Antioxidant Activities of Caragana sinica Flower Extracts and Their Main Chemical Constituents
Abstract
:1. Introduction
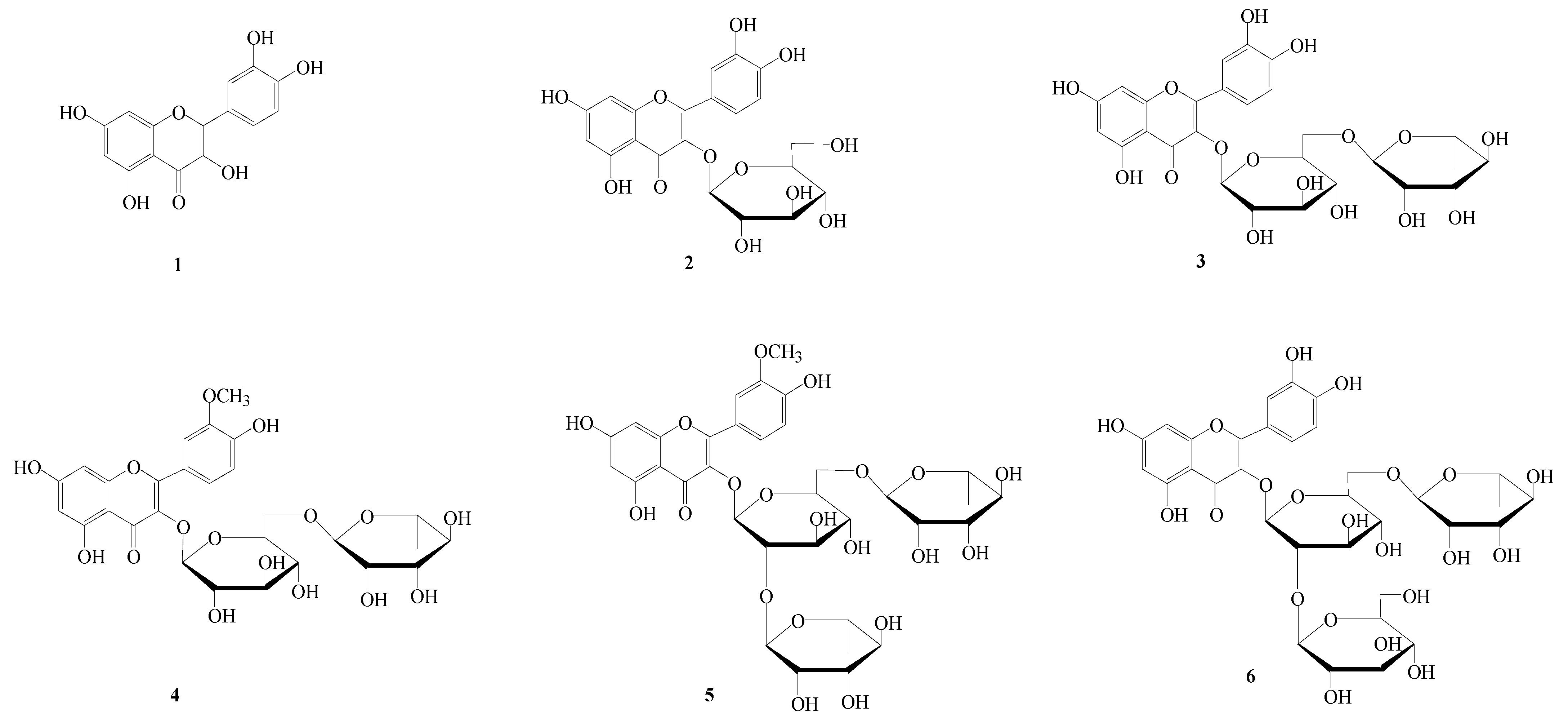
2. Results and Discussion
2.1. Antioxidant Activities
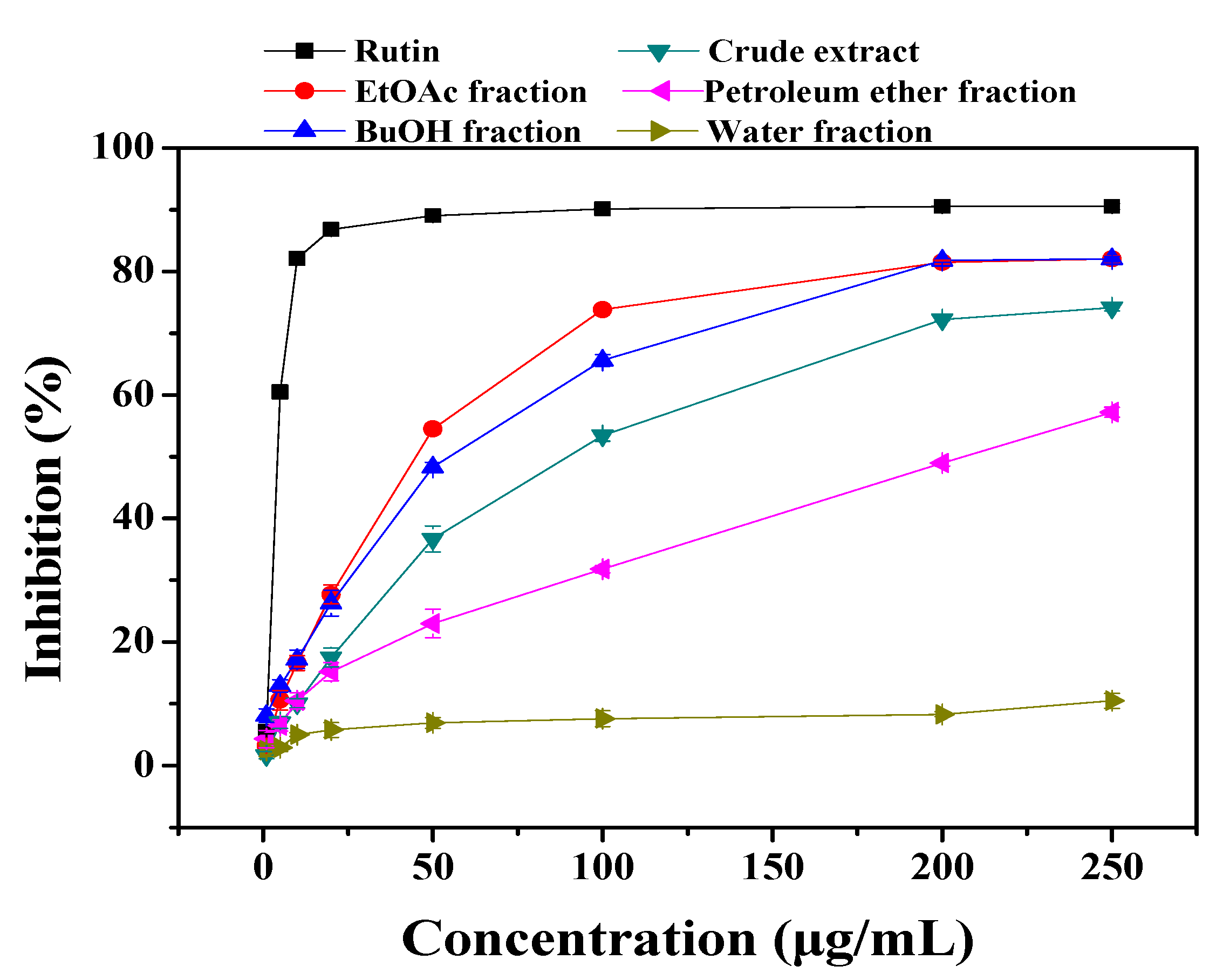
| Sample | Antioxidant activity (AA%) | Oxidation rate ratio (ROR) |
|---|---|---|
| EtOAc fraction | 73.57 ± 0.63 | 0.26 ± 0.004 |
| BuOH fraction | 59.12 ± 0.56 | 0.41 ± 0.005 |
| Crude extract | 47.85 ± 0.53 | 0.52 ± 0.005 |
| Petroleum ether fraction | 42.11 ± 0.46 | 0.58 ± 0.006 |
| Water fraction | 48.77 ± 0.49 | 0.51 ± 0.005 |
2.2. Contents of Total Phenolics and Total Flavonoids
| Sample | Total phenolic content (TPC)(mg gallic acid /g dry extract) | Total flavonoid content (TFC) (mg rutin /g dry extract) |
|---|---|---|
| EtOAc fraction | 140.0 ± 10.1 | 157.8± 11.4 |
| BuOH fraction | 117.5 ± 8.4 | 100.6 ± 6.8 |
| Crude extract | 68.0 ± 4.7 | 69.9 ± 3.4 |
| Petroleum ether fraction | 31.7 ± 1.8 | 15.5 ± 2.3 |
| Water fraction | 7.9 ± 0.7 | 4.9 ± 0.5 |
2.3. Correlation Analysis
| Assay | Equation | |||
|---|---|---|---|---|
| Total phenolic content (TPC) | R2 | Total flavonoid content (TFC) | R2 | |
| Total flavonoids content | y = 1.1042x - 10.8870 | 0.9566 | - | - |
| Frap assay | y = 17.8586x - 269.2349 | 0.9349 | y = 15.9845x - 79.9588 | 0.9546 |
| EC50 of DPPH radical-scavenging activity a | y = -1.3945x + 223.6241a | 0.8470 a | y = -1.1250x + 195.7892 a | 0.8186 a |
| β-carotene bleaching inhibition | y = 0.1931x + 40.1867 | 0.7534 | y = 0.1807x + 41.6832 | 0.8410 |
2.4. Isolation and Determination of Flavonoid Compounds
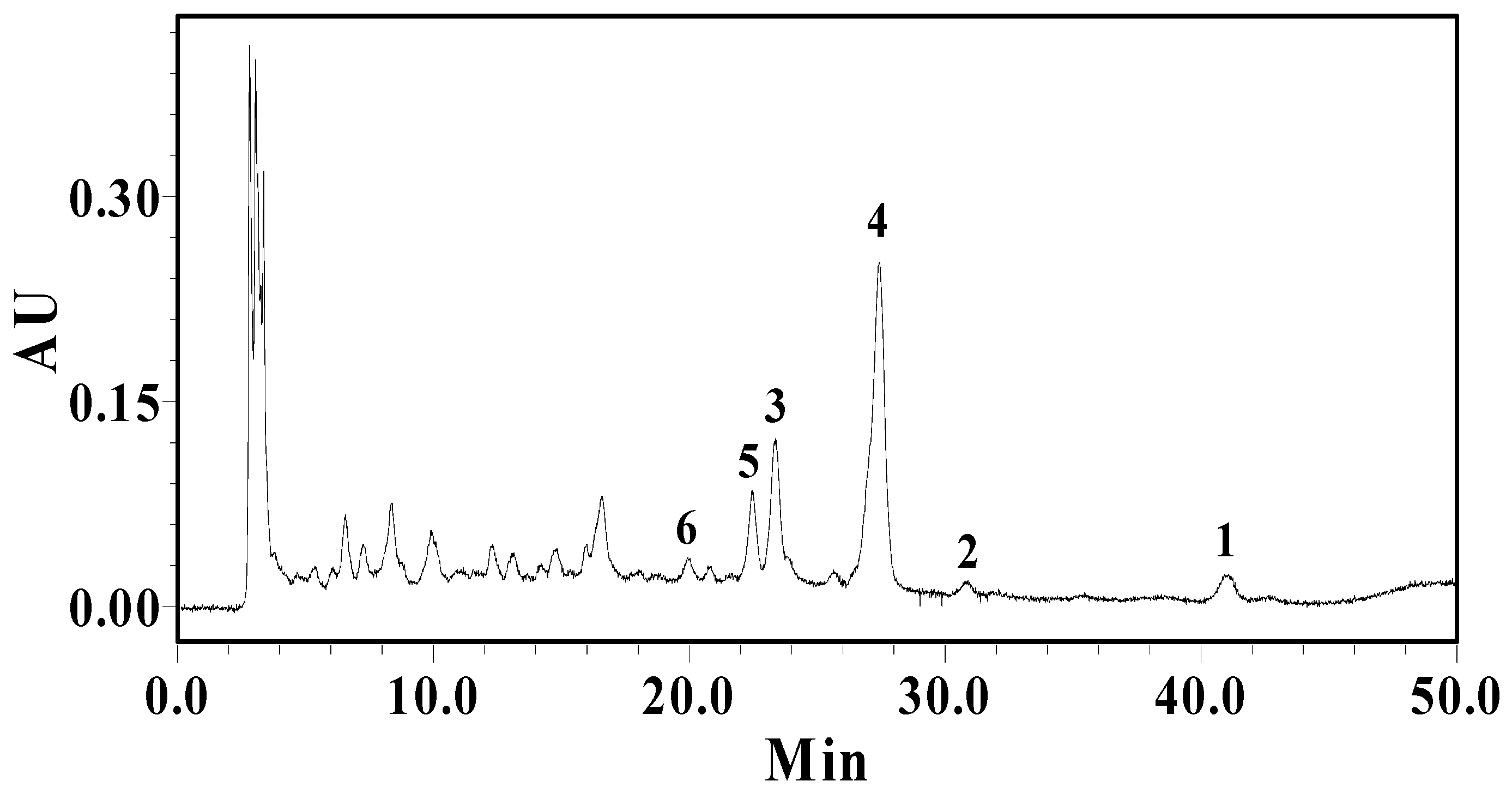
3. Experimental
3.1. Plant Material, Chemicals and Equipment
3.2. Extraction and Isolation of Compounds.
3.3. Determination of Antioxidant Activity
3.4. Determination of Total Antioxidant Content
3.5. HPLC Analysis
3.6. Statistical Analysis
4. Conclusions
Acknowledgements
- Samples Availability: Samples of the compounds are available from the authors.
References
- Halliwell, B. Antioxidants and human diseases: A general introduction. Nutrit. Rev. 1997, 55, S44–S55. [Google Scholar] [CrossRef]
- Halliwell, B.; Murcia, M.A.; Chirico, S.; Aruoma, O.I. Free radicals and antioxidants in food and in vivo: What they do and how they work. Crit. Rev. Food Sci. 1995, 35, 7–20. [Google Scholar] [CrossRef]
- Hou, W.C.; Lin, R.D.; Cheng, K.T.; Hung, Y.T.; Cho, C.H.; Chen, C.H.; Hwang, S.Y.; Lee, M.H. Free radical-scavenging activity of Taiwanese native plants. Phytomedicine 2003, 10, 170–175. [Google Scholar] [CrossRef]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomah, B.D. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J. Agr. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Shyu, Y.S.; Lin, J.T.; Chang, Y.T.; Chiang, C.J., Yang. Evaluation of antioxidant ability of ethanolic extract from dill (Anethum graveolens L.) flower. Food Chem. 2009, 115, 515–521. [Google Scholar] [CrossRef]
- Ruan, Z.P.; Zhang, L.L.; Lin, Y.M. Evaluation of the antioxidant activity of Syzygium cumini leaves. Molecules 2008, 13, 2545–2556. [Google Scholar] [CrossRef]
- Rop, O.; Reznicek, V.; Valsikova, M,; Jurikova, T.; Mlcek, J.; Kramarova, D. Antioxidant properties of European cranberrybush fruit (Viburnum opulus var. edule). Molecules 2010, 15, 4467–4477. [Google Scholar] [CrossRef]
- Ding, Z.T.; Fang, Y.S.; Tai, Z.G.; Yang, M.H.; Xu, Y.Q.; Li, F.; Cao, Q.E. Phenolic content and radical scavenging capacity of 31 species of ferns. Fitoterapia 2008, 79, 581–583. [Google Scholar] [CrossRef]
- Gao, K.P. Zhongguo shiliao bencao; Chemical Industry Press: Beijing, China, 2006; p. 404. [Google Scholar]
- Woo, E.R.; Kim, H.J.; Kwak, J.H.; Lim, Y.K.; Park, S.K.; Kim, H.S.; Lee, C.K.; Park, H. Anti-herpetic activity of various medicinal plant extracts. Arch. Pharm. Res. 1997, 20, 58–67. [Google Scholar] [CrossRef]
- Cheng, K.J.; Ma, D.Y.; Yang, G.X.; Hu, C.Q. A new tetrastilbene from Caragana sinica. Chin. Chem. Lett. 2008, 19, 711–715. [Google Scholar] [CrossRef]
- Luo, H.F.; Zhang, L.P.; Hu, C.Q. Five novel oligostilbenes from the roots of Caragana Sinica. Tetrahedron 2001, 57, 4849–4854. [Google Scholar] [CrossRef]
- Liang, G.L.; Bi, J.B.; Huang, H.Q.; Zhang, S.; Hu, C.Q. Metabolites and the pharmacokinetics of kobophenol A from Caragana sinica in rats. J. Ethnopharmacol. 2005, 101, 324–329. [Google Scholar] [CrossRef]
- Luo, H.F.; Zhang, L.P.; Hu, C.Q. Four stilbene oligomers from the roots of Caragana sinica. J. Chin. Pharm. Sci. 2000, 9, 162–164. [Google Scholar]
- Sánchez-Moreno, C. Review: Methods used to evaluate the free radical scanvenging activity in foods and biological systems. Food Sci.Technol. Inter. 2002, 8, 121–137. [Google Scholar]
- Duh, P.D.; Du, P.C.; Yen, G.C. Action of methanolic extract of mung bean hulls as inhibitors of lipid peroxidation and non-lipid oxidative damage. Food Chem. Toxicol. 1999, 37, 1055–1061. [Google Scholar] [CrossRef]
- Suja, K.P.; Jayalekshmy, A.; Arumughan, C. Antioxidant activity of sesame cake extract. Food Chem. 2005, 91, 213–219. [Google Scholar] [CrossRef]
- Barreira, J. C. M.; Ferreira, I. C. F. R.; Oliveira, M. B. P. P.; Pereira, J. A. Antioxidant activities of the extracts from chestnut flower, leaf, skins and fruit. Food Chem. 2008, 107, 1106–1113. [Google Scholar] [CrossRef]
- Liu, S.C.; Lin, J.T.; Wang, C.K.; Chen, H.Y.; Yang, D.J. Antioxidant properties of various solvent extracts from lychee (Litchi chinenesis Sonn.) flowers. Food Chem. 2009, 114, 577–581. [Google Scholar] [CrossRef]
- Kubola, J., Siriamornpun. Phenolic contents and antioxidant activities of bitter gourd (Momordica charantia L.) leaf, stem and fruit fraction extracts in vitro. Food Chem. 2008, 110, 881–890. [Google Scholar] [CrossRef]
- Matthaüs, B. Antioxidant activity of extracts obtained from residues of different oilseeds. J. Agr. Food Chem. 2002, 50, 3444–3452. [Google Scholar] [CrossRef]
- Teixeira, S.; Siquet, C.; Alves, C.; Boal, I.; Marques, M.P.; Borges, F.; Lima, J.L.F.C.; Reis, S. Structure-property studies on the antioxidant activity of flavonoids present in diet. Free Radical Bio. Med. 2005, 39, 1099–1108. [Google Scholar] [CrossRef]
- Arora, A.; Nair, M.G.; Strasburg, G.M. Structure-activity relationships for antioxidant activities of a series of flavonoids in a liposomal system. Free Radical Biol. Med. 1998, 24, 1355–1363. [Google Scholar] [CrossRef]
- Hung, P.V.; Maeda, T.; Miyatake, K.; Morita, N. Total phenolic compounds and antioxidant capacity of wheat graded flours by polishing method. Food Res. Inter. 2009, 42, 185–190. [Google Scholar] [CrossRef]
- Suárez, B.; Álvarez, Á.L.; García, Y.D.; Barrio, G.D.; Lobo, A.P.; Parra, F. Phenolic profiles, antioxidant activity and in vitro antiviral properties of apple pomace. Food Chem. 2010, 120, 339–342. [Google Scholar] [CrossRef]
- Wu, N.; Fu, K.; Fu, Y.J.; Zu, Y.G.; Chang, F.R.; Chen, Y.H.; Liu, X.L.; Kong, Y.; Liu, W.; Gu, C.B. Antioxidant activities of extracts and main components of pigeonpea [Cajanus cajan (L.) Millsp.] Leaves. Molecules 2009, 14, 1032–1043. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol.Viti. 1965, 16, 144–158. [Google Scholar]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agr. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tai, Z.-G.; Cai, L.; Dai, L.; Sun, W.-J.; Zhe, W.; Yang, Y.-B.; Cao, Q.-E.; Ding, Z.-T. Antioxidant Activities of Caragana sinica Flower Extracts and Their Main Chemical Constituents. Molecules 2010, 15, 6722-6732. https://doi.org/10.3390/molecules15106722
Tai Z-G, Cai L, Dai L, Sun W-J, Zhe W, Yang Y-B, Cao Q-E, Ding Z-T. Antioxidant Activities of Caragana sinica Flower Extracts and Their Main Chemical Constituents. Molecules. 2010; 15(10):6722-6732. https://doi.org/10.3390/molecules15106722
Chicago/Turabian StyleTai, Zhi-Gang, Le Cai, Lin Dai, Wei-Jun Sun, Wei Zhe, Ya-Bin Yang, Qiu-E Cao, and Zhong-Tao Ding. 2010. "Antioxidant Activities of Caragana sinica Flower Extracts and Their Main Chemical Constituents" Molecules 15, no. 10: 6722-6732. https://doi.org/10.3390/molecules15106722
APA StyleTai, Z.-G., Cai, L., Dai, L., Sun, W.-J., Zhe, W., Yang, Y.-B., Cao, Q.-E., & Ding, Z.-T. (2010). Antioxidant Activities of Caragana sinica Flower Extracts and Their Main Chemical Constituents. Molecules, 15(10), 6722-6732. https://doi.org/10.3390/molecules15106722




