2β-(Isobutyryloxy)florilenalin, a Sesquiterpene Lactone Isolated from the Medicinal Plant Centipeda minima, Induces Apoptosis in Human Nasopharyngeal Carcinoma CNE Cells
Abstract
:1. Introduction
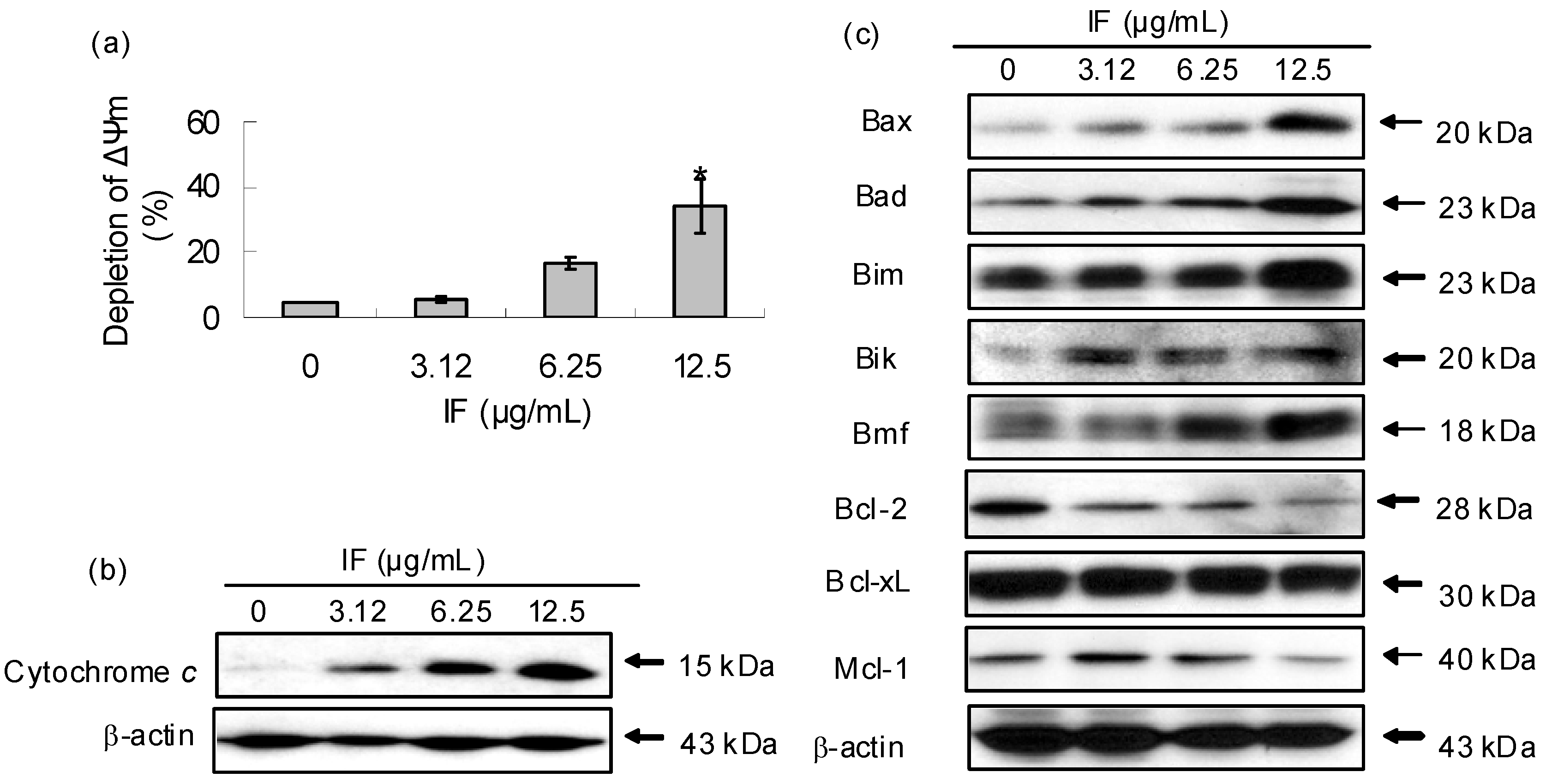
2. Results and Discussion
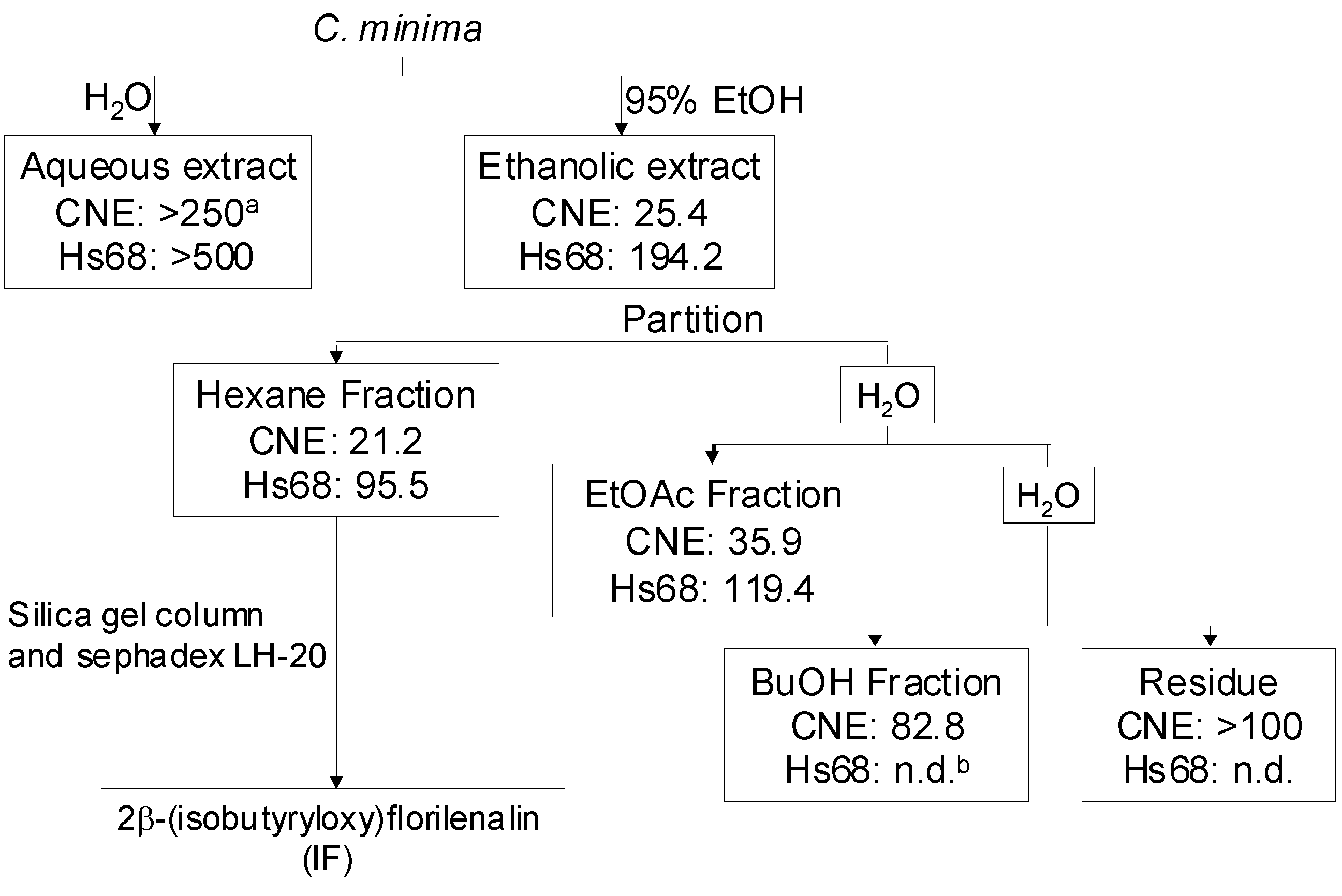
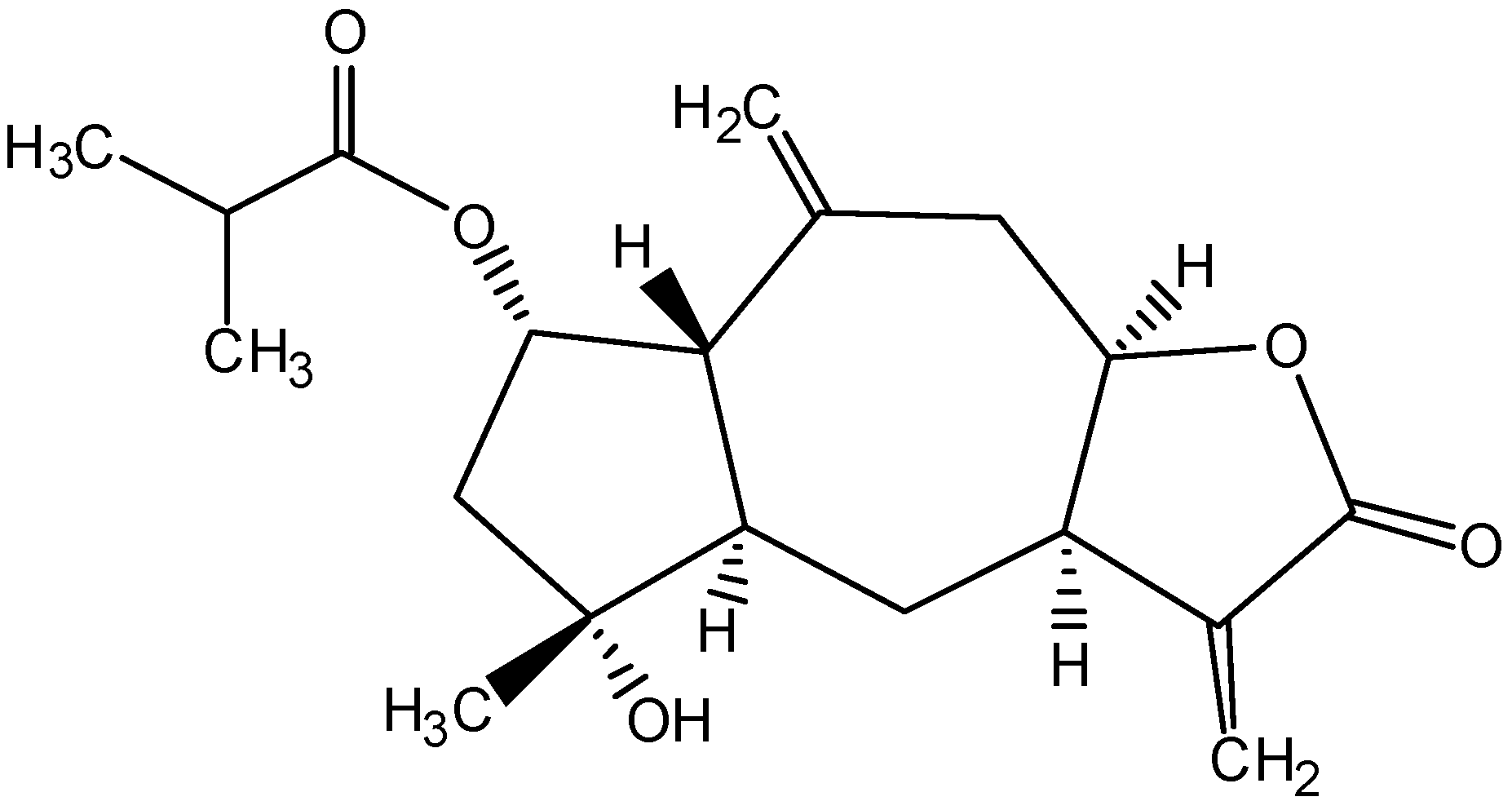
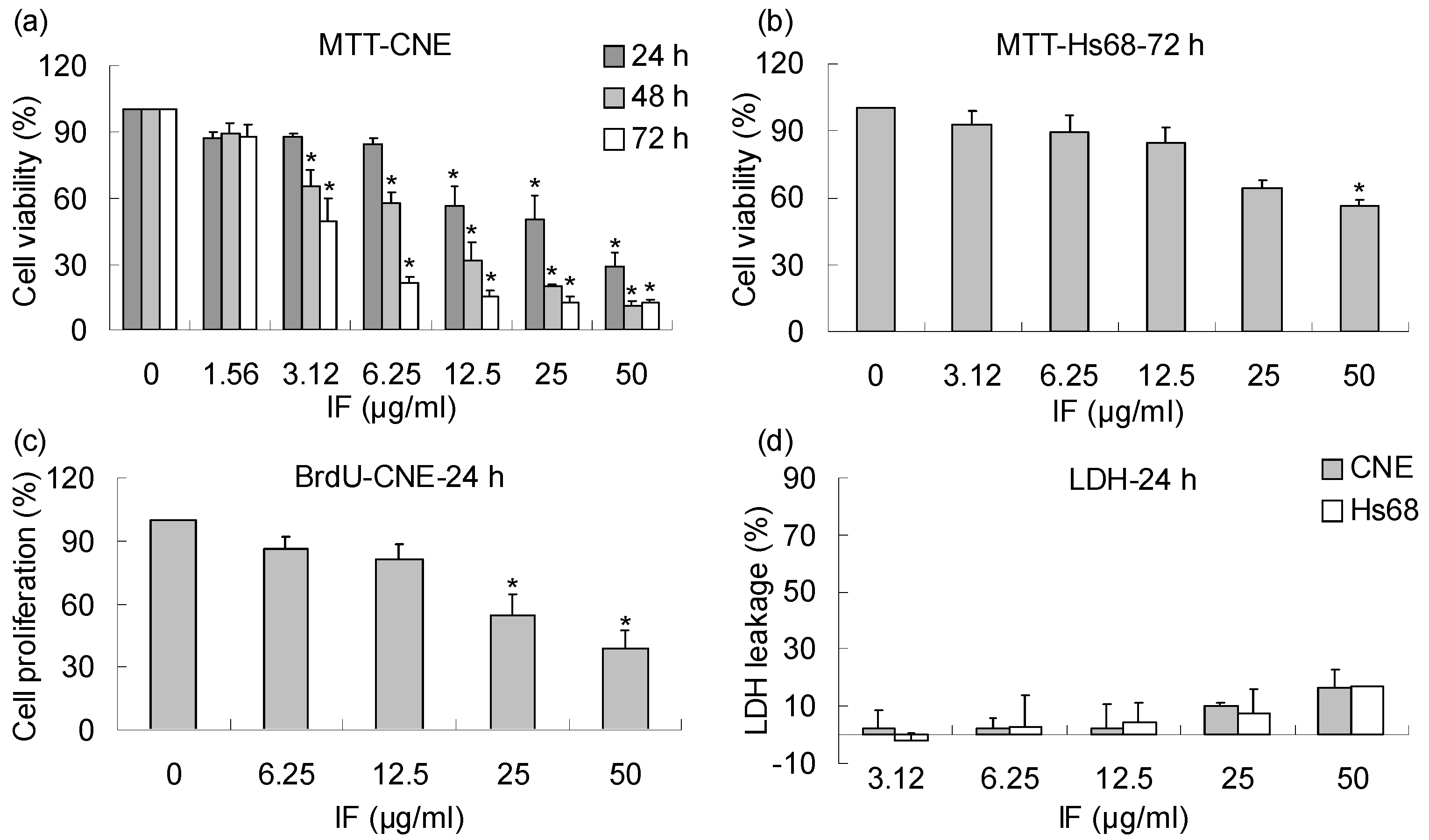

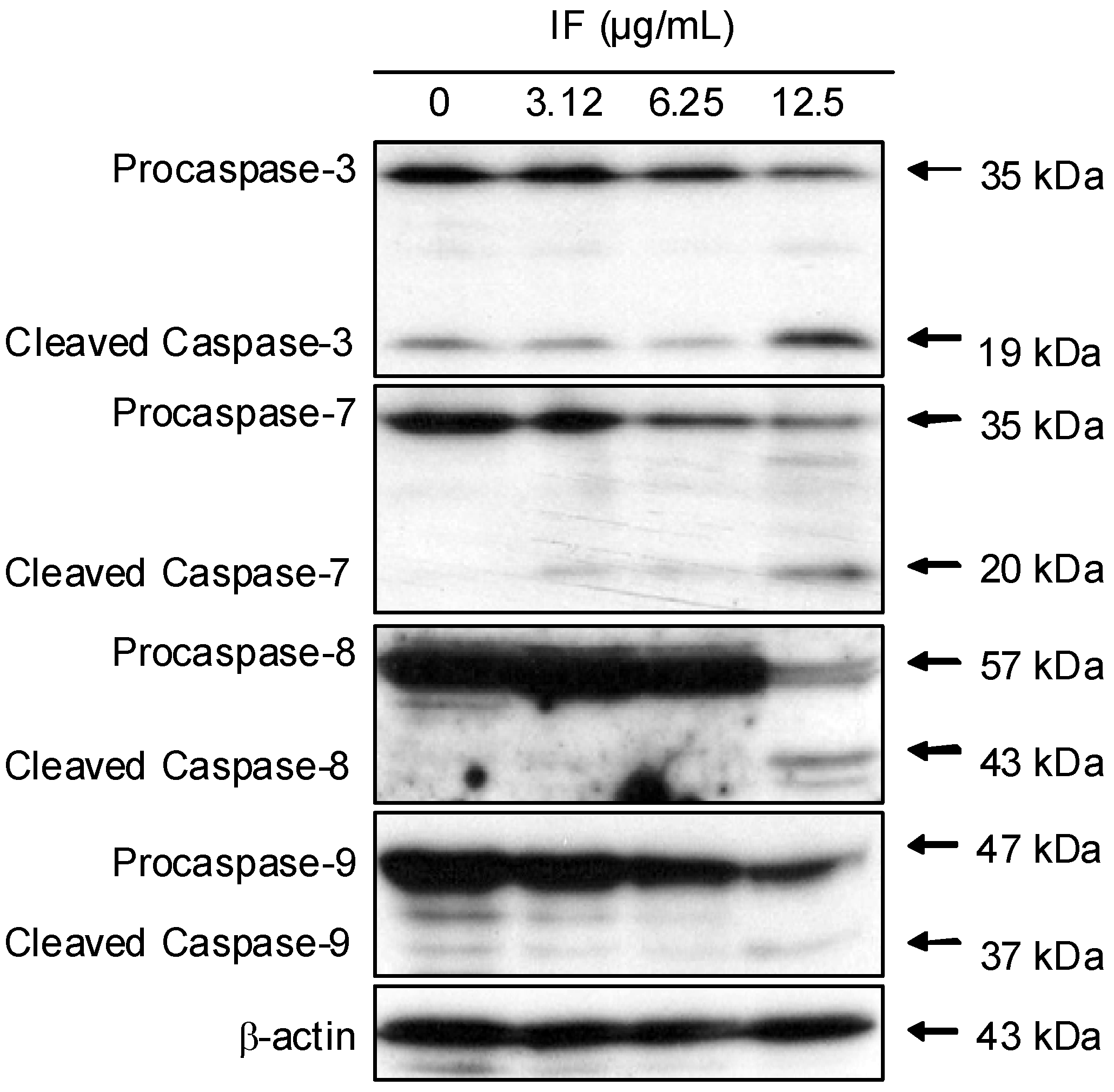
3. Conclusions
4. Experimental
4.1. General
4.2. Extraction, fractionation and isolation guided by in vitro cell viability assay
4.3. Cell culture
4.4. Cell viability analysis by MTT assay
4.5. Cell proliferation analysis by BrdU assay
4.6. Cytotoxicity determination by lactate dehydrogenase (LDH) assay
4.7. DNA contents analysis by flow cytometry
4.8. Nuclear staining with DAPI
4.9. Mitochondrial membrane potential (ΔΨm) assay
4.10. Caspase activity assay
4.11. Western blot analysis
4.12. Statistical analysis
Acknowledgements
References
- Shi, Z.; Fu, G.X. Flora of China; Science Publishing House: Beijing, China, 1983; Vol. 76, no. 1; pp. 132–133. [Google Scholar] [Green Version]
- Lin, R.; Shi, Z. Pharmacopoeia of China; Science Publishing House: Beijing, China, 2005; p. 132. [Google Scholar] [Green Version]
- Cheng, J.H.; Li, Y.B. Anti-tumor Herbal Medicines and Their Proved Recipes; Jiangxi Science & Technology Press: Jiangxi, China, 1998; p. 732. [Google Scholar] [Green Version]
- Zhang, Y.H. A Collection of Anticancer Chinese Medicines; Jiangsu Science and Technology Publishing House: Nanjing, China, 2000; p. 435. [Google Scholar] [Green Version]
- Wu, J.B.; Chun, T.T.; Ebizuka, Y.; Sankawa, U. Biologically active constituents of Centipeda minima: Isolation of a new plenolin ester and the anti-allergy activity of sesquiterpene lactones. Chem. Pharm. Bull. 1985, 33, 4091–4094. [Google Scholar] [CrossRef]
- Wu, J.B.; Chun, T.T.; Ebizuka, Y.; Sankawa, U. Biologically active constituents of Centipeda minima: Sesquiterpenes of potential anti-allergy activity. Chem. Pharm. Bull. 1991, 39, 3272–3275. [Google Scholar] [CrossRef]
- Taylor, R.S.L.; Towers, G.H.N. Antibacterial constituents of the Nepalese medicinal herb, Centipeda minima. Phytochemistry 1998, 47, 631–634. [Google Scholar] [CrossRef]
- Liang, H.X.; Bao, F.K.; Dong, X.P.; Zhu, H.J.; Lu, X.J.; Shi, M.; Lu, Q.; Cheng, Y.X. Two new antibacterial sesquiterpenoids from Centipeda minima. Chem. Biodivers. 2007, 4, 2810–2816. [Google Scholar] [CrossRef]
- Bohlmann, F.; Chen, Z.L. New guaianolides from Centipeda minima. Chin. Sci. Bull. 1984, 29, 900–903. [Google Scholar]
- Zhang, S.; Won, Y.K.; Ong, C.N.; Shen, H.M. Anti-cancer potential of sesquiterpene lactones: bioactivity and molecular mechanisms. Curr. Med. Chem. Anti-Cancer Agents 2005, 5, 239–249. [Google Scholar] [CrossRef]
- Li, C.L.; Wu, H.Z.; Huang, Y.P.; Yang, Y.F.; Liu, Y.W.; Liu, J.W. 6-O-Angeloylenolin induces apoptosis through a mitochondrial/caspase and NF-κB pathway in human leukemia HL60 cells. Biomed. Pharmacother. 2008, 62, 401–409. [Google Scholar] [CrossRef]
- Beekman, A.C.; Woerdenbag, H.J.; van Uden, W.; Pras, N.; Konings, A.W.; Wikstrom, H.V.; Schmidt, T.J. Structure-cytotoxicity relationships of some helenanolide-type sesquiterpene lactones. J. Nat. Prod. 1997, 60, 252–257. [Google Scholar] [CrossRef]
- Yu, H.W.; Wright, C.W.; Cai, Y.; Yang, S.L.; Phillipson, J.D.; Kirby, G.C.; Warhurst, D.C. Antiprotozoal activities of Centipeda minima. Phytother. Res. 1994, 8, 436–438. [Google Scholar] [CrossRef]
- Riedl, S.J.; Shi, Y. Molecular mechanisms of caspase regulation during apoptosis. Nat. Rev. Mol. Cell Biol. 2004, 5, 897–907. [Google Scholar] [CrossRef]
- Kim, R.; Emi, M.; Tanabe, K. Role of mitochondria as the gardens of cell death. Cancer Chemother. Pharmacol. 2006, 57, 545–553. [Google Scholar] [CrossRef]
- Mattson, M.P.; Kroemer, G. Mitochondria in cell death: novel targets for neuroprotection and cardioprotection. Trends Mol. Med. 2003, 9, 196–205. [Google Scholar] [CrossRef]
- Burlacu, A. Regulation of apoptosis by Bcl-2 family proteins. J. Cell. Mol. Med. 2003, 7, 249–257. [Google Scholar] [CrossRef]
- Borner, C. The Bcl-2 protein family: sensors and checkpoints for life-or-death decisions. Mol. Immunol. 2003, 39, 615–647. [Google Scholar] [CrossRef]
- Lu, Q.L.; Elia, G.; Lucas, S.; Thomas, J.A. Bcl-2 proto-oncogene expression in Epstein-Barr-virus-associated nasopharyngeal carcinoma. Int. J. Cancer 1993, 53, 29–35. [Google Scholar] [CrossRef]
- Fan, S.Q.; Ma, J.; Zhou, J.; Xiong, W.; Xiao, B.Y.; Zhang, W.L.; Tan, C.; Li, X.L.; Shen, S.R.; Zhou, M.; Zhang, Q.H.; Ou, Y.J.; Zhuo, H.D.; Fan, S.; Zhou, Y.H.; Li, G.Y. Differential expression of Epstein-Barr virus-encoded RNA and several tumor-related genes in various types of nasopharyngeal epithelial lesions and nasopharyngeal carcinoma using tissue microarray analysis. Hum. Pathol. 2006, 37, 593–60. [Google Scholar] [CrossRef]
- Sample Availability: Samples are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Su, M.; Li, Y.; Chung, H.Y.; Ye, W. 2β-(Isobutyryloxy)florilenalin, a Sesquiterpene Lactone Isolated from the Medicinal Plant Centipeda minima, Induces Apoptosis in Human Nasopharyngeal Carcinoma CNE Cells. Molecules 2009, 14, 2135-2146. https://doi.org/10.3390/molecules14062135
Su M, Li Y, Chung HY, Ye W. 2β-(Isobutyryloxy)florilenalin, a Sesquiterpene Lactone Isolated from the Medicinal Plant Centipeda minima, Induces Apoptosis in Human Nasopharyngeal Carcinoma CNE Cells. Molecules. 2009; 14(6):2135-2146. https://doi.org/10.3390/molecules14062135
Chicago/Turabian StyleSu, Miaoxian, Yaolan Li, Hau Yin Chung, and Wencai Ye. 2009. "2β-(Isobutyryloxy)florilenalin, a Sesquiterpene Lactone Isolated from the Medicinal Plant Centipeda minima, Induces Apoptosis in Human Nasopharyngeal Carcinoma CNE Cells" Molecules 14, no. 6: 2135-2146. https://doi.org/10.3390/molecules14062135
APA StyleSu, M., Li, Y., Chung, H. Y., & Ye, W. (2009). 2β-(Isobutyryloxy)florilenalin, a Sesquiterpene Lactone Isolated from the Medicinal Plant Centipeda minima, Induces Apoptosis in Human Nasopharyngeal Carcinoma CNE Cells. Molecules, 14(6), 2135-2146. https://doi.org/10.3390/molecules14062135




