Cytotoxic Metabolites from the Okinawan Ascidian Diplosoma virens
Abstract
:Introduction
Results and Discussion
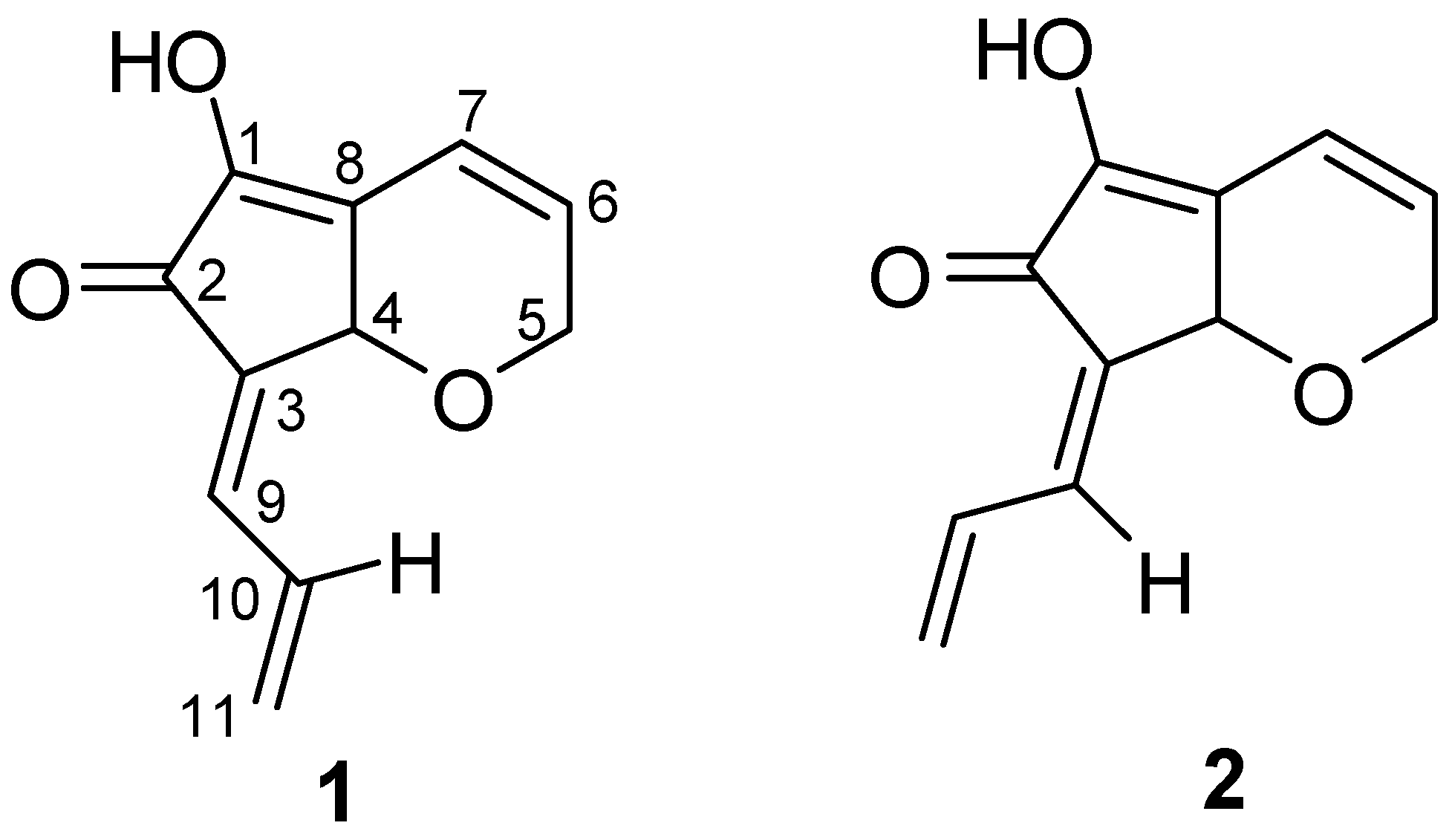
| 1 | 2 | ||||||
|---|---|---|---|---|---|---|---|
| C no. | δC | δH (mult, J in Hz) | δC | δH (mult, J in Hz) | |||
| 1 | 147.2 | 147.5 | |||||
| 2 | 187.9 | 187.8 | |||||
| 3 | 132.6 | 131.6 | |||||
| 4 | 71.3 | 5.01 (s) | 71.9 | 4.79 (s) | |||
| 5α | 67.1 | 4.50 (ddd, 18.5, 4.2, 2.0) | 67.2 | 4.48 (ddd, 18.3, 4.2, 1.7) | |||
| 5β | 4.61 (ddd, 18.5, 2.4, 2.4) | 4.57 (ddd, 18.3, 2.4, 2.4) | |||||
| 6 | 134.0 | 6.17 (ddd, 10.0, 4.2, 2.4) | 134.2 | 6.17 (ddd, 10.0, 4.2, 2.4) | |||
| 7 | 118.4 | 6.78 (br d, 10.0) | 118.4 | 6.77 (ddd, 10.0, 2.4, 1.7) | |||
| 8 | 129.1 | 128.3 | |||||
| 9 | 133.6 | 7.04 (br d, 11.7) | 136.6 | 6.65 (br d, 11.5) | |||
| 10 | 132.4 | 6.89 (ddd, 16.8, 11.7, 10.0) | 132.1 | 7.76 (ddd, 17.1, 11.5, 10.0) | |||
| 11α | 127.9 | 5.64 (br d, 10.0) | 127.3 | 5.57 (dd, 10.0, 1.7) | |||
| 11β | 5.71 (br d, 16.8) | 5.61 (dd, 17.1, 1.7) | |||||
| OH | 6.08 (br s) | 6.12 (br s) | |||||
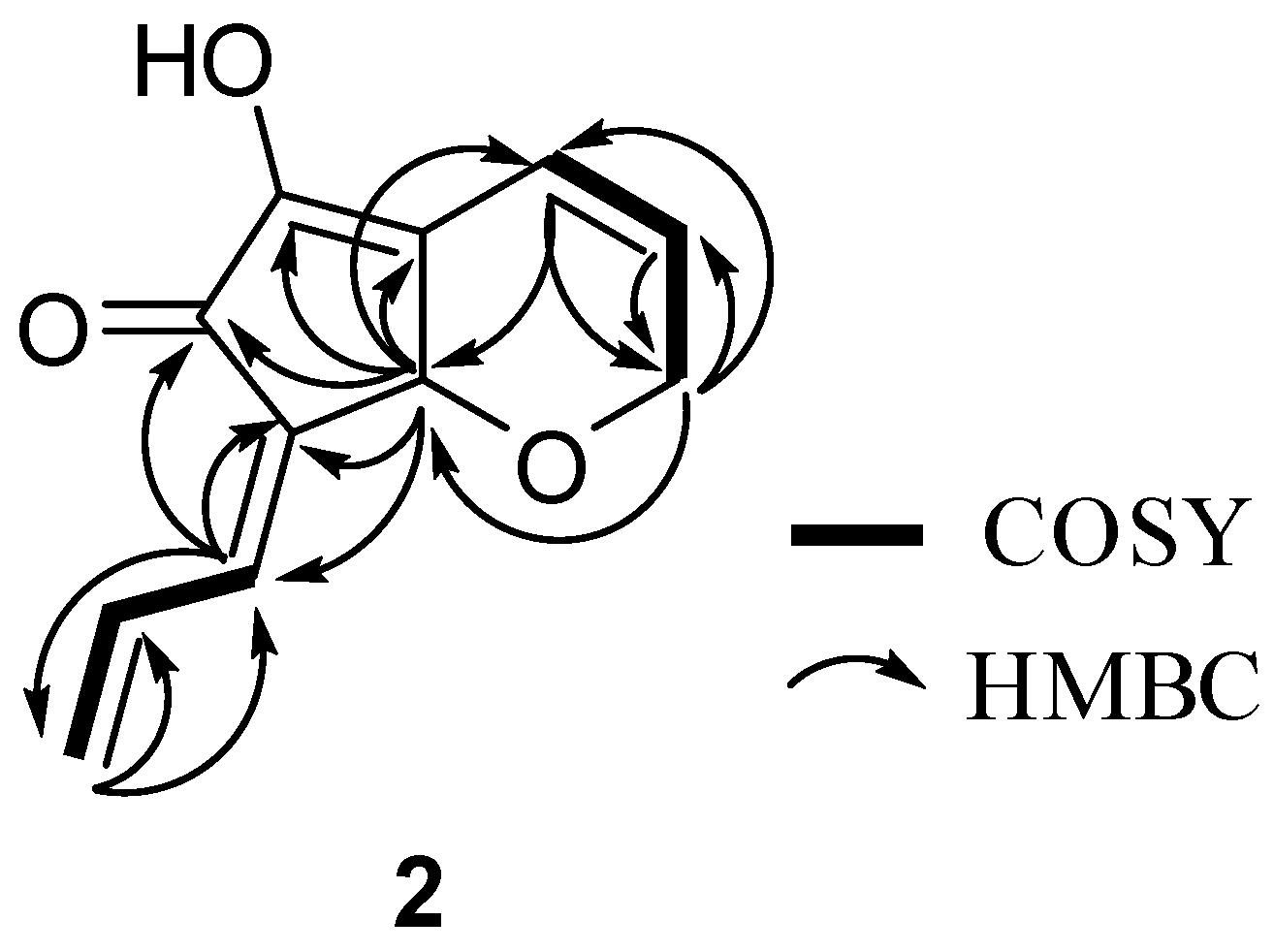
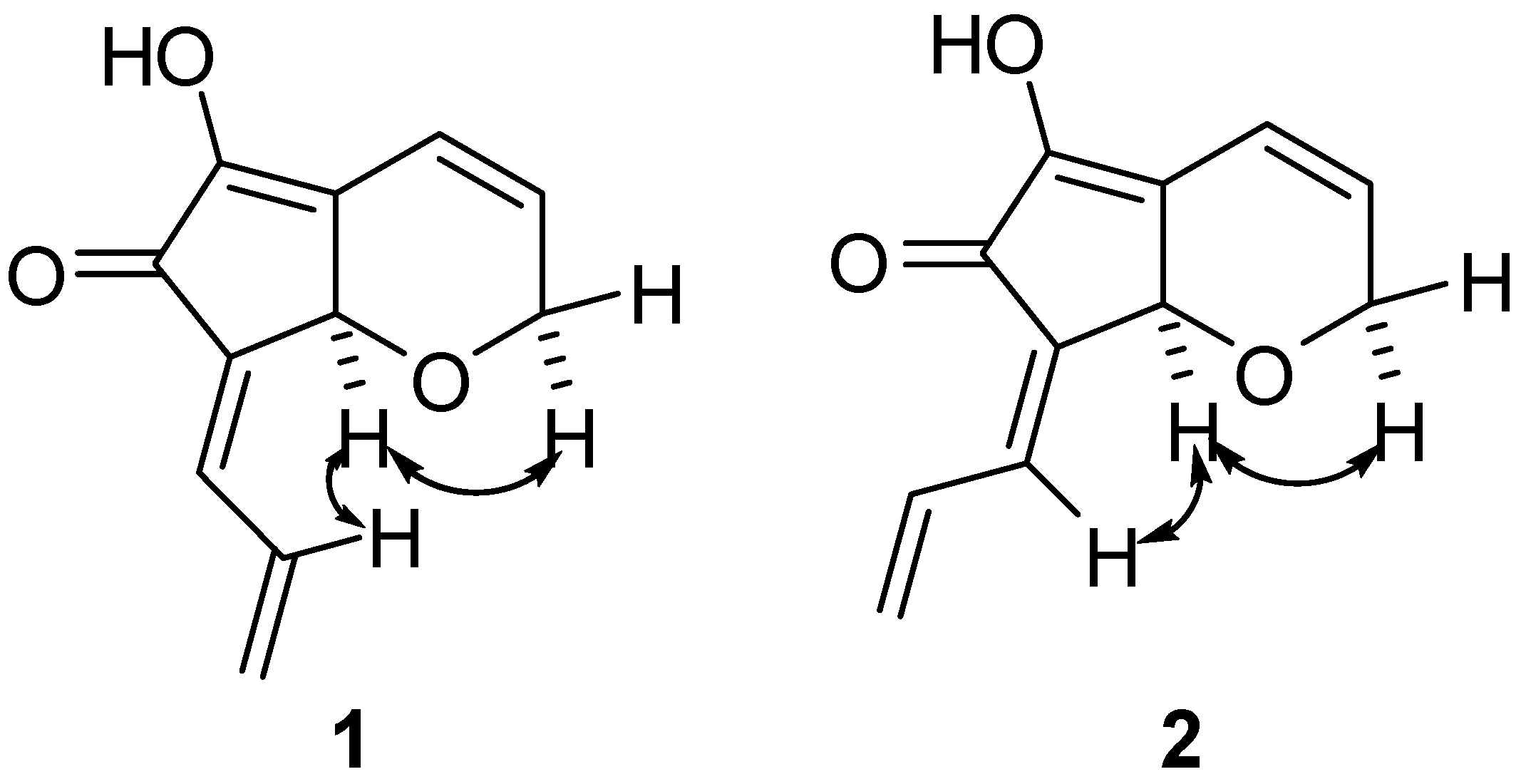
Biological Activities
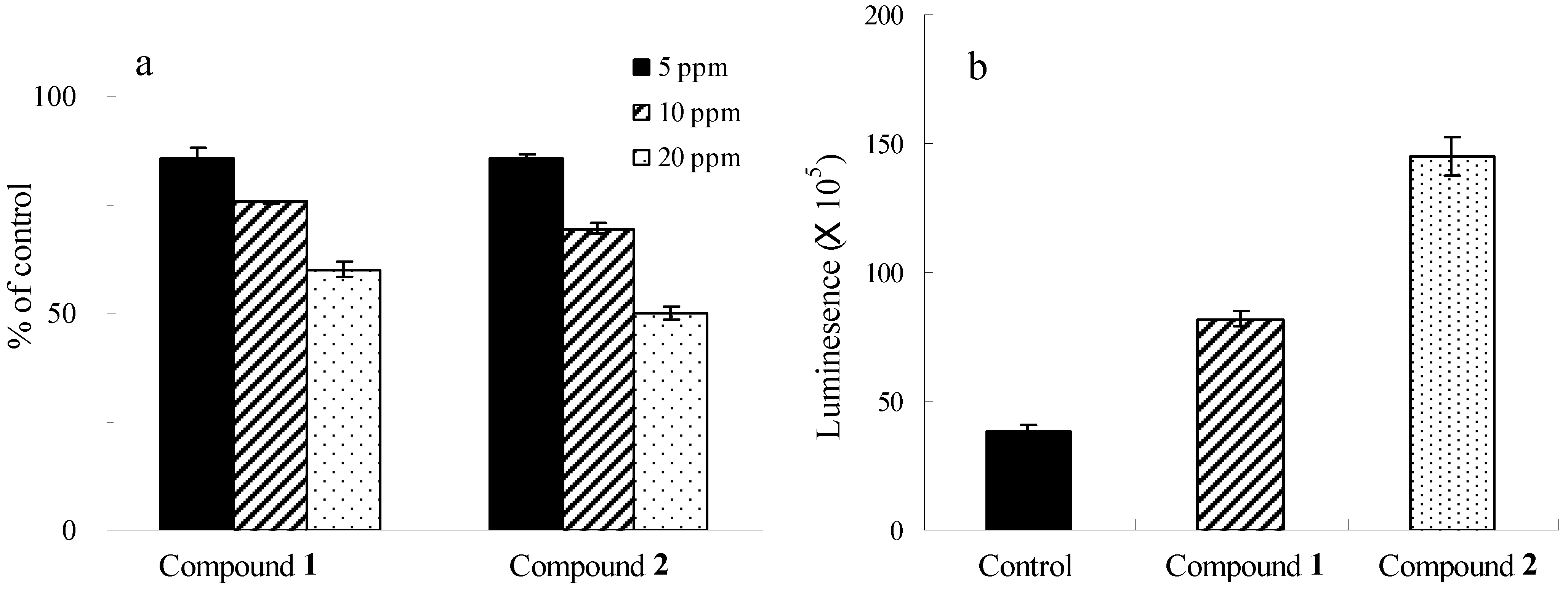
Conclusions
Experimental
General
Animal Collection
Extraction and Purification
Biological Assay
Acknowledgements
References
- Davidson, B.S. Ascidians: producers of amino acid-derived metabolites. Chem. Rev. 1993, 93, 1771–1791. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine Natural Products. Nat. Prod. Rep 2002, 19, 1–48, and previous reports in this series. [Google Scholar] Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R.J. Marine Natural Products. Nat. Prod. Rep 2006, 23, 26–78, and previous reports in this series. [Google Scholar]
- Lewin, R.A. Prochlorophyta as a proposed new division of algae. Nature 1976, 261, 697–698. [Google Scholar] [CrossRef]
- Withers, N.; Vidaver, W.; Lewin, R.A. Pigment Composition, Photosynthesis and Fine Structure of a Non-Blue-Green Prokaryotic Algal Symbiont (Prochloron Sp.) in a Didemnid Ascidian from Hawaiian Waters. Phycologia 1978, 17, 167–171. [Google Scholar] [CrossRef]
- Lewin, R.A.; Cheng, L. Prochrolon: A microbial Enigma; Chapman & Hall: New York, 1989; p. 129. [Google Scholar]
- Lindquist, N.; Fenical, W.; Sesin, D.F.; Ireland, C.M.; Duyne, G.D.V.; Forsyth, C.J.; Clardy, J. Isolation and structure determination of the didemnenones, novel cytotoxic metabolites from tunicates. J. Am. Chem. Soc. 1988, 110, 1308–1309. [Google Scholar] [CrossRef]
- Zabriskie, T.M.; Mayne, C.L.; Ireland, C.M. Patellazole C: a novel cytotoxic macrolide from Lissoclinum patella. J. Am. Chem. Soc. 1988, 110, 7919–7920. [Google Scholar] [CrossRef]
- Coreley, D.G.; Moore, R.E.; Paul, V.J. Patellazole B: a novel cytotoxic thiazole-containing macrolide from the marine tunicate Lissoclinum patella. J. Am. Chem. Soc. 1988, 110, 7920–7922. [Google Scholar] [CrossRef]
- Davidson, B.S.; Molinski, T.F.; Barrows, L.R.; Ireland, C.M. Varacin: a novel benzopentathiepin from Lissoclinum vareau that is cytotoxic toward a human colon tumor. J. Am. Chem. Soc. 1991, 113, 4709–4710. [Google Scholar] [CrossRef]
- Carroll, A.R.; Feng, Y.; Bowden, B.F.; Coll, J.C. Studies of Australian Ascidians. 5. Virenamides A-C, New Cytotoxic Linear Peptides from the Colonial Didemnid Ascidian Diplosoma virens. J. Org. Chem. 1996, 12, 4059–4061. [Google Scholar]
- Ueda, K.; Hu, Y. Haterumalide B: A new cytotoxic macrolide from an Okinawan ascidian Lissoclinum sp. Tetrahedron Lett. 1999, 40, 6305–6308. [Google Scholar] [CrossRef]
- Takada, N.; Sato, H.; Suenaga, K.; Arimoto, H.; Yamada, K.; Ueda, K.; Uemura, D. Isolation and structures of haterumalides NA, NB, NC, ND, and NE, novel macrolides from an Okinawan Sponge Ircinia sp. Tetrahedron Lett. 1999, 40, 6309–6312. [Google Scholar] [CrossRef]
- Kokubo, S.; Yogi, K.; Uddin, M.J.; Inuzuka, T.; Suenaga, K.; Ueda, K.; Uemura, D. Kohamaic Acids A and B, Novel Cytotoxic Sesterterpenic Acids, from the Marine Sponge Ircinia sp. Chem. Lett. 2001, 2, 176–177. [Google Scholar]
- Uddin, M.J.; Kokubo, S.; Ueda, K.; Suenaga, K.; Uemura, D. Haterumaimides F-I, Four New Cytotoxic Diterpene Alkaloids from an Ascidian Lissoclinum Species. J. Nat. Prod. 2001, 64, 1169–1173. [Google Scholar] [CrossRef]
- Wratten, S.J.; Faulkner, D.J. Antimicrobial metabolites from the marine sponge uloma sp. . Tetrahedron Lett. 1978, 19, 961–964. [Google Scholar] [CrossRef]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-Dependent Formation of Apaf-1/Caspase-9 Complex Initiates an Apoptotic Protease Cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Scaffidi, C.; Fulda, S.; Srinivasan, A.; Friesen, C.; Li, F.; Tomaselli, K.J.; Debatin, K.M.; Krammer, P.H.; Peter, M.E. Two CD95 (APO-1/Fas) signaling pathways. EMBO J. 1998, 17, 1675–1687. [Google Scholar] [CrossRef]
- Kuwana, T.; Smith, J.J.; Muzio, M.; Dixit, V.; Newmeyer, D.D.; Kornbluth, S. Apoptosis Induction by Caspase-8 Is Amplified through the Mitochondrial Release of Cytochrome c. J. Biol. Chem. 1998, 273, 16589–16594. [Google Scholar]
- Nagle, D.G.; Gerwick, W.H. Nakienones A-C and nakitriol, new cytotoxic cyclic C11 metabolites from an okinawan cyanobacterial (Synechocystis sp.) overgrowth of coral. Tetrahedron. Lett. 1995, 36, 849–852. [Google Scholar] [CrossRef]
- Teruya, T.; Nakagawa, S.; Koyama, T.; Suenaga, K.; Uemura, D. Terpiodiene: A Novel Tricyclic Alcohol from the Okinawan Sponge Terpios hoshinota. Chem. Lett. 2002, 38–39. [Google Scholar]
- Sample Availability: Samples of the compounds are available from authors.
© 2008 MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Ogi, T.; Taira, J.; Margiastuti, P.; Ueda, K. Cytotoxic Metabolites from the Okinawan Ascidian Diplosoma virens. Molecules 2008, 13, 595-602. https://doi.org/10.3390/molecules13030595
Ogi T, Taira J, Margiastuti P, Ueda K. Cytotoxic Metabolites from the Okinawan Ascidian Diplosoma virens. Molecules. 2008; 13(3):595-602. https://doi.org/10.3390/molecules13030595
Chicago/Turabian StyleOgi, Takayuki, Junsei Taira, Palupi Margiastuti, and Katsuhiro Ueda. 2008. "Cytotoxic Metabolites from the Okinawan Ascidian Diplosoma virens" Molecules 13, no. 3: 595-602. https://doi.org/10.3390/molecules13030595
APA StyleOgi, T., Taira, J., Margiastuti, P., & Ueda, K. (2008). Cytotoxic Metabolites from the Okinawan Ascidian Diplosoma virens. Molecules, 13(3), 595-602. https://doi.org/10.3390/molecules13030595




