Efficient Ce(NO3)3·6H2O-Catalyzed Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones
Abstract
:Introduction
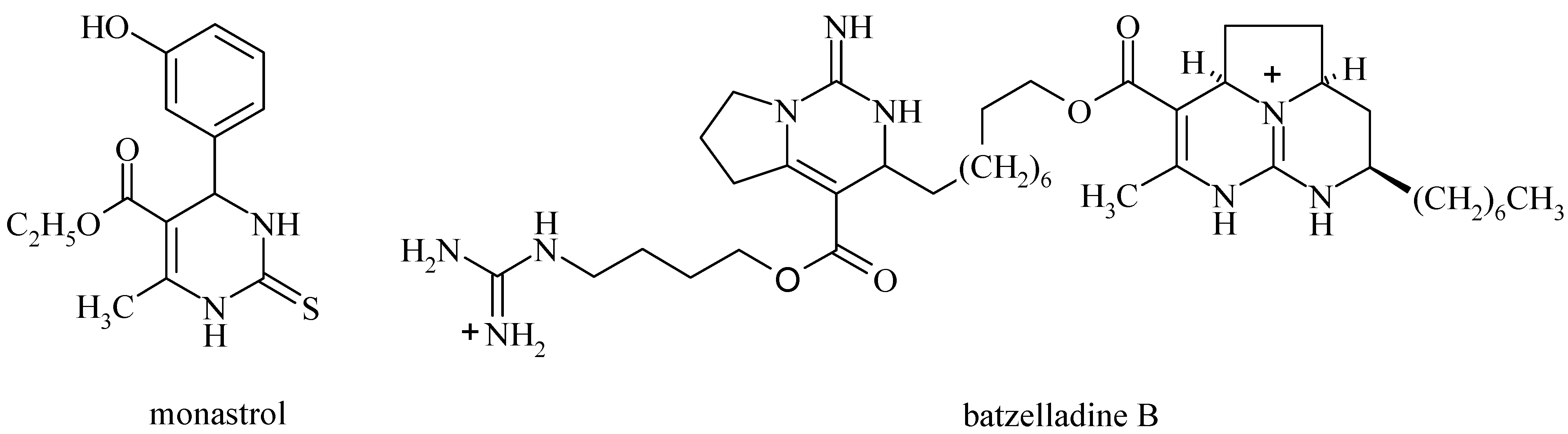
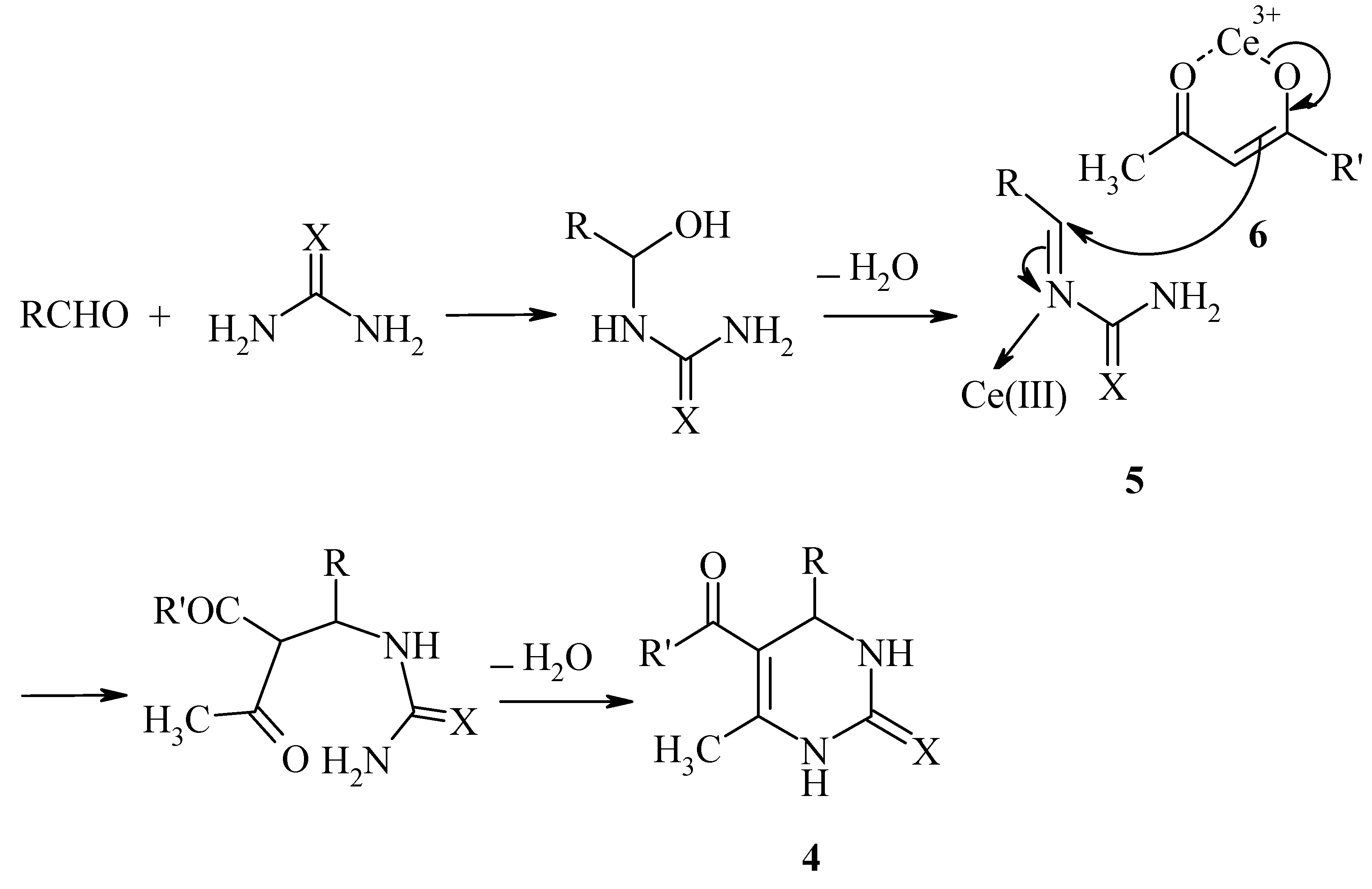
Results and Discussion
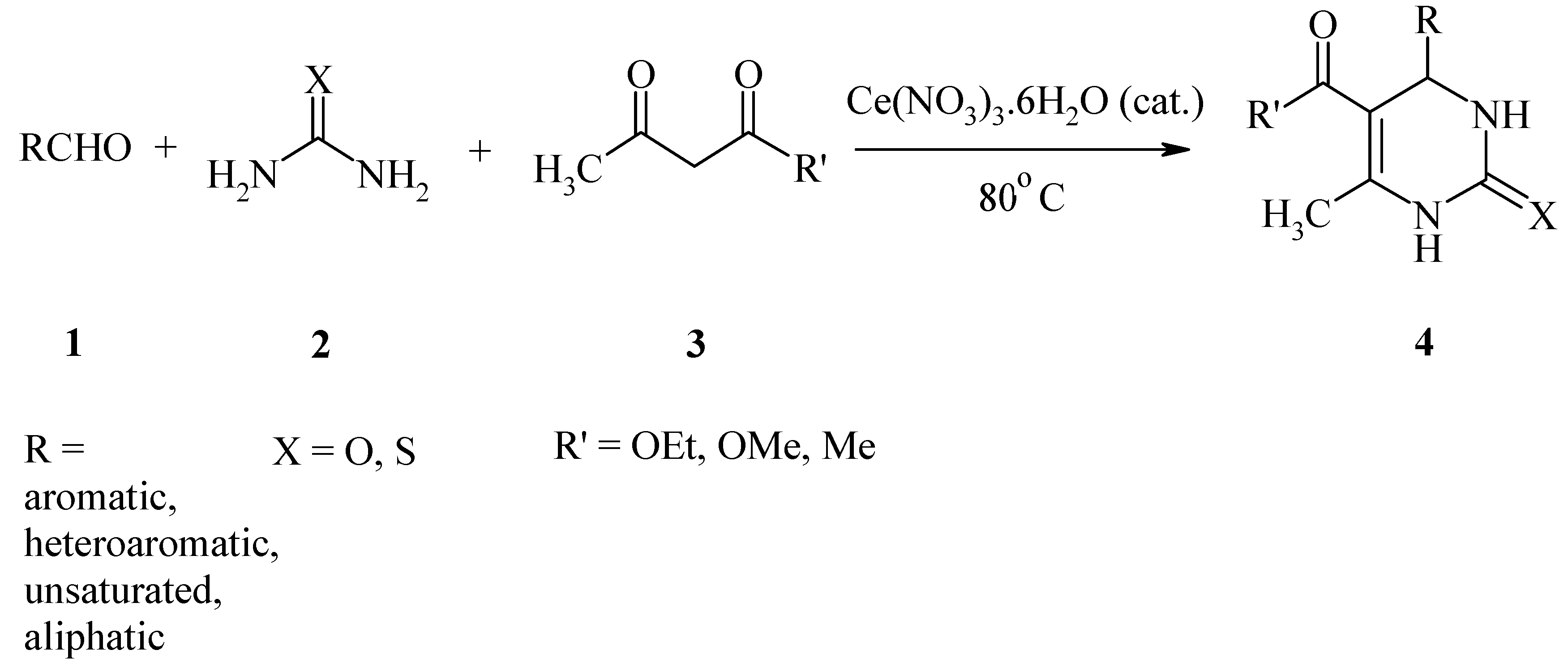
| Entry | R | R' | X | time (min) | Yield (%)b |
|---|---|---|---|---|---|
| 1 | C6H5 | OEt | O | 10 | 98 |
| 2 | 2-MeC6H4 | OEt | O | 15 | 95 |
| 3 | 4-MeC6H4 | OEt | O | 20 | 93 |
| 4 | 2-MeOC6H4 | OEt | O | 15 | 97 |
| 5 | 3-MeOC6H4 | OEt | O | 10 | 97 |
| 6 | 4-MeOC6H4 | OEt | O | 10 | 98 |
| 7 | 2-ClC6H4 | OEt | O | 25 | 90 |
| 8 | 3-ClC6H4 | OEt | O | 20 | 92 |
| 9 | 4-ClC6H4 | OEt | O | 25 | 94 |
| 10 | 4-BrC6H4 | OEt | O | 25 | 93 |
| 11 | 4-FC6H4 | OEt | O | 25 | 95 |
| 12 | 3-O2NC6H4 | OEt | O | 25 | 89 |
| 13 | 4-O2NC6H4 | OEt | O | 25 | 91 |
| 14 | 2,4-(MeO)2C6H3 | OEt | O | 10 | 97 |
| 15 | 1-Naphthyl | OEt | O | 30 | 91 |
| 16 | 2-Furyl | OEt | O | 15 | 93 |
| 17 | PhCH=CH | OEt | O | 25 | 84 |
| 18 | n-Pentyl | OEt | O | 35 | 87 |
| 19 | n-Hexyl | OEt | O | 40 | 84 |
| 20 | C6H5 | OMe | O | 10 | 96 |
| 21 | 4-O2NC6H4 | OMe | O | 20 | 93 |
| 22 | 4-MeOC6H4 | OMe | O | 10 | 97 |
| 23 | C6H5 | Me | O | 10 | 96 |
| 24 | 4-O2NC6H4 | Me | O | 30 | 90 |
| 25 | 4-MeOC6H4 | Me | O | 10 | 97 |
| 26 | C6H5 | OEt | S | 10 | 96 |
| 27 | 4-O2NC6H4 | OEt | S | 10 | 90 |
| 28 | 4-MeOC6H4 | OEt | S | 10 | 97 |
| 29 | 3-HOC6H4 | OEt | S | 30 | 96 |
| 30 | C6H5 | OMe | S | 10 | 95 |
| 31 | 4-MeOC6H4 | OMe | S | 10 | 96 |
| 32 | C6H5 | Me | S | 10 | 94 |
Conclusions
Acknowledgements
Experimental
General
Typical Procedure: 5-(Ethoxycarbonyl)-6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-one (Entry 1)
References and Notes
- Biginelli, P. Gazz. Chim. Ital. 1893, 23, 360.Kappe, C. O. Tetrahedron 1993, 49, 6937.
- Studer, A.; Jeger, P.; Wipf, P.; Curran, D. P. J. Org. Chem. 1997, 62, 2917.
- Kappe, C. O. Acc. Chem. Res. 2000, 33, 879. [CrossRef]
- Atwal, K. S.; Rovnyak, G. C.; O’Reilly, B. C.; Schwartz, J. J. Org. Chem. 1989, 54, 5898.
- Atwal, K. S.; Swanson, B. N.; Unger, S. E.; Floyd, D. M.; Moreland, S.; Hedberg, A.; O’Reilly, B. C. J. Med. Chem. 1991, 34, 806.Rovnyak, G. C.; Atwal, K. S.; Hedberg, A.; Kimball, S. D.; Moreland, S.; Gougoutas, J. Z.; O’Reilly, B. C.; Schwartz, J.; Malley, M. F. J. Med. Chem. 1992, 35, 3254.
- Rovnyak, G. C.; Kimbal, S. D.; Beyer, B.; Cucinotta, G.; Dimarco, J. D.; Gougoutas, J.; Hedberg, A.; Malley, M.; MaCarthy, J. P.; Zhang, R.; Mereland, S. J. Med. Chem. 1995, 38, 119.Kappe, C. O.; Fabian, W. M. F.; Semons, M. A. Tetrahedron 1997, 53, 2803.
- Barrow, J. C.; Nantermet, P. G.; Selnick, H. G.; Glass, K. L.; Rittle, K. E.; Gilbert, K. F.; Steele, T. G.; Homnick, C. F.; Freidinger, R. M.; Ransom, R. W.; Kling, P.; Reiss, D.; Broten, T. P.; Schorn, T. W.; Chang, R. S. L.; O’Malley, S. S.; Olah, T. V.; Ellis, J. D.; Barrish, A.; Kassahun, K.; Leppert, P.; Nagarathnam, D.; Forray, C. J. Med. Chem. 2000, 43, 2703.
- Mayer, T. U.; Kapoor, T. M.; Haggarty, S. J.; King, R. W.; Schreiber, S. L.; Mitchison, T. J. Science 1999, 286, 971.Haggarty, S. J.; Mayer, T. U.; Miyamoto, D. T.; Fathi, R.; King, R. W.; Mitchison, T. J.; Schreiber, S. L. Chem. Biol. 2000, 7, 275.
- Patil, A. D.; Kumar, N. V.; Kokke, W. C.; Bean, M. F.; Freyer, A. J.; De Brosse, C.; Mai, S.; Truneh, A.; Faulkner, D. J.; Carte, B.; Breen, A. L.; Hertzberg, R. P.; Johnson, R. K.; Westley, J. W.; Potts, B. C. M. J. Org. Chem. 1995, 60, 1182.
- (a) the use of water-based biphasic media, see: Bose, A. K.; Manhas, M. S.; Pednekar, S.; Ganguly, S. N.; Dang, H.; He, W.; Mandadi, A. Tetrahedron Lett. 2005, 46, 1901. (b) using Bi(NO3)3, see: Khodaei, M. M.; Khosropour, A. R.; Jowkar, M. Synthesis 2005, 1301. (c) using LiBr, see: Baruah, P. P.; Gadhwal, S.; Prajapati, D.; Sandhu, J. S. Chem. Lett. 2002, 1038. (d) using InBr3, see: Martins, M. A. P.; Teixeira, M. V. M.; Cunico, W.; Scapin, E.; Mayer, R.; Pereira, C. M. P.; Zanatta, N.; Bonacorso, H. G.; Peppe, C.; Yuan, Y.-F. Tetrahedron Lett. 2004, 45, 8991. (e) using I2, see: Bhosale, R. S.; Bhosale, S. V.; Bhosale, S. V.; Wang, T.; Zubaidha, P. K. Tetrahedron Lett. 2004, 45, 9111. (f) using FeCl3, see: Wang, Z.-T.; Xu, L.-W.; Xia, C.-G.; Wang, H.-Q. Tetrahedron Lett. 2004, 45, 7951. (g) using KHSO4, see: Tu, S.; Fang, F.; Zhu, S.; Li, T.; Zhang, X.; Zhuang, Q. Synlett 2004, 537. (h) using CdCl2, see: Narsaiah, A. V.; Basak, A. K.; Nagaiah, K. Synthesis 2004, 1253. (i) using ZnCl2 see: Sun, Q.; Wang, Y.-Q.; Ge, Z.-M.; Cheng, T.-M.; Li, R.-T. Synthesis 2004, 1047. (j) using Cu(OTf)2, see: Paraskar, A. S.; DewKar, G. K.; Sudalai, A. Tetrahedron Lett. 2003, 44, 3305. (k) using Me3SiI, see: Sabitha, G.; Reddy, G. S. K. K.; Reddy, C. S.; Yadav, J. S. Synlett 2003, 858. (l) using boric acid, see: Tu, S.; Fang, F.; Miao, C.; Jiang, H.; Feng, Y.; Shi, D.; Wang, X. Tetrahedron Lett. 2003, 44, 6153. (m) using ZrCl4, see: Reddy, C. V.; Mahesh, M.; Raju, P. V. K.; Romeshbabu, T.; Reddy, V. V. N. Tetrahedron Lett. 2002, 43, 2657.
- Lu, J.; Bai, Y.; Wang, Z.; Ma, B.; Yang, H. Tetrahedron Lett. 2000, 41, 9075.Ma, Y.; Qian, C.; Wang, L.; Yang, M. J. Org. Chem. 2000, 65, 3864.Bose, D. S.; Fatima, L.; Mereyala, H. B. J. Org. Chem. 2003, 68, 587.
- Kappe, C. O. J. Org. Chem. 1997, 62, 7201.
- Sample Availability: Samples are available from authors.
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Adib, M.; Ghanbary, K.; Mostofi, M.; Ganjali, M.R. Efficient Ce(NO3)3·6H2O-Catalyzed Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones. Molecules 2006, 11, 649-654. https://doi.org/10.3390/11080649
Adib M, Ghanbary K, Mostofi M, Ganjali MR. Efficient Ce(NO3)3·6H2O-Catalyzed Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones. Molecules. 2006; 11(8):649-654. https://doi.org/10.3390/11080649
Chicago/Turabian StyleAdib, Mehdi, Khadijeh Ghanbary, Manizheh Mostofi, and Mohammad Reza Ganjali. 2006. "Efficient Ce(NO3)3·6H2O-Catalyzed Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones" Molecules 11, no. 8: 649-654. https://doi.org/10.3390/11080649
APA StyleAdib, M., Ghanbary, K., Mostofi, M., & Ganjali, M. R. (2006). Efficient Ce(NO3)3·6H2O-Catalyzed Solvent-Free Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones. Molecules, 11(8), 649-654. https://doi.org/10.3390/11080649




