and recommendations. Microbiol. Mol. Biol. Rev. 2006, 70, 450–471. [Google Scholar] [CrossRef]
- Yamaguchi, I.; Fujimura, M. Recent topics on action mechanisms of fungicides. J. Pestic. Sci. 2005, 30, 67–74. [Google Scholar] [CrossRef]
- Margni, M.; Rossier, D.; Crettaz, P.; Jolliet, O. Life cycle impact assessment of pesticides on human health and ecosystems. Agr. Ecosyst. Environ. 2002, 93, 379–392. [Google Scholar] [CrossRef]
- Hadacek, F.; Greger, H. Testing of antifungal natural products: methodologies, comparability of results and assay choice. Phytochem. Anal. 2000, 11, 137–147. [Google Scholar] [CrossRef]
- Dixon, R.A. Natural products and plant disease resistance. Nature 2001, 411, 844–847. [Google Scholar]
- Gimenez, C.; Cabrera, R.; Reina, M.; Gonalez-Coloma, A. Fungal endophytes and their role in plant protection. Curr. Org. Chem. 2007, 11, 707–720. [Google Scholar] [CrossRef]
- Yen, T.; Chang, H.; Hsieh, C.; Chang, S. Antifungal properties of ethanolic extract and its active compounds from Calocedrus macrolepis var. formosana (Florin) heartwood. Bioresource Technol. 2008, 99, 4871–4877. [Google Scholar] [CrossRef]
- Zhou, L.; Li, D.; Wang, J.; Liu, S.; Wu, J. Antibacterial phenolic compounds from the spines of Gleditsia sinensis Lam. . Nat. Prod. Res. 2007, 21, 283–291. [Google Scholar] [CrossRef]
- Zhou, L.; Li, D.; Jiang, W.; Qin, Z.; Zhao, S.; Qiu, M.; Wu, J. Two ellagic acid glycosides from Gleditsia sinensis Lam. with antifungal activity on Magnaporthe grisea. Nat. Prod. Res. 2007, 21, 303–309. [Google Scholar] [CrossRef]
- Lahlou, M. Essential oils and fragrance compounds: bioactivity and mechanisms of action. Flav. Fragr. J. 2004, 19, 159–165. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Tripathi, P.; Dubey, N.K. Exploitation of natural products as an alternative strategy to control postharvest fungal rotting of fruit and vegetables. Postharvest Biol. Technol. 2004, 32, 235–245. [Google Scholar] [CrossRef]
- Reddy, M.V.B.; Angers, P.; Gosselin, A.; Arul, J. Characterization and use of essential oil from Thymus vulgaris against Botrytis cinerea and Rhizopus stolonifer in strawberry fruits. Phytochemistry 1998, 47, 1515–1520. [Google Scholar]
- Caccioni, D.R.L.; Guizzardi, M.; Biondi, D.M.; Renda, A.; Ruberto, G. Relationship between volatile components of Citrus fruits essential oil and antimicrobial action on Penicillium digitatum and Penicillium italicum. Int. J. Food Microbiol. 1998, 43, 73–79. [Google Scholar] [CrossRef]
- Tripathi, P.; Dubey, N.K.; Banerji, R.; Chansouria, J.P.N. Evaluation of some essential oils as botanical fungitoxicants in management of post-harvest rotting of citrus fruits. World J. Microbiol. Biotechnol. 2004, 20, 317–321. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, L.; Li, T.; Zhou, X.; Ding, L.; Yu, Y.; Yu, A.; Zhang, H. Rapid analysis of the essential oils from dried Illicium verum Hook. f. and Zingiber officinale Rosc. by improved solvent-free microwave extraction with three types of microwave-absorption medium. Anal. Bioanal. Chem. 2006, 386, 1863–1868. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils - A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Okuyama, E.; Nakamura, T.; Yamazaki, M. Convulsants from star anise (Illicium verum Hook. f.). Chem. Pharm. Bull. 1993, 41, 1670–1671. [Google Scholar] [CrossRef]
- Lee, S.; Li, G.; Lee, K.S.; Jung, J.; Xu, M.; Seo, C.; Chang, H.; Kim, S.; Song, D.; Son, J. Preventive agents against sepsis and new phenylpropanoid glucosides from the fruits of Illicium verum. Planta Med. 2003, 69, 861–864. [Google Scholar] [CrossRef]
- Singh, G.; Maurya, S.; de Lampasona, M.P.; Catalan, C. Chemical constituents, antimicrobial investigations and antioxidative potential of volatile oil and acetone extract of star anise fruits. J. Sci. Food Agr. 2006, 86, 111–121. [Google Scholar] [CrossRef]
- Padmashree, A.; Roopa, N.; Semwal, A.D.; Sharma, G.K.; Agathian, G.; Bawa, A.S. Star-anise (Illicium verum) and black caraway (Carum nigrum) as natural antioxidants. Food Chem. 2007, 104, 59–66. [Google Scholar] [CrossRef]
- De, M.; De, A.K.; Sen, P.; Banerjee, A.B. Antimicrobial properties of star anise (Illicium verum). Phytother. Res. 2002, 16, 94–95. [Google Scholar] [CrossRef]
- Ravid, U.; Putievsky, E.; Katzir, I. Chiral GC analysis of enantiomerically pure (R)-(-)-linalyl acetate in some Lamiaceae, myrtle and petigrain oils. Flav. Fragr. J. 1994, 9, 275–276. [Google Scholar] [CrossRef]
- Gudzic, B.; Djokovic, D.; Vajs, V.; Palic, R.; Stojanovic, G. Composition and antimicrobial activity of the essential oil of Hypericum maculatum Crantz. Flav. Fragr. J. 2002, 17, 392–394. [Google Scholar] [CrossRef]
- Sajjadi, S.E.; Rahiminezhad, M.R.; Mehregan, I.; Poorassar, A. Constituents of essential oil of Hypericum dogonbadanicum Assadi. J. Essent. Oil Res. 2001, 13, 43–44. [Google Scholar] [CrossRef]
- Cheng, S.; Liu, J.; Tsai, K.; Chen, W.; Chang, S. Chemical composition and mosquito larvicidal activity of essential oils from leaves of different Cinnamomum osmophloeum provenances. J. Agr. Food Chem. 2004, 52, 4395–4400. [Google Scholar] [CrossRef]
- Park, J.S.; Baek, H.H.; Bai, D.H.; Tae, K.; Lee, C.H. Antibacterial activity of fennel (Foeniculum vulgare Mill.) seed essential oil against the growth of Streptoccocus mutans. Food Sci. Biotechnol. 2004, 13, 581–585. [Google Scholar]
- Orav, A.; Raal, A.; Arak, E. Essential oil composition of Pimpinella aisum L. fruits from various European countries. Nat. Prod. Res. 2008, 22, 227–232. [Google Scholar] [CrossRef]
- Toth, M.; Schmera, D.; Imrei, Z. Optimization of a chemical attractant for Epicometis (Tropinota) hirta Poda. Z. Naturforsch. C 2004, 59, 288–292. [Google Scholar]
- Fujita, K.; Fujita, T.; Kubo, I. Anethole, a potential antimicrobial synergist, converts a fungistatic dodecanol to a fungicidal agent. Phytother. Res. 2007, 21, 47–51. [Google Scholar] [CrossRef]
- Knio, K.M.; Usta, J.; Dagher, S.; Zournajian, H.; Kreydivveh, S. Larvicidal activity of essential oils extracted from commonly used herbs in Lebanon against the seaside mosquito, Ochlerotatus caspius. Bioresource Technol. 2008, 99, 763–768. [Google Scholar] [CrossRef]
- Ravid, U.; Putievsky, E.; Katzir, I. Determination of the enantiomeric composition of α-terpineol in essential oils. Flav. Fragr. J. 1995, 10, 281–284. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oils Components by Gas Chromatography/Quadrupole Mass Spectroscopy; Allured Publishing Corporation: Carol Stream, IL, USA, 2001. [Google Scholar]
- Sacchetti, G.; Maietti, S.; Muzzoli, M.; Scaglianti, M.; Manfredini, S.; Radice, M.; Bruni, R. Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem. 2005, 91, 621–632. [Google Scholar] [CrossRef]
- Sokovic, M.D.; Vukojevic, J.; Marin, P.D.; Brkic, D.D.; Vajs, V.; van Griensven, L.J.L.D. Chemical composition of essential oils of Thymus and Mentha species and their antifungal activities. Molecules 2009, 14, 238–249. [Google Scholar] [CrossRef]
- Martino, L.D.; Bruno, M.; Formisano, C.; Feo, V.D.; Napolitano, F.; Rosselli, S.; Senatore, F. Chemical composition and antimicrobial activity of the essential oils from two species of Thymus growing wild in southern Italy. Molecules 2009, 14, 4614–4624. [Google Scholar] [CrossRef]
- Mohsen, H.; Ali, F. Essential oil composition of Artemisia herba-alba from southern Tunisia. Molecules 2009, 14, 1585–1594. [Google Scholar] [CrossRef]
- Wang, J.; Liu, H.; Zhao, J.; Gao, H.; Zhou, L.; Liu, Z.; Chen, Y.; Sui, P. Antimicrobial and antioxidant activities of the root bark essential oil of Periploca sepium and its main component 2-hydroxy-4-methoxybenzaldehyde. Molecules 2010, 15, 5807–5817. [Google Scholar] [CrossRef]
- Van den Dool, H.; Kratz, P.D. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Quiroga, E.N.; Sampietro, A.R.; Vattuone, M.A. Screening antifungal activities of selected medicinal plants. J. Ethnopharmacol. 2001, 74, 89–96. [Google Scholar] [CrossRef]
- Cakir, A.; Kordali, S.; Zengin, H.; Izumi, S.; Hirata, T. Composition and antifungal activity of essential oils isolated from Hypericum hyssopifolium and Hypericum heterophyllum. Flav. Fragr. J. 2004, 19, 62–68. [Google Scholar] [CrossRef]
- Song, W.; Zhou, L.; Yang, C.; Cao, X.; Zhang, L.; Liu, X. Tomato Fusarium wilt and its chemical control strategies in a hydroponic system. Crop Prot. 2004, 23, 243–247. [Google Scholar] [CrossRef]
- Gong, Y.; Huang, Y.; Zhou, L.; Shi, X.; Guo, Z.; Wang, M.; Jiang, W. Chemical composition and antifungal activity of the fruit oil of Zanthoxylum bungeanum Maxim. (Rutaceae) from China. J. Essen. Oil Res. 2009, 21, 174–178. [Google Scholar] [CrossRef]
- Finney, S. Probit Analysis; Cambridge University Press: Cambridge, UK, 1978. [Google Scholar]
- Letessier, M.P.; Svoboda, K.P.; Walters, S.D.R. Antifungal activity of the essential oil of hyssop (Hyssopus offcinalis). J. Phytopathol. 2001, 149, 673–678. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; Zhao, J.; Lu, S.; Wang, J.; Jiang, W.; Ma, Z.; Zhou, L. Isoquinoline alkaloids from Macleaya cordata active against plant microbial pathogens. Nat. Prod. Commun. 2009, 4, 1557–1560. [Google Scholar]
- Wang, J.; Zhao, J.; Liu, H.; Zhou, L.; Liu, Z.; Wang, J.; Han, J.; Yu, Z.; Yang, F. Chemical analysis and biological activity of the essential oils of two valerianaceous species from China: Nardostachys chinensis and Valeriana officinalis. Molecules 2010, 15, 6411–6422. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, Y.; Zhao, J.; Zhou, L.; Wang, J.; Gong, Y.; Chen, X.; Guo, Z.; Wang, Q.; Jiang, W. Antifungal Activity of the Essential Oil of Illicium verum Fruit and Its Main Component trans-Anethole. Molecules 2010, 15, 7558-7569. https://doi.org/10.3390/molecules15117558
Huang Y, Zhao J, Zhou L, Wang J, Gong Y, Chen X, Guo Z, Wang Q, Jiang W. Antifungal Activity of the Essential Oil of Illicium verum Fruit and Its Main Component trans-Anethole. Molecules. 2010; 15(11):7558-7569. https://doi.org/10.3390/molecules15117558
Chicago/Turabian StyleHuang, Yongfu, Jianglin Zhao, Ligang Zhou, Jihua Wang, Youwen Gong, Xujun Chen, Zejian Guo, Qi Wang, and Weibo Jiang. 2010. "Antifungal Activity of the Essential Oil of Illicium verum Fruit and Its Main Component trans-Anethole" Molecules 15, no. 11: 7558-7569. https://doi.org/10.3390/molecules15117558
APA StyleHuang, Y., Zhao, J., Zhou, L., Wang, J., Gong, Y., Chen, X., Guo, Z., Wang, Q., & Jiang, W. (2010). Antifungal Activity of the Essential Oil of Illicium verum Fruit and Its Main Component trans-Anethole. Molecules, 15(11), 7558-7569. https://doi.org/10.3390/molecules15117558





