Reactions of (Benzamidomethyl)triethylammonium Chloride with Some Inorganic Nucleophiles in Aqueous Media
Abstract
:Introduction
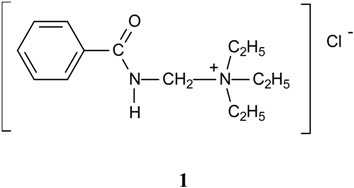
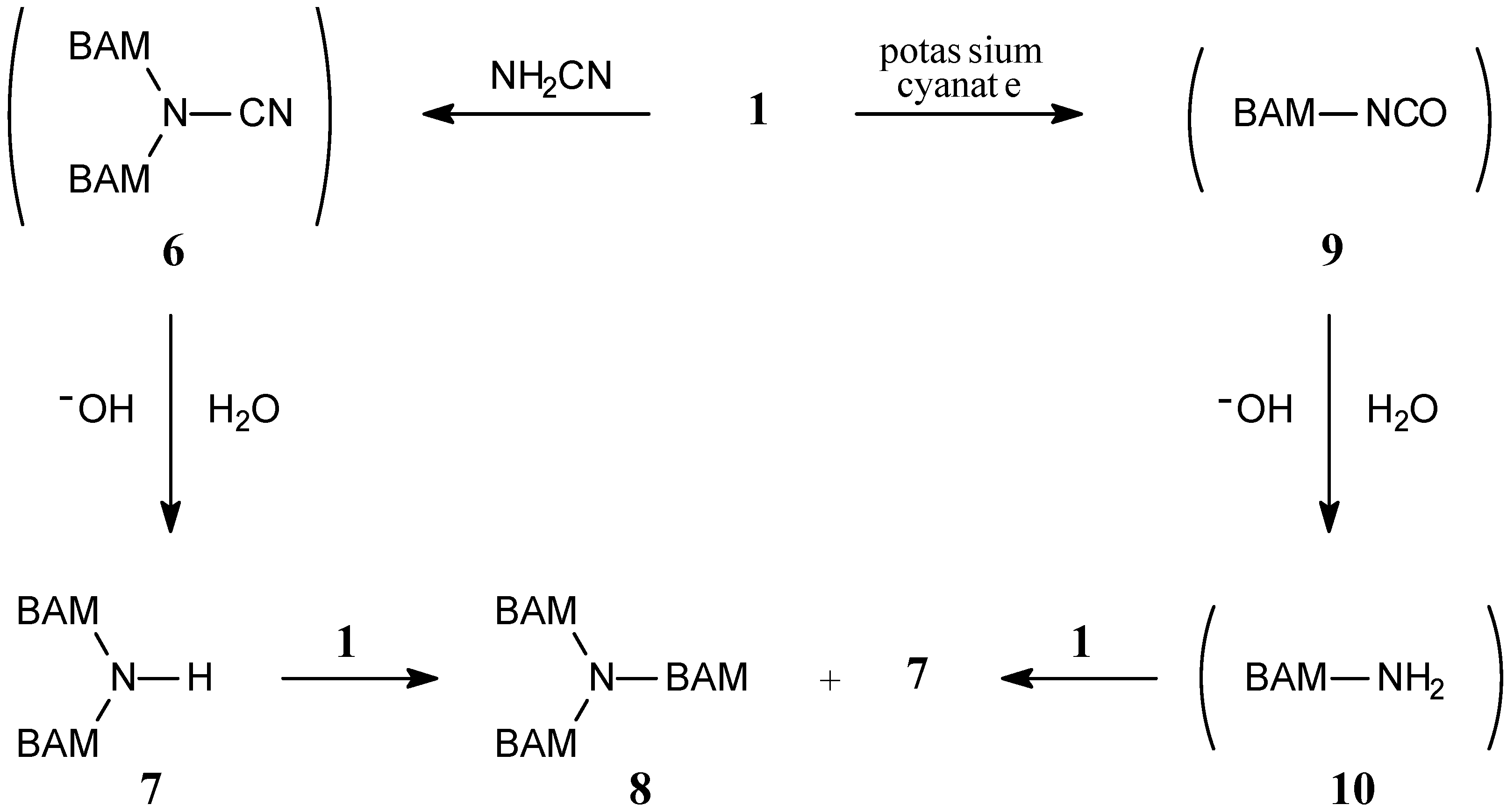
Results and Discussion
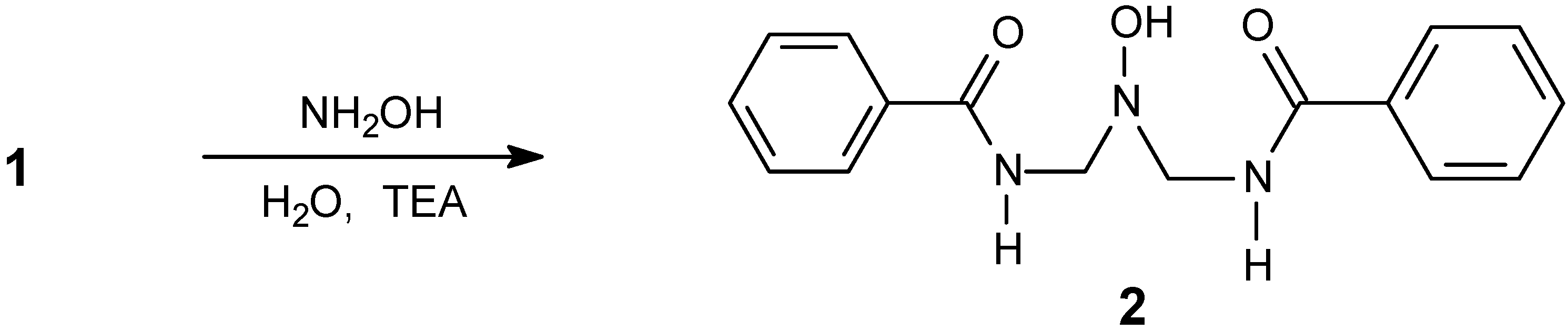
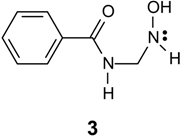
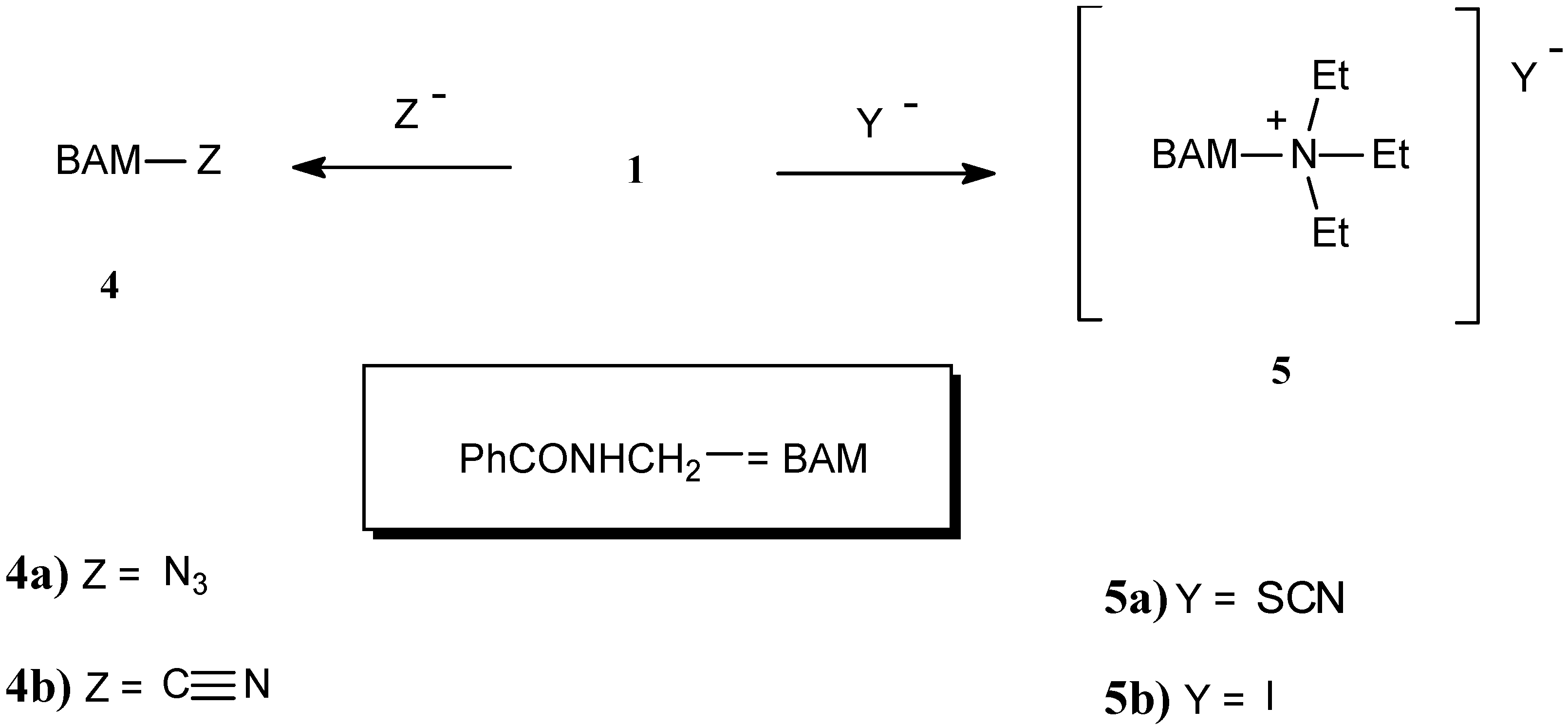
| Compound | Yield % | M.p. oC | Calc./found | FTIR (KBr) / cm-1 | |||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | N | νNHa | Amide I | Amide II | Other | |||
| 2 | 63 | 140-143 | 64.2 64.0 | 5.7 5.9 | 14.0 13.8 | 3315 3262 | 1639 1628 | 1549 1540 | |
| 4a | 48 | 62 | 54.5 54.3 | 4.6 4.2 | 31.8 31.6 | 3299 | 1645 | 1528 | 2125 and 2085 ν(N3) |
| 4b | 73 | 143-144b | 67.5 67.7 | 5.0 5.3 | 17.5 17.3 | 3244 | 1640 | 1538 | absence of ν(C≡N) bond |
| 5a | 96 | 127-128 | 61.4 61.3 | 7.9 8.2 | 14.3 14.0 | 3229 3180 | 1671 | 1545 | 2039 ν(SCN ¯ ) |
| 5b | 65 | 165 | 46.4 46.1 | 6.4 6.6 | 7.7 7.5 | 3207 3178 | 1678 | 1523 | |
Conclusions
| Comp. | 1H-NMR (DMSO-d6; δ in ppm) | 13C-NMR (DMSO-d6; δ in ppm) | |||
|---|---|---|---|---|---|
| CONH | Aromatic | NHCH2 | Other | ||
| 2 | s(t), 8.87 2H | m, 7.94-7.46 10H | d, 4.32 4H, J 4.2 | s, 8.28, 1H OH | 166.81 C=O; 60.00 CH2; Ar: 134.25; 131.34; 128.27; 127.38 |
| 4a | t, 9.55 1H, J 6.0 | m, 7.91-7.49 5H | d, 4.74 2H, J 6.3 | - | 167.35 C=O; 55.53 CH2;
Ar: 133.06; 132.14; 128.65; 127.49 |
| 4b | t, 8.87 1H, J 4.9 | m, 7.90-7.46 5H | d, 4.34 2H, J 5.6 | - | 166.79 C=O; 117.75 C≡N; 27.85 CH2;
Ar: 132.94; 132.02; 128.59; 127.45 |
| 5a | t, 9.40 1H, J 7.0 | m, 7.96-7.53 5H | d, 4.74 2H, J 7.2 | q, 3.23, 6H, 3 × CH2, J 7.2 t, 1.27, 9H 3 × CH3, J 7.1 | 168.64 C=O; 129.73 SCN; 59.68 N-CH2-N; 50.10 N-
CH2-C; 7.36 CH3 Ar: 132.61; 132.52; 128.45; 128.32 |
| 5b | t, 9.38 1H, J 7.0 | m, 7.96-7.52 5H | d, 4.74 2H, J 7.0 | q, 3.23, 6H, 3 × CH2, J 7.0 t, 1.26, 9H 3 × CH3, J 7.0 | 168.61 C=O; 59.69 N-CH2-N;
50.08 N-CH2-C; 7.39 CH3 Ar: 132.62; 132.52; 128.42; 128.14 |
Experimental Section
General
N,N-Di(benzamidomethyl)hydroxylamine (2).
Benzamidomethyl azide (4a).
Benzamidomethyl cyanide (4b).
Acknowledgements
References and Notes
- Jones, R. A. Quaternary Ammonium Salts: Their Use in Phase-Transfer Catalysed Reactions; Academic Press: San Diego, 2001; pp. 1–8. [Google Scholar]
- Popovski, E.; Klisarova, L.; Vikic-Topic, D. Simple Method for Benzamidomethylation of Phenols in Water Solution. Synth. Commun. 1999, 29, 3451–3458. [Google Scholar] [CrossRef]
- Popovski, E.; Klisarova, L.; Vikic-Topic, D. Benzamidomethylation with (Benzamidomethyl)-triethylammonium Chloride. 2. A Simple Method for Benzamidomethylation of Thiols, Amines and Carboxylic acids. Molecules 2000, 5, 927–936. [Google Scholar]
- Silverstein, R. M.; Bassler, G. C.; Morrill, T. C. Spectrometric Identification of Organic Compounds, 5th ed.; Wiley: New York, 1991; p. 126. [Google Scholar]
- Klages, A. Ueber Das Methylenamino-acetonitril. Chem Ber 1903, 36, 1506–1512. [Google Scholar] [CrossRef]
- Martin, A. R.; Ketcham, R. Heterocyclic Ring-Closure Reactions. I. A Novel Oxazole Synthesis from S,S'-Dialkyl or Diaryl Dithiooxaldiimidates and Aromatic Aldehydes. J. Org. Chem. 1966, 31, 3612–3615. [Google Scholar]
- Sun, Y. Z.; Martell, A. E.; Welch, M. J. Synthesis of a New Diaminodithiol Bifunctional Chelator for Radiolabeling Biomolecules with Indium(III). Tetrahedron 2000, 56, 5093–5103. [Google Scholar] [CrossRef]
- Popovski, E. unpublished results.
- Loos, R.; Kobayashi, S.; Mayr, H. Ambident Reactivity of the Thiocyanate Anion Revisited: Can the Product Ratio be Explained by the Hard Soft Acid Base Principle? J. Am. Chem. Soc. 2003, 125, 14126–14132. [Google Scholar] [CrossRef]
- The IR band (KBr disk) of the SCN anion of tetrabutyilammonium thiocyanate is at 2059 cm–1 which is comparable to 2039 cm-1 of 5b. The 13C-NMR chemical shift (CDCl3) of the SCN anion of tetrabutylammonium thiocyanate is at 129.73 ppm vs. 130.94 ppm for 5b. The spectral data for tetrabutylammonium thiocyanate were obtained from: SDBS Integrated Spectral Data Base System for Organic Compounds. http://www.aist.go.jp/RIODB/SDBS, National Institute of Advanced Industrial Science and Technology, 2/22/2006.
- Ritchie, C. D. Cation Anion Combination Reactions .23. Solvent Effects on Rates and Equilibria of Reactions. J. Am. Chem. Soc. 1983, 105, 3573–3578. [Google Scholar]
- Pearson, R. G.; Songstad, J. Application of Principle of Hard and Soft Acids and Bases to Organic Chemistry. J. Am. Chem. Soc. 1967, 89, 1827–1836. [Google Scholar] [CrossRef]
- Ohtani, N.; Murakawa, S.; Watanabe, K.; Tsuchimoto, D.; Sato, D. Thiocyanation of Alkyl Halides with Alkyl Thiocyanates in the Presence of Quaternary Phosphonium Halides. J. Chem. Soc.-Perkin Trans. 2 2000, 1851–1856. [Google Scholar]
- Noller, C. R. Chemistry of Organic Compounds, 2nd ed.; Saunders: Philadelphia, 1957; pp. 320–321. [Google Scholar]
- March, J. Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 4th ed.; Wiley: New York, 1992; pp. 429, 886. [Google Scholar]
- Sample Availability: Samples of the compounds are available from authors.
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Popovski, E.; Bogdanov, J.; Najdoski, M.; Hey-Hawkins, E. Reactions of (Benzamidomethyl)triethylammonium Chloride with Some Inorganic Nucleophiles in Aqueous Media. Molecules 2006, 11, 279-285. https://doi.org/10.3390/11040279
Popovski E, Bogdanov J, Najdoski M, Hey-Hawkins E. Reactions of (Benzamidomethyl)triethylammonium Chloride with Some Inorganic Nucleophiles in Aqueous Media. Molecules. 2006; 11(4):279-285. https://doi.org/10.3390/11040279
Chicago/Turabian StylePopovski, Emil, Jane Bogdanov, Metodija Najdoski, and Evamarie Hey-Hawkins. 2006. "Reactions of (Benzamidomethyl)triethylammonium Chloride with Some Inorganic Nucleophiles in Aqueous Media" Molecules 11, no. 4: 279-285. https://doi.org/10.3390/11040279
APA StylePopovski, E., Bogdanov, J., Najdoski, M., & Hey-Hawkins, E. (2006). Reactions of (Benzamidomethyl)triethylammonium Chloride with Some Inorganic Nucleophiles in Aqueous Media. Molecules, 11(4), 279-285. https://doi.org/10.3390/11040279




