Synthesis of Novel 3,5-Dichloro-2-arylpyridines by Palladium Acetate-Catalyzed Ligand-Free Suzuki Reactions in Aqueous Media
Abstract
:Introduction
Results and Discussion
Conclusions
Experimental
General
Typical procedure for the preparation of 3,5-dichloro-2-arylpyridines 3
Typical Procedure for the preparation of 2-phenylpyridine (4)
Acknowledgement
References
- Pozharskii, A.F.; Soldatenkov, A.T.; Katrizky, A.R. Heterocycles in Life and Society; Wiley: New York, NY, USA, 1997. [Google Scholar]
- Gravett, E.C.; Hilton, P.J.; Jones, K.; Peron, J.M. A new route to 5-aryl and 5-heteroaryl-2-pyrones via Suzuki coupling of a 2-pyrone-5-boronate ester. Synlett 2003, 253–255. [Google Scholar] [CrossRef]
- Arterburn, J.B.; Bryant, B.K.; Chen, D.J. Synthesis of (pyridin-2-yl)hydrazine conjugates as bifunctional chelates using the Suzuki–Miyaura reaction. Chem. Commun. 2003, 1890–1891. [Google Scholar] [CrossRef]
- Cossy, J.; Belotti, D.; Magner, A. Synthesis of Indatraline Using a Suzuki cross-coupling reaction and a chemoselective hydrogenation: a versatile approach. Synlett 2003, 1515–1517. [Google Scholar] [CrossRef]
- Li, J.H.; Deng, C.L.; Xie, Y.X. Solvent-free palladium-catalyzed Suzuki-Miyaura cross-couplings of aryl chlorides with arylboronic acids. Synth. Commun. 2007, 37, 2433–2448. [Google Scholar] [CrossRef]
- Kume, T.; Goshima, T.; Kaji, S.; Yanagi, A.; Hyakawa, H. Perparation of 2,4,6-substituted pyridines and pyrimidines as herbicides. Japan Patent 04049279, 1992. [Google Scholar]
- Schafer, P.; Hamprecht, G.; Heistracher, E.; Menke, O.; Zagar, C.; Rack, M.; Goetz, N.; Harreus, A.; Westphalen, K.O.; Walter, H.; Mislitz, U. Substituted 2-phenylpyridines as herbicides. German Patent 19534466, 1997. [Google Scholar]
- Menke, O.; Menges, M.; Sagasser, I.; HamPrecht, G.; Reinhard, R.; Zagar, C.; Westphalen, K.O.; Otten, M.; Walter, H. Substituted 2-phenylpyridines as herbicides. WO Patent 2000042015, 2000. [Google Scholar]
- Bringmann, G.; Gunther, C.; Ochse, M.; Schupp, O.; Tasler, S. Progress in the Chemistry of Organic Natural Products; Herz, W., Falk, H., Kirby, G.W., Moore, R.E., Eds.; Springer: New York, NY, USA, 2001; Volume 82, pp. 1–293. [Google Scholar]
- Hegedus, L.S. Organometallics in Synthesis; Schlosser, M., Ed.; Wiley: Chichester, UK, 2002. [Google Scholar]
- Sutherland, A.; Gallagher, T. Versatile synthesis of 3,5-disubstituted 2-fluoropyridines and 2-pyridones. J. Org. Chem. 2003, 68, 3352–3355. [Google Scholar] [CrossRef] [PubMed]
- Diederich, F.; Stang, P.J. Metal-Catalyzed Cross-Coupling Reactions; Wiley-VCH: Weinheim, Germany, 1998. [Google Scholar]
- Miyaura, N. Cross-Coupling Reaction; Springer: Berlin, Germany, 2002. [Google Scholar]
- Khanapure, S.P.; Garvey, D.S. Use of highly reactive, versatile and air-stable palladium–phosphinous acid complex [(t-Bu)2P(OH)]2PdCl2 (POPd) as a catalyst for the optimized Suzuki–Miyaura cross-coupling of less reactive heteroaryl chlorides and arylboronic acids. Tetrahedron Lett. 2004, 45, 5283–5286. [Google Scholar] [CrossRef]
- Miyaura, N.; Suzuki, A. Palladium-catalyzed cross-coupling reactions of organoboron compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef]
- Tan, J.Q.; Chang, J.H.; Deng, M.Z. Coupling reaction of organoboronic acids with chloropyrimidines and trichlorotriazine. Chinese J. Chem. 2004, 9, 941–944. [Google Scholar] [CrossRef]
- Ohta, H.; Tokunaga, M.; Obora, Y.; Iwai, T.; Iwasawa, T.; Fujihara, T.; Tsuji, Y. A bowl-shaped phosphine as a ligand in palladium-catalyzed Suzuki−Miyaura coupling of aryl chlorides: Effect of the depth of the bowl. Org. Lett. 2007, 9, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Hassan, J.; Sevignon, M.; Schulz, E.; Lemaire, M. Aryl−aryl bond formation one century after the discovery of the Ullmann reaction. Chem. Rev. 2002, 102, 1359–1470. [Google Scholar] [CrossRef] [PubMed]
- Kotha, S.; Lahiri, K.; Kashinath, D. Recent applications of the Suzuki–Miyaura cross-coupling reaction in organic synthesis. Tetrahedron 2002, 58, 9633–9695. [Google Scholar] [CrossRef]
- Alonso, F.; Beletskaya, I.P.; Yus, M. Non-conventional methodologies for transition-metal catalysed carbon–carbon coupling: A critical overview. Part 2: The Suzuki reaction. Tetrahedron 2008, 64, 3047–3101. [Google Scholar] [CrossRef]
- Marinak, M.J.; Simonson, J.L. Process for producing 2,3,5-trichloropyridine. US Patent 4563531, 1986. [Google Scholar]
- Seifert, G.; Sting, A.R.; Urwyler, B. Process for preparation of propionic acid derivatives. US Patent 6175018, 2001. [Google Scholar]
- Goodson, F.E.; Wallow, T.I.; Novak, B.M. Accelerated Suzuki coupling via a ligandless palladium catalyst: 4-methoxy-2’-methylbiphenyl: (1,1’-biphenyl, 4’-methoxy-2-methyl-). Org. Synth. 1998, 75, 61–68. [Google Scholar]
- Liu, L.F.; Zhang, Y.H.; Xin, B.W. Synthesis of biaryls and polyaryls by ligand-free Suzuki reaction in aqueous phase. J. Org. Chem. 2006, 71, 3994–3997. [Google Scholar] [CrossRef] [PubMed]
- Barder, T.E.; Walker, S.D.; Martinelli, J.R.; Buchwald, S.L. Catalysts for Suzuki−Miyaura coupling processes: Scope and studies of the effect of ligand structure. J. Am. Chem. Soc. 2005, 127, 4685–4696. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 3a-j are available from the authors. |
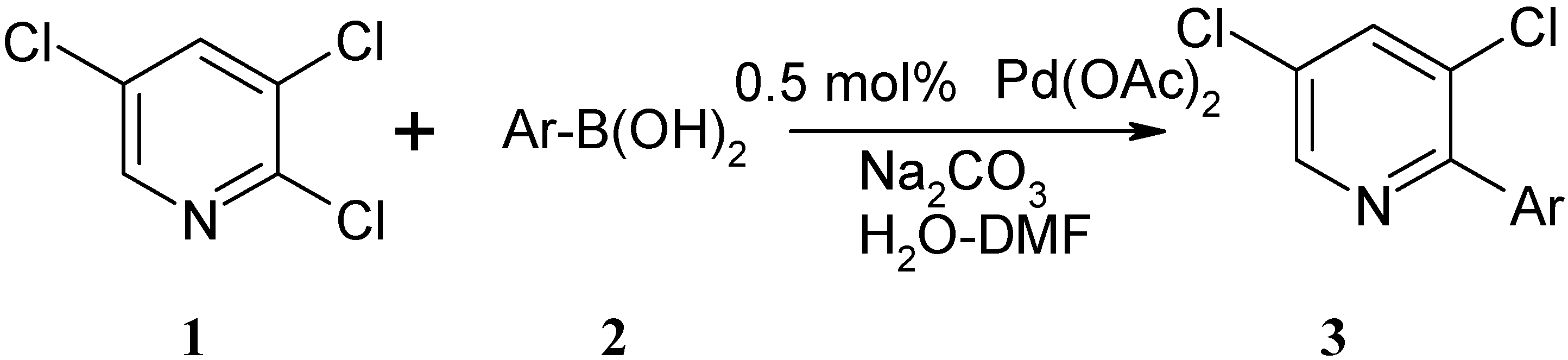
| Entry | Solvent (3:3.5 mL) | Yieldb (%) |
|---|---|---|
| 1 | acetone/H2O | 81 |
| 2 | ethanol/H2O | 80 |
| 3 | propanol/H2O | 79 |
| 4 | DMF/H2O | 83 |
| 5 | DCM/H2O | 29 |
| 6 | THF/H2O | 34 |
| 7 | Toluene/H2O | 21 |
| 8c | DMF/H2O | 89 |
| Time/h | 4 | 6 | 8 | 10 | 12 | 14 |
| Yieldb/% | 55 | 61 | 79 | 85 | 89 | 87 |
| Entry | Arylboronic acid | Product(3) | Yieldb (%) |
|---|---|---|---|
| 1 | 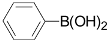 | 3a | 89 |
| 2 | 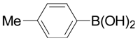 | 3b | 90 |
| 3 |  | 3c | 87 |
| 4 | 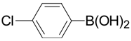 | 3d | 91 |
| 5 | 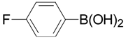 | 3e | 89 |
| 6 | 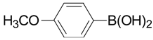 | 3f | 85 |
| 7 | 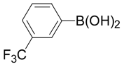 | 3g | 88 |
| 8 |  | 3h | 82 |
| 9 | 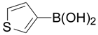 | 3i | 86 |
| 10 | 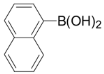 | 3j | 89 |
| Entry | chloropyridine | phenylboronic acid | Product | Yieldb(%) |
|---|---|---|---|---|
| 1 | 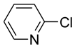 | 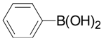 | 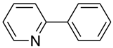 4 | 76 |
| 2 | 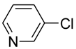 | 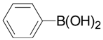 | 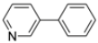 5 | trace |
| 3 | 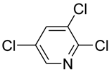 |  | 3a | 89 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, H.; Ge, C.; Zhang, A.; Ding, L. Synthesis of Novel 3,5-Dichloro-2-arylpyridines by Palladium Acetate-Catalyzed Ligand-Free Suzuki Reactions in Aqueous Media. Molecules 2009, 14, 3153-3160. https://doi.org/10.3390/molecules14093153
Hu H, Ge C, Zhang A, Ding L. Synthesis of Novel 3,5-Dichloro-2-arylpyridines by Palladium Acetate-Catalyzed Ligand-Free Suzuki Reactions in Aqueous Media. Molecules. 2009; 14(9):3153-3160. https://doi.org/10.3390/molecules14093153
Chicago/Turabian StyleHu, Huanan, Changhua Ge, Anjiang Zhang, and Lisheng Ding. 2009. "Synthesis of Novel 3,5-Dichloro-2-arylpyridines by Palladium Acetate-Catalyzed Ligand-Free Suzuki Reactions in Aqueous Media" Molecules 14, no. 9: 3153-3160. https://doi.org/10.3390/molecules14093153
APA StyleHu, H., Ge, C., Zhang, A., & Ding, L. (2009). Synthesis of Novel 3,5-Dichloro-2-arylpyridines by Palladium Acetate-Catalyzed Ligand-Free Suzuki Reactions in Aqueous Media. Molecules, 14(9), 3153-3160. https://doi.org/10.3390/molecules14093153





