RETRACTED: Antibacterial Effect of Five Zingiberaceae Essential Oils
Abstract
:Introduction
Results and Discussion
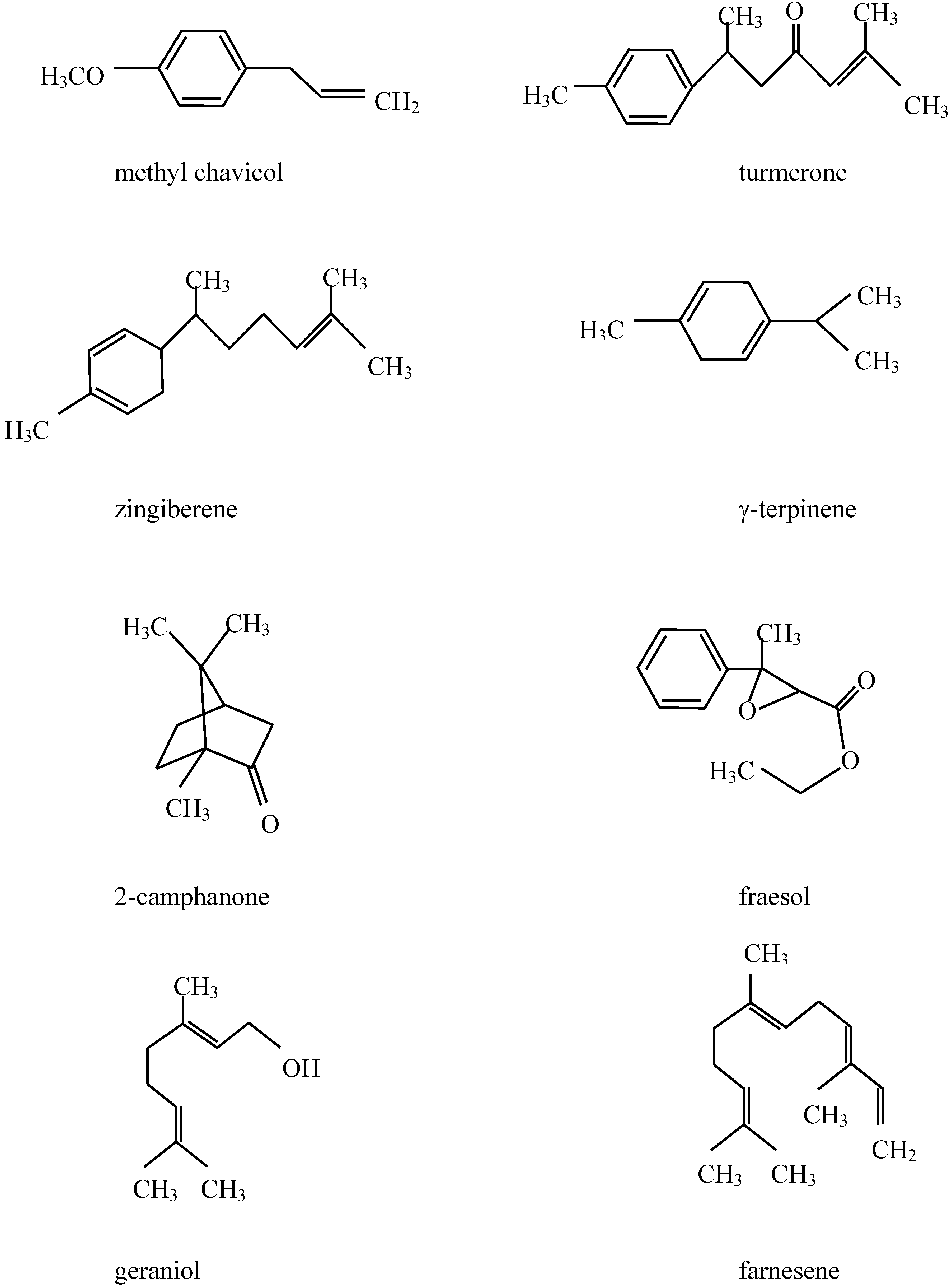
Chemical composition of the plant extracts.
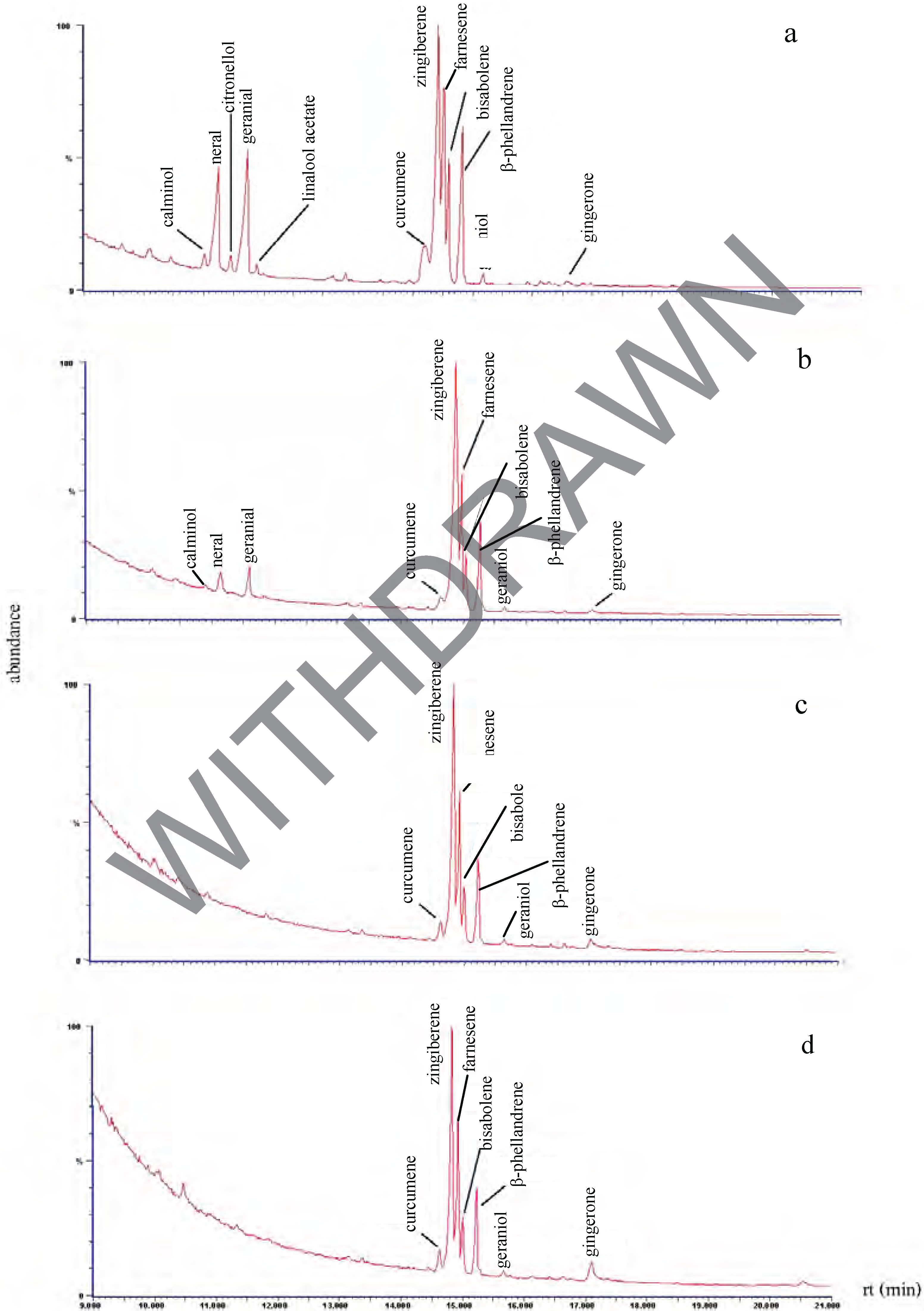
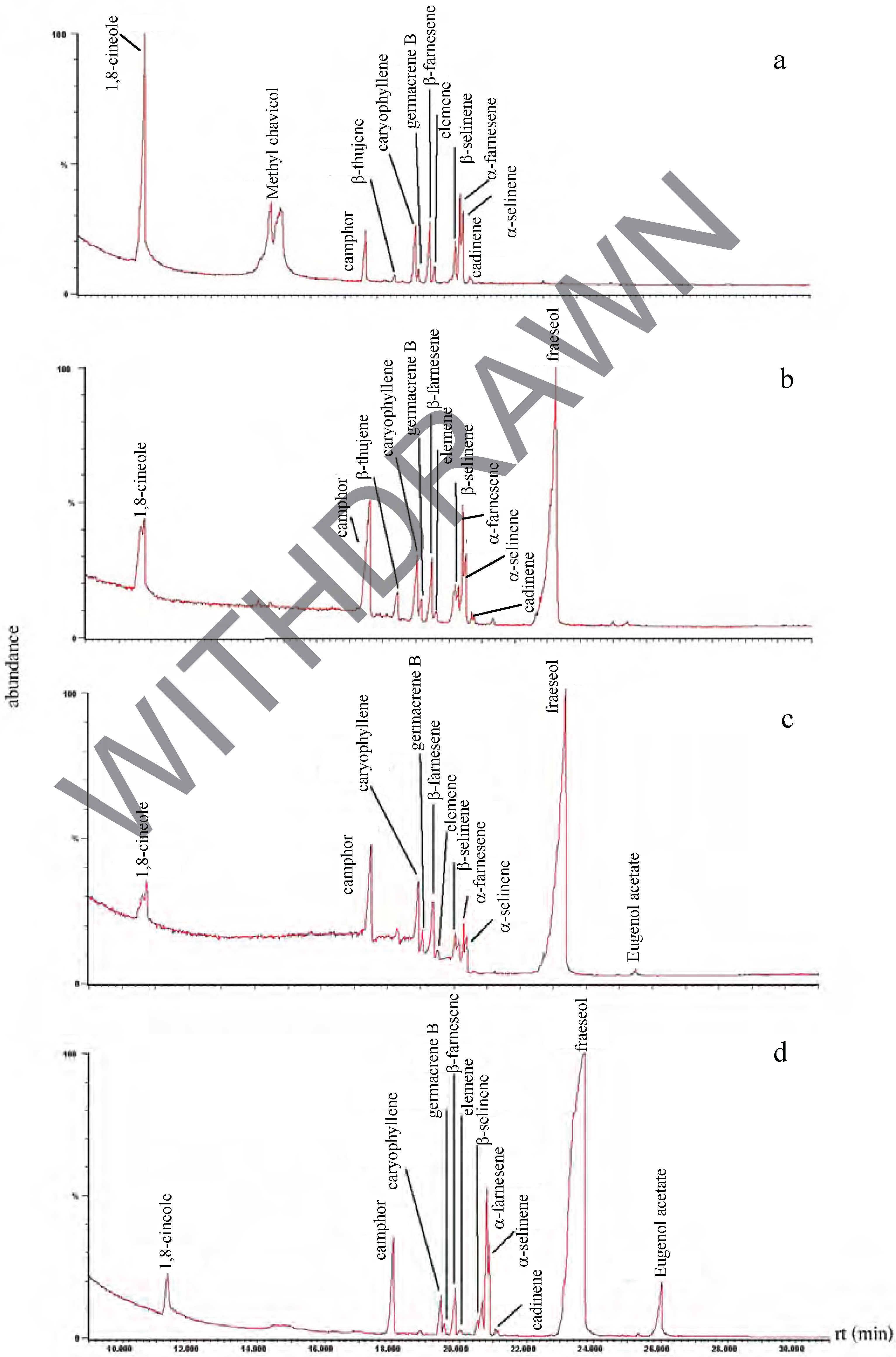
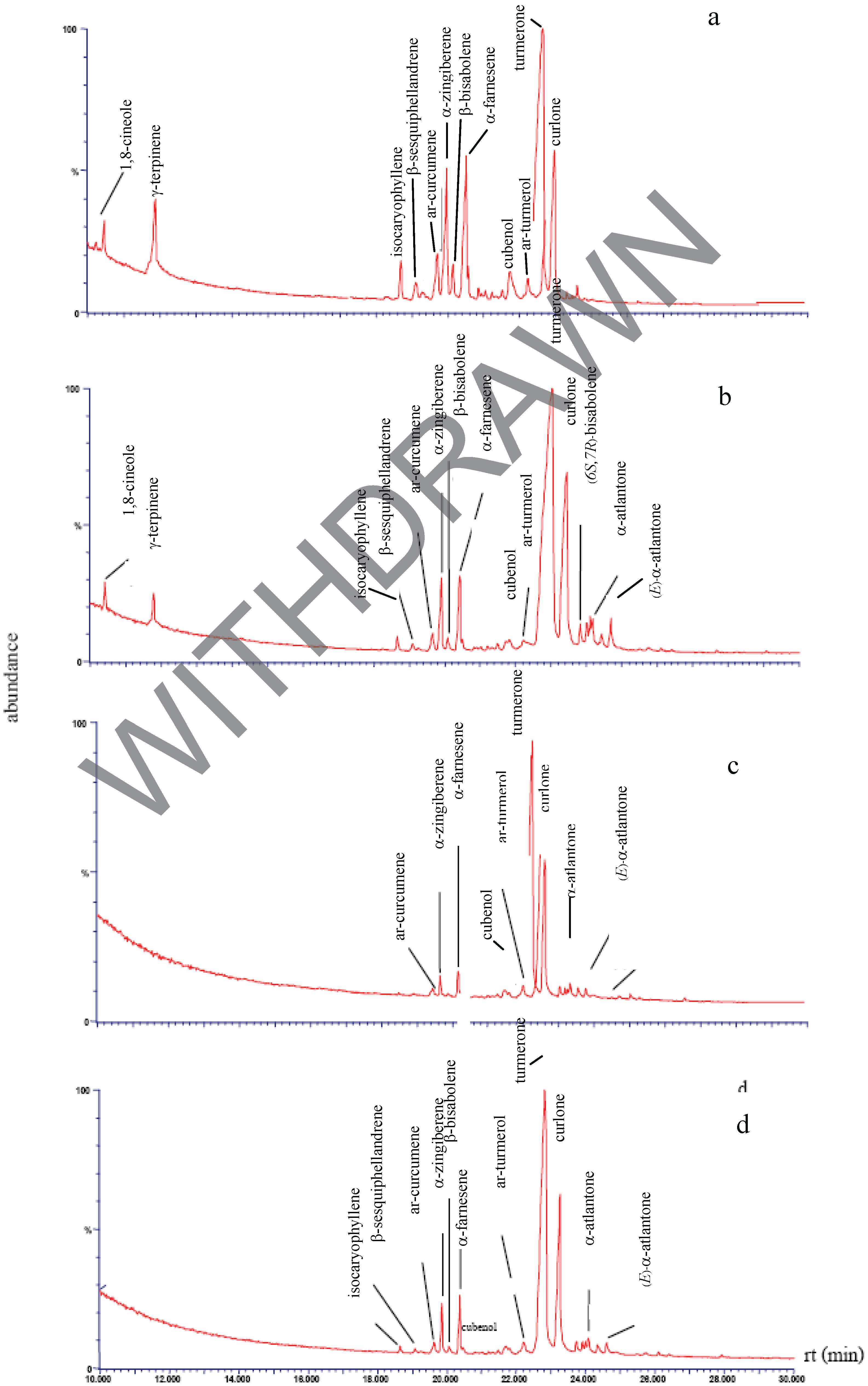

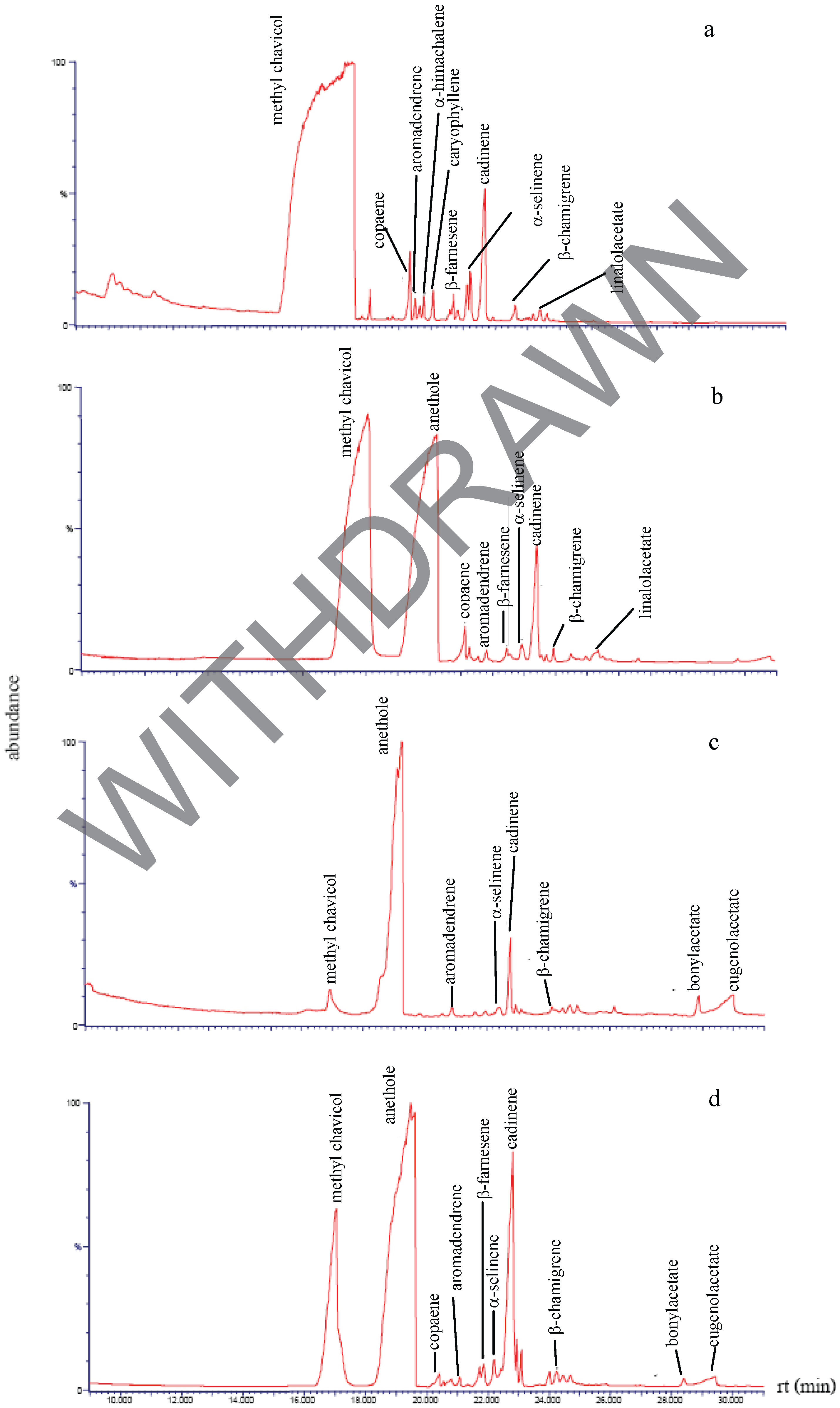
Antibacterial activity
| Plant species | Extract | Yield a*: % | Inhibition zones (mm)b* against | |||
|---|---|---|---|---|---|---|
| S. aureus | B. cereus | E.coli | L. mono cytogenes | |||
| Zingiber officinale (ginger) | Water | 0.27l | 16a | 20a | 0c | 22a |
| Ethanol | 3.55b | 9hi | 10h | 0c | 10i | |
| Secondary extraction | 2.65c | 8j | 11g | 0c | 8l | |
| Petroleum ether | 1.08fg | 10g | 12f | 0c | 11h | |
| Alpinia galangal (galanga) | Water | 0.18n | 8k | 9hi | 0c | 11h |
| Ethanol | 1.15f | 12e | 13d | 0c | 9k | |
| Secondary extraction | 1.03g | 13d | 14cd | 0c | 10i | |
| Petroleum ether | 0.48j | 12e | 12f | 0c | 9k | |
| Curcuma longa (turmeric) | Water | 0.64i | 9i | 10h | 0c | 16c |
| Ethanol | 3.65a | 11fg | 12f | 0c | 13f | |
| Secondary extraction | 3.60ab | 11fg | 11e | 0c | 11g | |
| Petroleum ether | 1.65d | 10h | 10g | 0c | 10i | |
| Boesenbergia pandurata (kaempferia) | Water | 0.26m | 15ba | 16b | 9b | 19b |
| Ethanol | 1.25e | 11f | 14c | 0c | 15d | |
| Secondary extraction | 0.83h | 14b | 16b | 0c | 14e | |
| Petroleum ether | 0.40kj | 14b | 12cf | 0c | 13f | |
| Amomum xanthioides (bastard cardamom) | Water | 0.27lm | 12c | 13d | 10a | 13f |
| Ethanol | 0.48j | 0l | 8i | 0c | 0m | |
| Secondary extraction | 0.35k | 0l | 0j | 0c | 0m | |
| Petroleum ether | 0.65i | 8j | 9hi | 0c | 0m | |
| Streptomycin, 5 mg/mL | – | 24 | 26 | 22 | 24 | |
Minimum inhibitory concentration (MIC)
| Bacterial species | Minimum inhibition concentration (mg mL- 1) | |||||||
|---|---|---|---|---|---|---|---|---|
| Hydrodistillation | Secondary extraction with ethanol | Ethanol | ||||||
| Z. o. | C. l. | B. p. | A. x. | A. g. | B. p. | B. p. | ||
| S. aureus | 12.5 | N.T. | 12.5 | N.T. | 100 | 50 | 12.5 | |
| B. cereus | 6.25 | N.T. | 12.5 | N.T. | 25 | 12.5 | 12.5 | |
| E. coli | N.T. | N.T. | 50 | 25 | N.T. | N.T. | N.T. | |
| L. monocytogenes | 6.25 | 25 | 6.25 | N.T. | N.T. | 6.25 | 6.25 | |
Experimental
1. Plant material
2. Extraction procedure
2.3 Gas chromatography/mass spectrometry analysis (GC-MS)
2.4 Preparation of bacterial strains
2.5 Antibacterial screening
2.6 Determination of minimum inhibitory concentrations (MICs)
2.7 Statistical analysis
Acknowledgements
References and Notes
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Breese, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food related illness and dead in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar]
- Betts, G.D.; Linton, P.; Betteridge, R.J. Food spoilage yeasts: effects of pH, NaCl and temperature on growth. Food Control 1999, 10, 27–33. [Google Scholar]
- Deak, T.; Beuchat, L.R. Handbook of Food Spoilage; CRC Press: New York, USA, 1996. [Google Scholar]
- Sagdıc, O.; Ozcan, M. Antibacterial activity of Turkish spice hydrosols. Food Control 2003, 14, 141–143. [Google Scholar]
- Smid, E.J.; Gorris, L.G.M. Natural antimicrobials for food preservation. In Handbook of Food Preservation; Rahman, M.S., Ed.; Marcel Dekker: New York, 1999; pp. 285–308. [Google Scholar]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 564–582. [Google Scholar]
- Kumarasamy, Y.; Cox, P.; Jaspars, M.; Nahar, L.; Sarker, S. Screening seeds of Scottish plants for antibacterial activity. J. Ethnopharmacol. 2002, 83, 73–77. [Google Scholar]
- Srinivasan, D.; Nathan, S.; Suresh, T.; Perumalsamy, L. Antimicrobial activity of certain Indian medicinal plants used in folkloric medicine. J. Ethnopharmacol. 2001, 74, 217–220. [Google Scholar]
- Burkill, I.H. A Dictionary of the Economic Products of the Malay Peninsula. Vol I: A-H, Vol II: I-Z; Art Printing Works: Kaula Lumpur, 1966; p. 2402. [Google Scholar]
- Leal, P.F.; Braga, M.E.M.; Sato, D.N.; Carvalho, J.E.; Marques, M.O.M.; Meireles, M.A.A. Functional properties of spice extracts obtained via supercritical fluid extraction. J. Agric. Food Chem. 2003, 51, 2520–2525. [Google Scholar]
- Sekiwa, Y; Kubota, K.; Kobayashi, A. Isolation of novel glucosides related to gingerdiol from ginger and their antioxidative activities. J. Agric. Food Chem. 2000, 48, 373–377. [Google Scholar]
- Nguefack, J.; Leth, V.; Amvam, P.H.; Mathur, S.B. Evaluation of five essential oil from aromatic plant of Cameroon for controlling food spoilage and mycotoxin producing fungi. Int. J. Food Microbiol. 2004, 94, 329–334. [Google Scholar]
- Jirovetz, L.; Buchbauer, G.; Pottachola, M.; Kalathil, N. Analysis of the essential oils of the leaves, stems, rhizomes and roots of the medicinal plant Alpinia galanga from southern India. Acta Pharma. 2003, 53, 73–81. [Google Scholar]
- Bendjeddou, D.; Lalaoui, K.; Satta, D. Immunostimulating activity of the hot water-soluble poly-saccharide extracts of Anacyclus pyrethrum, Alpinia galanga and Citrullus colocythis. J. Ethnopharmacol. 2003, 88, 155–160. [Google Scholar]
- Scartezzini, P.; Speroni, E. Review on some plants of Indian traditional medicine with antioxidant activity. J. Ethnopharmacol. 2000, 71, 23–43. [Google Scholar]
- Negi, P.S.; Jayaprakasha, G.K.; Jagan, M.R.L.; Sarariah, K.K. Antibacterial activity of turmeric oil: A byproduct from curcumin manufacture. J. Agric. Food Chem. 1999, 47, 4297–4300. [Google Scholar]
- Zancan, K.C.; Marques, M.O.M.; Petenate, A.J.; Meireles, M.A.A. Extraction of ginger (Zingiber officinale Roscoe) oleoresin with CO2 and co-solvent: A study of the antioxidant action of the extracts. J. Supercrit. Fluids 2002, 24, 57–76. [Google Scholar]
- Baydar, H.; Sagdic, O.; Ozkan, G.; Karadogan, T. Antibacterial activity and composition of essential oils of Origanum, Thymbra and Sajureja species with commercial importance in Turkey. Food Control 2004, 15, 169–172. [Google Scholar]
- Alzoreky, N.S.; Nakahara, K. Antibacterial activity of extracts from some edible plants commonly consumed in Asia. Int. J. Food Microbiol. 2003, 80, 223–230. [Google Scholar]
- Cosentino, S.; Tuberoso, C.I.G.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas, F. In-vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Appl. Microbiol. 1999, 29, 130–135. [Google Scholar]
- Karaman, I.; Sahin, F.; Gulluce, M.; Qgutcu, H.; Sengul, M.; Adiguzel, A. Antimicrobial activity of aqueous and methanol extracts of Juniperus oxycedrus L. J. Ethnopharmacol. 2003, 85, 231–235. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods: A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar]
- Hsieh, P.C.; Mau, J.L.; Huang, S.H. Antimicrobial effect of various combinations of plant extracts. Food Microbiol. 2001, 18, 35–43. [Google Scholar]
- Kumar, S.; Narain, U.; Tripathi, S.; Misra, K. Syntheses of curcumin bioconjugates and study of their antibacterial activities against β-lactamase-producing microorganisms. Bioconj. Chem. 2001, 12, 464–469. [Google Scholar]
- Gulluce, M.; Sokmen, M.; Deferera, D.; Agar, G.; Ozkan, H.; Kartal, N.; Polissiou, M.; Sokmen, A.; Sahin, F. In vitro antibacterial, antifungal, and antioxidant activities of the essential oil and methanol extracts of herbal parts and cullus cultures of Satureja hortensis L. J. Agric. Food Chem. 2003, 51, 3958–3965. [Google Scholar]
- SAS. SAS user’s guide: Statistics; SAS Institute, Inc.: Cary, NC, 1991; p. 558. [Google Scholar]
- Sample Availability: Contact the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Norajit, K.; Laohakunjit, N.; Kerdchoechuen, O. RETRACTED: Antibacterial Effect of Five Zingiberaceae Essential Oils. Molecules 2007, 12, 2047-2060. https://doi.org/10.3390/12082047
Norajit K, Laohakunjit N, Kerdchoechuen O. RETRACTED: Antibacterial Effect of Five Zingiberaceae Essential Oils. Molecules. 2007; 12(8):2047-2060. https://doi.org/10.3390/12082047
Chicago/Turabian StyleNorajit, Krittika, Natta Laohakunjit, and Orapin Kerdchoechuen. 2007. "RETRACTED: Antibacterial Effect of Five Zingiberaceae Essential Oils" Molecules 12, no. 8: 2047-2060. https://doi.org/10.3390/12082047
APA StyleNorajit, K., Laohakunjit, N., & Kerdchoechuen, O. (2007). RETRACTED: Antibacterial Effect of Five Zingiberaceae Essential Oils. Molecules, 12(8), 2047-2060. https://doi.org/10.3390/12082047




