New secoiridoid Glycosides from the Roots of Picrorhiza Scrophulariiflora
Abstract
:Introduction
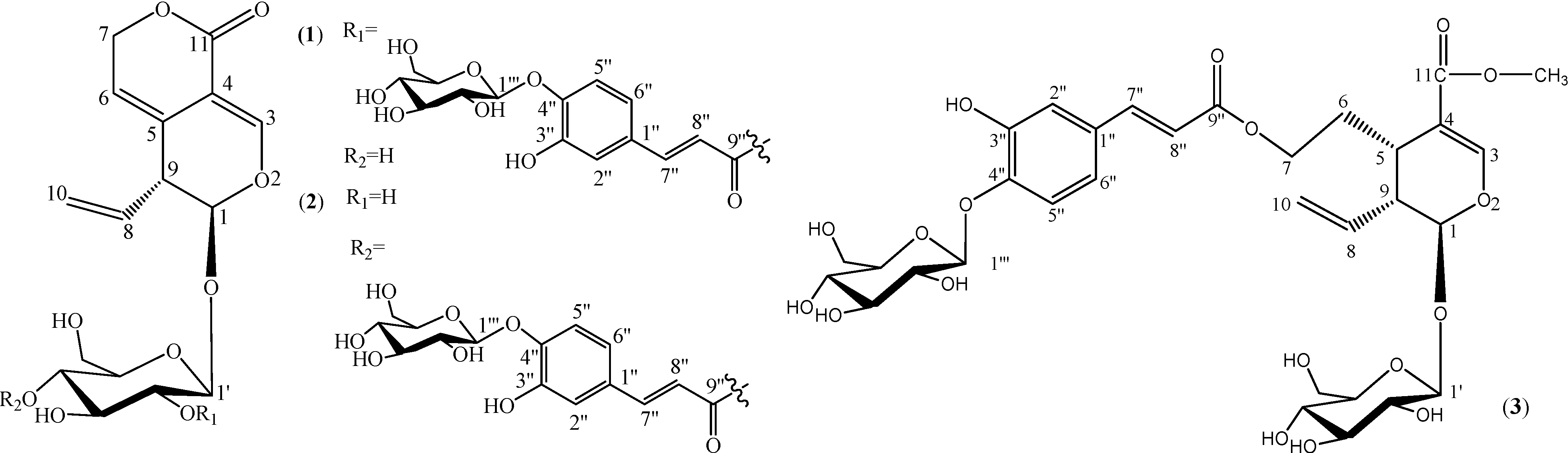
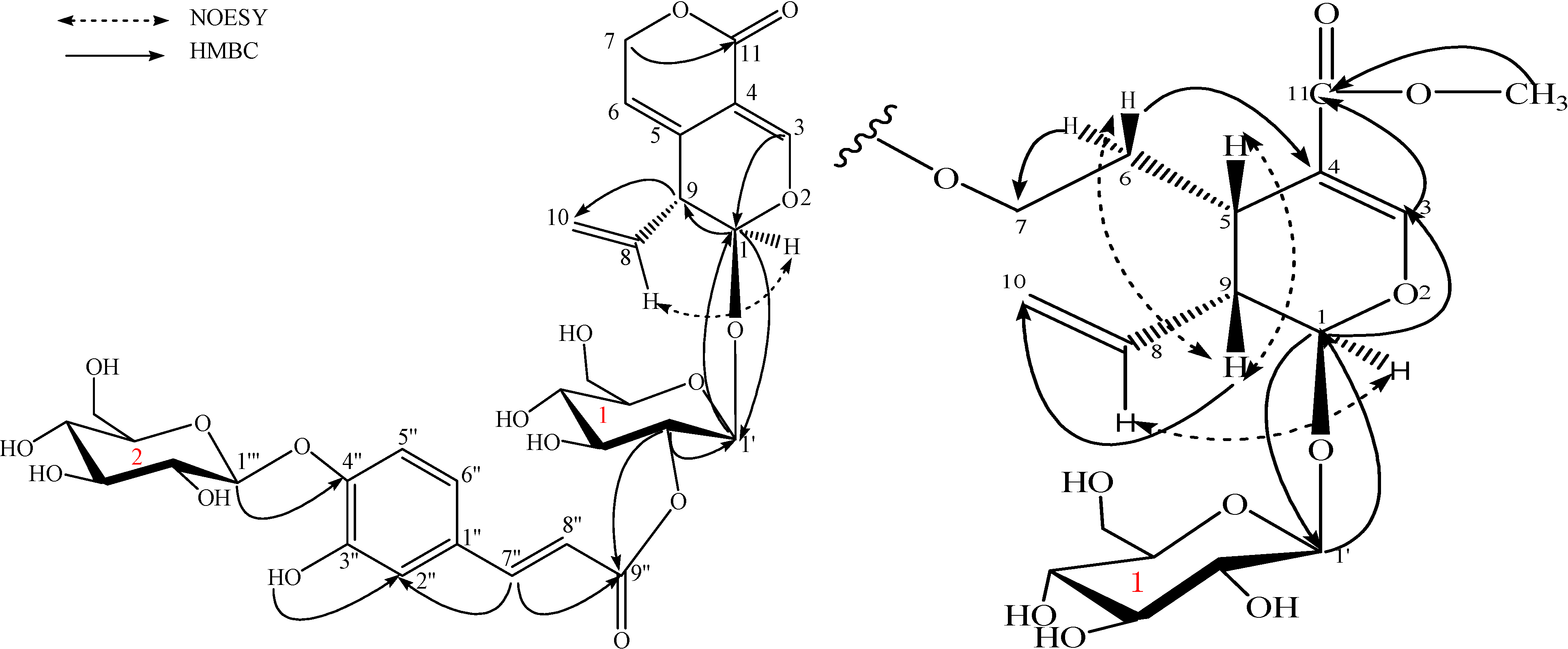
Results and Discussion
| position | picrogentioside A (1) | picrogentioside B (2) | picrogentioside C (3) | |||
|---|---|---|---|---|---|---|
| C | H (J, Hz) | C | H (J, Hz) | C | H (J, Hz) | |
| secoiridoid | ||||||
| 1 | 95.9 | 5.44, d, (3.2) | 95.5 | 5.60, d, (3.2) | 95.6 | 5.50, d, (6.4) |
| 3 | 150.8 | 7.28, br.s | 151.4 | 7.40, br.s | 152.0 | 7.49, s |
| 4 | 103.7 | 103.4 | 109.5 | |||
| 5 | 124.7 | 124.9 | 30.0 | 2.80, dd, (5.6,12.0) | ||
| 6 | 116.1 | 5.60, m | 116.3 | 5.65, m | 28.9 | 1.77, dd, (6.8, 13.6),1.94, dd, (6.8, 13.6) |
| 7 | 68.7 | 5.02, m5.07, m | 69.1 | 4.98, m5.05, m | 62.8 | 4.16, m4.24, m |
| 8 | 133.7 | 5.68, ddd, (6.8, 10.4, 16.8) | 134.0 | 5.71, ddd, (6.8, 10.4, 16.8) | 134.5 | 5.73, ddd, (8.4, 10.0, 18.0) |
| 9 | 44.0 | 3.40, m | 44.3 | 3.41, m | 43.1 | 2.60, dd, (6.4, 13.2) |
| 10 | 117.7 | 5.13, dd, (2.0, 10.4);5.20, dd, (2.0, 16.8) | 118.0 | 5.19, dd, (2.0, 10.4);5.24, dd, (2.0, 16.8) | 119.0 | 5.25, br.d, (11.2),5.27, br.d, (17.2) |
| 11 | 162.2 | 162.7 | 166.6 | |||
| CH3O | 51.0 | 3.61, s | ||||
| caffeoyl | ||||||
| 1'' | 128.7 | 128.7 | 128.6 | |||
| 2'' | 115.0 | 7.12, br.s | 114.9 | 7.15, br.s | 114.9 | 7.13, br. s |
| 3'' | 146.7 | 146.9 | 146.9 | |||
| 4'' | 147.4 | 147.4 | 147.4 | |||
| 5'' | 115.8 | 7.09, br.d, (8.0) | 116.1 | 7.11, br. d, (8.0) | 116.1 | 7.11, br. d, (8.4) |
| 6'' | 120.7 | 7.03, d, (8.0) | 120.6 | 7.11, br. d, (8.0) | 120.8 | 7.11, br. d, (8.4) |
| 7'' | 144.6 | 7.44, d, (16.0) | 144.1 | 7.51, d, (16.0) | 144.4 | 7.50, d, (16.0) |
| 8'' | 116.1 | 6.18, d, (16.0) | 115.9 | 6.40, d, (16.0) | 115.9 | 6.34, d, (16.0) |
| 9'' | 165.1 | 165.9 | 166.2 | |||
| Glc-1 | ||||||
| 1' | 95.7 | 4.78, d, (7.2) | 98.1 | 4.66, d, (7.6) | 98.7 | 4.53, d, (8.0) |
| 2' | 73.0 | 4.60, dd, (8.4, 7.2) | 72.5 | 3.17-3.49, m﹡ | 76.7 | 3.15, m |
| 3' | 73.7 | 3.15-3.51, m﹡ | 73.2 | 3.17-3.49, m﹡ | 73.0 | 2.97, t, (8.0) |
| 4' | 70.1 | 3.15-3.51, m﹡ | 71.2 | 4.61, m | 70.0 | 3.03, t, (9.2) |
| 5' | 77.5 | 3.15-3.51, m﹡ | 75.8 | 3.17-3.49, m﹡ | 77.3 | 3.30, m |
| 6' | 60.8 | 3.47, m; 3.71, m | 60.7 | 3.49, m﹡; 3.70, m | 61.1 | 3.43, m; 3.68, m |
| Glc-2 | ||||||
| 1''' | 101.6 | 4.75, d, (8.0) | 101.6 | 4.77, d, (7.2) | 101.6 | 4.78, d, (7.2) |
| 2''' | 75.8 | 3.15-3.48, m﹡ | 75.8 | 3.17-3.49, m﹡ | 75.8 | 3.28, m |
| 3''' | 73.2 | 3.15-3.48, m﹡ | 73.2 | 3.17-3.49, m﹡ | 73.2 | 4.29, m |
| 4''' | 69.7 | 3.15-3.48, m﹡ | 69.8 | 3.17-3.49, m﹡ | 69.8 | 3.15, m |
| 5''' | 77.2 | 3.15-3.48, m﹡ | 77.2 | 3.17-3.49, m﹡ | 77.2 | 3.31, m |
| 6''' | 60.7 | 3.48, m﹡; 3.72, m | 60.7 | 3.48, m﹡; 3.70, m | 60.7 | 3.43, m; 3.68,m |
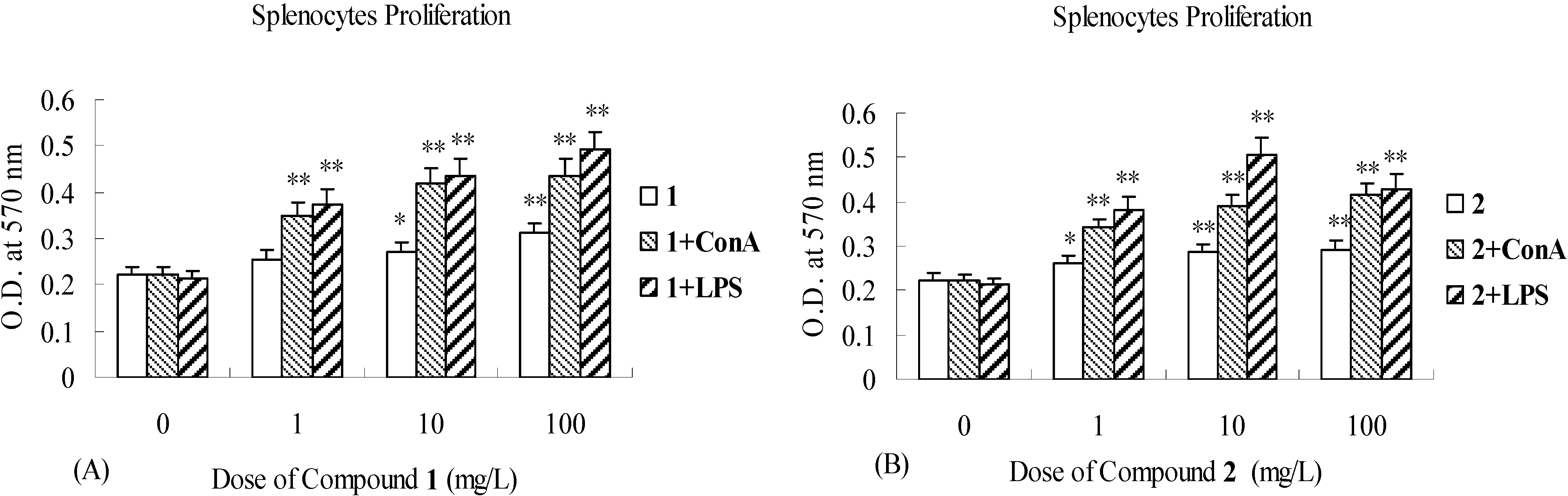
Biological activity
Experimental
General
Plant Material
Extraction and Isolation
Acid hydrolysis of 1, 2 and 3
Immunomodulatory effects study of 1, 2 and 3
Acknowledgements
References
- Wang, H.; Ye, W.C.; Jiang, R.W.; Wu, J.J.; Thomas, C.W.; Zhao, S.X.; Yao, X.S. Three new cyclopentanoid monoterpenes from Picrorhiza scrophulariiflora. Planta Med. 2004, 70, 382–384. [Google Scholar] [CrossRef]
- Wang, H.; Wu, J.J.; Liu, G.; Ye, W.C.; Zhao, Sh.X. Chemical Studies on Iridoids from Picrorh- iza scrophuariilflora. Chin. J. Nat. Med. 2006, 4, 36–41. [Google Scholar]
- Huang, S.X.; Zhou, Y.; Nie, Q.J.; Ding, L.S.; Peng, S.L. Two new iridoid glucosides from Picrorhiza scrophulariiflora. J. Asian Nat. Prod. Res. 2006, 8, 259–263. [Google Scholar] [CrossRef]
- Wang, H.; Ye, W.C.; Xiong, F.; Zhao, Sh.X. Phenylethanoid glycosides from root of Picrorhiza scrophulariiflora. Zhongguo Zhong Yao Za Zhi 2004, 29, 531–535. [Google Scholar]
- State Pharmacopeia Commission of P. R. China. Pharmacopeia of the P. R. China; People’s Health Publishing House: Beijing, P. R. China, 1995; Vol. I, p. 204. [Google Scholar]
- Hu, H.X.; Yang, P.M. Studies on chemical constituents of Picrorhiza scrophulariiflora Pennell. Chin. J. Pharm. 2005, 36, 336–339. [Google Scholar]
- Zhu, T.F.; Huang, K.Y.; Deng, X.M.; Zhang, Y.; Xiang, H.; Gao, H.Y.; Wang, D.C. Three New Caffeoyl Glycosides from the Roots of Picrorhiza scrophulariilflora. Molecules 2008, 13, 729–735. [Google Scholar] [CrossRef]
- Wang, M.Y.; Yang, L.; Tu, Y.Y. Phenylethanoid glycosides from stem of Chirita longgangensis var. hongyao. Zhongguo Zhong Yao Za Zhi 2005, 30, 1921–1923. [Google Scholar]
- Tian, J.; Zhao, Y.M.; Luan, X.H. Chemical Constituents of Verbena officinalis L. Nat. Prod. Res. Dev 2007, 19, 247–249. [Google Scholar]
- Cheng, B.C.; Tezuka, Y.; Nakano, H.; Tamaoki, T.; Jong, H.P. Constituents of a Fern, Davallia mariesii Moore. I isolated and structures of Davallialactone and a new flavanone glucuronide. Chem. Pharm. Bull. (Tokyo) 1990, 38, 3218–3225. [Google Scholar] [CrossRef]
- Oshima, R.; Yamauchi, Y.; Kumanotani, J. Resolution of the enantiomers of aldoses by liquid chromatography of diastereoisomeric 1-(N-acetyl-α-methylbenzylamino)-1-deoxyalditol acetates. Carbohydr. Res. 1982, 107, 169–176. [Google Scholar] [CrossRef]
- Jensen, S.R.; Lyse-Petersen, S.E.; Nielsen, B.J. Novel bis-iridoid glucosides from Dipsacus sylvestris. Phytochemistry 1979, 18, 273–277. [Google Scholar] [CrossRef]
- Mpondo, E.M.; Garcia, J. Secologanin and Derivatives from Gentiana verna. Planta Med. 1990, 52, 1146–1150. [Google Scholar]
- Chulia, A.J.; Vercauteren, J.; Mariotte, M. Iridoids and flavones from Gentiana Depressa. Phytochemistry 1996, 42, 139–143. [Google Scholar] [CrossRef]
- Müller, A.A.; Kufer, J.K.; Dietl, K.G.; Weigend, M.A. Dimeric iridoid from loasa acerifolia. Phytochemistry 1998, 49, 1705–1707. [Google Scholar] [CrossRef]
- Tomassini, L.; Foddai, S.; Serafini, M.; Cometa, M.F. Bis-iridoid glucosides from Abelia chinensis. J. Nat. Prod. 2000, 63, 998–999. [Google Scholar]
- Damtoft, S.; Franzyk, H.; Jensen, S.R. Iridoid glucosides from Picconia excelsa. Phytochemistry 1997, 45, 743–750. [Google Scholar] [CrossRef]
- Diana, C.H.; Richard, J.W.; Mary, E.H. Potentiation of rat lymphocyte proliferation by novel non- peptidic synthetic opioids. Int. Immunopharmacol. 2005, 5, 1271–1278. [Google Scholar] [CrossRef]
- Yang, T.H.; Jia, M.; Wu, H.; Mei, Q.B. Immunomodulatory activity of polysaccharide isolated from Angelica sinensis. Int. J. Biol. Macromol. 2006, 39, 179–184. [Google Scholar] [CrossRef]
- Sample Availability: Available from the authors.
© 2008 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zou, L.-C.; Zhu, T.-F.; Xiang, H.; Yu, L.; Yan, Z.-H.; Gan, S.-C.; Wang, D.-C.; Zeng, S.; Deng, X.-M. New secoiridoid Glycosides from the Roots of Picrorhiza Scrophulariiflora. Molecules 2008, 13, 2049-2057. https://doi.org/10.3390/molecules13092049
Zou L-C, Zhu T-F, Xiang H, Yu L, Yan Z-H, Gan S-C, Wang D-C, Zeng S, Deng X-M. New secoiridoid Glycosides from the Roots of Picrorhiza Scrophulariiflora. Molecules. 2008; 13(9):2049-2057. https://doi.org/10.3390/molecules13092049
Chicago/Turabian StyleZou, Lian-Chun, Tong-Fei Zhu, Hua Xiang, Lu Yu, Zhi-Hui Yan, Shu-Cai Gan, Da-Cheng Wang, Sheng Zeng, and Xu-Ming Deng. 2008. "New secoiridoid Glycosides from the Roots of Picrorhiza Scrophulariiflora" Molecules 13, no. 9: 2049-2057. https://doi.org/10.3390/molecules13092049
APA StyleZou, L.-C., Zhu, T.-F., Xiang, H., Yu, L., Yan, Z.-H., Gan, S.-C., Wang, D.-C., Zeng, S., & Deng, X.-M. (2008). New secoiridoid Glycosides from the Roots of Picrorhiza Scrophulariiflora. Molecules, 13(9), 2049-2057. https://doi.org/10.3390/molecules13092049




