Photoresponsive Block Copolymers Containing Azobenzenes and Other Chromophores
Abstract
:1. Introduction
2. Synthesis of Well-Defined PRBCs
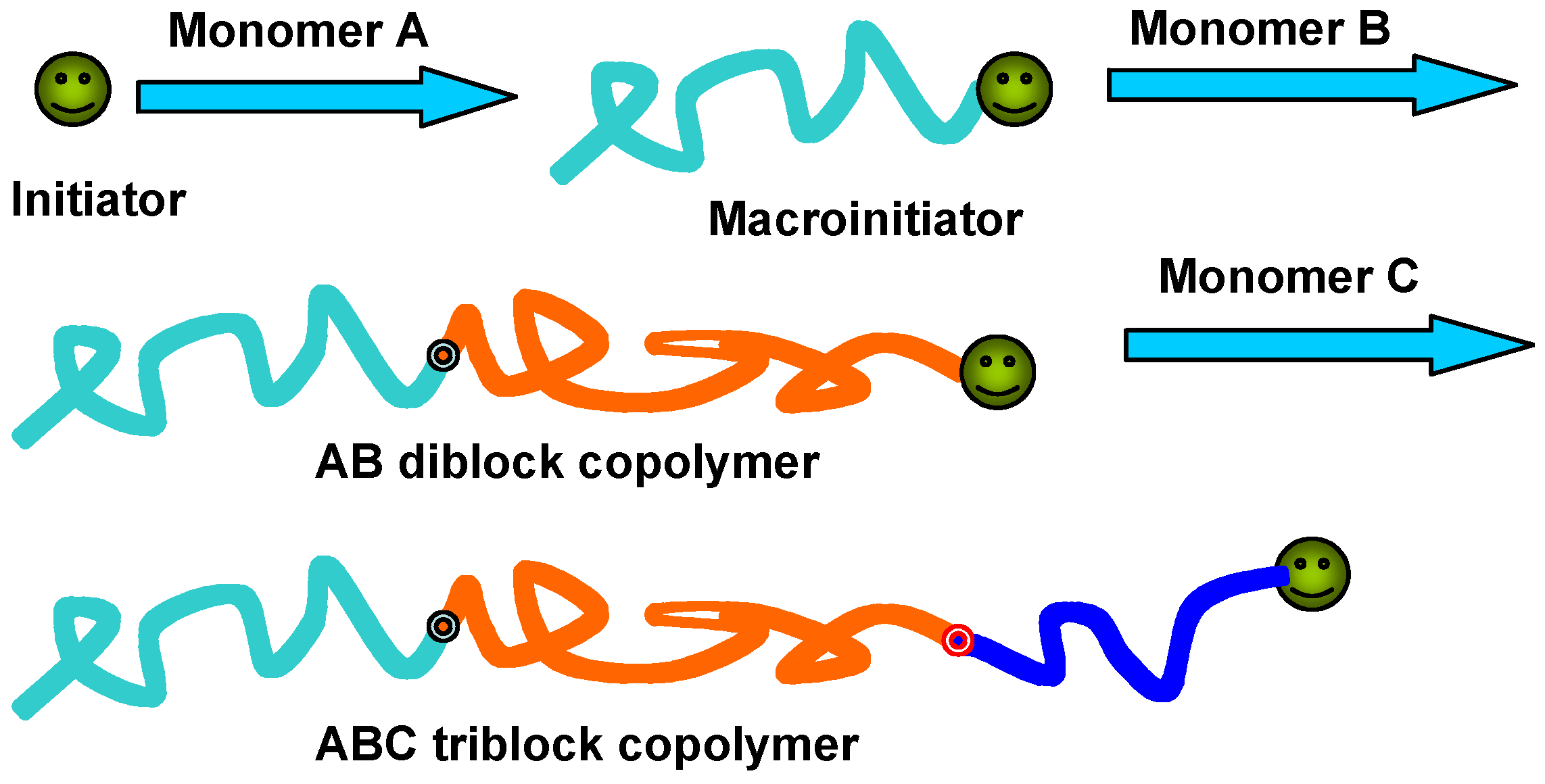
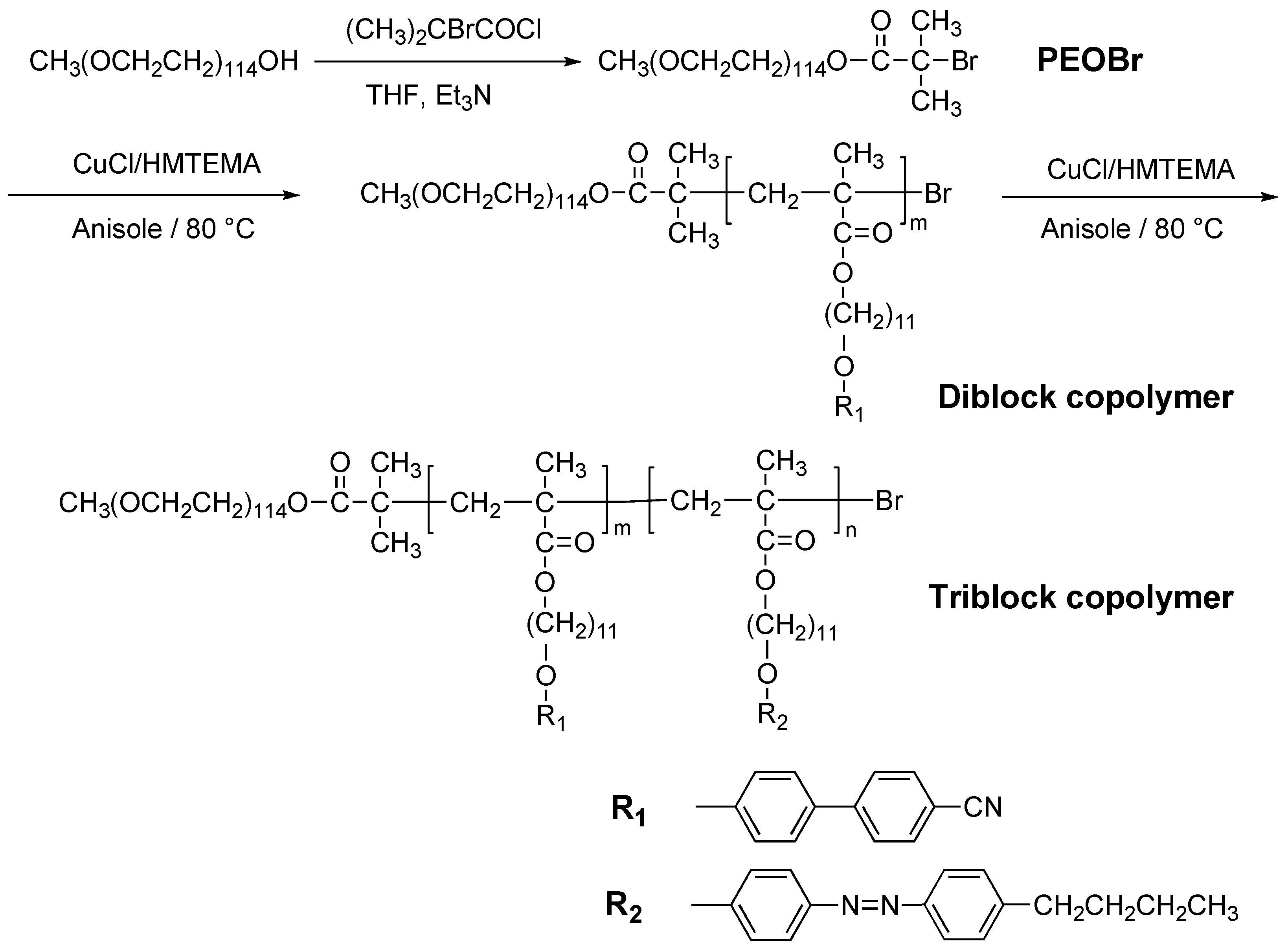
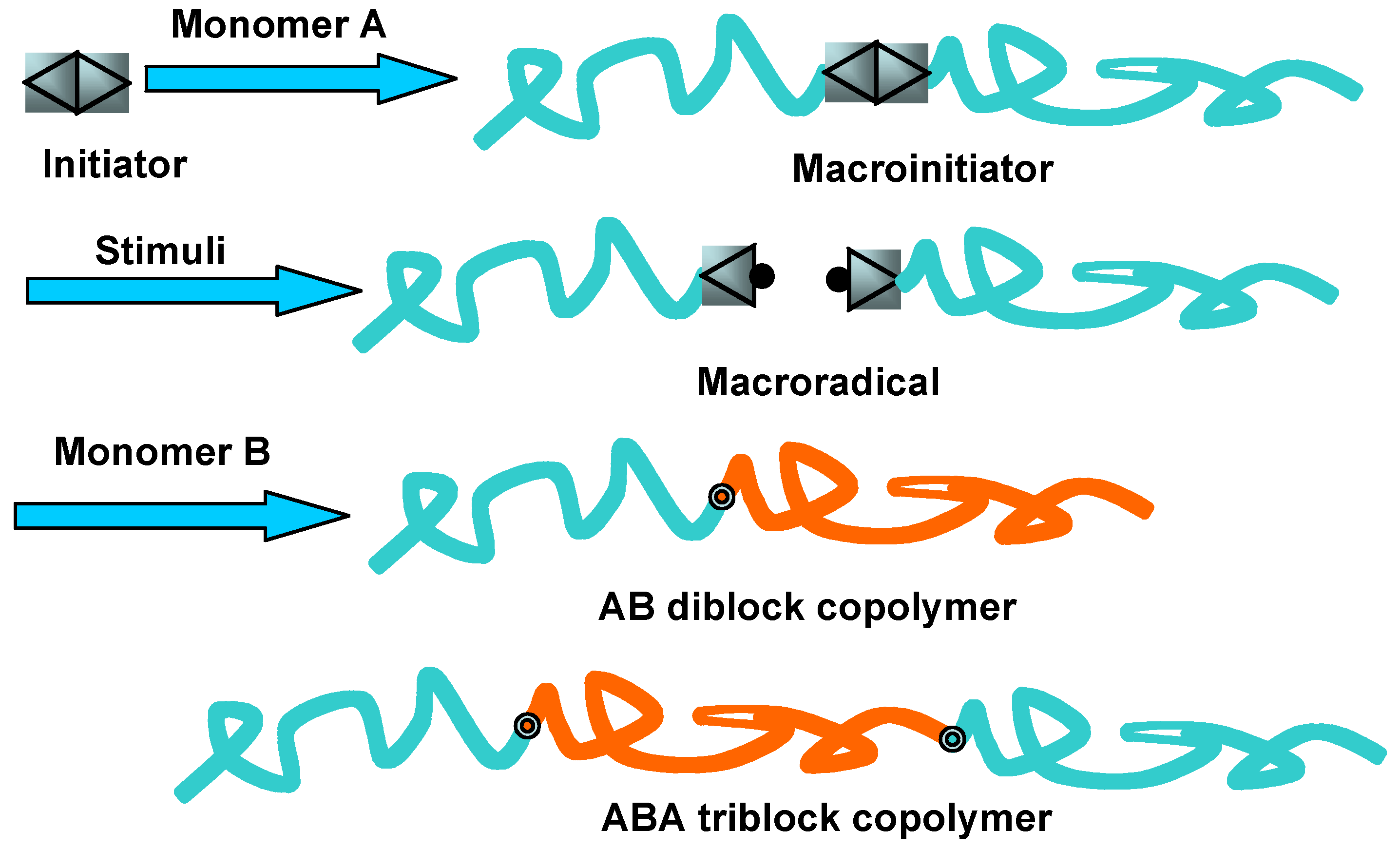
2.1. Direct Polymerization
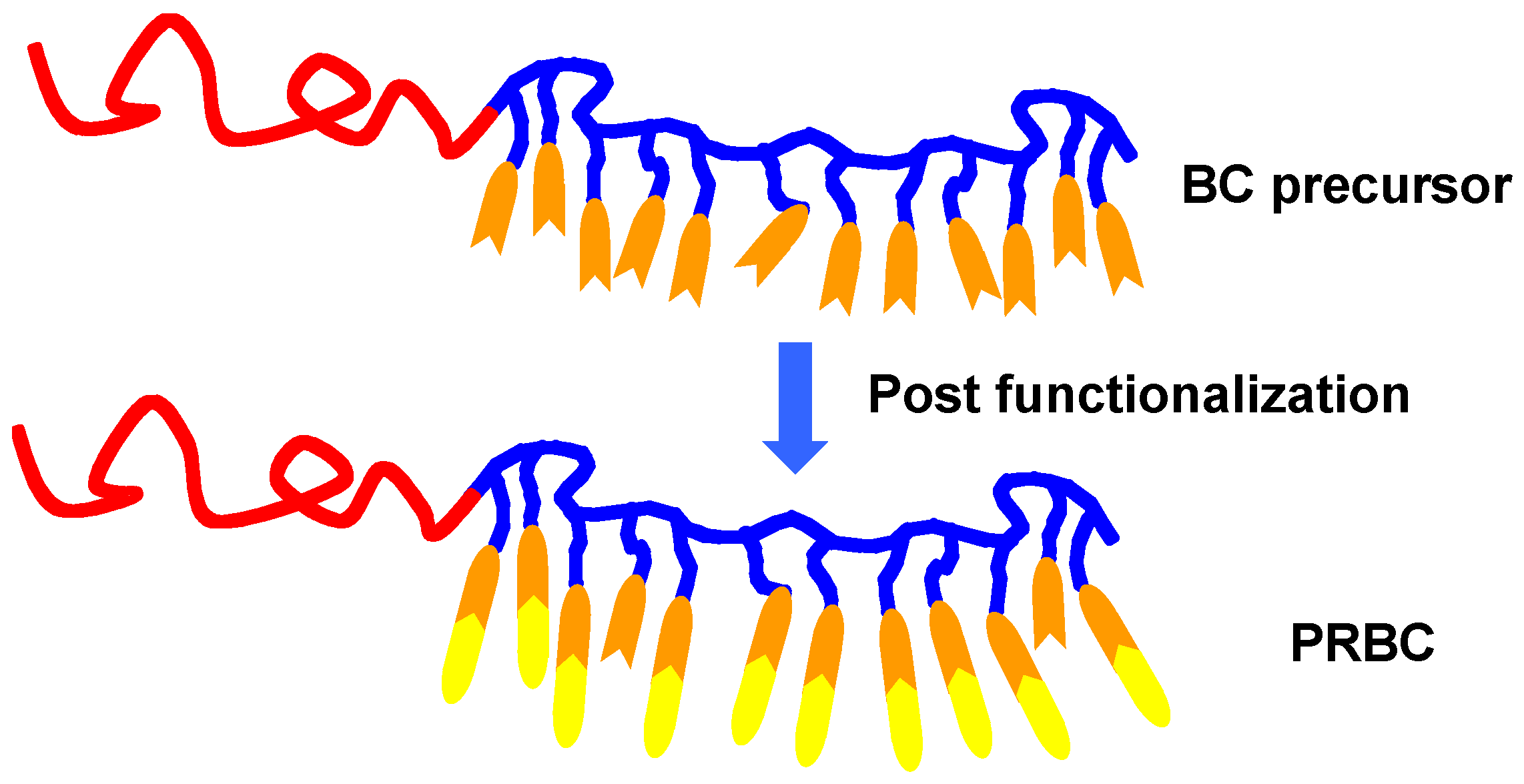
2.2. Post Functionalization
2.3. Supramolecular Self-Assembly
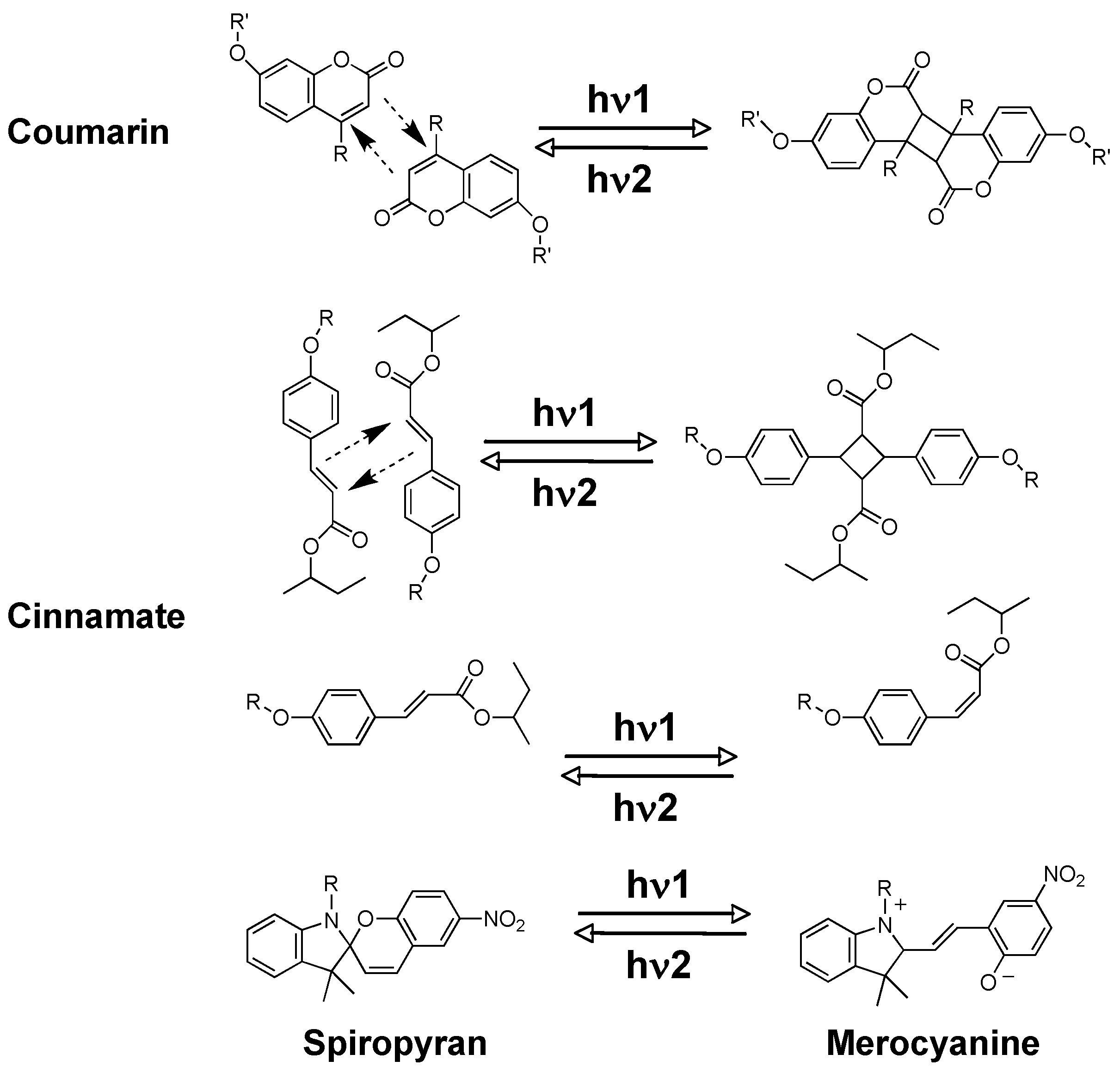
2.4. Special Reactions
3. Properties of PRBCs
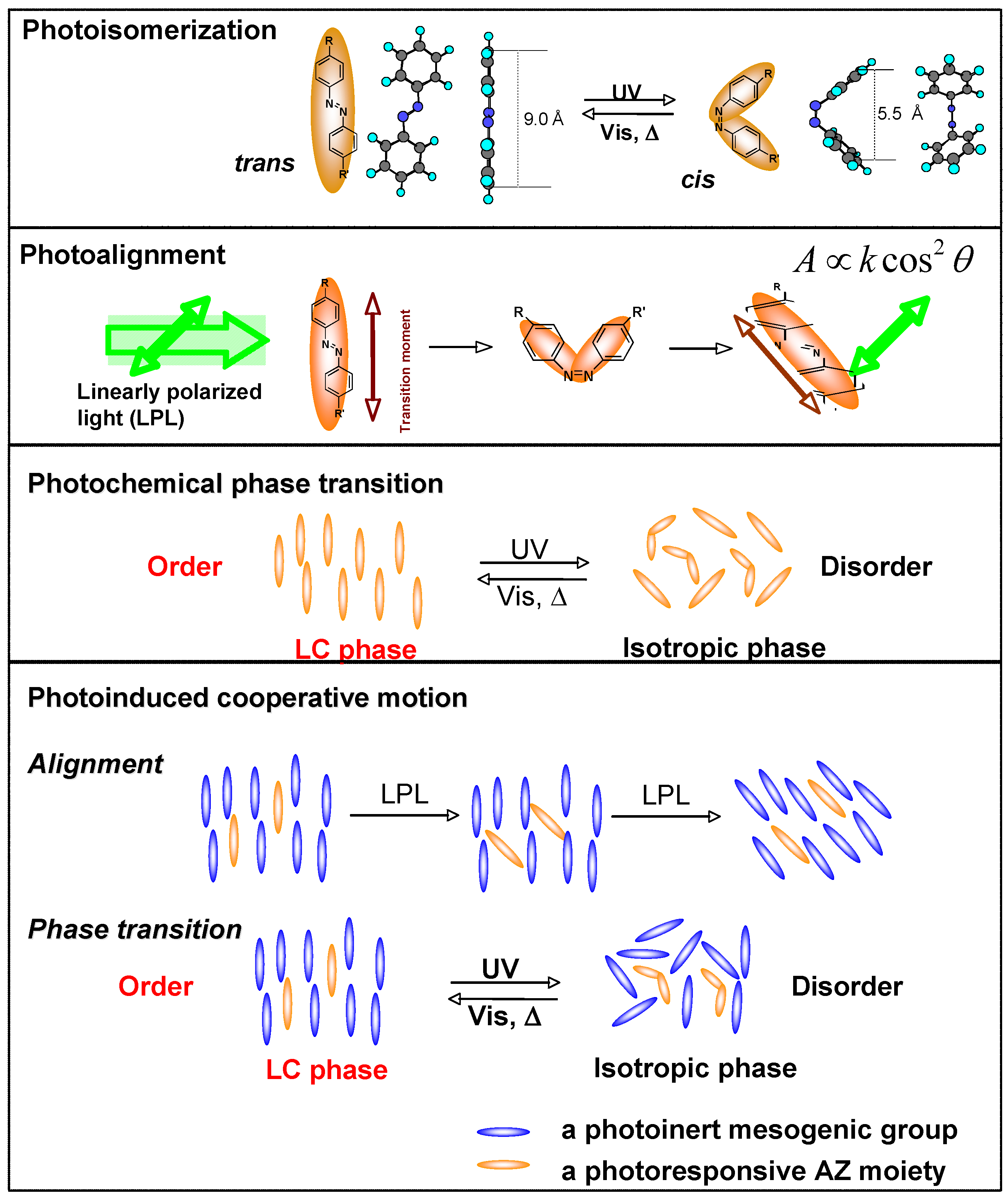
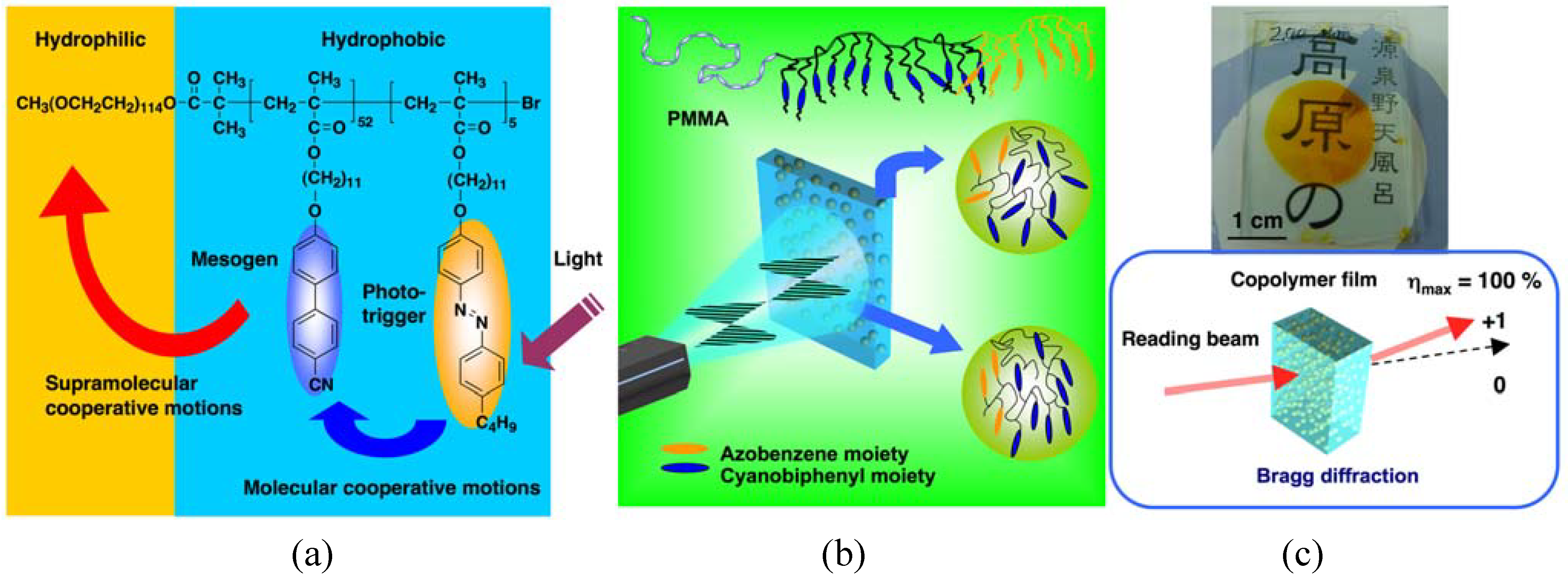
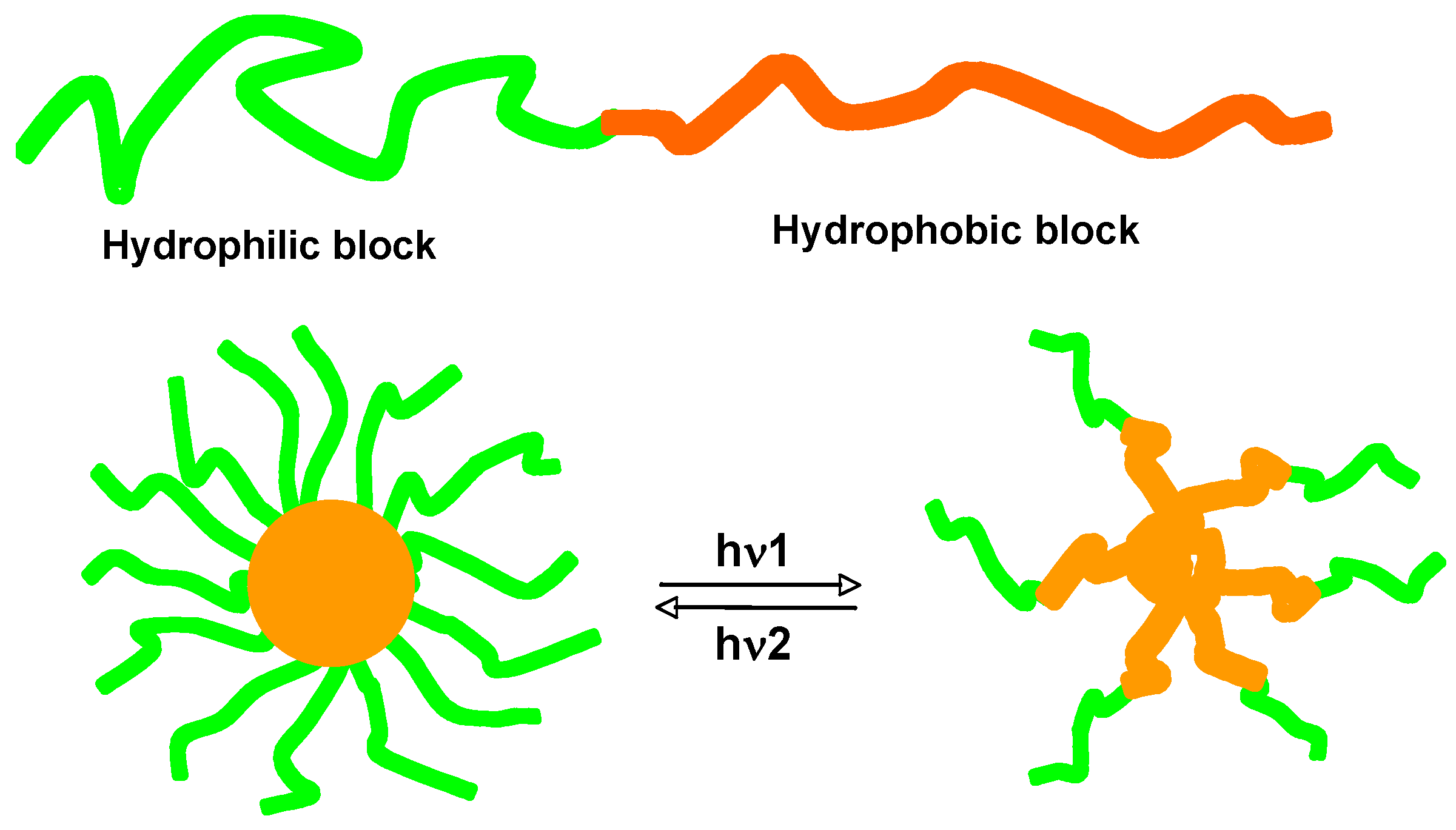
3.1. Photoresponsive Behaviors
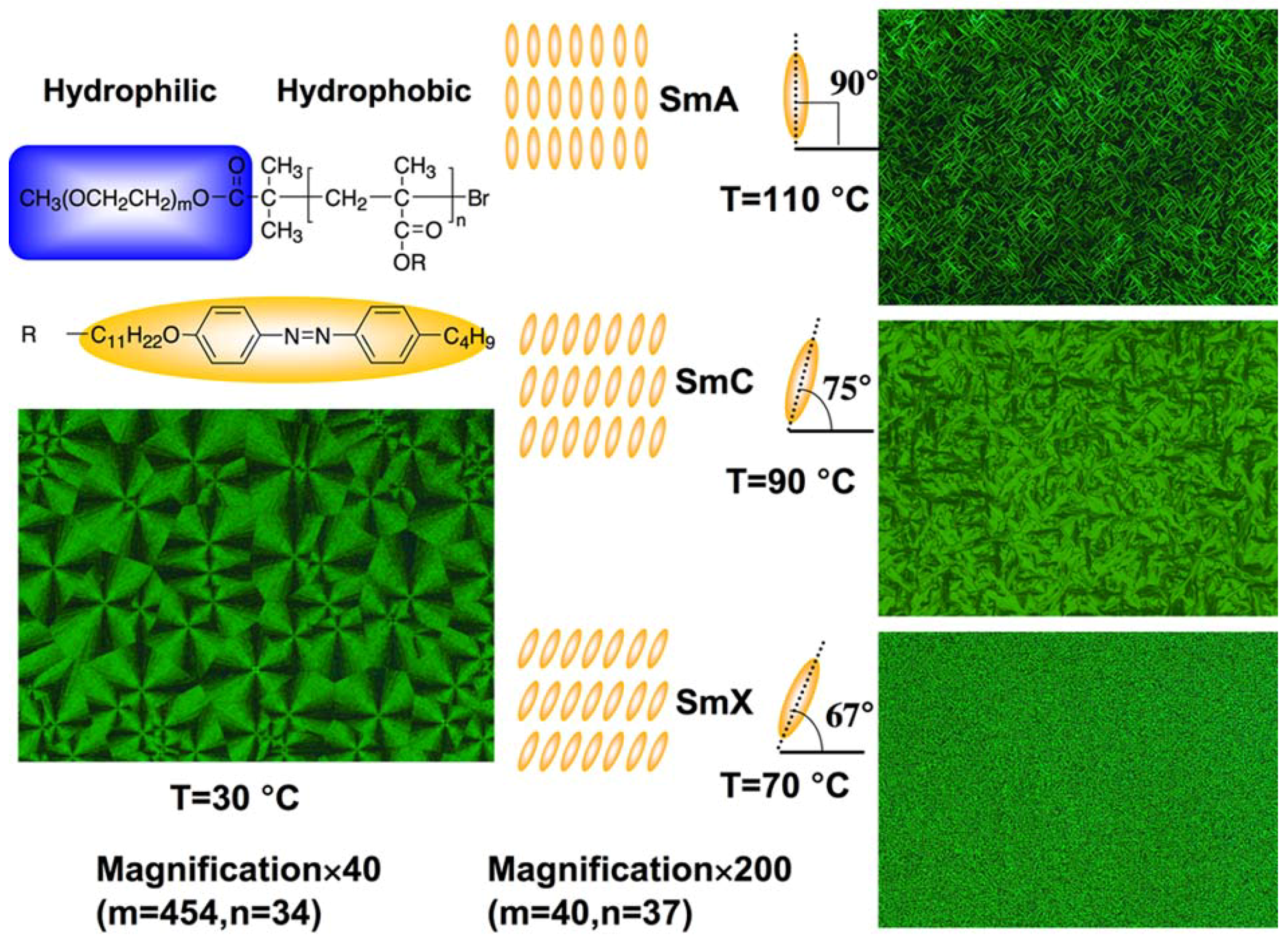
3.2. Non-Photoresponsive Properties
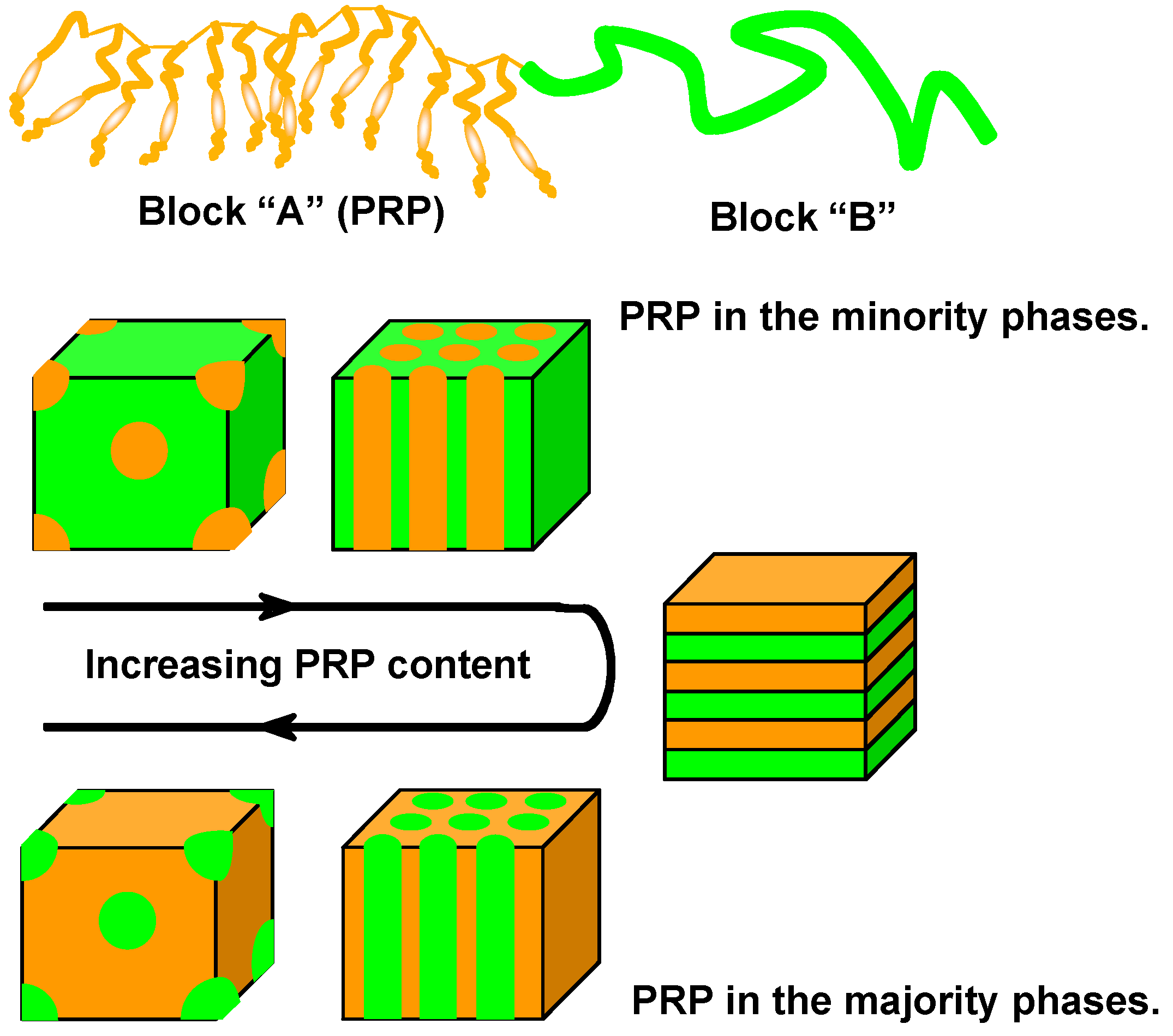
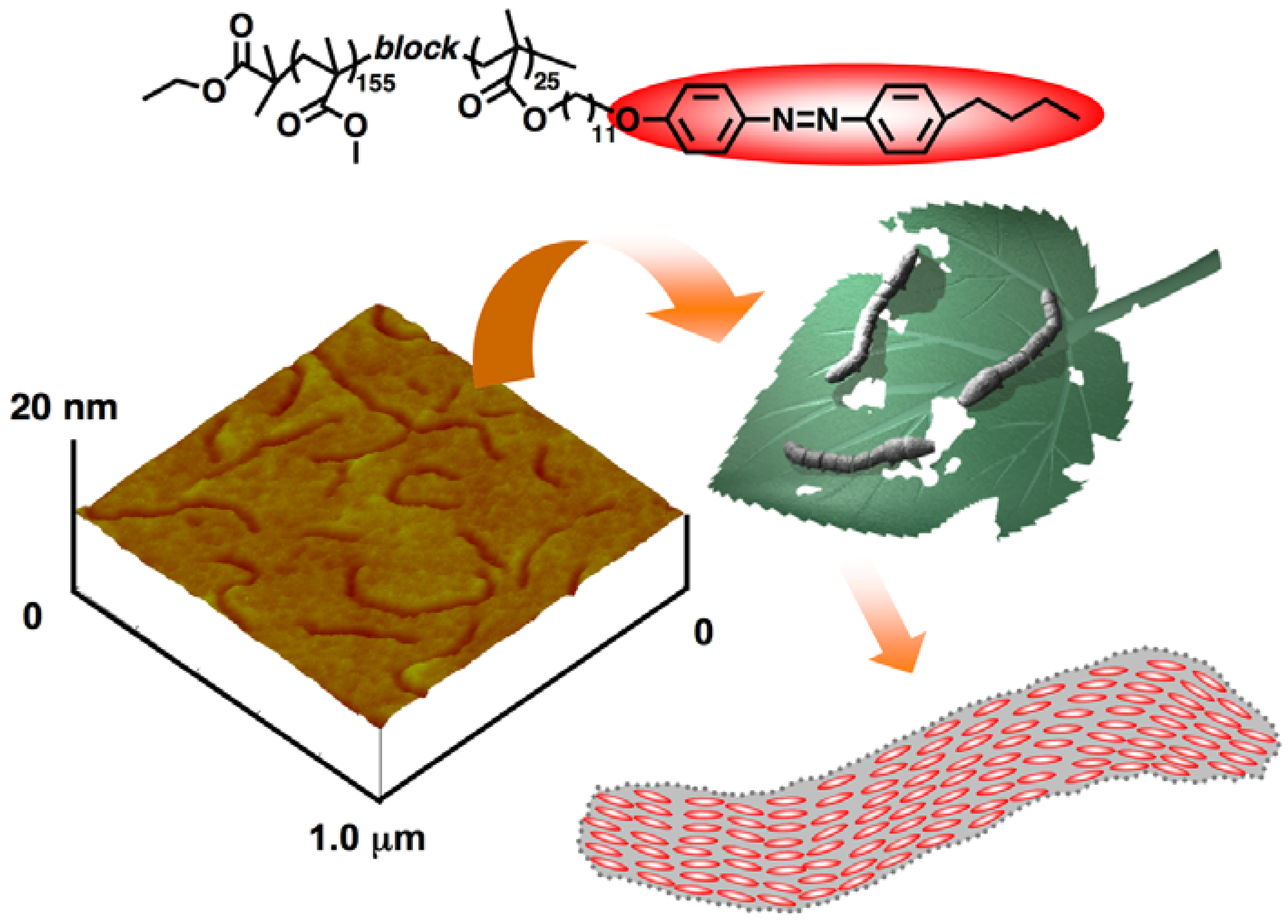
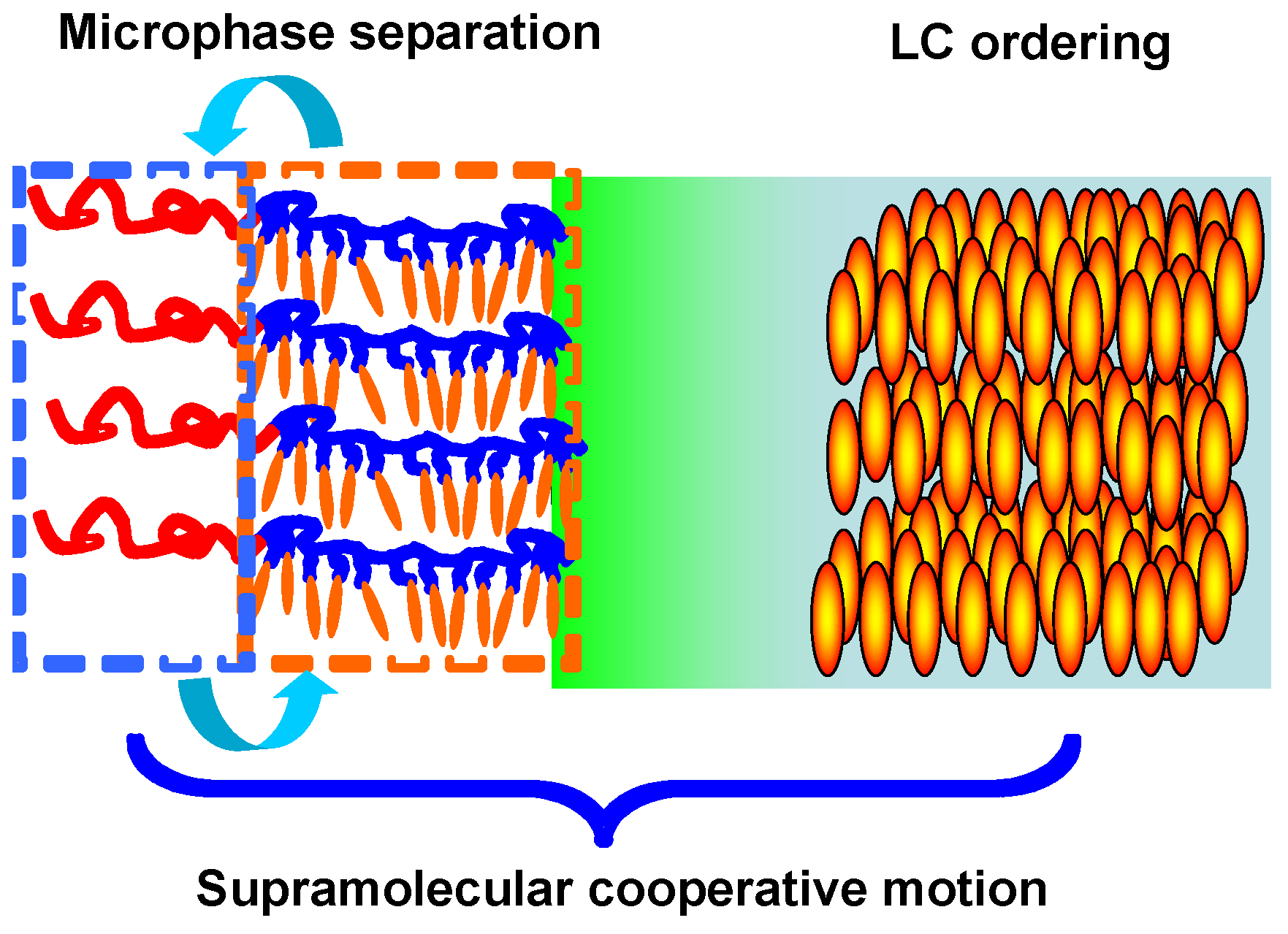
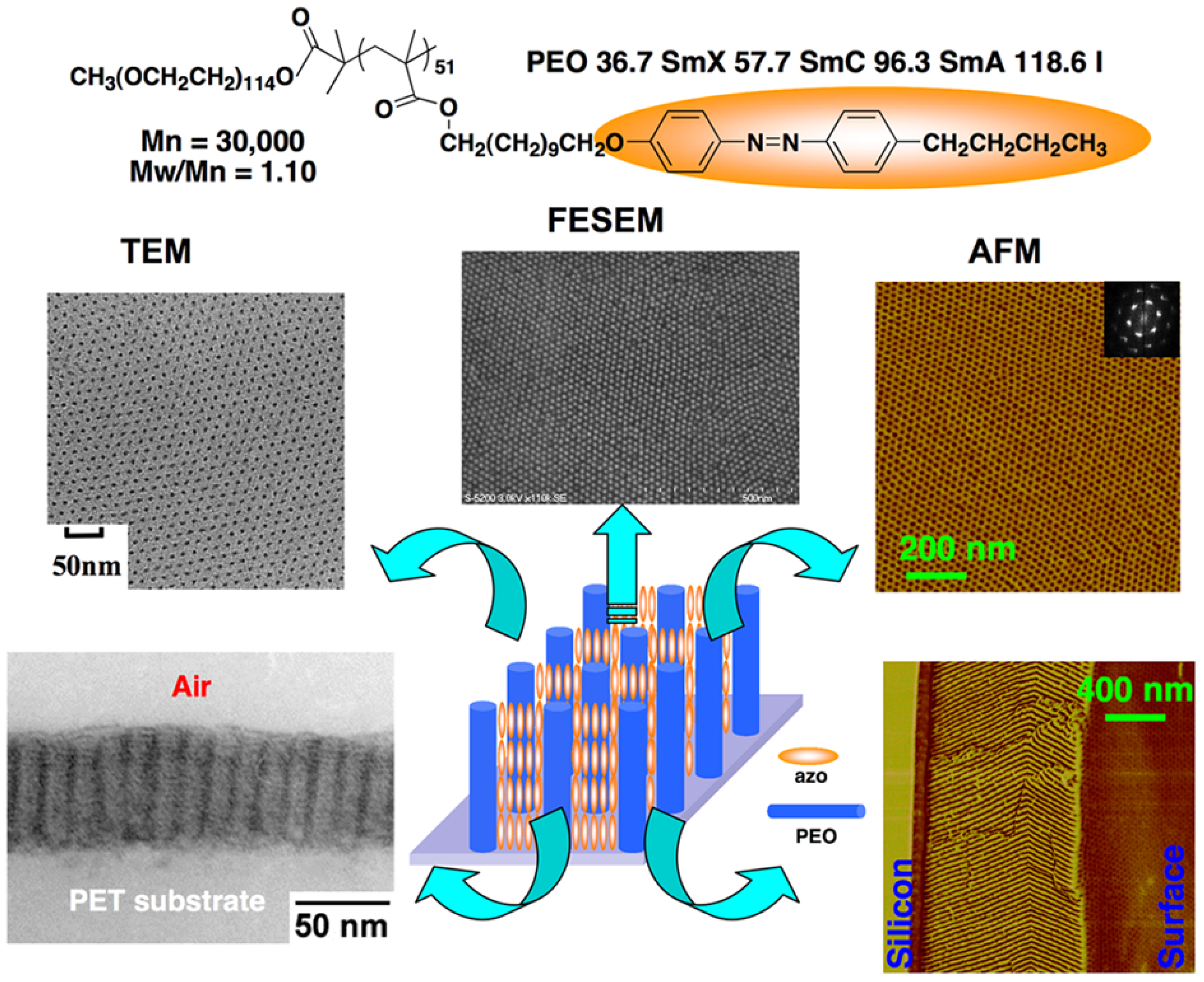
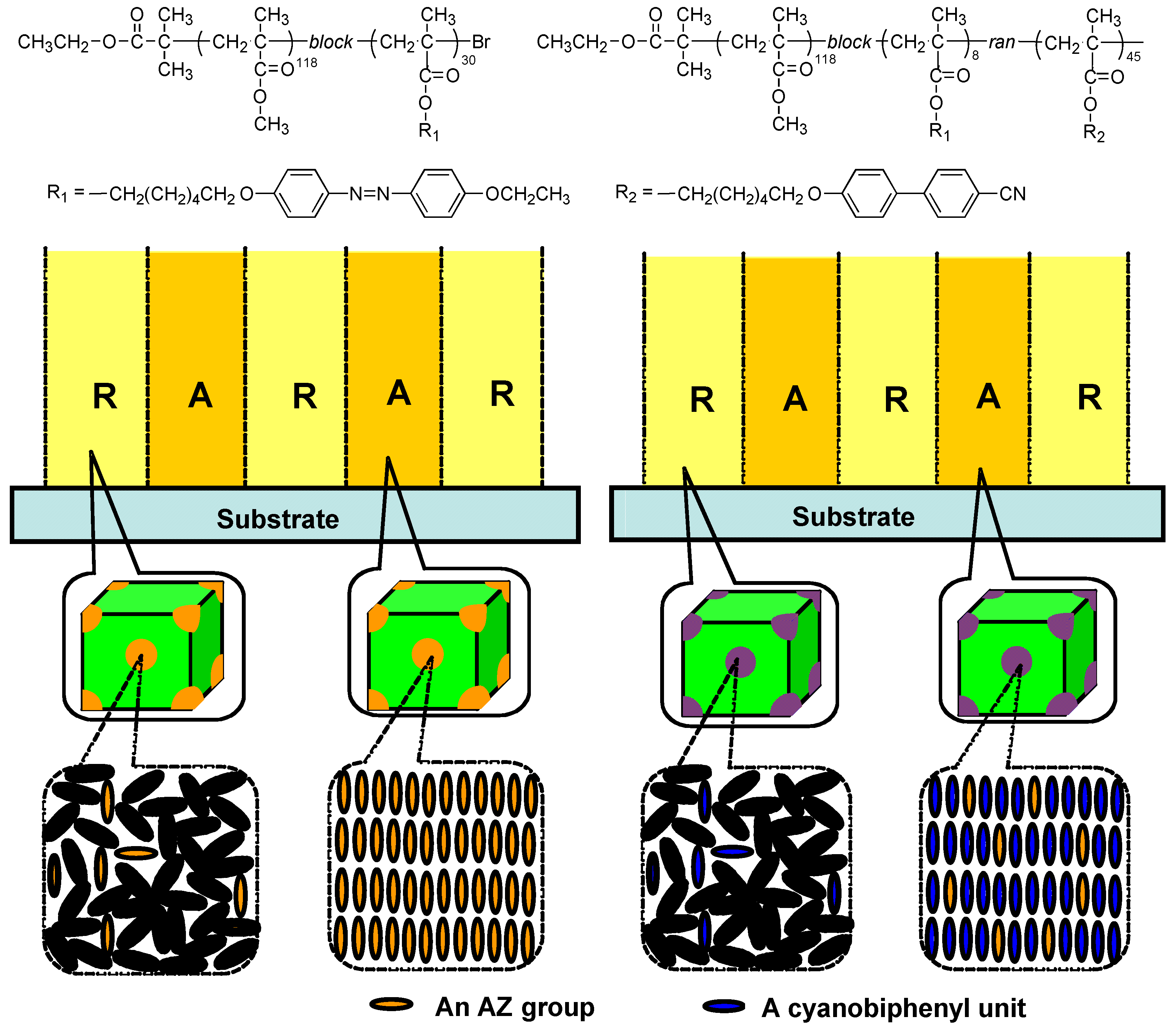
3.3. Microphase Separation of PRBCs
4. Control of Microphase Separation of PRBCs
4.1. Thermal Annealing
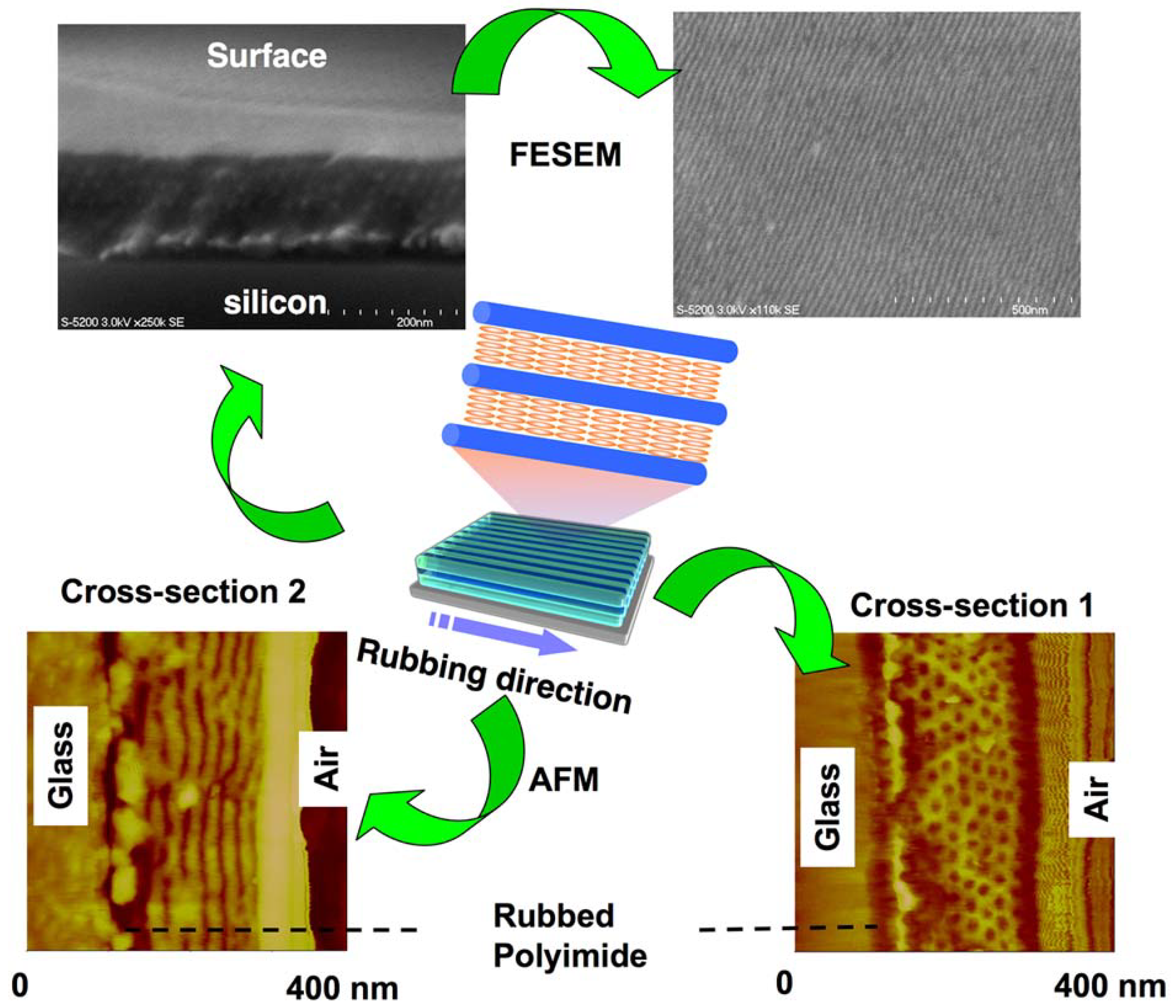
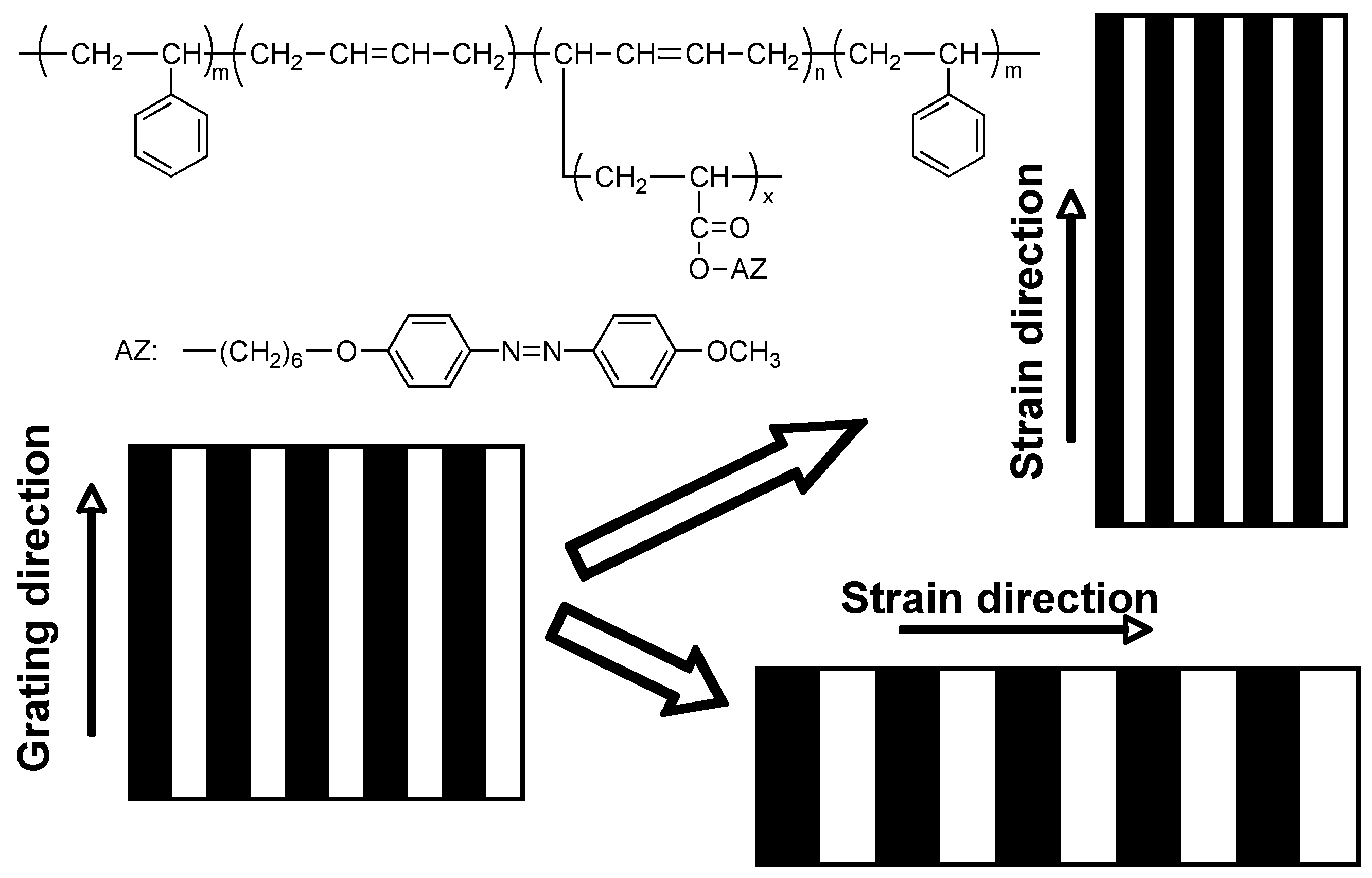
4.2. Mechanical Rubbing
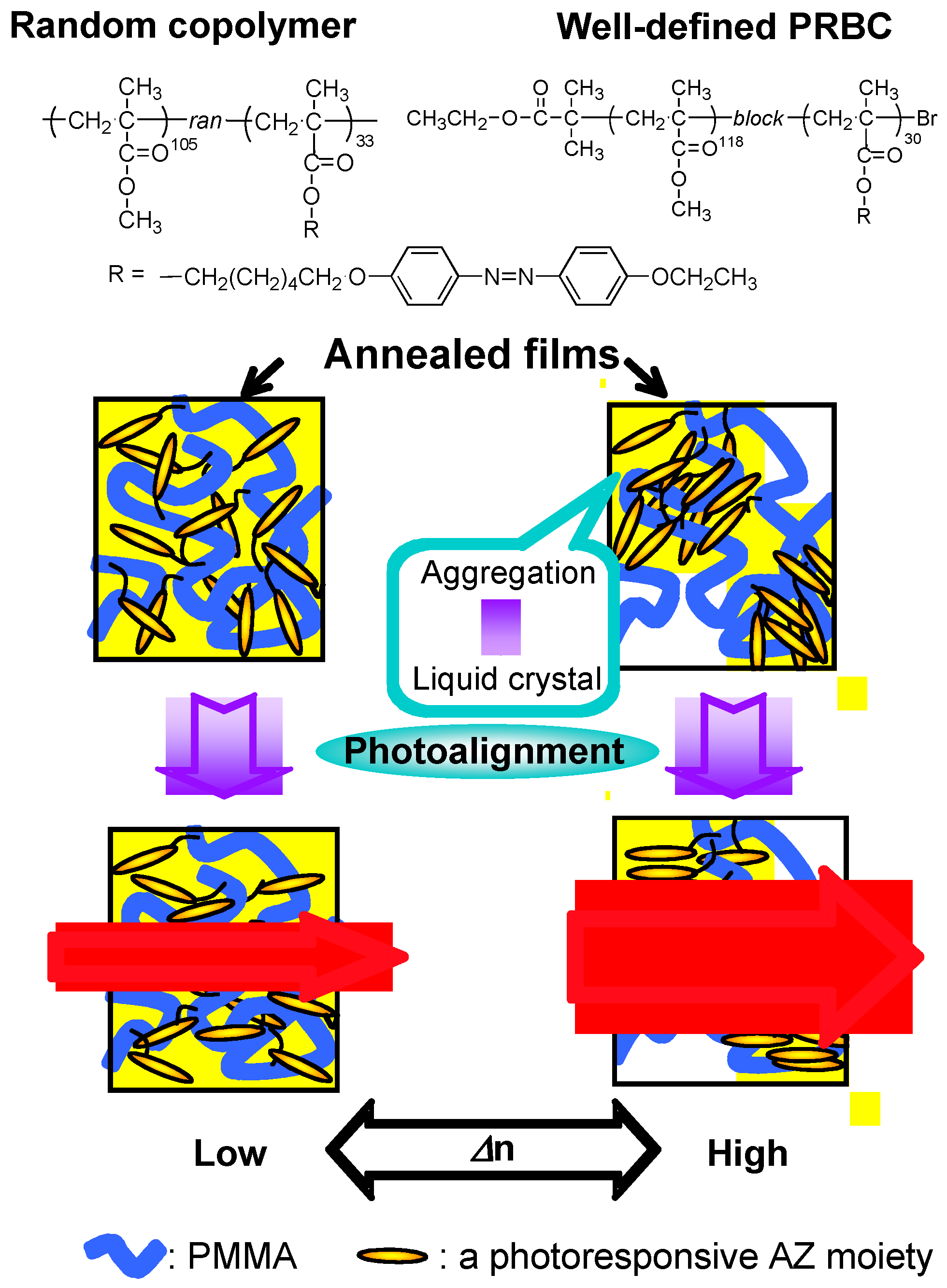
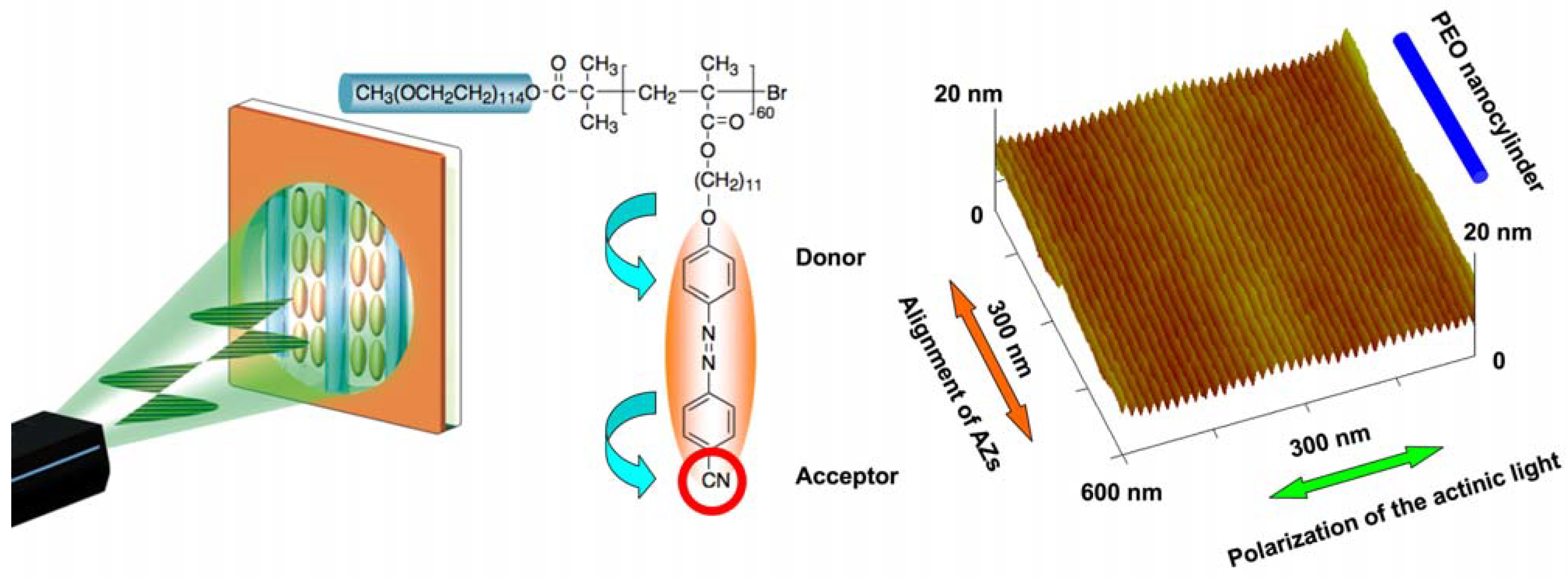
4.3. Photoalignment
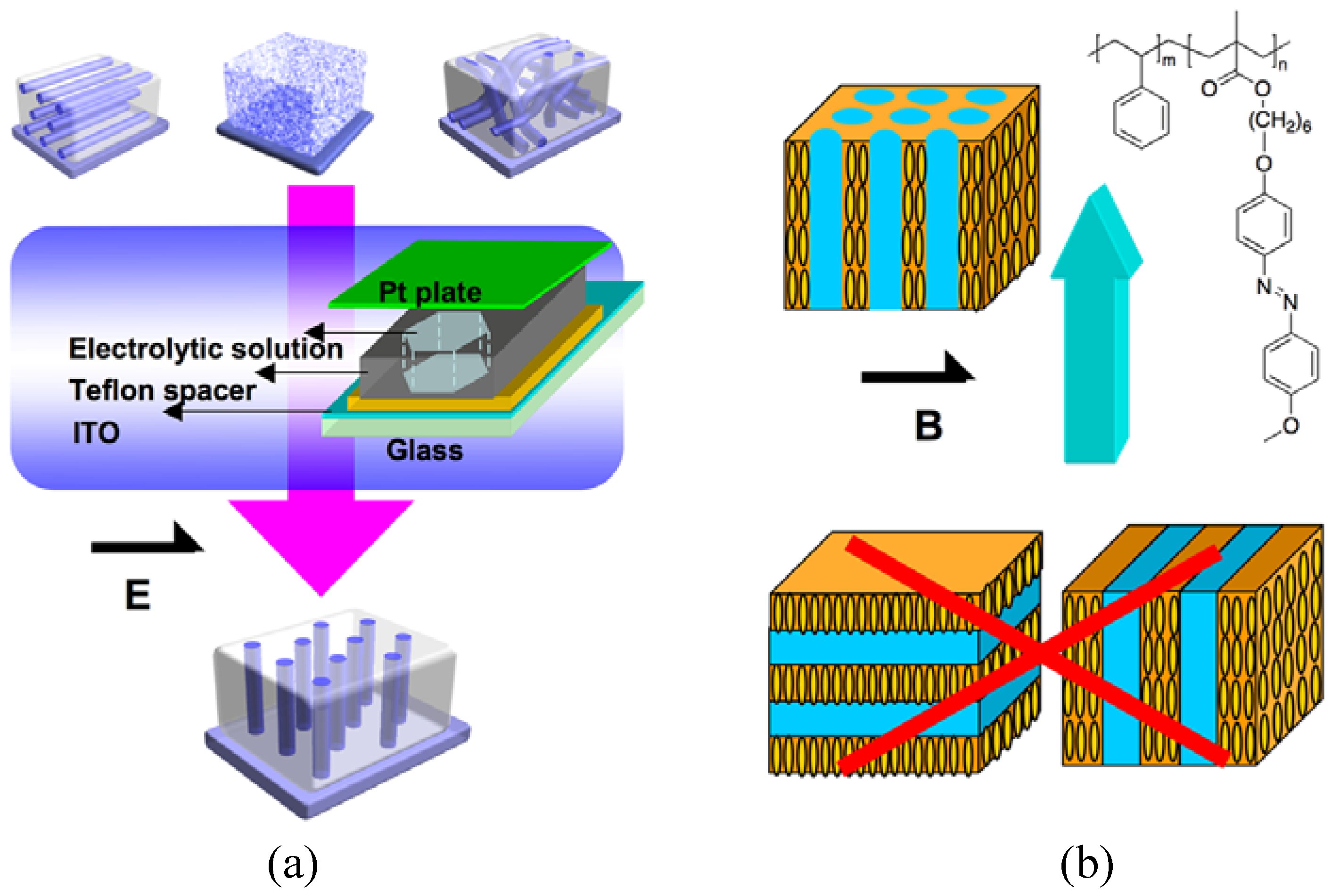
4.4. Electric and Magnetic Fields
4.5. Other Methods
5. Applications
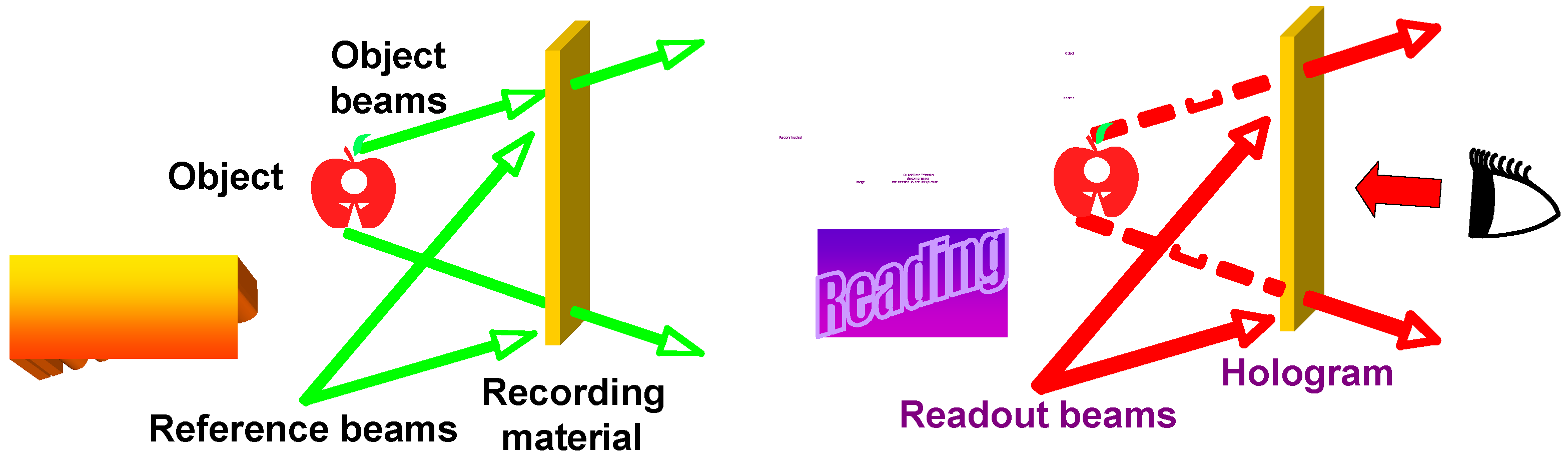
5.1. Holographic Gratings and Storage
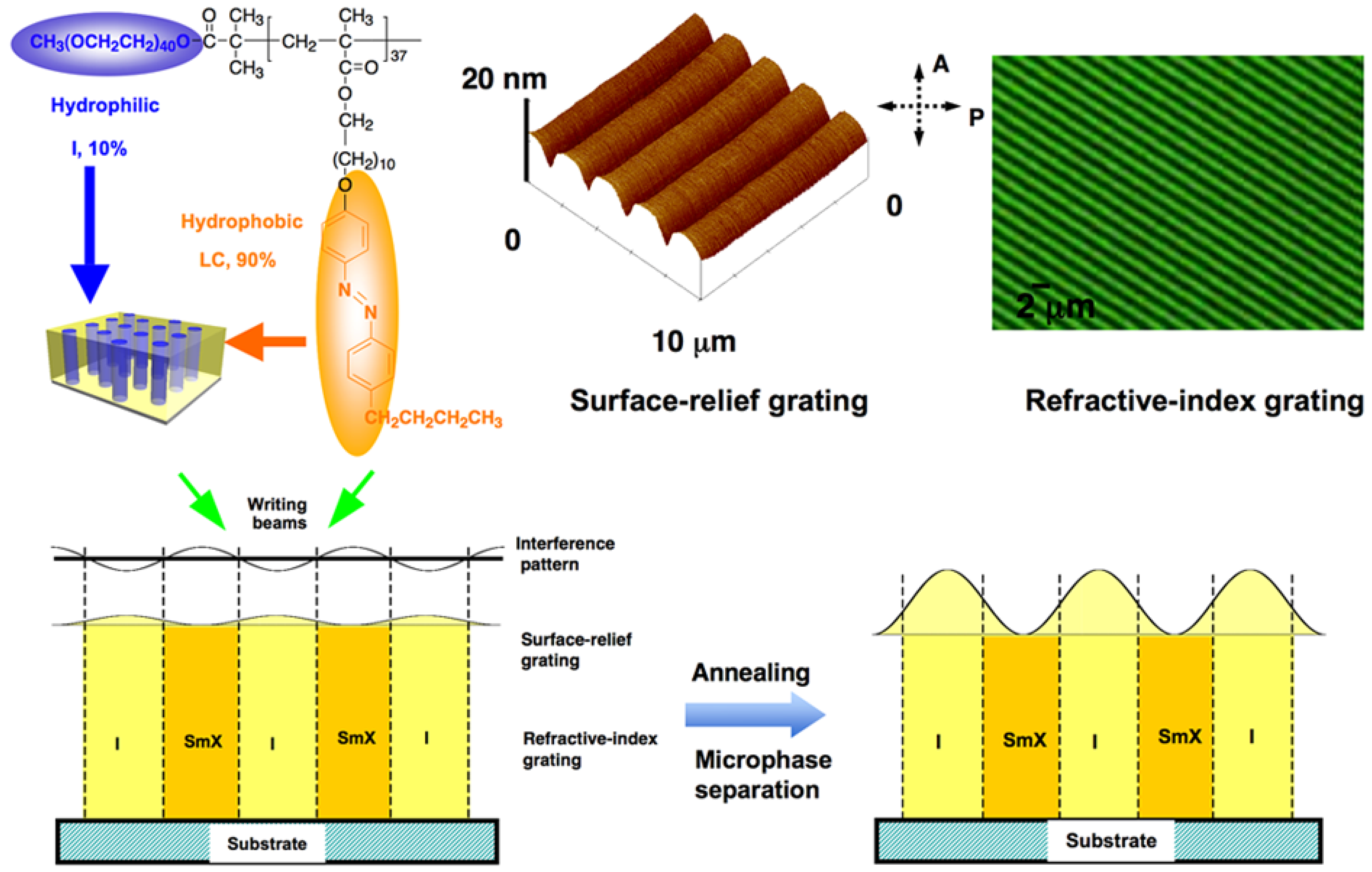
5.1.1. Enhancement of Surface Relief
5.1.2. Enhancement of Refractive-Index Modulation
5.1.3. Adjusting Fringe Spacing of Gratings
5.1.4. Volume Storage
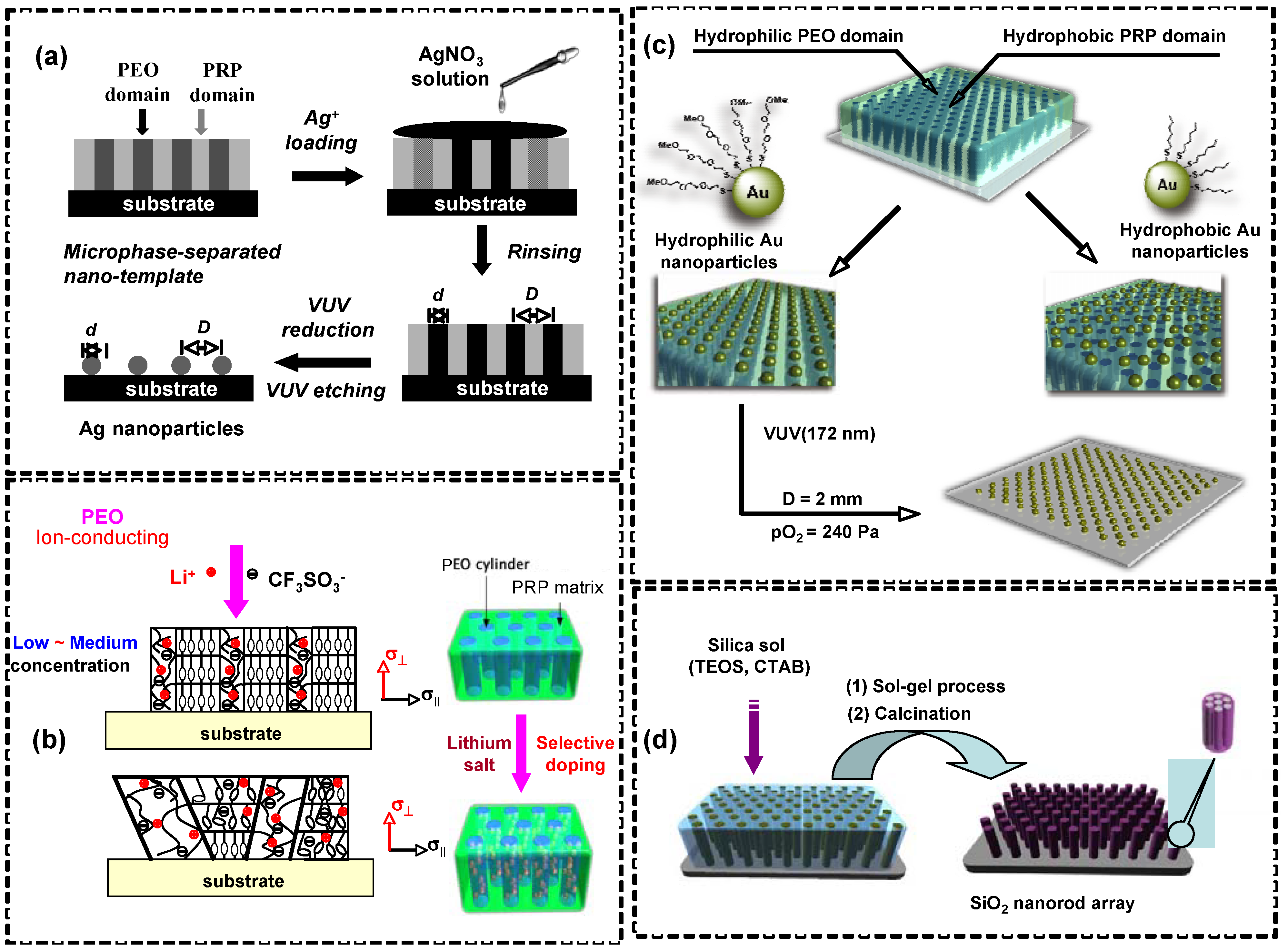
5.2. Nanotemplates
5.3. Photocontrolled Deformation
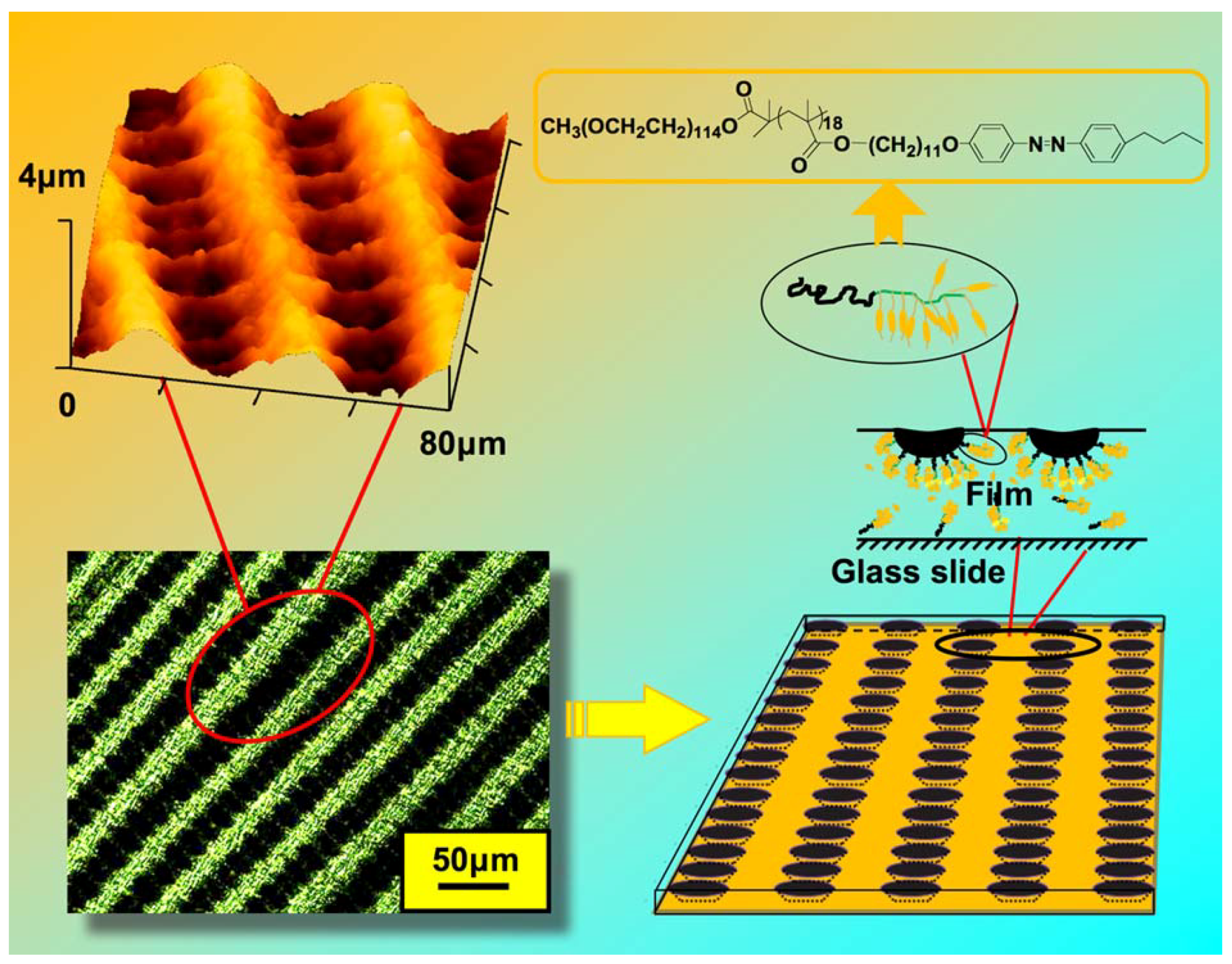
5.4. Microporous Structures
6. Conclusions
Acknowledgements
References
- Kumar, G.; Neckers, D. Photochemistry of azobenzene-containing polymers. Chem. Rev. 1989, 89, 1915–1925. [Google Scholar] [CrossRef]
- Xie, S.; Natansohn, A.; Rochon, P. Recent developments in aromatic azo polymers research. Chem. Mater. 1993, 5, 403–411. [Google Scholar] [CrossRef]
- Viswanathan, N.; Kim, D.; Bian, S.; Williams, J.; Liu, W.; Li, L.; Samuelson, L.; Kumar, J.; Tripathy, S. Surface relief structures on azo polymer films. J. Mater. Chem. 1999, 9, 1941–1955. [Google Scholar] [CrossRef]
- Ichimura, K. Photoalignment of liquid-crystal systems. Chem. Rev. 2000, 100, 1847–1873. [Google Scholar] [CrossRef] [PubMed]
- Hvilsted, S.; Ramanujam, P.S. The azobenzene optical storage puzzle-demands on the polymer scaffold? Monatshefte fur Chemie 2001, 132, 43–51. [Google Scholar] [CrossRef]
- Natansohn, A.; Rochon, P. Photoinduced motions in azo-containing polymers. Chem. Rev. 2002, 102, 4139–4175. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T. Photomodulation of liquid crystal orientations for photonic applications. J. Mater. Chem. 2003, 13, 2037–2057. [Google Scholar] [CrossRef]
- Zhao, Y.; He, J. Azobenzene-containing block copolymers: the interplay of light and morphology enables new functions. Soft. Mater. 2009, 5, 2686–2693. [Google Scholar] [CrossRef]
- Yager, K.; Barrett, C. Novel Photoswitching using azobenzene functional materials. J. Photochem. Photobio. A: Chem. 2006, 182, 250–261. [Google Scholar] [CrossRef]
- Seki, T. Smart photoresponsive polymer systems organized in two dimensions. Bull. Chem. Soc. Jpn. 2007, 80, 2084–2109. [Google Scholar] [CrossRef]
- Ikeda, T.; Mamiya, J.; Yu, Y. Photomechanics of liquid-crystalline elastomers and other polymers. Angew. Chem. Int. Ed. 2007, 46, 506–528. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.F.; Li, J.; Ikeda, T.; Iyoda, T. Macroscopic parallel nanocylinder array fabrication using a simple rubbing technique. Adv. Mater. 2006, 18, 2213–2215. [Google Scholar] [CrossRef]
- Yu, H.F.; Iyoda, T.; Ikeda, T. Photoinduced alignment of nanocylinders by supramolecular cooperative motions. J. Am. Chem. Soc. 2006, 128, 11010–11011. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Watanabe, K.; Kong, X.; Abe, J.; Iyoda, T. Synthesis, nanostructures, and functionality of amphiphilic liquid crystalline block copolymers with azobenzene moieties. Macromolecules 2002, 35, 3739–3747. [Google Scholar] [CrossRef]
- Thomas, E.; Lescanec, R. Phase morphology in block copolymer systems. Philosoph. transact. Roy. Soc. London. A 1994, 348, 149–166. [Google Scholar] [CrossRef]
- Bates, F.; Fredrichson, G. Block copolymer-designer soft materials. Phys. Today 1999, 32–38. [Google Scholar] [CrossRef]
- Hawker, C.; Thomas, P. Block copolymer lithography: merging “bottom-up” with “top-down” processes. MRS Bull. 2005, 30, 952–966. [Google Scholar] [CrossRef]
- Darling, S. Directing the self-assembly of block copolymers. Prog. Polym. Sci. 2007, 32, 1152–1204. [Google Scholar] [CrossRef]
- Weigert, F. Dichroism induced in a fine-grain silverchloride emulsion by a beam of linearly polarized light. Verh. Dtsch. Phys. Ges. 1919, 21, 479–483. [Google Scholar]
- Yu, H.F.; Asaoka, A.; Shishido, A.; Iyoda, T.; Ikeda, T. Photoinduced nanoscale cooperative motion in a novel well-defined triblock copolymer. Small 2007, 3, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.F.; Shishido, A.; Iyoda, T.; Ikeda, T. Novel wormlike nanostructure self-assembled in a well-defined liquid-crystalline diblock copolymer with azobenzene moieties. Macromol. Rapid Commun. 2007, 28, 927–931. [Google Scholar] [CrossRef]
- Cui, L.; Tong, X.; Yan, X.; Liu, G.; Zhao, Y. Photoactive thermoplastic elastomers of azobenzene-containing triblock copolymers prepared through atom transfer radical polymerization. Macromolecules 2004, 37, 7097–7104. [Google Scholar] [CrossRef]
- Qi, B.; Yavrian, A.; Galstian, T.; Zhao, Y. Liquid crystalline ionomers containing azobenzene mesogens: phase stability, photoinduced birefringence and holographic grating. Macromolecules 2005, 38, 3079–3086. [Google Scholar]
- Ravi, P.; Sin, S.; Gan, L.; Gan, Y.; Tam, K.; Xia, X.; Hu, X. New water soluble azobenzene-containing diblock copolymers: synthesis and aggregation behavior. Polymer 2005, 46, 137–146. [Google Scholar] [CrossRef]
- Zhao, Y. Rational design of light-controllable polymer micelles. Chem. Rec. 2007, 7, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Discher, D.; Eisenberg, A. Polymer vesicles. Science 2002, 297, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y. Photocontrollable block copolymer micelles: what can we control? J. Mater. Chem. 2009, 19, 4887–4895. [Google Scholar] [CrossRef]
- Park, C.; Yoon, J.; Thomas, E. Enabling nanotechnology with self assembled block copolymer patterns. Polymer 2003, 44, 6725–6760. [Google Scholar] [CrossRef]
- Cheng, J.Y.; Ross, C.; Smith, H.; Thomas, E. Templated self-assembly of block copolymers: top-down helps bottom-up. Adv. Mater. 2006, 18, 2505–2521. [Google Scholar] [CrossRef]
- Finkelmann, H.; Bohnert, R. Liquid-crystalline side-chain AB block copolymers by direct anionic polymerization of a mesogenic methacrylate. Macromol. Chem. Phys. 1994, 195, 689–700. [Google Scholar]
- Lehmann, O.; Forster, S.; Springer, J. Synthesis of new side-group liquid crystalline block copolymers by living anionic polymerization. Macromol. Rapid Commun. 2000, 21, 133–135. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Xia, J. Atom transfer radical polymerization. Chem. Rev. 2001, 101, 2921–2990. [Google Scholar] [CrossRef] [PubMed]
- Coessensm, V.; Pintauer, T.; Matyjaszewski, K. Functional polymers by atom transfer radical polymerization. Prog. Polym. Sci. 2001, 26, 337–377. [Google Scholar] [CrossRef]
- Han, Y.; Dufour, B.; Wu, W.; Kowalewski, T.; Matyjaszewski, K. Synthesis and characterization of new liquid-crystalline block copolymers with p-cyanoazobenzene moieties and poly(n-butyl acrylate) segments using atom-transfer radical polymerization. Macromolecules 2004, 37, 9355–9365. [Google Scholar] [CrossRef]
- Cui, L.; Dahmane, S.; Tong, X.; Zhu, L.; Zhao, Y. Using self-assembly to prepare multifunctional diblock copolymers containing azopyridine moiety. Macromolecules 2005, 38, 2076–2084. [Google Scholar] [CrossRef]
- Tong, X.; Cui, L.; Zhao, Y. Confinement effects on photoalignment, photochemical phase transition and thermochromic behavior of liquid crystalline azobenzene-containing diblock copolymers. Macromolecules 2004, 37, 3101–3112. [Google Scholar] [CrossRef]
- Cui, L.; Zhao, Y.; Yavrian, A.; Galstian, T. Synthesis of azobenzene-containing diblock copolymers using atom transfer radical polymerization and the photoalignment behavior. Macromolecules 2003, 36, 8246–8252. [Google Scholar] [CrossRef]
- Morikawa, Y.; Kondo, T.; Nagano, S.; Seki, T. Photoinduced 3D ordering and patterning of microphase-separated nanostructure in polystyrene-based block copolymer. Chem. Mater. 2007, 19, 1540–1542. [Google Scholar] [CrossRef]
- Yu, H.F.; Shishido, A.; Ikeda, T.; Iyoda, T. Novel amphiphilic diblock and triblock liquid-crystalline copolymers with well-defined structures prepared by atom transfer radical polymerization. Macromol. Rapid Commun. 2005, 26, 1594–1598. [Google Scholar] [CrossRef]
- Tang, X.; Gao, L.; Fan, X.; Zhou, Q. ABA-type amphiphilic triblock copolymers containing p-ethoxyazobenzene via atom transfer radical polymerization: synthesis, characterization, and properties. J. Polym. Sci. Part A: Polym. Chem. 2007, 45, 2225–2234. [Google Scholar] [CrossRef]
- Forcen, P.; Oriol, L.; Sanchez, C.; Alcala, R.; Hvilsted, S.; Jankova, K.; Loos, J. Synthesis, characterization and photoinduction of optical anisotropy in liquid crystalline diblock azo-copolymers. J. Polym. Sci. Part A: Polym. Chem. 2007, 45, 1899–1910. [Google Scholar] [CrossRef]
- He, X.; Zhang, H.; Yan, D.; Wang, X. Synthesis of side-Chain liquid-crystalline homopolymers and triblock copolymers with p-methoxyazobenzene moieties and poly(ethylene glycol) as coil segments by atom transfer radical polymerization and their thermotropic phase behavior. J. Polym. Sci. Part A: Polym. Chem. 2003, 41, 2854–2864. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheng, Z.; Chen, X.; Zhang, W.; Wu, J.; Zhu, J.; Zhu, X. Synthesis and photoresponsive behaviors of well-defined azobenzene-containing polymers via RAFT polymerization. Macromolecules 2007, 40, 4809–4817. [Google Scholar] [CrossRef]
- Zhao, Y.; Qi, B.; Tong, X.; Zhao, Y. Synthesis of double side-chain liquid crystalline block copolymers using RAFT polymerization and the orientational cooperative effect. Macromolecules 2008, 41, 3823–3831. [Google Scholar] [CrossRef]
- Yoshida, E.; Ohta, M. Preparation of micelles with azobenzene at their coronas or cores from nonamphiphilic diblock copolymers. Colloid Polym. Sci. 2005, 283, 521–531. [Google Scholar] [CrossRef]
- Adams, J.; Gronski, W. LC side chain AB-block copolymers with an amorphous A-block and a liquid-crystalline B-block. Macromol. Rapid Commun. 1989, 10, 553–557. [Google Scholar] [CrossRef]
- Mao, G.; Wang, J.; Clingman, S.; Ober, C.; Chen, J.; Thomas, E. Molecular design, synthesis, and characterization of liquid crystal-coil diblock copolymers with azobenzene side groups. Macromolecules 1997, 30, 2556–2567. [Google Scholar] [CrossRef]
- Frenz, C.; Fuchs, A.; Schmidt, H.W.; Theissen, U.; Haarer, D. Diblock copolymers with azobenzene side-groups and polystyrene matrix: synthesis, characterization and photoaddressing. Macromol. Chem. Phys. 2004, 205, 1246–1258. [Google Scholar] [CrossRef]
- Hayakawa, T.; Horiuchi, S.; Shimizu, H.; Kawazoe, T.; Ohtsu, M. Synthesis and characterization of polystyrene-b-poly(1,2-isoprene-ran–3,4-isoprene) block copolymers with azobenzene side groups. J. Polym. Sci. A: Polym. Chem. 2002, 40, 2406–2414. [Google Scholar] [CrossRef]
- Wang, D.; Ye, G.; Wang, X. Synthesis of aminoazobenzene-containing diblock copolymer and photoinduced deformation behavior of its micelle-like aggregates. Macromol. Rapid Commun. 2007, 28, 2237–2243. [Google Scholar] [CrossRef]
- Wang, X.; Chen, J.; Marturunkakul, S.; Li, L.; Kumar, J.; Tripathy, S. Epoxy-based nonlinear optical polymers functionalized with tricyanovinyl chromophores. Chem. Mater. 1997, 9, 45–50. [Google Scholar] [CrossRef]
- Wang, D.; Ye, G.; Zhu, Y.; Wang, X. Photoinduced mass-migration behavior of two amphiphilic side-chain azo diblock copolymers with different length flexible spacers. Macromolecules 2009, 42, 2651–2657. [Google Scholar] [CrossRef]
- Yu, H.F.; Kobayasi, T.; Ge, Z. Precise control of photoinduced birefringence in azobenzene-containing liquid-crystalline polymers by post functionalization. Macromol. Rapid Commun. 2009, 30, 1725–1730. [Google Scholar] [CrossRef] [PubMed]
- Makinen, R.; Ruokolainen, J.; Ikkala, O.; Moel, K.; Brinke, G.; Odorico, W.; Stamm, M. Orientation of supramolecular self-organized polymeric nanostructures by oscillatory shear flow. Macromolecules 2000, 33, 3441–3446. [Google Scholar] [CrossRef]
- Sidorenko, A.; Tokarev, I.; Minko, S.; Stamm, M. Ordered reactive nanomembranes/ nanotemplates from thin films of block copolymer supramolecular assembly. J. Am. Chem. Soc. 2003, 125, 12211–12216. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.; Li, X.; Ober, C.; Osuji, C.; Thomas, E. Orientational switching of mesogens and microdomains in hydrogen-bonded side-chain liquid-crystalline block copolymers using AC electric fields. Adv. Funct. Mater. 2004, 14, 364–370. [Google Scholar] [CrossRef]
- Serhatli, I.; Serhatli, M. Synthesis and characterization of amorphous-liquid crystalline poly(vinyl ether) block copolymers. Turk. J. Chem. 1998, 22, 279–287. [Google Scholar]
- Yu, H.F.; Iyoda, T.; Okano, K.; Shishido, A.; Ikeda, T.; Watanabe, K. Photoresponsive behavior and photochemical phase transition of amphiphilic diblock liquid-crystalline copolymer. Mol. Cryst. Liq. Cryst. 2005, 443, 191–199. [Google Scholar] [CrossRef]
- Trenor, S.; Shultz, A.; Love, B.; Long, T. Coumarins in polymers: from light harvesting to photo-cross-linkable tissue scaffolds. Chem. Rev. 2004, 104, 3059–3077. [Google Scholar] [CrossRef] [PubMed]
- Dilline, W. Polymerization of unsaturated compounds by photocyloaddition reaction. Chem. Rev. 1983, 83, 1–47. [Google Scholar] [CrossRef]
- Hasegawa, M. Photopolymerization of diolefin crystals. Chem. Rev. 1983, 83, 507–518. [Google Scholar] [CrossRef]
- Wu, Y.; Demachi, Y.; Tsutsumi, O.; Kanazawa, A.; Shiono, T.; Ikeda, T. Photoinduced alignment of polymer liquid crystals containing azobenzene moieties in the side chain. 1. effect of light intensity on alignment behavior. Macromolecules 1998, 31, 349–354. [Google Scholar] [CrossRef]
- Wu, Y.; Demachi, Y.; Tsutsumi, O.; Kanazawa, A.; Shiono, T.; Ikeda, T. Photoinduced alignment of polymer liquid crystals containing azobenzene moieties in the side chain. 3. effect of structure of photochromic moieties on alignment behavior. Macromolecules 1998, 31, 4457–4463. [Google Scholar] [CrossRef]
- Lin, S.; Lin, J.; Nose, T.; Iyoda, T. Micellar structures of block-copolymers with ordered cores in dilute solution as studied by polarized and depolarized light scattering. J. Polym. Sci. B: Polym. Phys. 2007, 45, 1333–1343. [Google Scholar] [CrossRef]
- Li, J.; Kamata, K.; Watanabe, S.; Iyoda, T. Template- and vacuum-ultraviolet-assisted fabrication of a Ag-nanoparticle array on flexible and rigid substrates. Adv. Mater. 2007, 19, 1267–1271. [Google Scholar] [CrossRef]
- Li, J.; Kamata, K.; Komura, M.; Yamada, T.; Yoshida, H.; Iyoda, T. Anisotropic ion conductivity in liquid crystalline diblock copolymer membranes with perpendicularly oriented PEO cylindrical domains. Macromolecules 2007, 40, 8125–8128. [Google Scholar] [CrossRef]
- Breiner, T.; Kreger, K.; Hagen, R.; Hackel, M.; Kador, L.; Muller, A.; Kramer, E.; Schmidt, H. Blends of poly(methacrylate) block copolymers with photoaddressable segments. Macromolecules 2007, 40, 2100–2108. [Google Scholar] [CrossRef]
- Paik, M.; Krishnan, S.; You, F.; Li, X.; Hexemer, A.; Ando, Y.; Kang, S.; Fischer, D.; Kramer, E.; Ober, C. Surface organization, light-driven surface changes, and stability of semifluorinated azobenzene polymers. Langmuir 2007, 23, 5110–5119. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Doi, M.; Kanaoka, S.; Aoshima, S. Polymer surface modification using diblock copolymers containing azobenzene. J. Polym. Sci. A: Polym. Chem. 2005, 43, 5704–5709. [Google Scholar] [CrossRef]
- Naka, Y.; Yu, H.F.; Shishido, A.; Ikeda, T. Photoalignment and holographic properties of azobenzene-containing block copolymers with oxyethylene and alkyl spacers. Mol. Cryst. Liq. Cryst. 2009, 498, 118–130. [Google Scholar] [CrossRef]
- Yu, H.F.; Naka, Y.; Shishido, A.; Ikeda, T. Well-defined liquid-crystalline diblock copolymers with an azobenzene moiety: synthesis, photoinduced alignment and their holographic properties. Macromolecules 2008, 41, 7959–7966. [Google Scholar] [CrossRef]
- Hamley, I. Nanostructure fabrication using block copolymers. Nanotechnology 2003, 14, R39–R54. [Google Scholar] [CrossRef]
- Mao, G.; Ober, C. Block copolymers containing liquid crystalline segements. Acta Polym. 1996, 48, 405–422. [Google Scholar] [CrossRef]
- Finkelmann, H.; Walther, M. Structure formation of liquid crystalline block copolymers. Prog. Polym. Sci. 1996, 21, 951–979. [Google Scholar]
- Whitesides, G.; Grzybowski, B. Self-assembly at all scales. Science 2002, 295, 2418–2421. [Google Scholar] [CrossRef] [PubMed]
- Komura, M.; Iyoda, T. AFM cross-sectional imaging of perpendicularly oriented nanocylinder structures of microphase-separated block copolymer films by crystal-like cleavage. Macromolecules 2007, 40, 4106–4108. [Google Scholar] [CrossRef]
- Watanabe, K.; Yoshida, H.; Kamata, K.; Iyoda, T. Direct TEM observation of perpendicularly oriented nanocylinder structure in amphiphilic liquid crystalline block copolymer thin films. Trans. Mater. Res. Soc. Jpn. 2005, 30, 377–381. [Google Scholar]
- Kim, S.; Solak, H.; Stoykovich, M.; Ferrier, N.; Pablo, J.; Nealey, P. Epitaxial self-assembly of block copolymers on lithographically defined nanopatterned substrates. Nature 2003, 424, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Berreman, D. Solid surface shape and the alignment of an adjacent nematic liquid crystal. Phys. Rev. Lett. 1972, 28, 1683–1686. [Google Scholar] [CrossRef]
- Demus, D.; Goodbye, J.; Gray, G.; Spiess, H.; Vill, V. Handbook of Liquid Crystals; Wiley-VCH Verlag: Weinheim, Germany, 1998. [Google Scholar]
- Ikeda, T.; Nakano, M.; Yu, Y.; Tsutsumi, O.; Kanazawa, A. Anisotropic bending and unbending behavior of azobenzene liquid-crystalline gels by light. Adv. Mater. 2003, 15, 201–205. [Google Scholar] [CrossRef]
- Reiter, G.; Gastelein, G.; Hoerner, P.; Riess, G.; Blumen, A.; Sommer, J. Nanometer-scale surface patterns with long-range order created by crystallization of diblock copolymers. Phys. Rev. Lett. 1999, 83, 3844–3847. [Google Scholar] [CrossRef]
- Ikeda, T.; Tsutsumi, O. Optical switching and image storage by means of azobenzene liquid-crystal films. Science 1995, 268, 1873–1875. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, Y.; Nagano, S.; Watanabe, K.; Kamata, K.; Iyoda, T.; Seki, T. Optical alignment and patterning of nanoscale microdomains in a block copolymer thin film. Adv. Mater. 2006, 18, 883–886. [Google Scholar] [CrossRef]
- Kamata, K.; Iyoda, T. Alignment control and templating process in amphiphilic block copolymer thin film. Res. Rep.-NIFS-PROC Series 2008, 70, 48–50. [Google Scholar]
- Tomikawa, N.; Lu, Z.; Itoh, T.; Imre, C.T.; Adachi, M.; Tokita, M.; Watanabe, J. Orientation of microphase-segregated cylinders in liquid crystalline diblock copolymer by magnetic field. Jpn. J. Appl. Phys. 2005, 44, L711–L714. [Google Scholar] [CrossRef]
- Osuji, C.; Ferreira, P.; Mao, G.; Ober, C.; Vander, J.; Thomas, E. Alignment of self-assembled hierarchical microstructure in liquid crystalline diblock copolymers using high magnetic fields. Macromolecules 2004, 37, 9903–9908. [Google Scholar] [CrossRef]
- Hamley, I.; Castelletto, V.; Lu, Z.; Imrie, C.; Itoh, T.; Al-Hussein, M. Interplay between smectic ordering and microphase separation in a series of side-group liquid-crystal block copolymers. Macromolecules 2004, 37, 4798–4807. [Google Scholar] [CrossRef]
- Osuji, C.; Zhang, Y.; Mao, G.; Ober, C.; Thomas, E. Transverse cylindrical microdomain orientation in an LC diblock copolymer under oscillatory shear. Macromolecules 1999, 32, 7703–7706. [Google Scholar] [CrossRef]
- Osuji, C.; Chen, J.; Mao, G.; Ober, C.; Thomas, E. Understanding and controlling the morphology of styrene-isoprene side-group liquid crystalline diblock copolymers. Polymer 2000, 41, 8897–8907. [Google Scholar] [CrossRef]
- Tokita, M.; Adachi, M.; Masuyama, S.; Takazawa, F.; Watanabe, J. Characteristic shear-flow orientation in LC block copolymer resulting from compromise between orientations of microcylinder and LC mesogen. Macromolecules 2007, 40, 7276–7282. [Google Scholar] [CrossRef]
- Tokita, M.; Adachi, M.; Takazawa, F.; Watanabe, J. Shear flow orientation of cylindrical microdomain in liquid crystalline diblock copolymer and its potentiality as anchoring substrate for nematic mesogens. Jpn. J. Appl. Phys. 2006, 45, 9152–9156. [Google Scholar] [CrossRef]
- Rochon, P.; Batalla, E.; Natanhson, A. Optically induced surface gratings on azoaromatic polymer films. Appl. Phys. Lett. 1995, 66, 136–138. [Google Scholar] [CrossRef]
- Kim, D.; Tripathy, S.; Li, L.; Kumar, J. Laser-induced holographic surface relief gratings on nonlinear optical polymer films. Appl. Phys. Lett. 1995, 66, 1166–1168. [Google Scholar] [CrossRef]
- Yu, H.F.; Okano, K.; Shishido, A.; Ikeda, T.; Kamata, K.; Komura, M.; Iyoda, T. Enhancement of surface-relief gratings recorded in amphiphilic liquid-crystalline diblock copolymer by nanoscale phase separation. Adv. Mater. 2005, 17, 2184–2188. [Google Scholar] [CrossRef]
- Yu, H.F.; Shishido, A.; Iyoda, T.; Ikeda, T. Effect of recording time on grating formation and enhancement in an amphiphilic diblock liquid-crystalline copolymer. Mol. Cryst. Liq. Cryst. 2009, 498, 29–39. [Google Scholar]
- Naka, Y.; Yu, H.F.; Shishido, A.; Ikeda, T. Photoalignment and holographic properties of azobenzene-containing block copolymers with oxyethylene and alkyl spacers. Mol. Cryst. Liq. Cryst. 2009, 513, 131–141. [Google Scholar] [CrossRef]
- Yu, H.F.; Shishido, A.; Ikeda, T. Subwavelength modulation of surface relief and refractive index in pre-irradiated liquid-crystalline polymer Films. Appl. Phys. Lett. 2008, 92, 103117/1–3. [Google Scholar] [CrossRef]
- Frenz, C.; Fuchs, A.; Schmidt, H.; Theissen, U.; Haarer, D. Diblock copolymers with azobenzene side-groups and polystyrene matrix: synthesis, characterization and photoaddressing. Macromol. Chem. Phys. 2004, 205, 1246–1258. [Google Scholar] [CrossRef]
- Pakula, T.; Saijo, K.; Kawai, H.; Hashimoto, T. Deformation behavior of styrene-butadiene-styrene triblock copolymer with cylindrical morphology. Macromolecules 1985, 18, 1294–1302. [Google Scholar] [CrossRef]
- Bai, S.; Zhao, Y. Azobenzene elastomers for mechanically tunable diffraction gratings. Macromolecules 2002, 35, 9657–9664. [Google Scholar] [CrossRef]
- Zhao, Y.; Bai, S.; Dumont, D.; Galstian, T. mechanically tunable diffraction gratings recorded on an azobenzene elastomer. Adv. Mater. 2002, 14, 512–514. [Google Scholar] [CrossRef]
- Bai, S.; Zhao, Y. Azobenzene-containing thermoplastic elastomers: coupling mechanical and optical effects. Macromolecules 2001, 34, 9032–9038. [Google Scholar] [CrossRef]
- Saishoji, A.; Sato, D.; Shishido, A.; Ikeda, T. Formation of bragg gratings with large angular multiplicity by means of photoinduced reorientation of azobenzene. Langmuir 2007, 23, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, M.; Sato, D.; Shishido, A.; Ikeda, T. Bragg-type polarization gratings formed in thick polymer films containing azobenzene and tolane moieties. Langmuir 2007, 23, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Minabe, J.; Maruyama, T.; Yasuda, S.; Kawano, K.; Hayashi, K.; Ogasawara, Y. Design of dye concentrations in azobenzene-containing polymer films for volume holographic storage. Jpn. J. Appl. Phys. 2004, 43, 4964–4967. [Google Scholar] [CrossRef]
- Häckel, M.; Kador, L.; Kropp, D.; Frenz, C.; Schmidt, H. Holographic gratings in diblock copolymers with azobenzene and mesogenic side groups in the photoaddressable dispersed phase. Adv. Funct. Mater. 2005, 15, 1722–1727. [Google Scholar] [CrossRef]
- Häckel, M.; Kador, L.; Kropp, D.; Schmidt, H. Polymer blends with azobenzene-containing block copolymers as stable rewritable volume holographic media. Adv. Mater. 2007, 19, 227–231. [Google Scholar] [CrossRef]
- Suzuki, S.; Kamata, K.; Yamauchi, H.; Iyoda, T. Selective doping of lead ions into normally aligned PEO cylindrical nanodomains in amphiphilic block copolymer thin films. Chem. Lett. 2007, 36, 978–979. [Google Scholar] [CrossRef]
- Watanabe, S.; Fujiwara, R.; Hada, M.; Okazaki, Y.; Iyoda, T. Site-specific recognition of nanophase-separated surfaces of amphiphilic block copolymers by hydrophilic and hydrophobic gold nanoparticles. Angew. Chem. Int. Ed. 2007, 46, 1120–1123. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Komura, M.; Kamata, K.; Iyoda, T. Highly ordered arrays of mesoporous silica nanorods with tunable aspect ratios from block copolymer thin films. Adv. Mater. 2008, 20, 763–767. [Google Scholar] [CrossRef]
- Kadota, S.; Aoki, K.; Nagano, S.; Seki, T. Photocontrolled microphase separation of block copolymers in two dimensions. J. Am. Chem. Soc. 2005, 127, 8266–8267. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Tong, X.; Zhao, Y. Preparation of azobenzene-containing amphiphilic diblock copolymers for light-responsive micellar aggregates. Macromolecules 2004, 37, 8911–8917. [Google Scholar] [CrossRef]
- Lee, H.; Wu, W.; Oh, J.K.; Mueller, L.; Sherwood, G.; Peteanu, L.; Kowalewski, T.; Matyjaszewski, K. Light-induced reversible formation of polymeric micelles. Angew. Chem. Int. Ed. 2007, 46, 2453–2457. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Tong, X.; Zhao, Y. A new design for light-breakable polymer micelles. J. Am. Chem. Soc. 2005, 127, 8290–8291. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Tong, X.; Morris, D.; Zhao, Y. Toward photocontrolled release using light-dissociable block copolymer micelles. Macromolecules 2006, 39, 4633–4640. [Google Scholar] [CrossRef]
- Ding, J.; Liu, G. Hairy, semi-shaved, and fully shaved hollow nanospheres from polyisoprene-block-poly(2-cinnamoylethyl methacrylate). Chem. Mater. 1998, 10, 537–542. [Google Scholar] [CrossRef]
- Tao, J.; Stewart, S.; Liu, G.; Yang, M. Star and cylindrical micelles of polystyrene-block-poly(2–cinnamoylethyl methacrylate) in cyclopentane. Macromolecules 1997, 30, 2738–2745. [Google Scholar] [CrossRef]
- Liu, G. Block copolymer nanofibers and nanotubes. In Block Copolymers in Nanoscienc; Lazzari, M., Liu, G., Lecommandoux, S., Eds.; Publisher: WILEY-VCH Verlag GmbH & C. KGaA; 2006; pp. 233–256. [Google Scholar]
- Babin, J.; Lepage, M.; Zhao, Y. Decoration of shell-crosslinked reverse polymer micelles using ATRP: a new route to stimuli-responsive nanoparticles. Macromolecules 2008, 41, 1246–1253. [Google Scholar] [CrossRef]
- He, J.; Tong, X.; Tremblay, L.; Zhao, Y. Corona-cross-linked polymer vesicles displaying a large and reversible temperature-responsive volume transition. Macromolecules 2009, 42, 7267–7270. [Google Scholar] [CrossRef]
- Wang, D.; Liu, J.; Ye, G.; Wang, X. Amphiphilic block copolymers bearing strong push-pull azo chromophores: Synthesis, micelle formation and photoinduced shape deformation. Polymer 2009, 50, 418–427. [Google Scholar] [CrossRef]
- Deng, W.; Albouy, P.; Lacaze, E.; Keller, P.; Wang, X.; Li, M. Azobenzene-containing liquid crystal triblock copolymers: synthesis, characterization, and self-assembly behavior. Macromolecules 2008, 41, 2459–2466. [Google Scholar] [CrossRef]
- Deng, W.; Li, M.; Wang, X.; Keller, P. Light-responsive wires from side-on liquid crystalline azo polymers. Liq. Cryst. 2009, 36, 1023–1029. [Google Scholar] [CrossRef]
- Chen, D.; Liu, H.; Kobayasi, T.; Yu, H.F. Fabrication of regularly-patterned microporous films by self-organization of an amphiphilic liquid-crystalline diblock Copolymer under a Dry Environment. Macromol. Mater. Eng. 2009, 295, 26–31. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yu, H.; Kobayashi, T. Photoresponsive Block Copolymers Containing Azobenzenes and Other Chromophores. Molecules 2010, 15, 570-603. https://doi.org/10.3390/molecules15010570
Yu H, Kobayashi T. Photoresponsive Block Copolymers Containing Azobenzenes and Other Chromophores. Molecules. 2010; 15(1):570-603. https://doi.org/10.3390/molecules15010570
Chicago/Turabian StyleYu, Haifeng, and Takaomi Kobayashi. 2010. "Photoresponsive Block Copolymers Containing Azobenzenes and Other Chromophores" Molecules 15, no. 1: 570-603. https://doi.org/10.3390/molecules15010570
APA StyleYu, H., & Kobayashi, T. (2010). Photoresponsive Block Copolymers Containing Azobenzenes and Other Chromophores. Molecules, 15(1), 570-603. https://doi.org/10.3390/molecules15010570