Synthesis and Antifungal Bioactivities of 3-Alkylquinazolin- 4-one Derivatives
Abstract
:Introduction
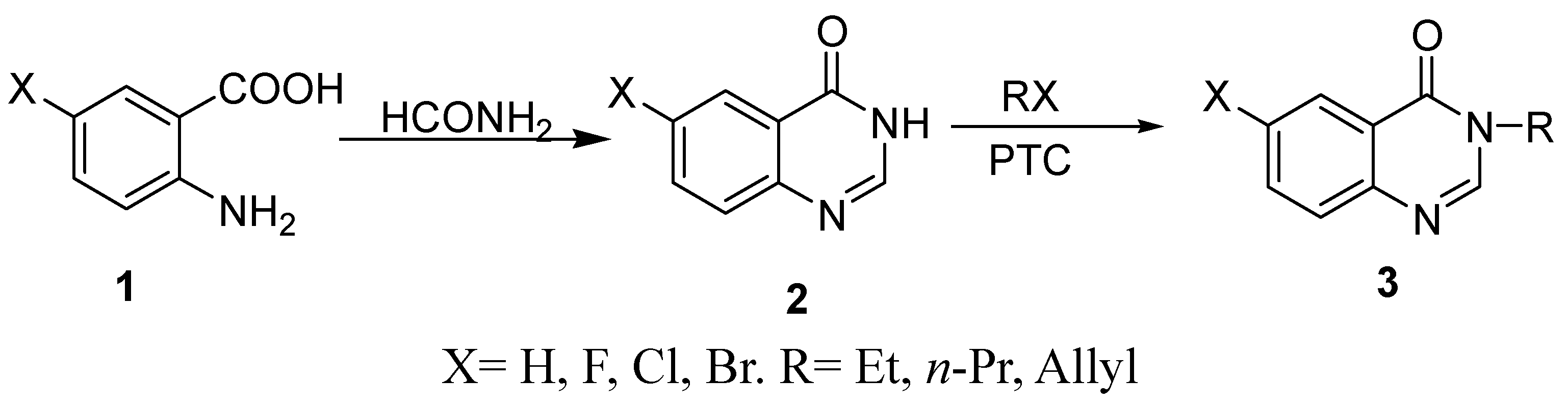
Results and Discussion
| Entry a | Reaction time / h | KOH concentration / % (w/w) | The amount of Bu4NBr / mol % | Reaction temperature / °C | Yield b / % |
| 1 | 1 | 30 | 3 | 88-90 | 38.8 |
| 2 | 1 | 30 | 5 | 88-90 | 62.2 |
| 3 | 1 | 30 | 8 | 88-90 | 77.1 |
| 4 | 1 | 30 | 10 | 88-90 | 77.4 |
| 5 | 1 | 30 | 0 | 88-90 | 8.5 |
| 6 | 1 | 10 | 8 | 88-90 | 48.5 |
| 7 | 1 | 20 | 8 | 88-90 | 69.1 |
| 8 | 1 | 35 | 8 | 88-90 | 83.0 |
| 9 | 1 | 40 | 8 | 88-90 | 74.0 |
| 10 | 1 | 35 | 8 | 20-25 | 4.3 |
| 11 | 1 | 35 | 8 | 40-45 | 13.3 |
| 12 | 1 | 35 | 8 | 60-65 | 65.0 |
| 13 | 2 | 35 | 8 | 88-90 | 83.8 |
| 14 | 0.75 | 35 | 8 | 88-90 | 74.5 |
| 15 | 0. 5 | 35 | 8 | 88-90 | 55.9 |
| Entry a | Reaction time / h | organic phase | The amount of solvent / mL | Yield b / % |
| 1 | 1 | CHCl3 | 10 | 25.5 |
| 2 | 1 | C6H6 | 10 | 45.2 |
| 3 | 1 | CH3COOEt | 10 | 54.3 |
| 4 | 1 | Toluene | 10 | 83.0 |
| Compd | 3a | 3b | 3c | 3d | 3e | 3f |
| X- | H | F | Cl | Br | H | F |
| R- | Et | Et | Et | Et | n-Pr | n-Pr |
| bYields /% | 79.5 | 74.5 | 74.5 | 77.0 | 83.0 | 92.2 |
| Compd | 3g | 3h | 3i | 3j | 3k | 3l |
| X- | Cl | Br | H | F | Cl | Br |
| R- | n-Pr | n-Pr | allyl | allyl | allyl | allyl |
| bYields /% | 83.1 | 87.0 | 70.0 | 72.7 | 82.5 | 70.9 |
Antifungal activity bioassay
| Compd (50 μg/mL) | Fusarium oxysporum | Gibberella zeae | Valsa mali |
| 3a | 17.9 | 17.6 | 8.7 |
| 3b | 14.2 | 11.8 | 6.5 |
| 3c | 37.5 | 55.0 | 24.4 |
| 3d | 33.3 | 35.4 | 21.7 |
| 3e | 21.2 | 17.3 | 16.9 |
| 3f | 4.9 | 17.0 | 14.5 |
| 3g | 20.4 | 5.5 | 15.9 |
| 3h | 47.2 | 50.3 | 40.9 |
| 3i | 15.3 | 19.9 | 0.9 |
| 3j | 13.9 | 26.7 | 13.4 |
| 3k | 36.4 | 49.1 | 28.3 |
| 3l | 32.4 | 37.0 | 30.4 |
| Hymexazol | 55.4 | 50.4 | 22.5 |
Conclusions
Experimental
General
Preparation of 3-Alkylquinazolin-4-one Derivatives 3a-3l
Bioassays
 , where A represents the diameter of fungi growth on untreated PDA, B represents the diameter of fungi on treated PDA, and CV represents the rate of inhibition.
, where A represents the diameter of fungi growth on untreated PDA, B represents the diameter of fungi on treated PDA, and CV represents the rate of inhibition.Acknowledgements
References
- Tobe, M.; Isobe, Y.; Tomizawa, H.; Nagasaki, T.; Obara, F.; Hayashi, H. Structure-Activity relationships of 6-fluoroquinazolines: Dual-acting compounds with inhibitory activities toward both TNF-α production and T cell proliferation. Bioorg. & Med. Chem. 2003, 11, 609–616. [Google Scholar]
- Lamberth, C.; Hillesheim, E.; Bassand, D.; Schaub, F. Synthesis and acaricidal activity of 4-pyrimidinyloxy- and 4-pyrimidinylaminoethylphenyl dioxolanes and oxime ethers. Pest Manag. Sci. 2000, 56, 94–100. [Google Scholar]
- Hung, R. Q.; Li, H.Y.; Ma, J. A.; Qiu, D. W. Synthesis of O-(4-Quinazolinyl)oxime Ethers and Their Antiviral Activity. Chem. J. Chin. Univ. 1996, 17, 571–575. [Google Scholar]
- Dandia, A.; Singh, R.; Sarawagi, P. Green chemical multi-component one-pot synthesis of fluorinated 2,3-disubstituted quinazolin-4(3H)-ones under solvent-free conditions and their anti-fungal activity. J. Fluorine Chem. 2004, 125, 1835–1840. [Google Scholar] [CrossRef]
- Khan, I. A.; Hassan, G.; Ihsanullah.; Khan, M. A. Efficacy of Post-emergence Herbicides for controlling Weeds in Canola. Asian J. Plant Sci. 2003, 2, 294–296. [Google Scholar] [CrossRef]
- Jin, Y.; Li, H.Y.; Lin, L. P.; Tan, J. Z.; Ding, J.; Luo, X. M.; Long, Y. Q. Synthesis and antitumor evaluation of novel 5-substituted-4-hydroxy-8-nitroquinazolines as EGFR signaling-targeted inhibitors. Bioorg. & Med. Chem. 2005, 13, 5613–5622. [Google Scholar]
- Wissner, A.; Berger, D. M.; Boschelli, D. H.; Floyd, M. B., Jr.; Greenberger, L. M.; Gruber, B. C.; Johnson, B. D; Mamuya, N.; Nilakantan, R.; Reich, M. F.; Shen, R.; Tsou, H. R.; Upeslacis, E.; Wang, Y. F.; Wu, B.; Ye, F.; Zhang, N. 4-Anilino-6,7-dialkoxyquinoline-3-carbonitrile Inhibitors of Epidermal Growth Factor Receptor Kinase and Their Bioisosteric Relationship to the 4-Anilino-6,7-dialkoxy- quinazoline Inhibitors. J. Med. Chem. 2000, 43, 3244–3256. [Google Scholar] [CrossRef]
- Maillard, J.; Morin, R. M.; Vincent, M. M. J.; Bernard, M. M. Quinazolone anti-inflammatory composition. US 3047462, 1962. [Chem. Abstr., 1963, 58, 1474g]. [Google Scholar]
- Liu, G.; Song, B. A.; Sang, W. J.; Yang, S.; Jin, L. H.; Ding, X. Synthesis and Bioactivity of N-Aryl-4-aminoquinazoline Compounds. Chin. J. Org. Chem. 2004, 24, 1296–1299. [Google Scholar]
- Chao, Q.; Deng, L.; Shih, H.; Leoni, L. M.; Genini, D.; Carson, D.A.; Cottam, H. B. Substituted isoquinolines and quinazolines as potential antiinflammatory agents: Synthesis and biological evaluation of inhibitors of tumor necrosis factor. J. Med. Chem. 1999, 42, 3860–3873. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Sun, X.-L.; Zhang, S.-Y. An improved Niementowski reaction. Chin. J. Org. Chem. 2001, 21, 1161–1163. [Google Scholar]
- El-Hiti, G. A. Application of Organolithium in Organic Synthesis: A Convenient Synthesis of More Complex 6-Substituted 3H-Quinazolin-4-ones. Monatsh. Chem. 2004, 135, 323–331. [Google Scholar]
- Aquila, B.; Dakin, L.; Ezhuthachan, J.; Lee, S.; Lyne, P.; Pontz, T.; Zheng, X,-L. Preparation of quinazolinone derivatives as B-Raf inhibitors. WO 2006024834, 2006. [Chem. Abstr., 2006, 144. 292772]. [Google Scholar]
- Song, S. Q.; Zhou, L. G.; Li, D.; Tang, D.; Li, J. Q.; Jiang, W. B. Antifungal activity of five plants from Xinjiang. Nat. Prod. Res. & Dev. 2004, 16, 157–159. [Google Scholar]
- Sample Availability: Samples of the compounds are available from authors.
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Ouyang, G.; Zhang, P.; Xu, G.; Song, B.; Yang, S.; Jin, L.; Xue, W.; Hu, D.; Lu, P.; Chen, Z. Synthesis and Antifungal Bioactivities of 3-Alkylquinazolin- 4-one Derivatives. Molecules 2006, 11, 383-392. https://doi.org/10.3390/11060383
Ouyang G, Zhang P, Xu G, Song B, Yang S, Jin L, Xue W, Hu D, Lu P, Chen Z. Synthesis and Antifungal Bioactivities of 3-Alkylquinazolin- 4-one Derivatives. Molecules. 2006; 11(6):383-392. https://doi.org/10.3390/11060383
Chicago/Turabian StyleOuyang, Guiping, Peiquan Zhang, Gangfang Xu, Baoan Song, Song Yang, Linhong Jin, Wei Xue, Deyu Hu, Ping Lu, and Zhuo Chen. 2006. "Synthesis and Antifungal Bioactivities of 3-Alkylquinazolin- 4-one Derivatives" Molecules 11, no. 6: 383-392. https://doi.org/10.3390/11060383
APA StyleOuyang, G., Zhang, P., Xu, G., Song, B., Yang, S., Jin, L., Xue, W., Hu, D., Lu, P., & Chen, Z. (2006). Synthesis and Antifungal Bioactivities of 3-Alkylquinazolin- 4-one Derivatives. Molecules, 11(6), 383-392. https://doi.org/10.3390/11060383





