A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood
Abstract
:1. Introduction
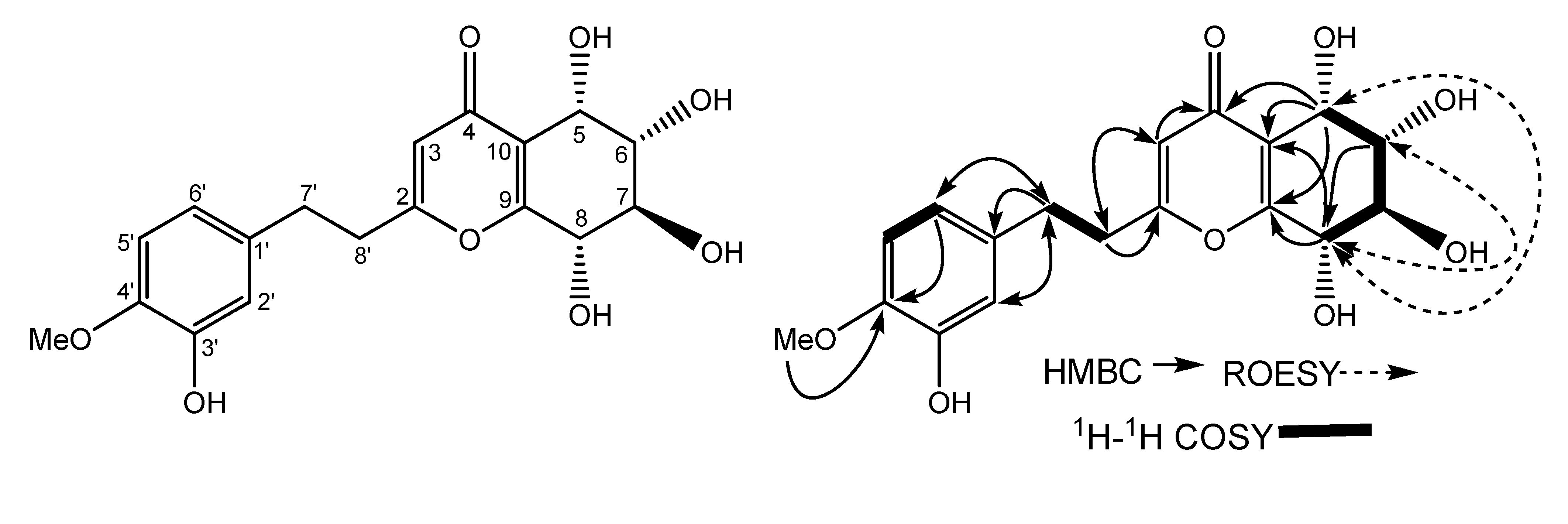
2. Results and Discussion
| Position | δH | δC | Position | δH | δC |
|---|---|---|---|---|---|
| 2 | 168.0 | 1' | 132.6 | ||
| 3 | 6.07 (1H, s) | 112.7 | 2' | 6.67 (1H, d, 1.9) | 115.6 |
| 4 | 178.5 | 3' | 146.0 | ||
| 5 | 4.48 (1H, d, 4.2) | 64.7 | 4' | 146.3 | |
| 6 | 3.74 (1H, dd, 6.9, 4.2 ) | 72.7 | 5' | 6.80 (1H, d, 8.0) | 112.2 |
| 7 | 3.83 (1H, t, 6.9) | 70.6 | 6' | 6.60 (1H, dd, 8.0, 1.9) | 118.7 |
| 8 | 4.31 (1H, d, 6.9) | 68.4 | 7' | 2.80 (2H, overlapped) | 31.1 |
| 9 | 163.1 | 8' | 2.80 (2H, overlapped) | 34.3 | |
| 10 | 120.7 | 4'-OCH3 | 3.71 (3H, s) | 55.6 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
4. Conclusions
Acknowledgements
References and Notes
- Institute of Materia Medica of Chinese Academy of Medical Sciences. Modern Research on Chinese Herbal Medicine, 3; Peking Medical College and Peking Union Medical College Associated Press: Beijing, China, 1997; p. 1. [Google Scholar]
- Dai, H.F.; Mei, W.L. Modern Research on Medicinal Plants in Hainan; China Science and Technology Press: Beijing, China, 2007; p. 31. [Google Scholar]
- Mei, W.L.; Zeng, Y.B.; Liu, J.; Dai, H.F. GC-MS analysis of volatile constituents from five different kinds of Chinese eaglewood. J. Chin. Med. Mat. 2007, 30, 551–555. [Google Scholar]
- Mei, W.L.; Zeng, Y.B.; Wu, J.; Cui, H.B.; Dai, H.F. Chemical composition and anti-MRSA activity of the essential oil from Chinese eaglewood. J. Chin. Pharm. Sci. 2008, 17, 225–229. [Google Scholar]
- Liu, J.; Wu, J.; Zhao, Y.X.; Deng, Y.Y.; Mei, W.L.; Dai, H.F. A new cytotoxic 2-(2-phenylethyl)chromone from Chinese eaglewood. Chin. Chem. Lett. 2008, 19, 934–936. [Google Scholar] [CrossRef]
- Yagura, T.; Shibayama, N.; Ito, M.; Kiuchi, F.; Honda, G. Three novel diepoxy tetrahydrochromones from agarwood artificially produced by intentional wounding. Tetrahedron Lett. 2005, 46, 4395–4398. [Google Scholar] [CrossRef]
- Yagura, T.; Ito, M.; Kiuchi, F.; Honda, G.; Shimada, Y. Four new 2-(2-phenylethyl)chromone derivatives from withered wood of Aquilaria sinensis. Chem. Pharm. Bull. 2003, 51, 560–564. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compound 1 are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dai, H.-F.; Liu, J.; Zeng, Y.-B.; Han, Z.; Wang, H.; Mei, W.-L. A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood. Molecules 2009, 14, 5165-5168. https://doi.org/10.3390/molecules14125165
Dai H-F, Liu J, Zeng Y-B, Han Z, Wang H, Mei W-L. A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood. Molecules. 2009; 14(12):5165-5168. https://doi.org/10.3390/molecules14125165
Chicago/Turabian StyleDai, Hao-Fu, Jun Liu, Yan-Bo Zeng, Zhuang Han, Hui Wang, and Wen-Li Mei. 2009. "A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood" Molecules 14, no. 12: 5165-5168. https://doi.org/10.3390/molecules14125165
APA StyleDai, H.-F., Liu, J., Zeng, Y.-B., Han, Z., Wang, H., & Mei, W.-L. (2009). A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood. Molecules, 14(12), 5165-5168. https://doi.org/10.3390/molecules14125165





