Results and Discussion
The
C-aryl-glycosides are a family of
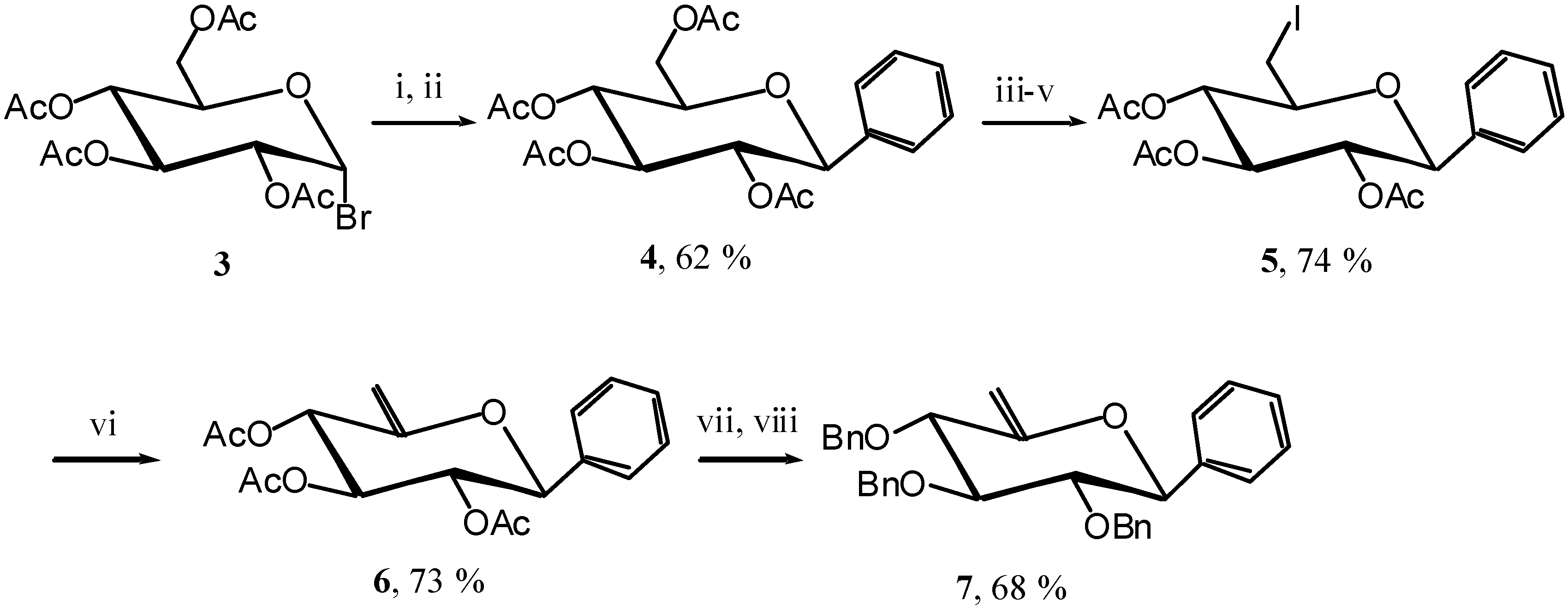
C-glycosides possessing an electron-donating aglycon, and their carbocyclisation could constitute a new entry into pancratistatine and similar structures. To test this hypothesis we first synthesised the unsaturated
C-phenyl glycoside
7. Starting from acetobromoglucose
3 [
5] we introduced the phenyl group using a Grignard reagent and then reacetylated to obtain the
C-glucoside
4 [
6]. Selective iodination, elimination, and protecting group exchange afforded the unsaturated C-phenyl glucoside
7 (
Scheme 3).
Scheme 3.
Reagents and conditions: i) PhMgBr, Et2O, reflux, 1 hour; ii) Ac2O, Pyr., R.T., 12 hours; iii) MeONa, MeOH, R.T., 5 hours; iv) I2, PPh3, DMF, R.T., 45 min.; v) Ac2O, Pyr., R.T., 4 hours; vi) DBU, THF, reflux, 3 hours; vii) MeONa, MeOH, 12 hours; viii) BnBr, NaH, DMF, R.T., 5 hours.
Compound
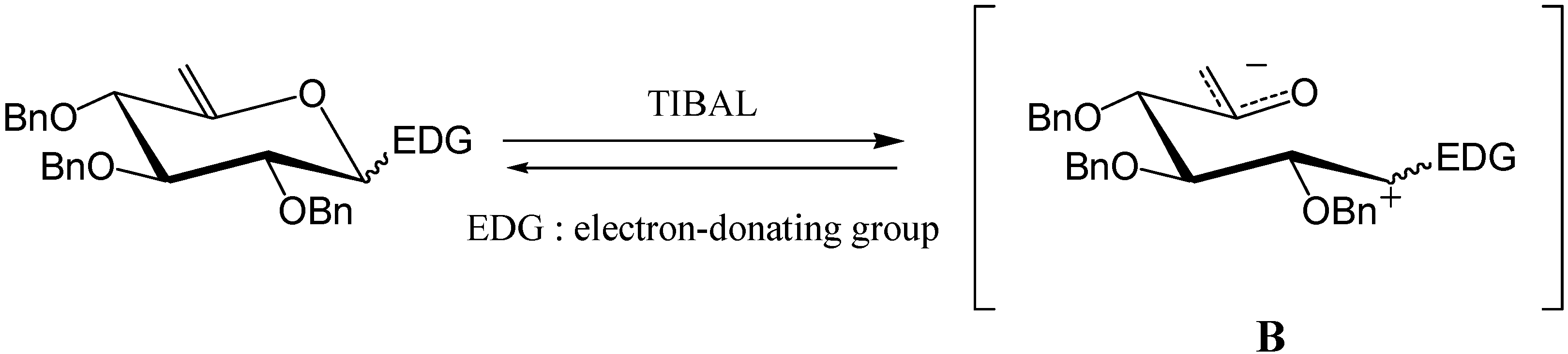
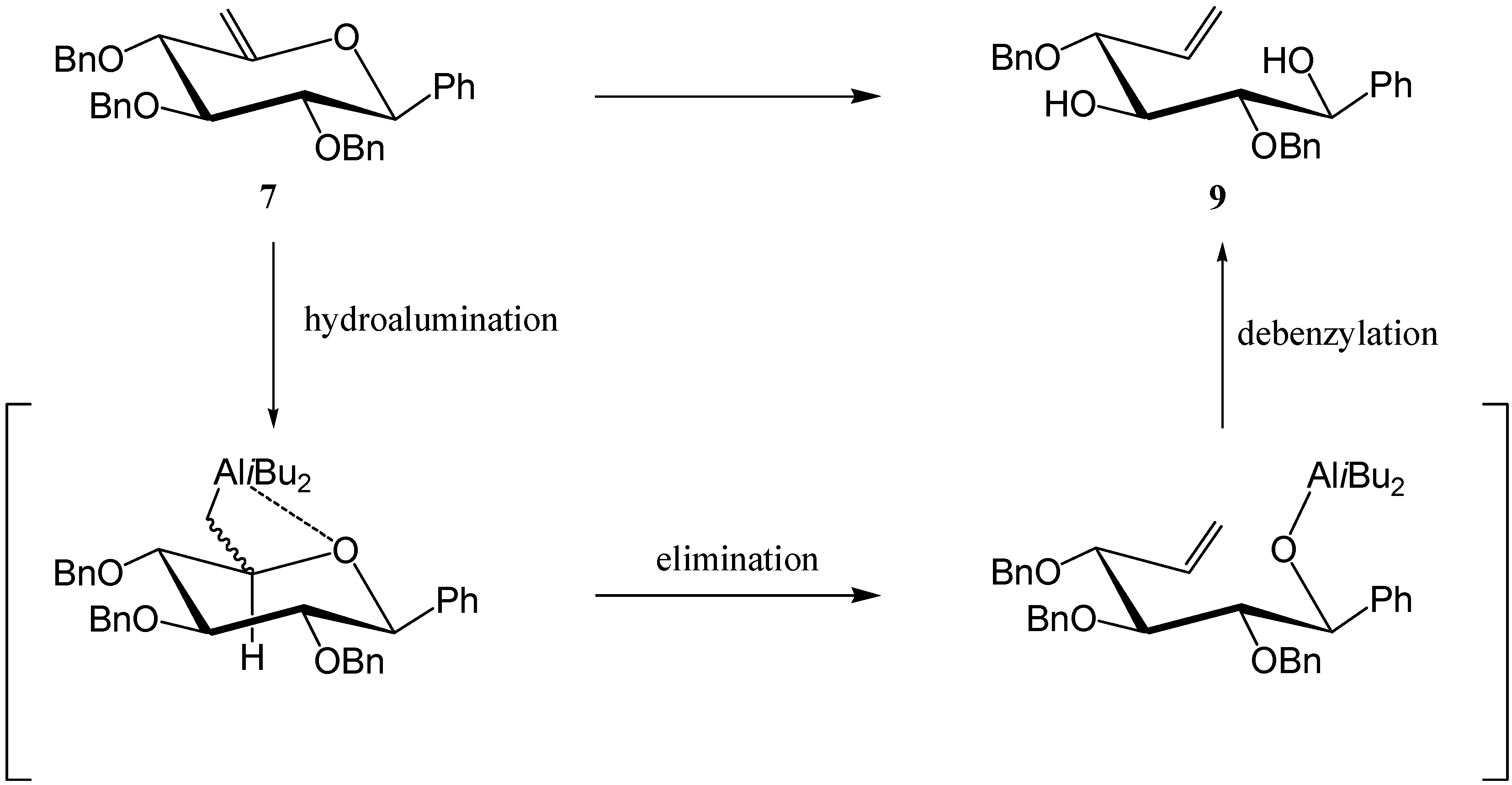
7 was then reacted with TIBAL and gave the desired carbocycle
8 (35 %) along with the de-
O-benzylated [
7] open-chain product
9 (35 %).
Scheme 4.
Reagents and conditions: i) TIBAL (10 eq.), Toluene, 50 °C, 12 hours
Product
9 is the result of an overall reductive cleavage of the endocyclic C5-O bond, a reaction which has already been described in enol ethers [
8,
9]. A hydro-alumination-elimination mechanism, as shown in
Scheme 5, may explain this process.
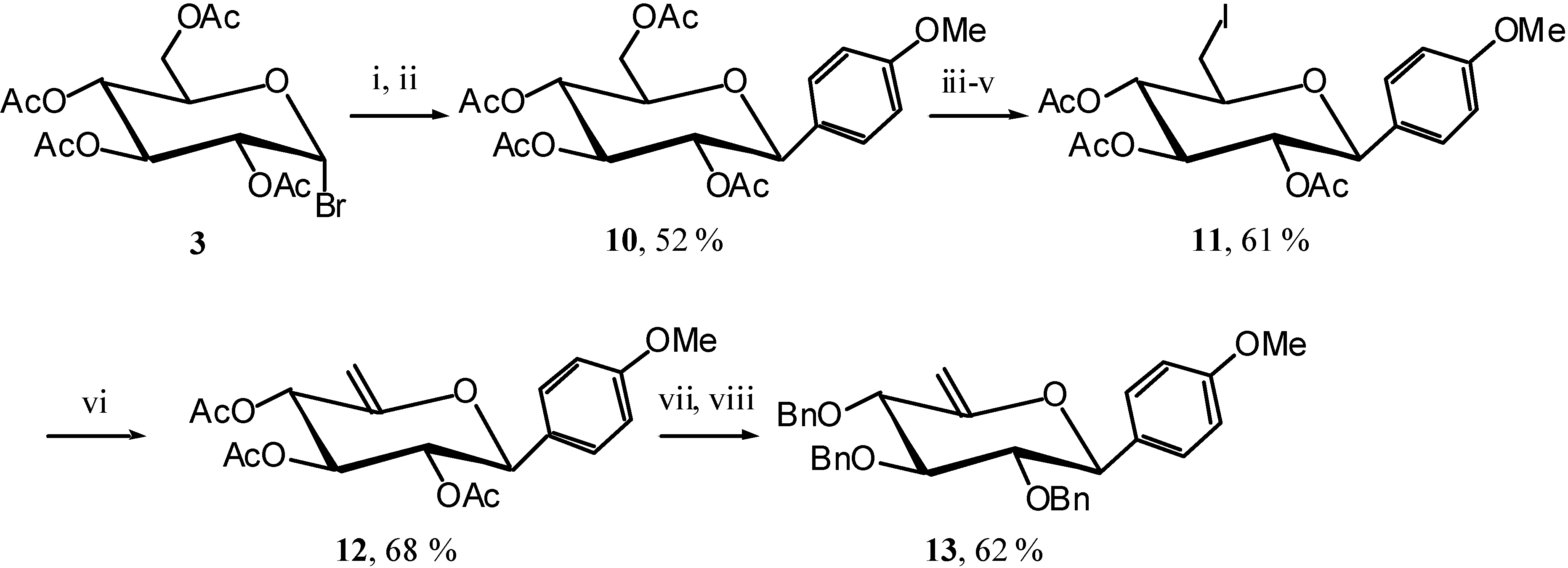
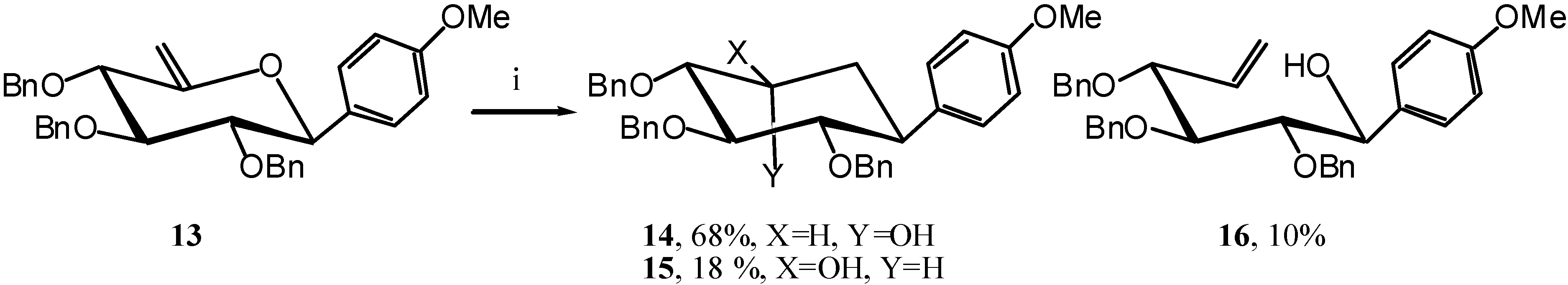
The reaction was here slower than usual, indicating that the phenyl might not be a strong enough EDG. We therefore turned our attention to anisole as EDG and synthesised unsaturated compound
13 in the same manner as
7 via the known
10 [
10]. (
Scheme 6)
Scheme 6.
Reagents and conditions: i) (MeO)PhMgBr, Et2O, reflux, 1 hour; ii) Ac2O, Pyr., R.T., 12 hours; iii) MeONa, MeOH, R.T., 5 hours; iv) I2, PPh3, DMF, R.T., 45 min.; v) Ac2O, Pyr., R.T., 8 hours; vi) DBU, THF, reflux, 3 hours; vii) MeONa, MeOH, 12 hours; viii) BnBr, NaH, DMF, R.T., 5 hours.
Under the action of TIBAL compound
13 only gave 10 % of the open-chain product
16 and 86 % of a mixture of expected carbocycles
14 and
15 in a 4/1 ratio. (
Scheme 7)
Scheme 7.
Reagents and conditions: i) TIBAL (6 eq.), Toluene, 50 °C, 1 hour
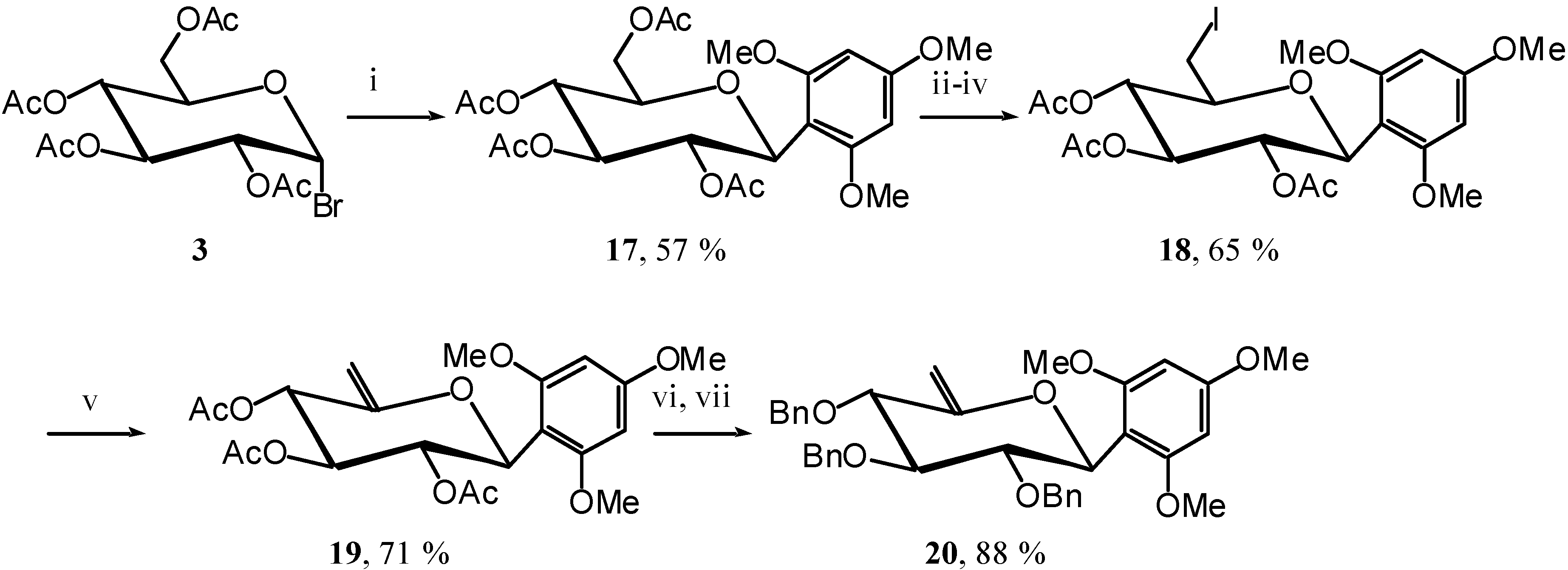
To achieve the demonstration that a better EDG gives a better reaction we synthesised compound
20, using again the same methodology, except for the
C-glycosylation of trimethoxybenzene to give
C-glucoside
17 [
11]. (
Scheme 8)
Scheme 8.
Reagents and conditions: i) Ph(OMe)3, ZnO, M.S., ClCH2CH2Cl, reflux, 30 min.; ii) MeONa, MeOH, R.T., 5 hours; iii) I2, PPh3, DMF, R.T., 45 min.; iv) Ac2O, Pyr., R.T., 4 hours; v) DBU, THF, reflux, 2 hours; vi) MeONa, MeOH, 12 hours; vii) BnBr, NaH, DMF, R.T., 2 hours.
Indeed, when compound
20 is submitted to the action of TIBAL, carbocyclic products
21 and
22 are the only ones obtained in a 3:2 molar ratio. (
Scheme 9)
Scheme 9.
Reagents and conditions: i) TIBAL (5 eq.), Toluene, 50 °C, 30 min.
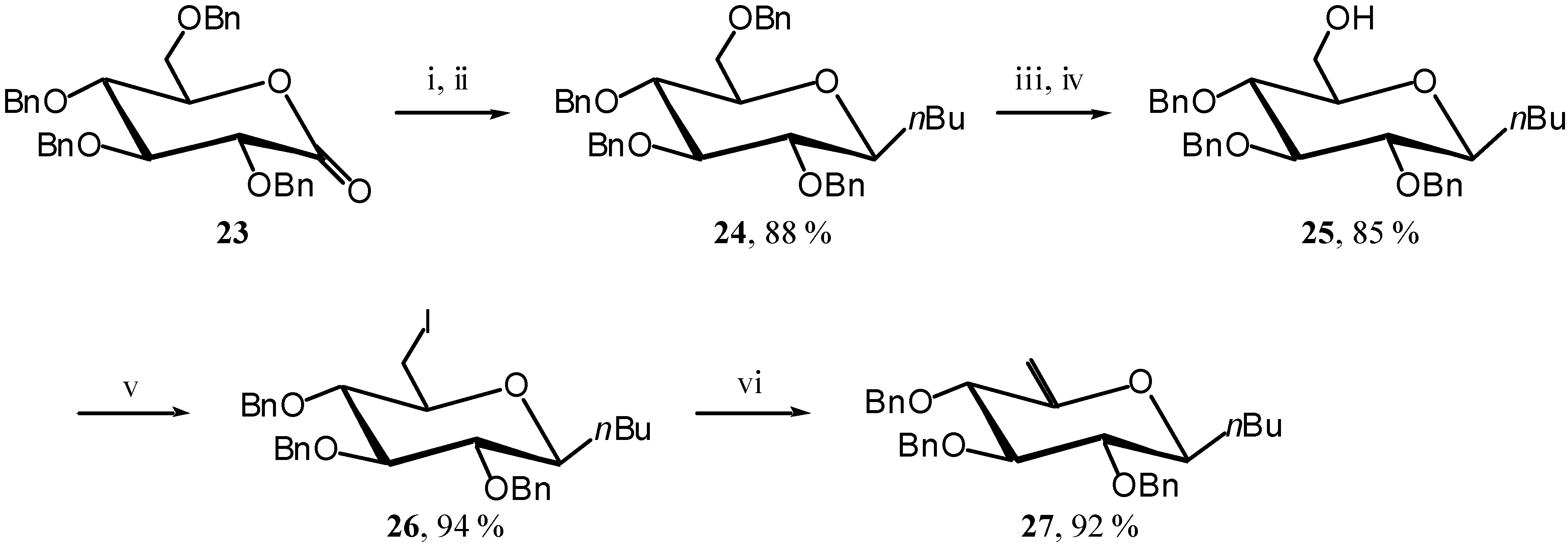
To really complete the demonstration, we also need to show that a weak EDG only induces the ring opening. To that end we selected the known
C-butyl unsaturated derivative
27 [
12]. Its synthesis, shown in
Scheme 10, is slightly different from the previous ones and starts from lactone
23 [
13] (
Scheme 10).
Scheme 10.
Reagents and conditions: i) BuLi, Et2O, -78 °C, 30 min.; ii) Et3SiH, BF3.OEt2, CH2Cl2, -78 °C, 30 min.; iii) TFA, Ac2O, R.T., 2 hours, iv) MeOH, MeONa, R.T., 10 min.; v) I2, PPh3, Imidazole, toluene, 70 °C, 30 min.; vi) DBU, THF, 70 °C, 1 hour.
Compound
27 reacts with TIBAL to give a single open-chain product
28 [14], hence achieving our demonstration. (
Scheme 11)
Scheme 11.
Reagents and conditions: i)TIBAL (5 eq.), toluene, 50 °C, 2 hours 30 min.
Experimental Section
General
Melting points were recorded on a Büchi 510 apparatus and are uncorrected. Optical rotations were measured on a Perkin Elmer 241 digital polarimeter with a path length of 1 dm. Mass spectra were recorded on a Nermag R10-10 spectrometer, using chemical ionisation with ammonia. Elemental analyses were performed by the Service d'Analyse de l'Université Pierre et Marie Curie, 75252 Paris Cedex 05, France. NMR spectra were recorded on a Brüker AM-400 (400 MHz and 100.6 MHz, for 1H and 13C, respectively) or Brüker AC-250 (250 MHz and 63 MHz, for 1H and 13C, respectively) using CDCl3 as solvent and TMS as internal standard. TLC was performed on silica gel 60 F254 (Merck) and developed by charring with conc. H2SO4. Flash column chromatography was performed using silica gel 60 (230-400 mesh, Merck).
(2,3,4-Tri-O-acetyl-6-deoxy-6-iodo-β-D-glucopyranosyl) benzene (5)
A catalytic amount of sodium is added to a solution 4 (3.74 g, 6.7 mmol) in methanol (40 mL), and the solution is stirred for 5 hours. It is then neutralised using IR 120 (H+) resin, filtered and evaporated in vacuo. The residue is dissolved in anhydrous DMF (15 mL), then PPh3 (4.8 g, 13.4 mmol) is added and the solution is cooled to 0 °C under argon. A solution of diiodine (4.6 g, 13.4 mmol) in anhydrous DMF (5 mL) is then added dropwise and the solution stirred for 45 min at R.T, until the completion of the reaction is detected by TLC (9:1 dichloromethane/MeOH). The solvent is evaporated and the residue is next dissolved in pyridine (20 mL) and acetic anhydride (10 mL). The reaction, monitored by TLC (7:3 cyclohexane/AcOEt), is complete in 4 hours at R.T. The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:4 AcOEt/cyclohexane) to afford compound 5 (2.9 g, 74 %) as a white solid. = + 6 (c = 1.25, CHCl3); m.p. = 162-163 °C (AcOEt/cyclohexane); 1H-NMR (250 MHz): δ = 7.31-7.20 (5H, H arom.), 5.29 (t, 1H, J2,3 = 9.4 Hz, J3,4 = 9.4 Hz, H-3), 5.06 (dd, 1H, J4,5 = 9.4 Hz, H-4), 5.05 (dd, 1H, J1,2 = 9.7 Hz, H-2), 4.38 (d, 1H, H-1), 3.46 (ddd, 1H, J5,6a = 3.0 Hz, J6b,5 = 5.7 Hz, H-5), 3.34 (dd, 1H, J6b,6a = 11.1 Hz, H-6a), 3.16 (dd, 1H, H-6b), 2.02, 1.94, 1.74 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 170.2, 169.2, 168.6 (3 x Ac), 136.1 (C arom. quat.), 128.7, 128.3, 126.9 (5 C arom.), 79.6 (C-1), 76.3 (C-5), 73.7 (C-3), 72.7 (C-2), 72.4 (C-4), 20.6, 20.5, 20.2 (3 x Ac), 3.9 (C-6); MS m/z 494 (M+NH4)+; Anal. calc. C, 45.39; H, 4.44; found. C, 45.51; H, 4.51.
(2,3,4-Tri-O-acetyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) benzene (6)
Compound 5 (2.9 g, 6.7 mmol) is dissolved in dry THF (30 mL) and DBU (6.1 mL, 40.6 mmol) is added. The mixture is heated at 70 °C until after 3 hours the end of the reaction is detected by TLC (7:3 cyclohexane/AcOEt). The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:5 AcOEt/cyclohexane) to afford compound 6 (1.7 g, 73 %) as a white solid. = -53 (c = 1.2, CHCl3); m.p. = 132 °C (AcOEt/cyclohexane); 1H-NMR (250 MHz): δ = 7.32-7.22 (5H, H arom.), 5.59-5.51 (m, 1H, H-4), 5.29-5.12 (m, 2H, H-3, H-2), 4.79 (s, 1H, H-6a), 4.51 (s, 1H, H-6b), 4.49-4.39 (m, 1H, H-1), 2.09, 1.96, 1.74 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 170.1, 169.3, 168.8 (3 x Ac), 153.8 (C-5), 135.9 (C arom. quat.), 129.1, 128.5, 127.1 (5 C arom.), 96.5 (C-6), 80.8 (C-1), 73.4 (C-3), 72.7 (C-2), 69.6 (C-4), 20.7, 20.6, 20.3 (3 x Ac); MS: m/z 366 (M+NH4)+; 349 (M+H)+; Anal. calc. C, 62.06; H, 5.79; found. C, 62.05; H, 5.88.
(2,3,4-Tri-O-benzyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) benzene (7)
A catalytic amount of sodium is added to a solution 6 (1.1 g, 3.2 mmol) in methanol (10 mL) and the solution is stirred for 12 hours. The solvent is evaporated without neutralisation and the residue dissolved in dry DMF (100 mL) to which benzyl bromide (4.4 mL, 32 mmol) and NaH (0.9 g, 60% in suspension in oil, 19.2 mmol) are added. The mixture is stirred at R.T. until the end of the reaction is detected after 5 hours by TLC (9:1 cyclohexane/AcOEt ). Excess NaH is quenched with methanol, and the mixture is extracted with ether and washed with water. The organic layer is dried (MgSO4), filtered and concentrated. The residue is purified by silica gel flash column chromatography (1:99 AcOEt/cyclohexane) to afford compound 7 (1.05 g, 68 %) as a white solid. = -51 (c = 1.1, CHCl3); m.p. = 71 °C (AcOEt/cyclohexane); 1H-NMR (250 MHz): δ = 7.31-6.92 (20H, H arom.), 4.70 (d, 1H, J = 11.3 Hz, -CHPh), 4.65 (d, 1H, J = 11.7 Hz, -CHPh), 4.63 (s, 1H, H-6a), 4.59 (d, 1H, J = 11.2 Hz, -CHPh), 4.53 (d, 1H, J = 11.5 Hz, -CHPh), 4.53 (s, 1H, H-6b), 4.37 (d, 1H, J1,2 = 9.7 Hz, H-1), 4.19 (d, 1H, J = 10.5 Hz, -CHPh), 3.91 (d, 1H, J4,3 = 7.4 Hz, H-4), 3.64 (d, 1H, J = 11.9 Hz, -CHPh), 3.62 (t, 1H, J2,3 = 7.7 Hz, H-3), 3.47 (dd, 1H, H-2); 13C-NMR (63 MHz): δ = 156.6 (C-5), 138.7, 138.5, 138.0, 137.7 (4 C arom. quat.), 128.6-127.7 (20 C arom.), 95.2 (C-6), 84.7 (C-3), 83.7 (C-2), 81.3 (C-1), 79.3 (C-4), 74.7, 74.6, 73.0 (3 CHPh); MS :m/z 510 (M+NH4)+; Anal. calc. C, 80.46; H, 6.64; found. C, 80.45; H, 6.59.
(1R,2S,3S,4R,5R)-2,3,4-Tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (8) and 2,4-di-O-benzyl- 5,6-dehydro-1-C(S)-phenyl-D-glucitol (9)
A solution of TIBAL (0.6 mL, 0.6 mmol, 1M in toluene) is added to a solution of 7 (55 mg, 0.11 mmol) in dry toluene (1 mL) at R.T. under argon. The mixture is stirred at 50 °C for 12 hours, until the end of the reaction is detected by TLC (4:1 cyclohexane/AcOEt). The mixture is cooled to R.T. and water (2 mL) is slowly added, and stirred for 15 min. After extraction with AcOEt (3 x 10 mL) and washing with water (10 mL) the organic layer is dried (MgSO4), filtered and the solvent is evaporated. The residue purified by silica gel flash column chromatography (1:9 AcOEt/cyclohexane) to afford two major products 8 (18 mg, 35 %), as a white solid, and 9 (16 mg, 35 %) as an oil.
(1R,2S,3S,4R,5R)-2,3,4-Tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (8)
= -24 (c = 0.6, CHCl
3); m.p. = 96 °C (AcOEt/cyclohexane);
1H-NMR (250 MHz): δ = 7.42-6.90 (20H, arom. H), 4.85 (d, 1H,
J = 10.8 Hz, -CHPh), 4.79 (d, 1H,
J = 10.8 Hz, -CHPh), 4.72 (d, 1H, J = 11.5 Hz, -CHPh), 4.64 (d, 1H, J = 11.5 Hz, -CHPh), 4.41 (d, 1H, J = 10.2 Hz, -CHPh), 4.17-4.09 (m, 1H, H-5), 3.88 (t, 1H, J
2,3 = 9.2 Hz, J
3,4 = 9.2 Hz, H-3), 3.82 (d, 1H, J = 10.2 Hz, -CHPh), 3.53 (dd, 1H, J
4,5 = 3.0 Hz, H-4), 3.47 (dd, 1H, J
1,2 = 9.1 Hz, H-2), 3.27 (ddd, 1H, J
1,6a = 11.0 Hz, J
1,6e = 3.7 Hz, H-1), 2.50 (br s, 1H, -OH), 2.04 (dt, 1H, J
6e,6a = 14.5 Hz, J
6e,5 = 3.7 Hz, H-6e), 1.65 (br t, 1H, H-6a);
13C-NMR (63 MHz):δ = 142.4, 139.0, 138.3, 138.1 (4 C arom. quat.), 128.6-126.7 (20 C arom.), 86.0 (C-2), 83.4 (C-3), 83.3 (C-4), 76.0, 75.4, 72.8 (3 CHPh), 66.8 (C-5), 42.8 (C-1), 34.3 (C-6); MS: m/z 512 (M+NH
4)
+; Anal. calc. C, 80.13; H, 6.93; found. C, 79.95; H, 7.08.
2,4-Di-O-benzyl-5,6-dehydro-1-C(S)-phenyl-D-glucitol (9)
= -2 (c = 0.3, CHCl
3);
1H-NMR (400 MHz): δ = 7.39-7.29 (15H, arom. H), 5.54 (ddd, 1H, J
5,6a = 10.5 Hz, J
5,6b = 17 Hz, J
5,4 = 8 Hz, H-5), 5.33 (dd, 1H, J
6a,6b = 1.5 Hz, H-6a), 5.23 (dd, 1H, H-6b), 5.20 (dd, 1H,
J1,2 = 5.1 Hz,
J1,OH = 6.7 Hz, H-1), 4.60 (d, 1H,
J = 11.3 Hz, -CHPh), 4.58 (d, 1H,
J = 11.2 Hz, -CHPh), 4.46 (d, 1H,
J = 11.3 Hz, -CHPh), 4.34 (d, 1H,
J = 11.2 Hz, -CHPh), 4.06 (t, 1H,
J4,3 = 8 Hz, H-4), 3.94 (d, 1H,
J = 6.7 Hz, 1-OH), 3.72 (ddd,
J3,2 = 1.8 Hz,
J3,OH = 3.2 Hz, H-3), 3.60 (dd, 1H, H-2), 3.12 (d, 1H, 3-OH);
13C-NMR (100 MHz): δ = 141.6, 137.7, 137.6 (3 C arom. quat.), 134.3 (C-5), 128.5-126.1 (15 C arom.), 120.9 (C-6), 81.3 (C-4), 79.4 (C-2), 73.0 (C-1), 72.9 (C-3), 71.7, 70.6 (2 CHPh); MS: m/z 422 (M+NH
4)
+; Anal. calc. C, 77.20; H, 6.98; found. C, 77.56; H, 7.09.
4-(2,3,4-Tetra-O-acetyl-6-desoxy-6-iodo-β-D-glucopyranosyl) anisole (11)
A catalytic amount of sodium is added to a solution 10 (3.7 g, 8.4 mmol) in methanol (50 mL), and the solution is stirred for 12 hours. It is then neutralised using IR 120 (H+) resin, filtered and evaporated in vacuo. The residue is dissolved in anhydrous DMF (20 mL), then PPh3 (4.7 g, 17.9 mmol) is added, and the solution is cooled to 0 °C under argon. A solution of diiodine (4.5 g, 17.9 mmol) in anhydrous DMF (5 mL) is then added dropwise and the solution stirred for 30 min at R.T., until the end of the reaction is detected by TLC (9:1 dichloromethane/MeOH). The solvent is evaporated and the residue is dissolved in pyridine (20 mL) and acetic anhydride (10 mL). The reaction is monitored by TLC (7:3 cyclohexane/AcOEt) for 4 hours at R.T. The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:4 AcOEt/cyclohexane) to afford compound 11 (2.6 g, 61 %) as a white solid. = -8 (c = 1.8, CHCl3); m.p. = 99 °C (EtOH); 1H-NMR (250 MHz): δ = 7.21 (d, 2H, J = 8.8 Hz, 2 H arom.), 6.81 (d, 2H, J = 8.8 Hz, 2 H arom.), 5.29 (t, 1H, J2,3 = 9.4 Hz, J3,4 = 9.4 Hz, H-3), 5.12-4.98 (m, 2H, H-4 H-2), 4.33 (d, 1H, J1,2 = 9.7 Hz, H-1), 3.73 (s, 3H, OMe), 3.44 (ddd, 1H, J5,6a = 3.0, J6b,5 = 5.7 Hz, J5,4 = 9.3 Hz, H-5), 3.32 (dd, 1H, J6b,6a = 11.2 Hz, H-6a), 3.13 (dd, 1H, H- 6b), 2.03, 1.94, 1.75 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 170.3, 169.3, 168.8 (3 x Ac), 159.9 (C arom. quat.), 128.4, 113.8 (5 C arom.), 79.4 (C-1), 76.3 (C-5), 73.8 (C-3), 72.7 (C-2), 72.4 (C- 4), 55.2 (OMe), 20.7, 20.6, 20.4 (3 x Ac), 4.2 (C-6); MS: m/z 524 (M+NH4)+; Anal. calc. C, 45.07; H, 4.58; found. C, 44.98; H, 4.54.
(2,3,4-Tri-O-acetyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) anisole (12)
Compound 11 (1.6 g, 3.2 mmol) is dissolved in dry THF (15 mL) and DBU (2.9 mL, 19 mmol) is added. The mixture is heated at 70 °C until the end of the reaction is detected by TLC (7:3 cyclohexane/AcOEt) after 3 hours. The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:5 AcOEt/cyclohexane) to afford 12 (812 mg, 68 %) as a white solid. = -51 (c = 1.0, CHCl3); m.p. = 94 °C (Et2O/pentane); 1H-NMR (250 MHz): δ = 7.14 (d, 2H, J = 8.8 Hz, 2 H arom.), 6.72 (d, 2H, J = 8.8 Hz, 2 H arom.), 5.47-5.39 (m, 1H, H-4), 5.17-5.04 (m, 2H, H-3, H-2), 4.69 (sl, 1H, H-6a), 4.43 (sl, 1H, H-6b), 4.35-4.22 (m, 1H, H-1), 3.64 (s, 3H, OMe), 1.99, 1.87, 1.65 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 169.9, 169.1, 168.6 (3 x Ac), 159.9 (C arom. quat.), 153.6 (C-5), 128.3, 127.7, 113.7 (5 C arom.), 96.1 (C-6), 80.4 (C-1), 73.3 (C-3), 72.4 (C-2), 69.4 (C-4), 55.0 (OMe), 20.5, 20.4, 20.2 (3 x Ac); MS: m/z 396 (M+NH4)+, 379 (M+H)+; Anal. calc. C, 60.31; H, 5.86; found. C, 60.17; H, 5.98.
(2,3,4-Tri-O-benzyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) anisole (13)
A catalytic amount of sodium is added to a solution 12 (350 mg, 0.92 mmol) in methanol (3 mL), and the solution is stirred for 12 hours. The solvent is evaporated without neutralisation and the residue dissolved in dry DMF (30 mL) to which benzyl bromide (1.1 mL, 9.2 mmol) and (222 mg, 60% in suspension in oil, 5.5 mmol) are added. The mixture is stirred at R.T. until the end of the reaction is detected by TLC (9:1 cyclohexane/AcOEt) after 5 hours. Excess NaH is quenched with methanol, and the mixture is extracted with ether and washed with water. Organic layer is dried (MgSO4), filtered and concentrated. The residue purified by silica gel flash column chromatography (1:99 AcOEt/cyclohexane) to afford 13 (299 g, 62 %) as a white solid. = -53 (c = 1.0, CHCl3); m.p. = 77-78 °C (Et2O/pentane); 1H-NMR (250 MHz): δ = 7.33-6.75 (19H, H arom.), 4.81 (d, 1H, J = 11.3 Hz, -CHPh), 4.54 (d, 1H, J = 12.0 Hz, -CHPh), 4.72 (s, 1H, H-6a), 4.69 (d, 1H, J = 11.3 Hz, -CHPh), 4.64 (d, 1H, J = 11.5 Hz, -CHPh), 4.63 (s, 1H, H-6b), 4.42 (d, 1H, J1,2 = 9.7 Hz, H-1), 4.31 (d, 1H, J = 10.5 Hz, -CHPh), 4.00 (d, 1H, J4,3 = 7.4 Hz, H-4), 3.81 (d, 1H, J = 10.5 Hz, -CHPh), 3.76 (s, 3H, OMe), 3.71 (t, 1H, J2,3 = 7.7 Hz, H-3), 3.53 (dd, 1H, H-2); 13C-NMR (63 MHz): δ = 156.7 (C-5), 159.9, 138.5, 138.0, 137.7 (4 C arom. quat.), 130.9-113.9 (20 C arom.), 95.2 (C-6), 84.7 (C-3), 83.8 (C-2), 81.1 (C-1), 79.4 (C-4), 74.8, 74.6, 73.0 (3 CHPh), 55.5 (OMe); MS: m/z 540 (M+NH4)+; Anal. calc. C, 77.62; H, 6.71; found. C, 77.61; H, 6.67.
1R,2S,3S,4R,5S)-2,3,4-Tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) anisole (14), (1R,2S,3S,4R,5R)- 2,3,4-tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) anisole (15) and 2,3,4-tri-O-benzyl-5,6-dehydro-1- C(S)-anisyl-D-glucitol (16)
A solution of TIBAL (1 mL, 1 mmol, 1M in toluene) is added to a solution of 13 (85 mg, 0.16 mmol) in dry toluene (1 mL) at R.T. under argon. The mixture is stirred at 50 °C for 1 hour, until the end of the reaction is detected by TLC (7:3 cyclohexane/AcOEt). The mixture is cooled to R.T. and water (2 mL) is slowly added, and stirred for 15 min. After extraction with AcOEt (3 x 10 mL) and washing with water (10 mL) the organic layer is dried (MgSO4), filtered and the solvent is evaporated. The residue purified by silica gel flash column chromatography (1/9 AcOEt/cyclohexane) to afford three major products 14 (58 mg, 68 %) as a white solid, 15 (58 mg, 68 %) as a white solid and 16 (9 mg, 10 %) as an oil.
(1R,2S,3S,4R,5R)-2,3,4-Tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) anisole (14)
= -34 (c = 0.9, CHCl
3); m.p. = 110-111 °C (AcOEt/cyclohexane);
1H-NMR (400 MHz): δ = 7.31-6.69 (19H, arom. H), 4.97 (d, 1H, J = 10.8 Hz, -CHPh), 4.92 (d, 1H, J = 10.8 Hz, -CHPh), 4.83 (d, 1H, J = 11.5 Hz, -CHPh), 4.79 (d, 1H,
J = 11.5 Hz, -CHPh), 4.55 (d, 1H,
J = 10.2 Hz, -CHPh), 4.27-4.21 (m, 1H, H-5), 4.00 (t, 1H,
J2,3 = 10.3 Hz,
J3,4 = 10.3 Hz, H-3), 3.98 (d, 1H,
J = 10.3 Hz, -CHPh), 3.96 (s, 3H, OMe), 3.64 (dd, 1H,
J4,5 = 3.1 Hz, H-4), 3.54 (dd, 1H,
J1,2 = 9.1 Hz, H-2), 3.27 (ddd, 1H,
J1,6a = 11.0 Hz,
J1,6e = 3.7 Hz, H-1), 2.66 (br s, 1H, -OH), 2.13 (dt, 1H,
J6e,6a = 14.5 Hz,
J6e,5 = 3.7 Hz, H-6e), 1.73 (br t, 1H, H-6a);
13C-NMR (100 MHz): δ = 158.3, 138.9, 138.2, 138.0, 134.3 (5 C arom. quat.), 128.9-113.7 (19 C arom.), 86.0 (C-2), 83.6 (C-3), 83.2 (C-4), 75.9, 75.3, 72.6 (3 CHPh), 66.6 (C-5), 55.2 (OMe), 41.8 (C-1), 34.3 (C-6); MS: m/z 542 (M+NH
4)
+; Anal. calc. C, 77.31; H, 7.08; found. C, 77.26; H, 7.16.
(1R,2S,3S,4R,5S)-2,3,4-Tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) anisole (15)
= -6 (c = 1.0, CHCl
3); m.p. = 122 °C (AcOEt/cyclohexane);
1H-NMR (400 MHz): δ = 7.41-6.88 (19H, arom. H), 5.09 (d, 1H,
J = 11.3 Hz, -CHPh), 5.00 (d, 1H,
J = 10.8 Hz, -CHPh), 4.92 (d, 1H,
J = 10.8 Hz, -CHPh), 4.77 (d, 1H,
J = 11.3 Hz, -CHPh), 4.50 (d, 1H,
J = 10.1 Hz, -CHPh), 3.91 (d, 1H,
J = 10.1 Hz, -CHPh), 3.86 (s, 3H, OMe), 3.71 (dddd, 1H,
J5,OH = 1.7 Hz,
J5,4 = 9.1 Hz,
J5,6e = 4.6 Hz,
J5,6a = 13.4 Hz, H-5), 3.67 (t, 1H,
J3,2 = 9.1 Hz,
J3,4 = 9.1 Hz, H-3), 3.58 (dd, 1H,
J2,1 = 10.2 Hz, H-2), 3.48 (t, 1H, H-4), 2.75 (ddd, 1H,
J1,6a = 13.0 Hz,
J1,6e = 3.6 Hz, H-1), 2.40 (d, 1H, -OH), 2.11 (dt, 1H,
J6e,6a = 13.2 Hz,
J6e,5 = 4.2 Hz, H-6e), 1.77 (td, 1H, H-6a);
13C-NMR (100 MHz): δ = 158.5, 138.6, 137.9, 138.0, 133.7 (5 C arom. quat.), 128.7-113.9 (19 C arom.), 86.4 (C-2), 86.3 (C-4), 86.0 (C-3), 75.7, 75.5, 75.4 (3 CHPh), 71.2 (C-5), 55.3 (OMe), 44.6 (C-1), 35.6 (C-6); MS: m/z 542 (M+NH
4)
+; Anal. calc. C, 77.31; H, 7.08; found. C, 77.19; H, 7.34.
2,3,4-Tri-O-benzyl-5,6-dehydro-1-C(S)-anisyl-D-glucitol (16)
= -10 (c = 0.2, CHCl
3);
1H-NMR (400 MHz): δ = 7.39-6.88 (19H, arom. H), 5.78 (ddd, 1H,
J5,6a = 10.3 Hz,
J5,6b = 17.4 Hz,
J5,4 = 7.8 Hz, H-5), 5.31 (dd, 1H,
J6a,6b = 1.4 Hz, H-6a), 5.24 (dd, 1H, H-6b), 4.91 (bt, 1H,
J1,2 = 5.1 Hz,
J1,OH = 5.1 Hz, H-1), 4.86 (d, 1H,
J = 11.4 Hz, -CHPh), 4.66 (d, 1H,
J = 11.4 Hz, -CHPh), 4.63 (d, 1H,
J = 11.6 Hz, -CHPh), 4.40 (d, 1H,
J = 11.6 Hz, -CHPh), 4.34 (d, 1H,
J = 11.5 Hz, -CHPh), 4.29 (d, 1H,
J = 11.5 Hz, -CHPh), 4.19 (dd, 1H,
J = 7.6 Hz,
J = 6.5 Hz, H-4), 3.85 (s, 3H, OMe), 3.69-3.62 (m, 2H, H-3 H-2), 3.38 (d, 1H, -OH);
13C-NMR (100 MHz): δ = 158.9, 138.2, 138.1, 138.0, 133.9 (5 C arom. quat.), 135.3 (C-5), 128.5-113.6 (19 C arom.), 127.6 (C-6), 81.3 (C-4), 81.0, 80.7 (C-2 C-3), 72.6 (C-1), 74.3, 72.6, 70.6 (3 CHPh); HRMS: m/z calc. 542.2906 (M+NH
4)
+; found. 542.2905 (M+NH
4)
+.
1,3,5-Trimethoxy-2-(2,3,4-tri-O-acetyl-6-desoxy-6-iodo-β-D-glucopyranosyl) benzene (18)
A catalytic amount of sodium is added to a solution 17 (4.5 g, 9.0 mmol) in methanol (50 mL), and the solution is stirred for 5 hours. It is then neutralised using IR 120 (H+) resin, filtered and evaporated in vacuo. The residue is dissolved in anhydrous DMF (20 mL), then PPh3 (4.7 g, 18 mmol) is added, and the solution is cooled to 0 °C under argon. A solution of diiodine (4.6 g, 18 mmol) in anhydrous DMF (20 mL) is then added dropwise and the solution stirred for 45 min at R.T., until the end of the reaction is detected by TLC (9:1 dichloromethane/MeOH). Solvent is evaporated. The residue is next dissolved in pyridine (20 mL) and acetic anhydride (10 mL). The reaction is monitored by TLC (1:1 cyclohexane/AcOEt) for 3 hours at R.T. The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:4 AcOEt/cyclohexane) to give compound 18 (3.32 g, 65 %) as a white solid. = -9 (c = 1.1, CHCl3); m.p. = 157 °C (iPrOH); 1H-NMR (250 MHz): δ = 6.05-5.96 (m, 2H, 2 H arom.), 5.84 (t, 1H, J1,2 = 9.7 Hz, J2,3 = 9.7 Hz, H-2), 5.30 (t, 1H, J3,4 = 9.4 Hz, H-3), 5.04 (d, 1H, J4,5 = 9.4 Hz, H-4), 4.98 (d, 1H, H-1), 3.79 (s, 3H, OMe), 3.72 (s, 6H, 2 x OMe), 3.39-3.31 (m, 1H, H-5), 3.28 (dd, 1H, J5,6a = 3.2, J6b,6a = 10.8 Hz, H-6a), 3.13 (dd, J6b,5 = 4.8 Hz, 1H, H-6b), 2.02, 1.94, 1.67 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 170.4, 169.4, 169.1 (3 x Ac), 161.8, 161.1, 160.1 (4 x C arom. quat.), 91.4, 90.6 (2 C arom.), 76.1 (C-5), 74.8 (C-3), 73.0 (C-4), 71.6 (C-1), 69.8 (C-2), 56.2, 55.3 (3 x OMe), 20.9, 20.8, 20.6 (3 x Ac), 5.0 (C-6) ; MS: m/z 584 (M+NH4)+; Anal. calc. C, 44.53; H, 4.80; found. C, 44.44; H, 4.76
1,3,5-Trimethoxy-2-(2,3,4-tri-O-acetyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) benzene (19)
Compound 18 (1.5 g, 2.6 mmol) is dissolved in dry THF (15 mL) and DBU (2.5 mL, 16 mmol) is added. The mixture is heated at 70 °C until the end of the reaction is detected after 2 hours by TLC (7:3 cyclohexane/AcOEt). The solvent is then removed in vacuo and the residue purified by silica gel flash column chromatography (1:5 AcOEt/cyclohexane) to compound 19 (826 mg, 71 %) as a white solid. = -43 (c = 1.2, CHCl3); m.p. = 142 °C (Et2O/pentane); 1H-NMR (250 MHz): δ = 6.03 (s, 2H, 2 H arom.), 5.98 (dd, 1H, J1,2 = 10.2 Hz, J2,3 = 9.6 Hz, H-2), 5.54 (dl, 1H, J4,3 = 9.6 Hz, H-4), 5.17 (t, 1H, H-3), 5.02 (d, 1H, H-1), 4.72 (sl, 1H, H-6a), 4.45 (sl, 1H, H-6b), 3.75, 3.73 (2 x s, 9H, 3 x OMe), 2.08, 1.97, 1.68 (3 x s, 3 x 3H, 3 x Ac); 13C-NMR (63 MHz): δ = 169.9, 169.2, 167.0 (3 x Ac), 161.9, 160.3, 154.1, 103.4, 90.7 (6 C arom. C-5), 95.0 (C-6), 73.9 (C-3), 72.7 (C-1), 69.5 (C-4), 69.3 (C-2), 55.8, 55.0 (3 x OMe), 20.6, 20.2 (3 x Ac); MS: m/z 456 (M+NH4)+; 439 (M+H)+; Anal. calc. C, 57.53; H, 5.98; found. C, 57.55; H, 6.01.
1,3,5-Trimethoxy-2-(2,3,4-tri-O-benzyl-6-desoxy-β-D-xylo-hex-5-enopyranosyl) benzene (20)
A catalytic amount of sodium is added to a solution 19 (300 mg, 0.68 mmol) in methanol (3 mL), and the solution is stirred for 12 hours. The solvent is evaporated without neutralisation and the residue dissolved in dry DMF (30 mL) to which benzyl bromide (0.8 mL, 6.8 mmol) and NaH (165 mg, 60% in suspension in oil, 4.1 mmol) are added. The mixture is stirred at R.T. until the end of the reaction is detected after 2 hours by TLC (9:1 cyclohexane/AcOEt). Excess NaH is quenched with methanol, and the mixture is extracted with ether and washed with water. Organic layer is dried (MgSO4), filtered and concentrated. The residue purified by silica gel flash column chromatography (1:99 AcOEt/cyclohexane) compound 20 (350 mg, 88 %) as an oil. = -28 (c = 1.4, CHCl3); 1H-NMR (250 MHz): δ = 7.42-6.04 (17H, H arom.), 5.05 (d, 1H, J1,2 = 10.0 Hz, H-1), 4.86 (d, 1H, J = 10.7 Hz, -CHPh), 4.77 (d, 1H, J = 10.3 Hz, -CHPh), 4.73 (d, 1H, J = 10.8 Hz, CHPh), 4.70 (sl, 1H, H-6a), 4.68 (sl, 1H, H-6b), 4.67 (d, 1H, J = 11.4 Hz, -CHPh), 4.45 (d, 1H, J = 10.8 Hz, -CHPh), 4.39 (dd, 1H, J2,3 = 9.9 Hz, H-2), 4.06 (d, 1H, J = 10.7 Hz, -CHPh), 4.02 (dl, 1H, J4,3 = 8.7 Hz, H-4), 3.76, 3.70 (2 s, 9H, 3 x OMe), 3.64 (t, 1H, H-3); 13C-NMR (63 MHz): δ = 161.6, 160.4, 157.5, 138.6, 138.3, 138.2, 128.3- 127.3, 106.4, 91.0 (25 C arom. C-5), 94.2 (C-6), 85.6 (C-3), 79.8 (C-4), 79.2 (C-2), 73.5 (C-1), 75.2, 74.2, 73.4 (3 CHPh), 55.8, 55.2 (3 x OMe); MS: m/z 600 (M+NH4)+, 583 (M+H)+; Anal. calc. C, 74.20; H, 6.57; found. C, 74.56; H, 6.86.
1,3,5-Trimethoxy-2-(1R,2S,3S,4R,5R)-2,3,4-tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (21) and 1,3,5-trimethoxy-2-(1R,2S,3S,4R,5S)-2,3,4-tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (22)
A solution of TIBAL (0.9 mL, 0.9 mmol, 1M in toluene) is added to a solution of 20 (100 mg, 0.17 mmol) in dry toluene (1 mL) at R.T. under argon. The mixture is stirred at 50 °C for 30 min, until the end of the reaction is detected by TLC (7:3 cyclohexane/AcOEt). The mixture is cooled to R.T. and water (2 mL) is slowly added, and stirred for 15 min. After extraction with AcOEt (3 x 10 mL) and washing with water (10 mL) the organic layer is dried (MgSO4), filtered and the solvent is evaporated. The residue purified by silica gel flash column chromatography (3:7 AcOEt/cyclohexane) to afford a 2/1 mixture of two major unseparable products 21 and 22 (95 mg, 95 %) as an oil.
1,3,5-Trimethoxy-2-((1R,2S,3S,4R,5R)-2,3,4-tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (21)
(Extracted from the NMR of the mixture of isomers)
1H-NMR (400 MHz): δ = 7.45-6.92 (15H, arom. H), 6.25 (d, 1H, J = 2.3 Hz, arom. H), 6.12 (d, 1H, J = 2.3 Hz, arom. H), 5.16-4.78 (m, 4H, 4 -CHPh), 4.66 (d, 1H, J = 10.5 Hz, -CHPh), 4.27 (dd, 1H, J1,2 = 10.9 Hz, J2,3 = 9.1 Hz, H-2), 4.26-4.22 (m, 1H, H-5), 4.21 (d, 1H, J = 10.6 Hz, -CHPh), 4.04 (ddd, 1H, J1,6a = 13.2 Hz, J1,6e = 3.9 Hz, H-1), 3.98 (t, 1H, J3,4 = 9.2 Hz, H-3), 3.87 (s, 3H, OMe), 3.84 (s, 3H, OMe), 3.77 (s, 3H, OMe), 3.65 (dd, 1H, J4,5 = 3.3 Hz, H-4), 2.59 (br s, 1H, -OH), 2.36-2.24 (m, 1H, H- 6a), 1.87 (dt, 1H, J6e,6a = 14.0 Hz, J6e,5 = 3.6 Hz, H-6e); 13C-NMR (100 MHz): δ = 160.1-90.7 (24 C arom.), 86.6 (C-4), 83.8 (C-3), 82.2 (C-2), 76.7-72.5 (3 CHPh), 67.0 (C-5), 56.2-54.9 (3 OMe), 31.7 (C-1), 30.7 (C-6); MS: m/z 602 (M+NH4)+; Anal. for the mixture of isomers calc. C, 73.95; H, 6.89; found. C, 73.89; H, 6.91.
1,3,5-Trimethoxy-2-((1R,2S,3S,4R,5S)-2,3,4-tri-O-benzyl-2,3,4,5-tetrahydroxycyclohexyl) benzene (22)
(Extracted from the NMR of the mixture of isomers)
1H-NMR (400 MHz): δ = 7.45-6.92 (15H, arom. H), 6.25 (d, 1H, J = 2.3 Hz, arom. H), 6.15 (d, 1H, J = 2.3 Hz, arom. H), 5.16-4.78 (m, 4H, 4 -CHPh), 4.65 (d, 1H, J = 10.4 Hz, -CHPh), 4.36 (dd, 1H, J1,2 = 10.8 Hz, J2,3 = 9.0 Hz, H-2), 4.19 (d, 1H, J = 10.6 Hz, -CHPh), 3.88 (s, 3H, OMe), 3.83 (s, 3H, OMe), 3.81 (s, 3H, OMe), 3.71 (ddd, 1H, J5,6a = 11.6 Hz, J5,6e = 5 Hz, J5,4 = 9.1 Hz, H-5), 3.66 (t, 1H, J3,4 = 9.2 Hz, H-3), 3.55 (ddd, 1H, J1,6a = 13.2 Hz, J1,6e = 3.9 Hz, H-1), 3.52 (t, 1H, H-4), 2.45 (br s, 1H, -OH), 2.36-2.24 (m, 1H, H-6a), 1.87 (dt, 1H, J6e,6a = 14.0 Hz, J6e,5 = 3.6 Hz, H-6e); 13C-NMR (100 MHz): δ = 160.1-90.7 (24 C arom.), 86.6 (C-4), 83.6 (C-3), 82.4 (C-2), 76.7-72.5 (3 CHPh), 71.6 (C-5), 56.2-54.9 (3 OMe), 34.3 (C-1), 32.2 (C-6); MS: m/z 602 (M+NH4)+; Anal. for the mixture of isomers calc. C, 73.95; H, 6.89; found. C, 73.89; H, 6.91.
2,3,4-Tri-O-benzyl-5,6-dehydro-1-C(S)-butyl-D-glucitol (28)[14].
A solution of TIBAL (1.2 mL, 1.2 mmol, 1M in toluene) is added to a solution of 27 (110 mg, 0.23 mmol) in dry toluene (1 mL) at R.T. under argon. The mixture is stirred at 50 °C for 2 hours 30 min, until the end of the reaction is detected by TLC (7:3 cyclohexane/AcOEt). The mixture is cooled to R.T. and water (2 mL) is slowly added, and stirred for 15 min. After extraction with AcOEt (3 x 10 mL) and washing with water (10 mL) the organic layer is dried (MgSO4), filtered and the solvent is evaporated. The residue purified by silica gel flash column chromatography (3:7 AcOEt/cyclohexane) to afford a major product 28 (90 mg, 82 %) as an oil. = -17 (c = 1.8, CHCl3), lit.[14] = -21 (c = 18.7, CHCl3); 1H-NMR (250 MHz): δ = 7.51-7.12 (15H, arom. H), 5.85 (ddd, 1H, J5,6a = 11.5 Hz, J5,6b = 16.4 Hz, J5,4 = 7.6 Hz, H-5), 5.31 (dd, 1H, J6a,6b = 1.5 Hz, H-6a), 5.28 (dd, 1H, H-6b), 4.73 (d, 1H, J = 11.3 Hz, -CHPh), 4.65 (d, 1H, J = 11.2 Hz, -CHPh), 4.56 (d, 2H, J = 11.6 Hz, 2 -CHPh), 4.42 (d, 1H, J = 11.4 Hz, -CHPh), 4.31 (d, 1H, J = 11.5 Hz, -CHPh), 4.18 (dd, 1H, J4,3 = 5.7 Hz, H-4), 3.85-3.75 (m, 1H, H-1), 3.74 (dd, J3,2 = 4.0 Hz, H-3), 3.47 (dd, J2,1 = 5.4 Hz, H-2), 2.86 (d, 1H, J = 6.1 Hz, -OH), 1.6-0.7 (m, 9H, butyl); MS :m/z 492 (M+NH4)+