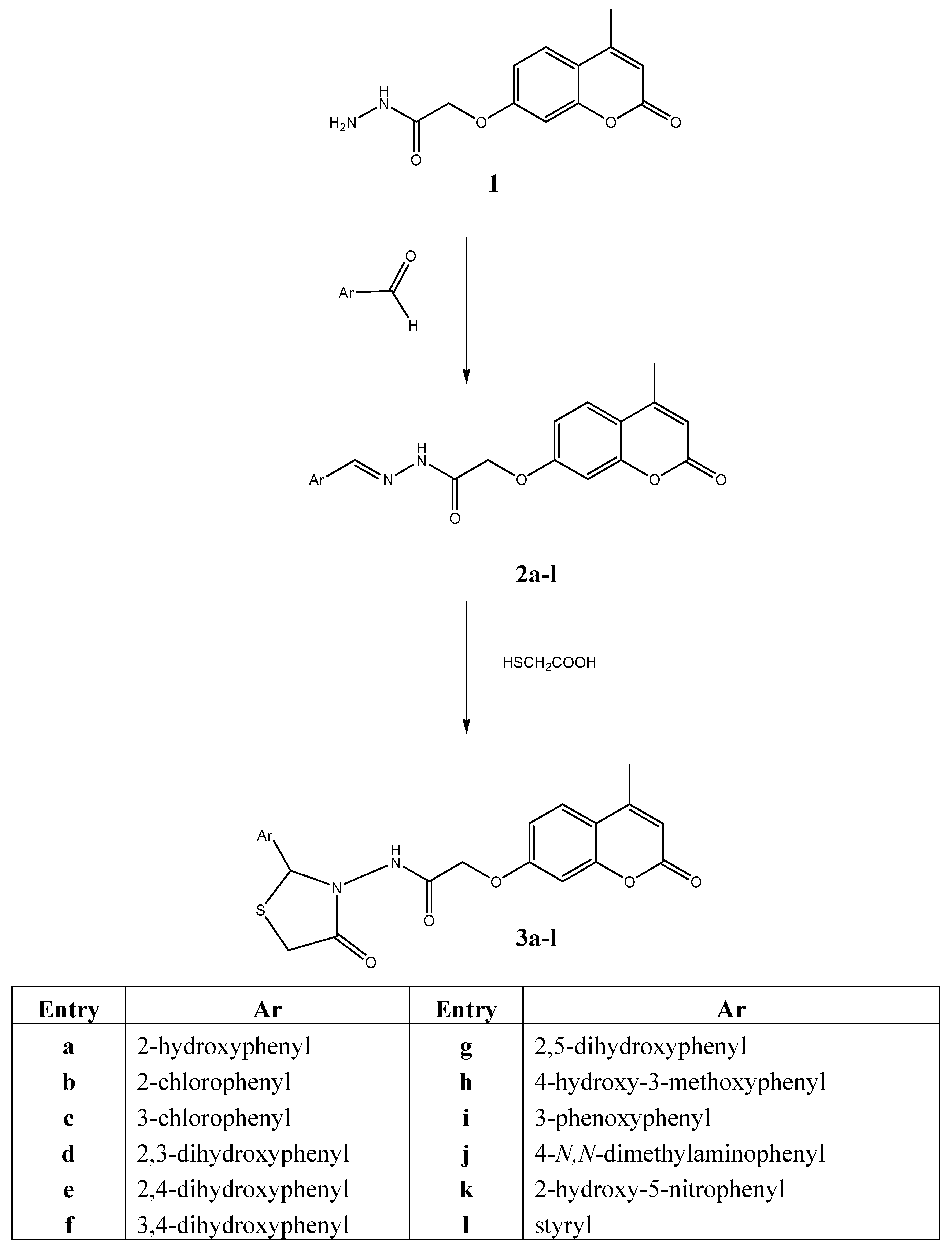
3.2. General procedure for preparation of (E)-Ń’-arylidene-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)-acetohydrazides 2a-l
A mixture of compound 1 (0.01 mole) and a suitable aromatic aldehyde (Ar/a-k; 0.01 mole) was refluxed in absolute ethanol (30 mL) in presence of a catalytic amount of glacial acetic acid for 2 to 4 hours. The reaction mixture was cooled and the precipitate was filtered and recrystallized from methanol to give compounds 2a-l.
(E)-Ń’-(2-Hydroxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (
2a) [
5]. M.p. 284–286 °C; yield (72%); FT-IR: ν
max 3,440; 3,282; 3,101; 2,916; 2,853; 1,724; 1,685; 1,621; 1,537; 1,489; 1,393; 1,300 and 1,154 cm
-1.
1H-NMR (δ, ppm): 2.41(s, 3H, CH
3, C-4); 4.85 (s, 2H, CH
2); 6.25 (s, 1H, C-3); 6.87 (s, 1H, H-8), 6.98 (d, 1H, H-6); 7.85 (d, 1H, H-5); 7.25-7.75 (m, 4H, arom.,) 8.33 (s, 1H, NH); 8.56 (s, 1H, HC=N-); 10.99 (s,1H, OH).
13C-NMR (δ, ppm): 21.4 (CH
3); 69.10 (OCH
2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 116.1 (C-3, Ar-); 118.4 (C-3, Ar-); 121.7 (C-5, Ar-); 127.8 (C-5); 130.5 (C-6, Ar-); 132.4 (C-1, Ar-); 161.2 (C-2, Ar-); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-4); 172.42 (CO-NH). Anal. Calcd. for C
19H
16N
2O
5 (352.34): C, 64.77; H, 4.58; N, 8.95. Found: C, 64.76; H, 4.55; N, 7.92
(E)-N´-(2-Chlorobenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (
2b) [
13]. M.p. 248–250 °C; yield (82%). FT-IR: ν
max 3,289; 2,950; 2,864; 1,712; 1,700; 1,624; 1,560; 1,525; 1,398; 1,290 and 1,150 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 4.84 (s, 2H, CH
2); 6.22 (s, 1H, H-3); 6.99 (s, 1H, H-8); 7.05 (d, 1H, H-6); 7.39-7.75 (m, 4H, arom.); 8.06 (d, 1H, H-5); 8.41(s, 1H, NH); 8.74 (s, 1H, HC=N-).
13C-NMR (δ, ppm): 21.4 (CH
3); 69.10 (OCH
2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 127.1 (C-5, Ar-); 127.8 (C-5); 129.1 (C-3, Ar-); 130.4 (C-6, Ar‑); 132.5 (C-4, Ar-); 133.2 (C-1, Ar-); 134.2 (C-2, Ar-); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C
19H
15ClN
2O
4 (370.79): C, 61.55; H, 4.08; N, 7.56. Found: C, 61.57; H, 4.08; N, 7.53.
(E)-N´-(3-Chlorobenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (
2c) [
5]. M.p. 242–244 °C; yield (80%). FT-IR: ν
max 3,448; 3,201; 3,102; 2,997; 1,713; 1,683; 1,617; 1,511; 1,434; 1,391; 1,274 and 1,136 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 4.82 (s, 2H, CH
2); 6.23 (s, 1H, H-3); 6.98 (s, 1H, H-8); 7.02 (d, 1H, H-6); 7.20 (d, 1H, H-5); 7.41-7.74 (m, 4H, arom.); 8.00 (s, 1H, NH); 8.31 (s, 1H, HC=N-).
13C-NMR (δ, ppm): 21.4 (CH
3); 69.10 (OCH
2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 127.3 (C-6, Ar-); 127.8 (C-5); 129.4 (C-2, Ar-); 130.6 (C-5, Ar-); 131.4 (C-4, Ar-); 134.5 (C-3, Ar-); 135.2 (C-1, Ar-); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.32 (CO-NH). Anal. Calcd. for C
19H
15ClN
2O
4 (370.79): C, 61.55; H, 4.08; N, 9.56. Found: C, 61.54; H, 4.07; N, 7.53.
(E)-N´-(2,3-Dihydroxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (2d). M.p. 270–274 °C; yield (63%). FT-IR: νmax 3,481; 3,436; 3,332; 3,280; 2,915; 1,719; 1,695; 1,620; 1,541; 1,473; 1,392; 1,265 and 1,153 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.19 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 9.27 (s, br., 1H, OH); 11.59 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 69.10 (OCH2 ); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 119.8 (C-4, Ar-); 120.1 (C-1, Ar-); 122.6 (C-5, Ar-); 123.5 (C-6, Ar-); 127.8 (C-5); 143.2 (C=N); 147.5 (C-3, Ar-); 151.4 (C-9); 151.8 (C-2, Ar-); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C19H16N2O6 (368.34): C, 61.95; H, 4.38; N, 7.61. Found: C, 61.93; H, 4.36; N, 7.59.
(E)-N´-(2,4-Dihydroxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (2e). M.p. 261–262 °C; yield (48%). FT-IR: νmax 3,357; 3,273; 3,169; 3,086; 2,924; 1,716; 1,671; 1,613; 1,512; 1,425; 1,391; 1,264 and 1,152 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 10.12 (s, br., 1H, OH); 11.40 (s, br., 1H, OH). 13C- NMR (δ, ppm): 21.4 (CH3); 69.10 (OCH2 ); 103.5 (C-3, from Ph); 107.6 (C-8); 108.6 (C-5, Ar-); 111.0 (C-6); 111.3 (C-1, Ar-); 112.8 (C-3); 113.5 (C-9); 127.8 (C-5); 132.5 (C-6, Ar-); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 162.4 (C-4, Ar-); 162.8 (C-2, Ar-); 172.40 (CO-NH). Anal. Calcd. for C19H16N2O6 (368.34): C, 61.95; H, 4.38; N, 7.61. Found: C, 61.93; H, 4.39; N, 7.59.
(E)-N´-(3,4-Dihydroxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (2f). M.p. 243–244 °C; yield (38%). FT-IR: νmax 3,393; 3,239; 3,075; 3,071; 2,980; 1,716; 1,694; 1,612; 1,541; 1,509; 1,494; 1,393; 1,262 and 1,159 cm-1. 1H-NMR(δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 9.35 (s, br., 1H, OH); 10.14 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 69.10 (OCH2 ); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 116.8 (C-2, Ar‑); 117.6 (C-5, Ar); 123.5 (C-6, Ar-); 127.3 (C-1, Ar-); 127.8 (C-5); 143.2 (C=N); 143.5 (C-3, Ar-); 149.8 (C-4, Ar-); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C19H16N2O6 (368.34): C, 61.95; H, 4.38; N, 7.61. Found: C, 61.90; H, 4.35; N, 7.62.
(E)-N´-(2,5-Dihydroxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (2g). M.p. 273–274 °C; yield (41%). IR: νmax 3,418; 3,276; 3,075; 2,915; 1,705; 1,686; 1,658; 1,622; 1,611; 1,584; 1,550; 1,512; 1,428; 1,392; 1,288 and 1,149 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.,); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 9.26 (s, br., 1H, OH); 11.93 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 116.5 (C-6, Ar-); 117.5 (C-3, Ar-); 119.4 (C-4, Ar-); 120.1 (C-1, Ar-); 127.8 (C-5); 143.2 (C=N); 151.4 (C-9); 151.3 (C-5, Ar-); 152.9 (C-4); 153.8 (C-2, Ar-); 160.3 (C-7); 160.8 (C-2); 172.44 (CO-NH). Anal. Calcd. for C19H16N2O6 (368.34): C, 61.95; H, 4.38; N, 7.61. Found: C, 61.92; H, 4.37; N, 7.62.
(E)-N´-(4-Hydroxy-3-methoxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetohydrazide (
2h) [
5]. M.p. 253–254 °C; yield (76%). FT-IR: ν
max 3,530; 3,481; 3,319; 3,268; 2,914; 1,720; 1,695; 1,618; 1,540; 1,472; 1,390; 1,265 and 1,153 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 3.82 (3H, s, OCH
3); 4.84 (s, 2H, OCH
2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 11.50 (s, br., 1H, OH).
13C-NMR (δ, ppm): 21.4 (CH
3); 56.2 (OCH
3); 69.10 (OCH
2 ); 107.6 (C-8);); 111.0 (C-6); 112.8 (C-3); 113.5 (C‑9); 114.8 (C-2, Ar-); 117.3 (C-5, Ar-); 123.0 (C-6, Ar-); 127.2 (C-1, Ar-); 127.8 (C-5); 143.2 (C=N); 148.1 (C-4, Ar-); 151.4 (C-9); 151.5 (C-3, Ar-); 152.9 (C-4); 160.3 (C-7); 160.7 (C-2); 172.42 (CO-NH). Anal. Calcd. for C
20H
18N
2O
6 (382.37): C, 62.82; H, 4.74; N, 7.33. Found: C, 62.79; H, 4.73; N, 7.31.
(E)-N´-(3-Phenoxybenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)-acetohydrazide (2i). M.p. 178–180 °C; yield (72%). FT-IR: νmax 3,526; 3,476; 3,329; 3,288; 2,916; 1,723; 1,690; 1,623; 1,544; 1,470; 1,392; 1,265 and 1,153 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 9H, arom.,); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-). 13C-NMR (δ, ppm): 21.4 (CH3); 56.2 (OCH3); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 116.8 (C-2, Ar-Ph); 117.4 (C-2,6, Ar- PhO); 119.7 (C-4, Ar- Ph); 121.8 (C-4, Ar-PhO); 122.3 (C-6, Ar- Ph); 127.8 (C-5); 128.5 (C-3,5, Ar-PhO); 128.8 (C-5, Ar- Ph); 133.6 (C-1, Ar- Ph); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 157.1 (C-1, Ar- PhO); 157.3 (C-3, Ar- Ph); 160.3 (C-7); 160.8 (C-2); 172.40 (CO-NH). Anal. Calcd. for C25H20N2O5 (428.44): C, 70.08; H, 4.71; N, 6.54. Found: C, 70.10; H, 4.72; N, 6.55.
(E)-N´-(4-(Dimethylamino)benzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetohydrazide (
2j) [
6]. M.p. 260–262 °C; yield (86%). FT-IR: ν
max 3,528; 3,471; 3,329; 3,285; 2,912; 1,721; 1,695; 1,621; 1,541; 1,473; 1,392; 1,265 and 1,153 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 2.86 (s, 6H, N(CH
3)
2); 4.84 (s, 2H, CH
2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 4H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-)
13C-NMR, (δ, ppm): 21.4 (CH
3); 41.4 (N(CH
3 )
2); 69.10 (OCH
2 ); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 114.5 (C-3,5, Ar‑); 123.04 (C-1, Ar-); 127.8 (C-5); 130.2 (C-2,6, Ar-); 143.2 (C=N); 151.4 (C-10); 151.8 (C-4, Ar-); 152.8 (C-4,coum.); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. For C
21H
21N
3O
4 (379.41): C, 66.48; H, 5.58; N, 11.08. Found: C, 66.50; H, 5.57; N, 11.10.
(E)-N´-(2-Hydroxy-5-nitrobenzylidene)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)acetohydrazide (2k). M.p. 288–290 °C; yield (90%). FT-IR: νmax 3,536; 3,481; 3,329; 3,268; 2,910; 1,724; 1,690; 1,622; 1,539; 1,474; 1,390; 1,265 and 1,153 cm-1. 1H NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 4.84 (s, 2H, CH2); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-); 11.59 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 117.1 (C-3, Ar-); 119.6 (C-1, Ar-); 120.1 (C-1, Ar-); 124.5 (C-4, Ar-); 125.7 (C-6, Ar-); 127.8 (C-5); 141.3 (C-5, Ar-); 143.2 (C=N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.8 (C-2); 167.8 (C-2, Ar-); 172.41 (CO-NH). Anal. Calcd. for C19 H15N3O7 (397.34): C, 57.43 ; H, 3.81; N,10.58. Found: C, 57.40; H, 3.82; N, 10.54.
(E)-2-(4-Methyl-2-oxo-2H-chromen-7-yloxy)-N´-[(E)-3-phenylallylidene]acetohydrazide (
2l) [
6]. M.p. 249–250 °C; yield (46%). FT-IR: ν
max 3,530; 3,481; 3,339; 3,288; 2,916; 1,720; 1,695; 1,621; 1,541; 1,473; 1,392; 1,265 and 1,153 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 4.84 (s, 2H, CH
2); 5.72 (d, 1H, styryl); 6.24 (s, 1H, H-3); 6.63 (d, 1H, styryl); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 8.52 (s, 1H, HC=N-).
13C-NMR (δ, ppm): 21.4 (CH
3); 69.10 (OCH
2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 126.1 (C-1, Ar-ethenyl); 126.6 (C-2,6, Ar-); 127.8 (C-5); 128.5 (C-4, Ar-); 128.9 (C-3,5, Ar-); 137.2 (C=N); 139.2 (C-2, Ar-ethenyl); 151.4 (C-10); 152.9 (C-4); 160.3 (C-7); 161.0 (C-2); 172.42 (CO-NH). Anal. Calcd. For C
21H
18N
2O
4 (362.38): C, 69.60; H, 5.01; N, 7.73. Found. C, 69.59; H, 4.99; N, 7.70.
3.3. General procedure for preparation of N-(2-(substituted)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamides 3a-l [33]
To a solution of (E)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)-N´-[(E)-3-arylidene]acetohydrazide 2a-l (0.01 mol) in dry 1,4-dioxane (15 mL), freshly distilled mercaptoacetic acid (0.01 mol) and anhydrous ZnCl2 (0.1 g) were added and the mixture was heated under reflux 10 to 12 hours. The solvent was removed (reduced pressure) and residue washed with 5% sodium bicarbonate solution (3 × 20 mL) and water (2 × 20 mL), dried, and recrystallized from an appropriate solvent.
N-(2-(2-Hydroxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3a). M.p. 284–286 °C; yield (72%). FT-IR: νmax 3,418; 3,182; 3,100; 2,906; 2,853; 1,725; 1,681; 1,623; 1,527; 1,489; 1,393; 1,300 and 1,144 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.34 (s, 2H, COCH2S); 4.84 (s, 2H, OCH2); 5.28 (s, 1H, SCHN); 6.24 (s, 1H, C-3); 6.87 (s, 1H, H-8), 6.98 (d, 1H, H-6); 7.25–7.75 (m, 4H, arom.); 8.06 (d, 1H, H-5); 8.33(s, 1H, NH); 10.99 (s, br.,.1H, OH). 13C- NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 69.10 (OCH2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.6 (C-10); 116.1 (C-3, Ar-); 118.4 (C-3, Ar-); 121.7 (C-5, Ar-); 127.8 (C-5); 130.5 (C-6, Ar-); 132.4 (C-1, Ar-); 161.2 (C-2, Ar-); 143.2 (CO-N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-4); 172.45 (CO-NH). Anal. Calcd. for C21H18N2O6S (426.44), C, 59.15; H, 4.25; N, 6.57; S, 7.52. Found: C, 59.10; H, 4.27; N, 6.54; S, 7.49.
N-(2-(2-Chlorophenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (
3b) [
26]. M.p. 248–250 °C; yield (82%). FT-IR: ν
max 3,280; 2,940; 2,854; 1,722; 1,710; 1,621; 1,580; 1,515; 1,394; 1,295 and 1,146 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 3.35 (s, 2H, COCH
2S); 4.84 (s, 2H, OCH
2); 5.29 (s, 1H, SCHN); 6.22 (s, 1H, H-3); 6.99 (s, 1H, H-8); 7.05 (d, 1H, H-6); 7.39–7.75 (m, 4H, arom.); 8.06 (d, 1H, H-5); 8.41 (s, 1H, NH).
13C-NMR (δ, ppm): 21.4 (CH
3); 32.92 (COCH
2S); 47.81 (SCHN); 69.10 (OCH
2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.4 (C-10); 127.1 (C-5, Ar-); 127.8 (C-5); 129.1 (C-3, Ar-); 130.4 (C-6, Ar-); 132.5 (C-4, Ar-); 133.2 (C-1, Ar-); 134.2 (C-2, Ar-); 143.2 (CO-N); 151.2 (C-19); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C
21H
17Cl N
2O
5S (444.89), C, 56.69; H, 3.85; N, 6.30; S, 7.21. Found: C, 56.70; H, 3.83; N, 6.31; S, 7.20.
N-(2-(3-Chlorophenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3c). M.p. 242–244 °C; yield (86%). FT-IR: νmax 3,438; 3,200; 3,112; 2,987; 1,723; 1,688; 1,627; 1,516; 1,430; 1,390; 1,264 and 1,136 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.38 (s, 2H, COCH2S); 4.82 (s, 2H, CH2); 5.28 (s, 1H, SCHN); 6.23 (s, 1H, H-3); 6.98 (s, 1H, H-8); 7.02 (d, 1H, H-6); 7.41–7.74 (m, 4H, arom.); 8.09 (d, 1H, H-5); 8.40 (s, 1H, NH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.65 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 127.3 (C-6, Ar-); 127.8 (C-5); 129.4 (C-2, Ar-); 130.6 (C-5, Ar-); 131.4 (C-4, Ar-); 134.5 (C-3, Ar-); 135.2 (C-1, Ar-); 143.3 (CO-N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C21H17Cl N2O5S (444.89), C, 56.69; H, 3.85; N, 6.30; S, 7.21.Found: C, 56.62; H, 3.82; N, 6.31; S, 7.24.
N-(2-(2,3-Dihydroxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3d). M.p. 270–271 °C; yield (63%). FT-IR: νmax 3,531; 3,471; 3,309; 3,268; 2,926; 1,725; 1,698; 1,626; 1,551; 1,483; 1,388; 1,260 and 1,143 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.34 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.28 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90–7.75 (m, 3H, arom.); 8.32 (s, 1H, NH); 9.27 (s, br., 1H, OH); 11.59 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 119.8 (C-4, Ar-); 120.1 (C-1, Ar-); 122.6 (C-5, Ar-); 123.5 (C-6, Ar-); 127.8 (C-5); 143.2 (CO-N); 147.5 (C-3, Ar-); 151.4 (C-9); 151.8 (C-2, Ar-); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.43 (CO-NH). Anal. Calcd. for C21H18N2O7S (442.44), C, 57.01; H, 4.10; N, 6.33; S, 7.25. Found: C, 57.00; H, 4.08; N, 6.30; S, 7.23.
N-(2-(2,4-Dihydroxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3e). M.p. 261–262 °C; yield (48%). FT-IR: νmax 3,345; 3,282; 3,172; 3,106; 2,912; 1,726; 1,691; 1,622; 1,510; 1,430; 1,382; 1,274 and 1,141 cm-1. 1H NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.38 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.29 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90–7.70 (m, 3H, arom.); 8.32 (s, 1H, NH); 10.12 (s, br., 1H, OH); 11.40 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.30 (OCH2); 102.7 (C-3, Ar-); 107.9 (C-8); 108.6 (C-5, Ar-); 111.5 (C-6); 111.7 (C-1, Ar-); 112.8 (C-3); 113.6 (C-10); 127.0 (C-5); 132.5 (C-6, Ar-); 143.2 (CO-N); 151.4 (C-9); 152.9 (C-4); 160.4 (C-7); 160.9 (C-2); 162.4 (C-4, Ar-); 162.8 (C-2, Ar-); 172.43 (CO-NH). Anal. Calcd. for C21H18N2O7S (442.44), C, 57.01; H, 4.10; N, 6.33; S, 7.25. Found: C, 57.04; H, 4.97; N, 6.32; S, 7.29.
N-(2-(3,4-Dihydroxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3f). M.p. 243–244 °C; yield (38%). FT-IR: νmax 3,389; 3,209; 3,045; 3,037; 2,974; 1,726; 1,698; 1,612; 1,548; 1,521; 1,489; 1,396; 1,262 and 1,150 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.35 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.28 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.65 (m, 3H, arom.); 8.32 (s, 1H, NH); 9.35 (s, br., 1H, OH); 10.14 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 116.8 (C-2, Ar-); 117.6 (C-5, Ar-); 123.5 (C-6, Ar-); 127.3 (C-1, Ar-); 127.8 (C-5); 143.2 (C-N); 143.5 (C-3, Ar-); 149.8 (C-4, Ar-); 151.4 (C‑9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.42 (CO-NH). Anal. Calcd. for C21H18N2O7S (442.44), C, 57.01; H, 4.10; N, 6.33; S, 7.25. Found: C, 56.99; H, 3.89; N, 6.32; S, 7.24.
N-(2-(2,5-Dihydroxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3g). M.p. 278–279 °C; yield (41%). FT-IR: νmax 3,420; 3,281; 3,065; 2,915; 1,708; 1,716; 1,688; 1,648; 1,627; 1,581; 1,554; 1,521; 1,418; 1,390; 1,278 and 1,149 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.38 (s,2H,COCH2S); 4.84 (s, 2H, CH2); 5.28 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.72 (m, 3H, arom.); 8.32 (s, 1H, NH); 9.26 (s, br., 1H, OH); 11.93 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 116.5 (C-6, Ar-); 117.5 (C-3, Ar-); 119.4 (C-4, Ar-); 120.1 (C-1, Ar-); 127.8 (C-5); 143.2 (C-N); 151.4 (C-9); 151.3 (C-5, Ar‑); 152.9 (C-4); 153.8 (C-2, Ar-); 160.3 (C-7); 160.9 (C-2); 172.43 (CO-NH). Anal. Calcd. for C21H18N2O7S (442.44), C, 57.01; H, 4.10; N, 6.33; S, 7.25. Found: C, 57.00; H, 4.08; N, 6.31; S, 7.20.
N-(2-(4-hydroxy-3-methoxyphenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (
3h) [
23]. M.p. 233–234 °C; yield (76%). FT-IR: ν
max 3,517; 3,478; 3,329; 3,274; 2,915; 1,723; 1,698; 1,629; 1,536; 1,469; 1,393; 1,255 and 1,145 cm
-1.
1H-NMR (δ, ppm): 2.40 (s, 3H, CH
3, C-4); 3.37 (s, 2H, COCH
2S); 3.76 (s, 3H, OCH
3); 4.84 (s, 2H, CH
2); 5.27 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.71 (m, 3H, arom.); 8.32 (s, 1H, NH); 11.59 (s, br., 1H, OH).
13C-NMR (δ, ppm): 21.4 (CH
3); 32.92 (COCH
2S); 47.81 (SCHN); 56.2 (OCH
3); 69.10 (OCH
2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 114.8 (C-2, Ar-); 117.3 (C-5, Ar-); 123.0 (C-6, Ar-); 127.2 (C-1, Ar-); 127.8 (C-5); 143.2 (C-N); 148.1 (C-4, Ar-); 151.4 (C‑9); 151.5 (C-3, Ar-); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 172.40 (CO-NH). Anal. Calcd. for C
22H
20N
2O
7S (456.47), C, 57.89; H, 4.42; N, 6.14; S, 7.02. Found: C, 57.88; H, 4.39; N, 6.13; S, 7.01.
2-(4-Methyl-2-oxo-2H-chromen-7-yloxy)-N-(4-oxo-2-(3-phenoxyphenyl)thiazolidin-3-yl) acetamide (3i). M.p. 217–218 °C; yield (72%). FT-IR: νmax 3,500; 3,471; 3,329; 3,279; 2,910; 1,720; 1,697; 1,619; 1,531; 1,467; 1,392; 1,264 and 1,151 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.38 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.29 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.76 (m, 9H, arom.); 8.32 (s, 1H, NH). 13C-NMR (δ, ppm), 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 115.7 (C-4, Ar-Ph); 116.8 (C-2, Ar- Ph); 117.4 (C-2,6, Ar- PhO); 121.8 (C‑4, Ar- PhO); 122.3 (C-6, Ar- Ph); 127.8 (C-5); 128.5 (C-3,5, Ar- PhO); 128.8 (C-5, Ar- Ph); 133.6 (C-1, Ar- Ph); 151.4 (C-9); 152.9 (C-4); 157.1 (C-1, Ar- PhO); 157.3 (C-3, Ar- Ph); 160.3 (C-7); 160.9 (C-2); 166.1 (CO-NH); 168.2 (CO-N). Anal. Calcd. for C27H22N2O6S (502.54), C, 64.53; H, 4.41; N, 5.57; S, 6.38. Found: C, 64.50; H, 4.39; N, 5.53; S, 6.39.
N-(2-(4-(Dimethylamino)phenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy)-acetamide (3j). M.p. 227–228 °C; yield (76%). FT-IR: νmax 3,526; 3,473; 3,329; 3,269; 2,926; 1,729; 1,690; 1,624; 1,539; 1,473; 1,378; 1,260 and 1,150 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 2.86 (s, 6H, N(CH3)2); 3.36 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.29 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90-7.7 (m, 4H, arom.); 8.32 (s, 1H, NH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 41.4(N(CH3)2); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 114.5 (C-3,5, Ar-); 123.04 (C-1, Ar); 130.2 (C‑2,6, Ar); 143.2 (C-N); 151.4 (C-9); 151.8 (C-4, Ar-); 160.2 (C-7); 160.9 (C-2); 172.43 (CO-NH). Anal. Calcd. for C23H23N3O5S (453.51), C, 60.91; H, 5.11; N, 9.27; S, 7.07. Found: C, 60.92; H, 5.09; N, 9.25; S, 7.01.
N-(2-(2-Hydroxy-5-nitrophenyl)-4-oxothiazolidin-3-yl)-2-(4-methyl-2-oxo-2H-chromen-7-yloxy) acetamide (3k). M.p. 288–290 °C; yield (90%). FT-IR: νmax 3,468; 3,459; 3,329; 3,218; 2,910; 1,720; 1,692; 1,625; 1,536; 1,469; 1,386; 1,260 and 1,147 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.37 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.28 (s, 1H, SCHN); 6.24 (s, 1H, H-3); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.90–7.82 (m, 3H, arom.); 8.32 (s, 1H, NH); 11.54 (s, br., 1H, OH). 13C-NMR (δ, ppm): 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-10); 117.1 (C-3, from Ph); 120.1 (C-1, from Ph); 124.5 (C-4, from Ph); 125.7 (C-6, from Ph); 127.8 (C-5); 141.3 (C-5, from Ph); 143.3 (C-N); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C-2); 167.8 (C-2, from Ph); 174.0 (CO-NH). Anal. Calcd. for C21H17N3O8S (471.44), C, 53.50; H, 3.63; N, 8.91; S, 6.80. Found: C, 53.47; H, 3.64; N, 8.90; S, 6.79.
(E)-2-(4-Methyl-2-oxo-2H-chromen-7-yloxy)-N-(4-oxo-2-styrylhiazolidin-3-yl)acetamide (3l). M.p. 249–250 °C; yield (46%). FT-IR: νmax 3,489; 3,416; 3,309; 3,292; 2,906; 1,728; 1,695; 1,621; 1,543; 1,468; 1,389; 1,254 and 1,131 cm-1. 1H-NMR (δ, ppm): 2.40 (s, 3H, CH3, C-4); 3.36 (s, 2H, COCH2S); 4.84 (s, 2H, CH2); 5.26 (s, 1H, SCHN); 6.13 (d, 1H, styryl); 6.24 (s, 1H, H-3); 6.41 (d, 1H, styryl); 6.73 (s, 1H, H-8); 6.85 (d, 1H, H-6); 7.20 (d, 1H, H-5); 6.94–7.70 (m, 5H, arom.); 8.32 (s, 1H, NH). 13C-NMR (δ, ppm) 21.4 (CH3); 32.92 (COCH2S); 47.81 (SCHN); 69.10 (OCH2); 107.6 (C-8); 111.0 (C-6); 112.8 (C-3); 113.5 (C-9); 126.1 (C-1, styryl); 126.6 (C-2,6, Ar-); 127.8 (C-5); 128.5 (C-4, Ar-); 128.9 (C-3,5, Ar-); 137.2 (C-N); 139.2 (C-2, styryl); 151.4 (C-9); 152.9 (C-4); 160.3 (C-7); 160.9 (C‑2); 174.4 (CO-NH). Anal. Calcd. for C23H20N2O5S (436.48), C, 63.29; H, 4.62; N, 6.42; S, 7.35. Found: C, 63.27; H, 4.61; N, 6.39; S, 7.31.