Chemical Constituents from the Stems of Diospyros maritima
Abstract
1. Introduction
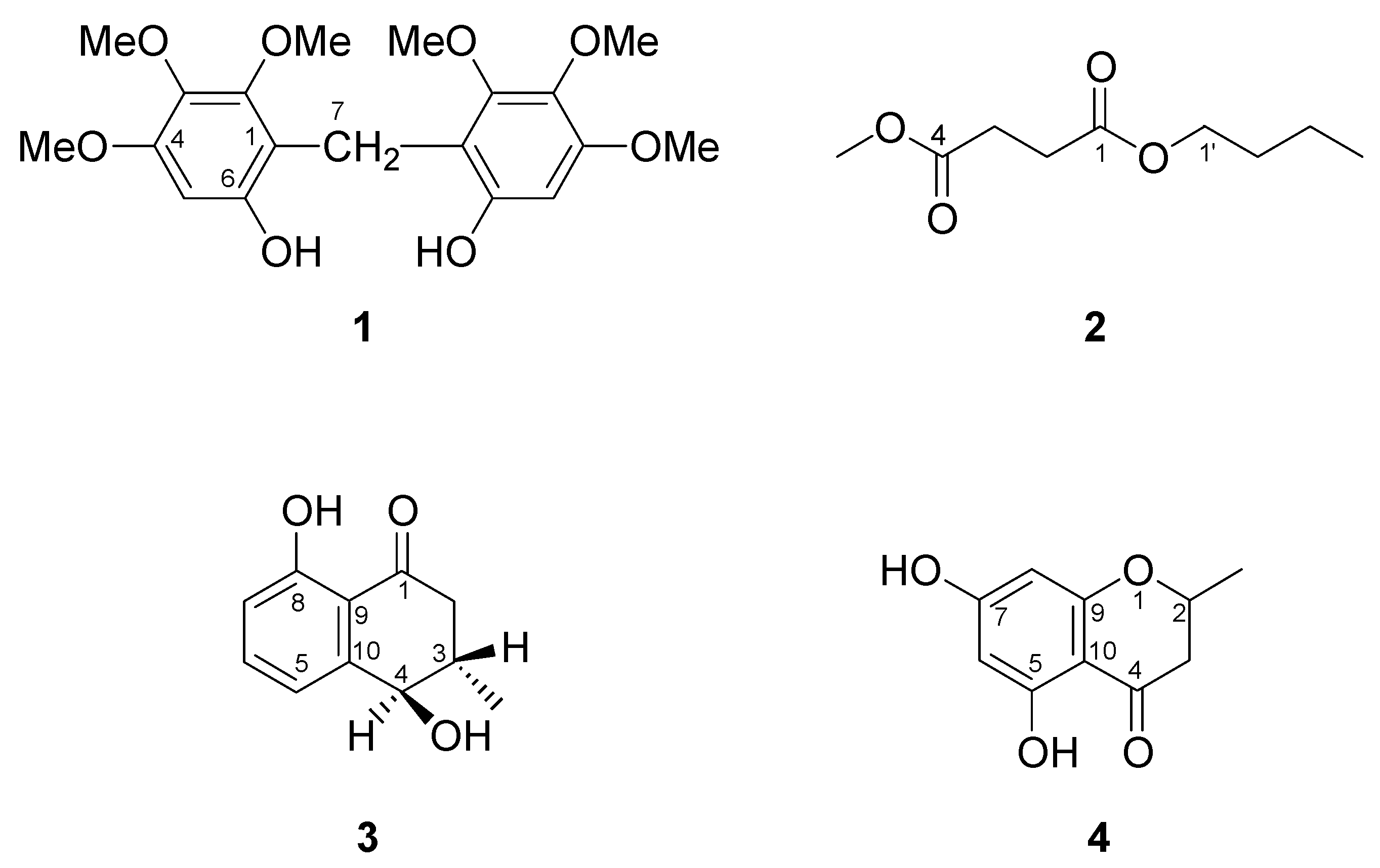
2. Results and Discussion
3. Experimental
3.1. General
3.2. Plant material
3.3. Extraction and isolation
3.4. Spectroscopic data
4. Conclusions
Acknowledgements
References and Notes
- Higa, M.; Ogihara, K.; Yogi, S. Bioactive naphthoquinone derivatives from Diospyros maritima Blume. Chem. Pharm. Bull. 1998, 46, 1189–1193. [Google Scholar] [CrossRef]
- Higa, M.; Noha, N.; Yokaryo, H.; Ogihara, K.; Yogi, S. Three new naphthoquinone derivatives from Diospyros maritima BLUME. Chem. Pharm. Bull. 2002, 50, 590–593. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Chang, C.I; Li, S.Y.; Chou, C.J.; Chen, C.F.; Kuo, Y.H.; Lee, K.H. Cytotoxic constituents from the stems of Diospyros maritima. Planta Med. 1997, 63, 363–364. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.Q.; Graf, T.N.; Lee, D.; Chai, H.B.; Mi, Q.; Kardono, L.B.; Setyowati, F.M.; Ismail, R.; Riswan, S.; Farnsworth, N.R.; Cordell, G.A.; Pezzuto, J.M.; Swanson, S.M.; Kroll, D.J.; Falkinham, J.O., 3rd; Wall, M.E.; Wani, M.C.; Kinghorn, A.D.; Oberlies, N.H. Cytotoxic and antimicrobial constituents of the bark of Diospyros maritima collected in two geographical locations in Indonesia. J. Nat. Prod. 2004, 67, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- Kan, W.S. Pharmaceutical Botany; National Research Institute of Chinese Medicine: Taipei, Taiwan, 1997; p. 440. [Google Scholar]
- Tezuka, M.; Takahashi, C.; Kuroyanagi, M.; Satake, M.; Yoshihira, K.; Natori, S. New naphthoquinones from Diospyros. Phytochemistry 1973, 12, 175–183. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Chang, C.I.; Kuo, Y.H. New naphthoquinones from the stem of Diospyros maritima Blume. J. Chin Chem. Soc. 1996, 43, 511–514. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Chang, C.I.; Kuo, Y.H.; Huang, S.L. Three new naphthoquinones from the stem of Diospyros maritima Blume. J. Chin. Chem. Soc. 1998, 45, 111–114. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Chang, C.I.; Kuo, Y.H. Triterpenes from the stem of Diospyros maritima Blume. Phytochemistry 1997, 46, 1135–1137. [Google Scholar]
- Kuo, Y.H.; Chang, C.I.; Kuo, Y.H. A novel trisnorlupane, diospyrolide, from Diospyros maritima. Chem. Pharm. Bull. 1997, 45, 1221–1222. [Google Scholar] [CrossRef]
- Chang, C.I.; Kuo, Y.H. Three new lupane-type triterpenes from Diospyros maritima. Chem. Pharm. Bull. 1998, 46, 1627–1629. [Google Scholar] [CrossRef][Green Version]
- Chang, C.I.; Kuo, Y.H. Two new lupane-type triterpenes from Diospyros maritima. J. Nat. Prod. 1999, 62, 309–310. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Chang, C.I. Six new compounds from the heartwood of Diospyros maritima. Chem. Pharm. Bull. 2000, 48, 1211–1214. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kuo, Y.H.; Huang, S.L.; Chang, C.I. A phenolic and an aliphatic lactone from the Diospyros maritima. Phytochemistry 1998, 49, 2505–2507. [Google Scholar] [CrossRef]
- Bhattacharyya, J.; De Carvalho, V.R. epi-Isoshinanolone from Plumbago scandens. Phytochemistry 1986, 25, 764–765. [Google Scholar] [CrossRef]
- Chandler, I.M.; Mclntyre, C.R.; Simpson, T.J. Structural revision and synthesis of ll-d253 and related chromanone fungal metabolites. J. Chem. Soc. Perkin Trans. 1 1992, 2271–2284. [Google Scholar] [CrossRef]
- Anderson, J.R.; Edwards, R.L.; Whalley, A.J.S. 3-Methyl-3,4-dihydroisocoumarins and related compounds from the ascomycete family Xylariaceae. J. Chem. Soc. Perkin Trans. 1 1983, 2185–2192. [Google Scholar] [CrossRef]
- Auricchio, S.; Morrocchi, S.; Ricca, A. Phenol compounds from isoxazoles, considered as masked β-polyketones. Tetrahedron Lett. 1974, 15, 2793–2796. [Google Scholar] [CrossRef]
- Gramatica, P.; Gianotti, M.P.; Speranra, G.; Manitto, P. Synthesis of naturally occurring 2,5-dialkylchromones. Part 1: Synthesis of aloesone and aloesol. Heterocycles 1986, 24, 743–750. [Google Scholar]
- Lee, C.K.; Chang, M.H. The chemical constituents from the heartwood of Eucalyptus citrodora. J. Chin. Chem. Soc. 2000, 47, 555–560. [Google Scholar] [CrossRef]
- Ito, J.; Chang, F.R.; Wang, H.K.; Park, Y.K.; Ikegaki, M.; Kilgore, N.; Lee, K.H. Anti-AIDS agents. 48. anti-HIV activity of moronic acid derivatives and the new melliferone-related triterpenoid isolated from Brazilian Propolis. J. Nat. Prod. 2001, 64, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Crestini, C.; D’Auria, M. Singlet oxygen in the photodegradation of lignin models. Tetrahedron 1997, 53, 7877–7888. [Google Scholar] [CrossRef]
- Carpinella, M.C.; Giorda, L.M.; Ferrayoli, C.G.; Palacios, S.M. Antifungal effects of different organic extracts from Melia azedarach L. on phytopathogenic fungi and their isolated active components. J. Agric. Food Chem. 2003, 51, 2506–2511. [Google Scholar] [CrossRef] [PubMed]
- Daubresse, N.; Francesch, C.; Mhamdi, F.; Rolando, C. A mild synthesis of coumaryl, coniferyl, sinapyl aldehydes and alcohols. Synthesis 1994, 4, 369–371. [Google Scholar] [CrossRef]
- Otsuka, H.; Takeuchi, M.; Inoshiri, S.; Sato, T.; Yamasaki, K. Phenolic compounds from Coix lachrymal-jobi var. ma-yuen. Phytochemistry 1989, 28, 883–886. [Google Scholar] [CrossRef]
- Fuchino, H.; Saton, T.; Tanaka, N. Chemical evaluation of Betula species in Japan. I. constituents of Betula ermanii. Chem. Pharm. Bull. 1995, 43, 1937–1942. [Google Scholar] [CrossRef]
- Saraswathy, A.; Sasikala, E. betulin-28-acetate from Capparis sepiaria L. J. Indian Chem. Soc. 1991, 68, 633–634. [Google Scholar]
- Rashid, M.A.; Gray, A.I.; Waterman, P.G.; Armstrong, J.A. Coumarins from Phebalium tuberculosum ssp. megaphyllum and Phebalium filifolium. J. Nat. Prod. 1992, 55, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.M.; Waterman, P.G.; Jeffreys, J.A.D. Naphthoquinones and triterpenes from African Diospyros species. Phytochemistry 1984, 23, 1067–1072. [Google Scholar] [CrossRef]
- Kuo, Y.H.; Chu, P.H. Studies on the constituents from the bark of Bauhinia purpurea. J. Chin. Chem. Soc. 2002, 49, 269–274. [Google Scholar] [CrossRef]
- Kundu, A.B.; Barik, B.R.; Mondal, D.N.; Dey, A.K.; Banerji, A. Zizyberanalic acid, a pentacyclic triterpenoid of Zizyphus jujuba. Phytochemistry 1989, 28, 3155–3158. [Google Scholar] [CrossRef]
- Barba, B.; Diaz, J.G.; Herz, W. Cassanes and anthraquinones from Chamaecrista greggii. Phytochemistry 1994, 37, 837–845. [Google Scholar] [CrossRef]
- Ahman, V.U.; Basha, A.; Rahman, A. Identification and C-13 N.M.R. spectrum of stachydrine from Cadaba fruticosa. Phytochemistry 1975, 14, 292–293. [Google Scholar] [CrossRef]
- Majumder, P.L; Bagchi, A. Oxidative transformations of triterpenoids of the ursane and oleanane skeleta with hydrogen peroxide. introduction of 11,12-double bond and 13(28)oxide moiety in the ursane system. Tetrahedron 1983, 39, 649–655. [Google Scholar] [CrossRef]
- Elujoba, A.A; Fell, A.F.; Linley, P.A.; Maitland, D.J. Triterpenoid saponins from fruit of Lagenaria breviflora. Phytochemistry 1990, 29, 3281–3285. [Google Scholar] [CrossRef]
- Tkachev, A.V.; Denisov, A.Y. Oxidative decarboxylation by hydrogen peroxide and a mercury (II) salt: A simple route to nor-derivatives of acetyloleanolic, acetylursolic and dehydroabietic acids. Tetrahedron 1994, 50, 2591–2598. [Google Scholar] [CrossRef]
- Tkachev, A.V.; Denisov, A.Y.; Gatilov, Y.V.; Bagryanskaya, I.Y.; Shevtsov, S.A.; Rybalova, T.V. Stereochemistry of hydrogen peroxide-acetic acid oxidation of ursolic acid and related compounds. Tetrahedron 1994, 50, 11459–11488. [Google Scholar] [CrossRef]
- Errington, S.G.; Jefferies, P.R. Triterpenoid sapogenins of Pittosporum phillyraeoides. Phytochemistry 1988, 27, 543–545. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| position | δC | δH | position | δC | δH |
| 1, 1’ | 110.4 | 7 | 18.3 | 3.67 s | |
| 2, 2’ | 149.5 | 2, 2’ -OCH3 | 62.1 | 4.07 s | |
| 3, 3’ | 135.0 | 3, 3’ -OCH3 | 61.1 | 3.75 s | |
| 4, 4’ | 152.8 | 4, 4’ -OCH3 | 55.9 | 3.77 s | |
| 5, 5’ | 97.7 | 6.30 s | 6, 6’ -OH | 8.10 s | |
| 6, 6’ | 151.6 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, C.-I.; Chen, C.-R.; Chiu, H.-L.; Kuo, C.-L.; Kuo, Y.-H. Chemical Constituents from the Stems of Diospyros maritima. Molecules 2009, 14, 5281-5288. https://doi.org/10.3390/molecules14125281
Chang C-I, Chen C-R, Chiu H-L, Kuo C-L, Kuo Y-H. Chemical Constituents from the Stems of Diospyros maritima. Molecules. 2009; 14(12):5281-5288. https://doi.org/10.3390/molecules14125281
Chicago/Turabian StyleChang, Chi-I, Chiy-Rong Chen, Hsi-Lin Chiu, Chao-Lin Kuo, and Yueh-Hsiung Kuo. 2009. "Chemical Constituents from the Stems of Diospyros maritima" Molecules 14, no. 12: 5281-5288. https://doi.org/10.3390/molecules14125281
APA StyleChang, C.-I., Chen, C.-R., Chiu, H.-L., Kuo, C.-L., & Kuo, Y.-H. (2009). Chemical Constituents from the Stems of Diospyros maritima. Molecules, 14(12), 5281-5288. https://doi.org/10.3390/molecules14125281




