Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †
Abstract
:Introduction
Results and Discussion
Chemical composition of oximation products
| Structures and names | ||||
|---|---|---|---|---|
 |  |  |  |  |
| 1a-d | 1a | 1b | 1c | 1d |
| Citronellal | Neral | Geranial | Perillaldehyde | |
| 2a-d (E) | 2a (E) | 2b (E) | 2c (E) | 2d (E) |
 | (E) Citronellal oxime | (E) Neral oxime | (E) Geranial oxime | (E) Perillaldehyde oxime or (E) Perillartine |
| 2a-d (Z) | 2a (Z) | 2b (Z) | 2c (Z) | --- |
 | (Z) Citronellal oxime | (Z) Neral oxime | (Z) Geranial oxime | |
| 3a-d | 3a | 3b | 3c | 3d |
 | Citronellal nitrile | Neral nitrile | Geranial nitrile | Perillaldehyde nitrile |

| Compounds | R. T.(min) | m/z a (70 eV) | Relative concentration (%)b | |||||
|---|---|---|---|---|---|---|---|---|
| E. citriodora | C. citratus | L. multiflora | ||||||
| CE. | OP | CE. | OP | CE. | OP | |||
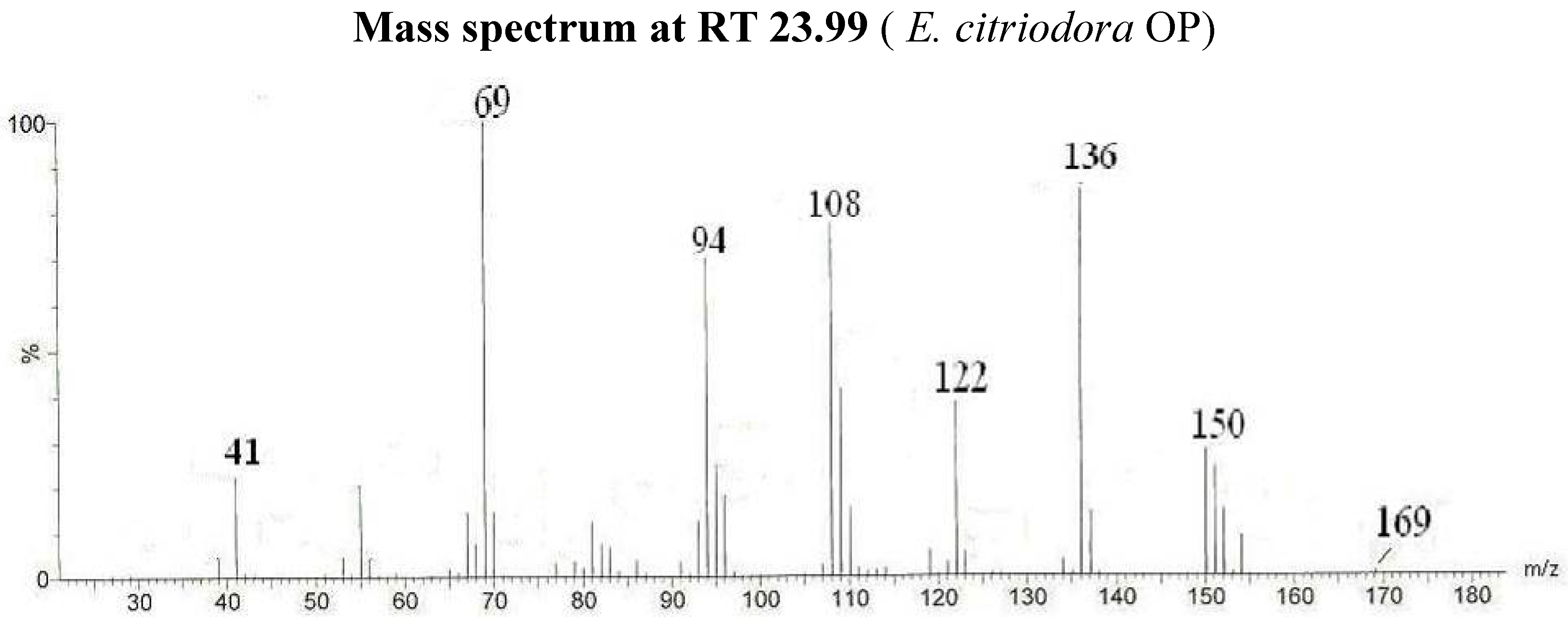
| Citronellal | 15.70 | 121/95/136/154/69/111/139/41/55 | 98.9 | 0.1 | ||||
| Neral nitrile | 18.67 | 69/134/148/149/81/121/41/135 | 1.2 | 1.6 | ||||
| Citronellal nitrile | 18.79 | 136/69/108/94/122/151/150/41/55 | 11.7 | |||||
| Neral | 19.52 | 69/41/94/84/109/119/137/134/123 | 42.1 | -- | 34.1 | 0.8 | ||
| Geranial nitrile | 20.20 | 69/134/148/149/81/121/41/135 | 3.7 | 3.4 | ||||
| Geranial | 20.85 | 69/41/84/94/137/123/109/119/134 | 56.7 | 0.1 | 45.1 | 1.9 | ||
| Perilaldehyde | 21.12 | 79/68/67/107/135/93/121/150 | 19.2 | 0.5 | ||||
| Perilaldehyde nitrile | 22.50 | 105/68/147/104/132/118/67/146 | 1.8 | |||||
| Citronellal oxime (Isomer I) | 23.99 | 69/136/108/94/122/150/41/55/169 | 45.3 | |||||
| Citronellal oxime (Isomer II) | 24.86 | 41.6 | ||||||
| Neral oxime (Isomer I) | 26.02 | 69/150/84/41/94/108/53/167 | 31.3 | 23.2 | ||||
| Neral oxime (Isomer II) | 27.32 | 7.3 | 05.6 | |||||
| Geranial oxime (Isomer I) | 27.51 | 69/84/41/150/108/53/94/167 | 38.3 | 26.1 | ||||
| Geranial oxime (Isomer II) | 28.27 | 17.2 | 14.4 | |||||
| Perillaldehyde oxime | 29.13 | 148/105/132/68/91/165 | 18.8 | |||||
| TOTAL | 98.9 | 98.7 | 98.8 | 99.1 | 98.4 | 98.1 | ||
Stability and volatility studies
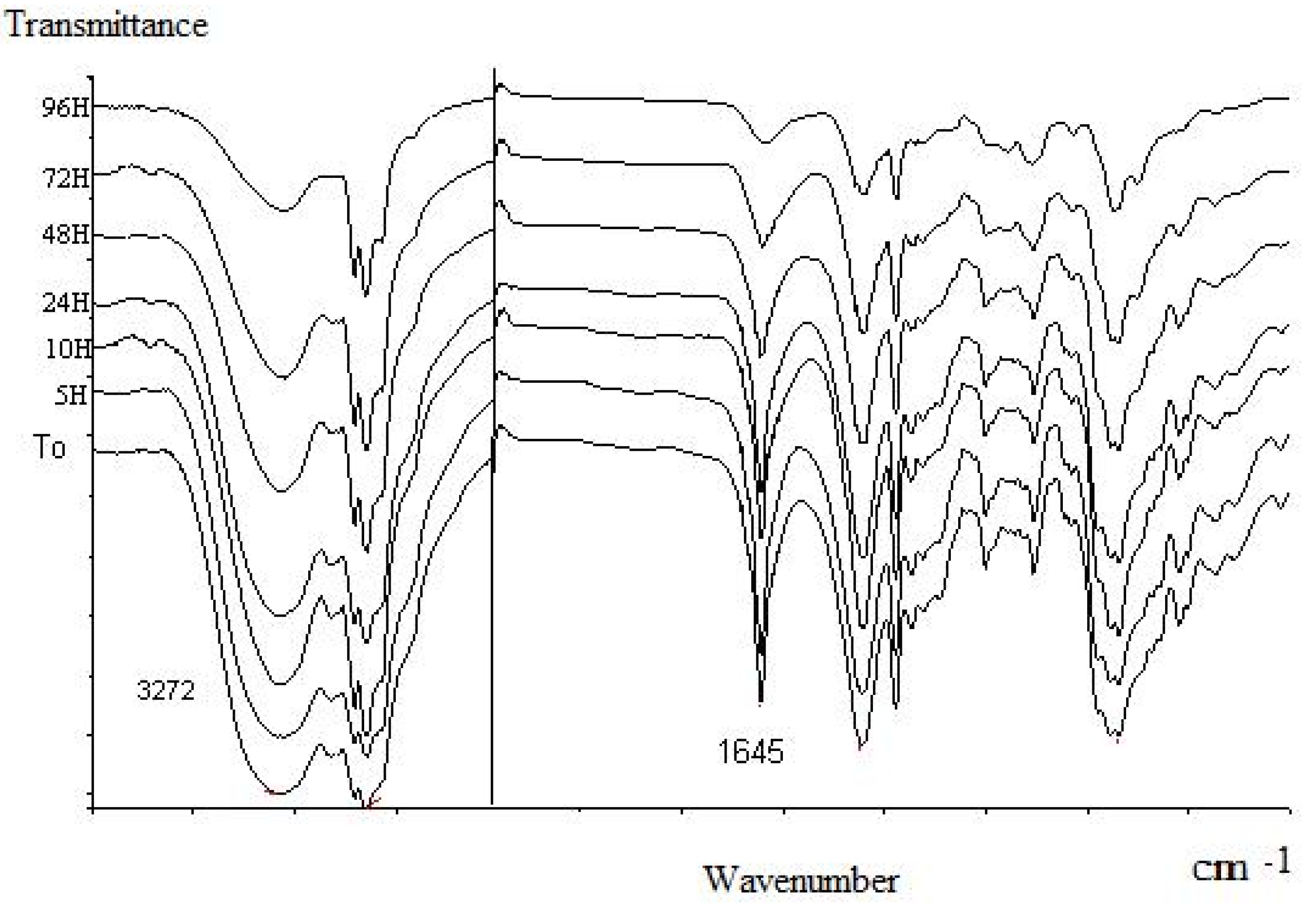
| Main components in Carbonyl Extract (Aromatic plant) | Time* (hour) CE/OP |
|---|---|
| Citronellal ( E. citriodora ) | 3/5 |
| Neral, geranial (C. citratus) | 5/96 |
| Neral, geranial, perialdehyde (L. multiflora) | 5/96 |
Experimental
Samples and Materials
Oxime preparation
Gas chromatography/mass spectrometry analysis (GC-MS)
Infra-red (IR) analysis
Conclusions
Acknowledgment
References
- Levrand, B.; Ruff, Y.; Lehn, J.M.; Herrmann, A. Controlled release of volatile aldehydes and ketones by reversible hydrazone formation-classical profragrances are getting dynamic. Chem. Commun. 2006, 2965–2967. [Google Scholar]
- Narula, A.P.S. The search for new fragrance ingredients for functional perfumery. Chem. Biodivers. 2004, 1, 1992–2000. [Google Scholar] [CrossRef]
- Dikusar, E.A.; Zhukovskaya, N.A.; Moiseichuk, K.L.; Zalesskaya, E.G.; Kurman, P.V.; Vyglazov, O.G. Preparative synthesis of veratraldehyde and citral oxime esters. Russ. J. Appl. Chem. 2008, 81, 643–646. [Google Scholar]
- Bonikowski, R.; Balawejder, M.; Sikora, M.; Kula, J. 4-furanyl and 4-thiophenylbutan-2-one oximes. Sci. Bulletin Tech. Univ. Lodz, Number 1008, Food Chem. Biotechnol. 2007, 71, 05–11. [Google Scholar]
- Wang, T.C.; Chen, I.L.; Lu, C.M.; Kuo, D.H.; Liao, C.H. Synthesis and cytotoxic and antiplatelet activities of oxime- and methyloxime-containing flavone, isoflavone, and xanthone derivatives. Chem. Biodivers. 2005, 2, 253–263. [Google Scholar] [CrossRef]
- Li, H.Q.; Xiao, Z.P.; Yin-Luo; Yan, T.; Lv, P.C.; Zhu, H.L. Amines and oximes derived from deoxybenzoins as Helicobacter pylori urease inhibitors. Eur. J. Med. Chem. 2009, 44, 2246–2251. [Google Scholar] [CrossRef]
- Karakurt, A.; Sevim, D.; Özalp, M.; Özbey, S.; Kendi, E.; Stables, J.P. Synthesis of some 1-(2-naphtyl)-2-imidazole-1-yl)ethanone oxime and oxime ether derivatives and their anticonvulsant and microbial activities. Eur. J. Med. Chem. 2001, 36, 421–433. [Google Scholar] [CrossRef]
- Puntel, G.O.; de Carvalho, N.R.; Gubert, P.; Palma, A.S.; Corte, C.L.D.; Ávila, D.S.; Pereira, M.E.; Carratu, V.S.; Bresolin, L.; J. Da Rocha, B.T.; Soares, F.A.A. Butane-2,3-dionethiosemicarbazone: an oxime with antioxidant properties. Chem. Biol. Interact. 2009, 177, 153–160. [Google Scholar] [CrossRef]
- De Sousa, D.P.; Schefer, R.R.; Brocksom, U.; Brocksom, T.J. Synthesis and antidepressant evaluation of three para-benzoquinone mono-oximes and their oxy derivatives. Molecules 2006, 11, 148–155. [Google Scholar] [CrossRef]
- Ouyang, G.; Chen, Z.; Cai, X.J.; Song, B.A.; Bhadury, P.S.; Yang, S.; Jin, L.H.; Xue, W.; Hu, D.Y.; Zeng, S. Synthesis and antiviral activity of novel pyrazole derivatives containing oxime esters group. Bioorg. Med. Chem. 2008, 16, 9699–9707. [Google Scholar] [CrossRef]
- Chamjangali, M.A.; Soltanpanah, S.; Goudarzi, N. Development and characterization of a copper optical sensor based on immobilization of synthesized 1-phenyl-1,2-propanedione-2-oxime thiosemicarbazone on a triacetylcellulose membrane. Sens. Actuat. B Chem. 2009, 138, 251–256. [Google Scholar] [CrossRef]
- Shokrollahi, A.; Ghaedi, M.; Rajabi, H.R.; Niband, M.S. Potentiometric study of binary complexes of methyl 2-pyridyl ketone oxime, phenyl 2-pyridyl ketone oxime and diacetyl monooxime with some transition and heavy metal ions in aqueous solution. Spectrochim. Acta Part A 2008, 71, 655–662. [Google Scholar] [CrossRef]
- Lapcik, O.; Copikova, J.; Uher, M.; Moravcova, J.; Draar, P. Necukerné prirodni latky sladké chute. Chem. Listy 2007, 101, 44–54. [Google Scholar]
- Zhukovskaya, N.A.; Dikusar, E.A.; Moiseichuk, K.L.; Vyglazov, O.G. Preparative synthesis of menthone oxime esters. Russ. J. Appl. Chem. 2006, 79, 634–636. [Google Scholar]
- Dikusar, E.A.; Zhukovskaya, N.A.; Moiseichuk, K.L.; Zalesskaya, E.G.; Vyglazov, O.G.; Kurman, P.V. Synthesis and structure-aroma correlation of citral oxime esters. Chem. Nat. Comp. 2008, 44, 81–83. [Google Scholar] [CrossRef]
- Ouédraogo, I.W.; Sassiémiké, S.; Bonzi-Coulibaly, Y.L. Chemical extraction via bisulphite adduct of carbonyl compounds from essential oils. Phys. Chem. News 2009, 50, 104–110. [Google Scholar]
- Cui, J.G.; Fan, L.; Huang, L.L.; Liu, H.L.; Zhou, A.M. Synthesis and evaluation of some steroidal oximes as cytotoxic agents: Structure/activity studies. Steroids 2009, 74, 62–72. [Google Scholar] [CrossRef]
- Chakkaravarthy, J.; Muthukumaran, G.; Pandiarajan, K. Conformational study of some N-acyl-2r, 6c-diphenylpiperidin-4-one oximes using NMR spectra. J. Mol. Struct. 2008, 889, 297–307. [Google Scholar] [CrossRef]
- Stepanenko, T.; Lapinski, L.; Nawork, M.J.; Adanmowiicz, L. Infrared spectra of syn and anti isomers of benzaloxime and pyridine-4-aldoxime: an experimental matrix isolation and theorical density theory study. Vibrat. Spectr. 2001, 26, 65–82. [Google Scholar] [CrossRef]
- Acton, E.M.; Leaffer, M.A.; Oliver, S.M.; Stone, H. Structure –taste relations in oximes related to perillartine. J. Agr. Food Chem. 1970, 18, 1061–1068. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the oximation products are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ouédraogo, I.W.; Boulvin, M.; Flammang, R.; Gerbaux, P.; Bonzi-Coulibaly, Y.L. Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †. Molecules 2009, 14, 3275-3285. https://doi.org/10.3390/molecules14093275
Ouédraogo IW, Boulvin M, Flammang R, Gerbaux P, Bonzi-Coulibaly YL. Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †. Molecules. 2009; 14(9):3275-3285. https://doi.org/10.3390/molecules14093275
Chicago/Turabian StyleOuédraogo, Igor W., Michael Boulvin, Robert Flammang, Pascal Gerbaux, and Yvonne L. Bonzi-Coulibaly. 2009. "Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †" Molecules 14, no. 9: 3275-3285. https://doi.org/10.3390/molecules14093275
APA StyleOuédraogo, I. W., Boulvin, M., Flammang, R., Gerbaux, P., & Bonzi-Coulibaly, Y. L. (2009). Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis †. Molecules, 14(9), 3275-3285. https://doi.org/10.3390/molecules14093275





