Energy Conservation in Absorption Refrigeration Cycles Using DES as a New Generation of Green Absorbents
Abstract
1. Introduction
2. Methods
2.1. Selected Deep Eutectic Solvents
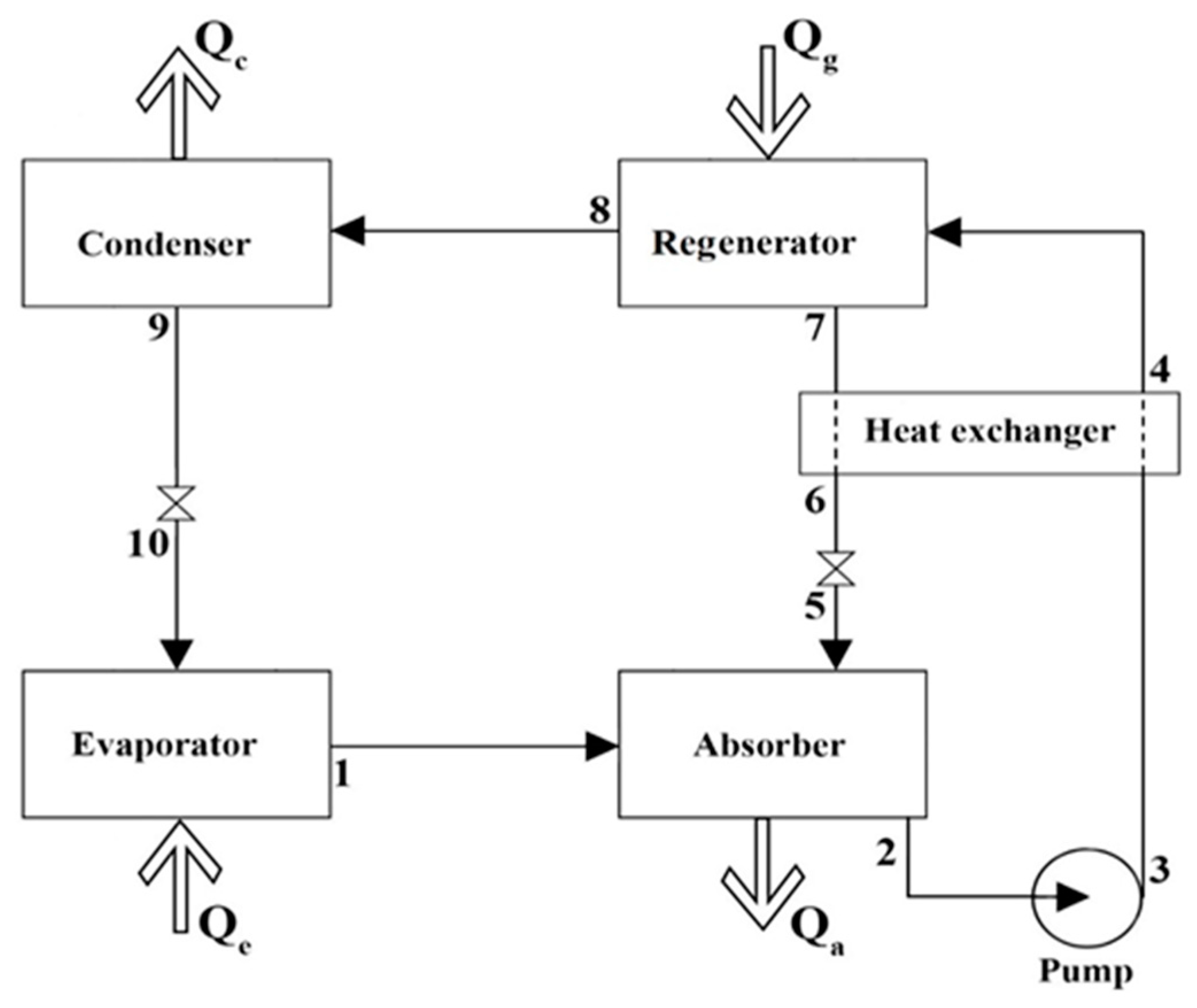
2.2. Absorption Refrigeration Cycle
2.3. Thermodynamic Calculations
2.3.1. Specific Enthalpy
2.3.2. The Cubic-Plus-Association Equation of State
2.4. Simulation of the Process
3. Results and Discussion
3.1. CPA Equation of State Parameterization
3.2. Base Case
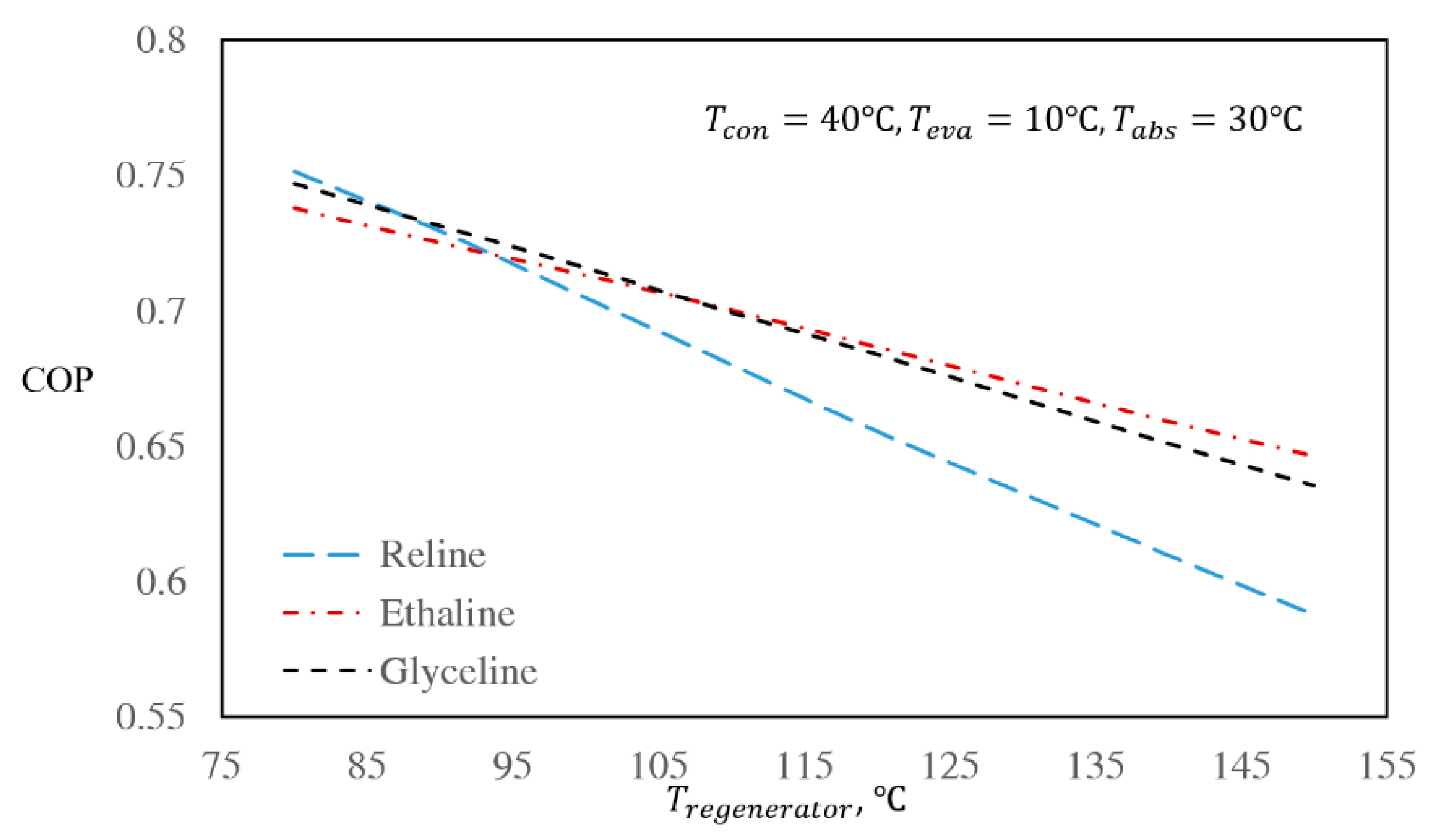
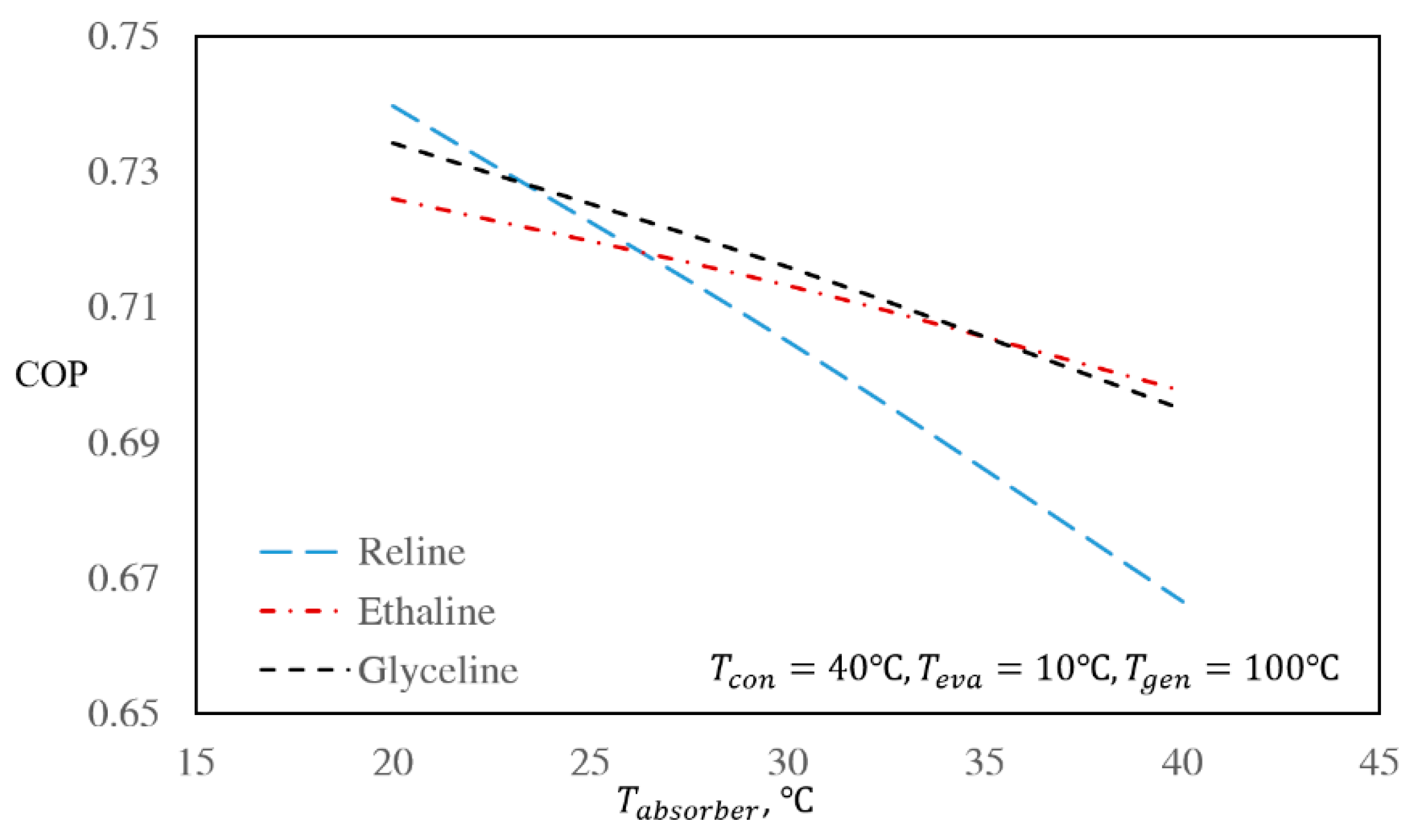
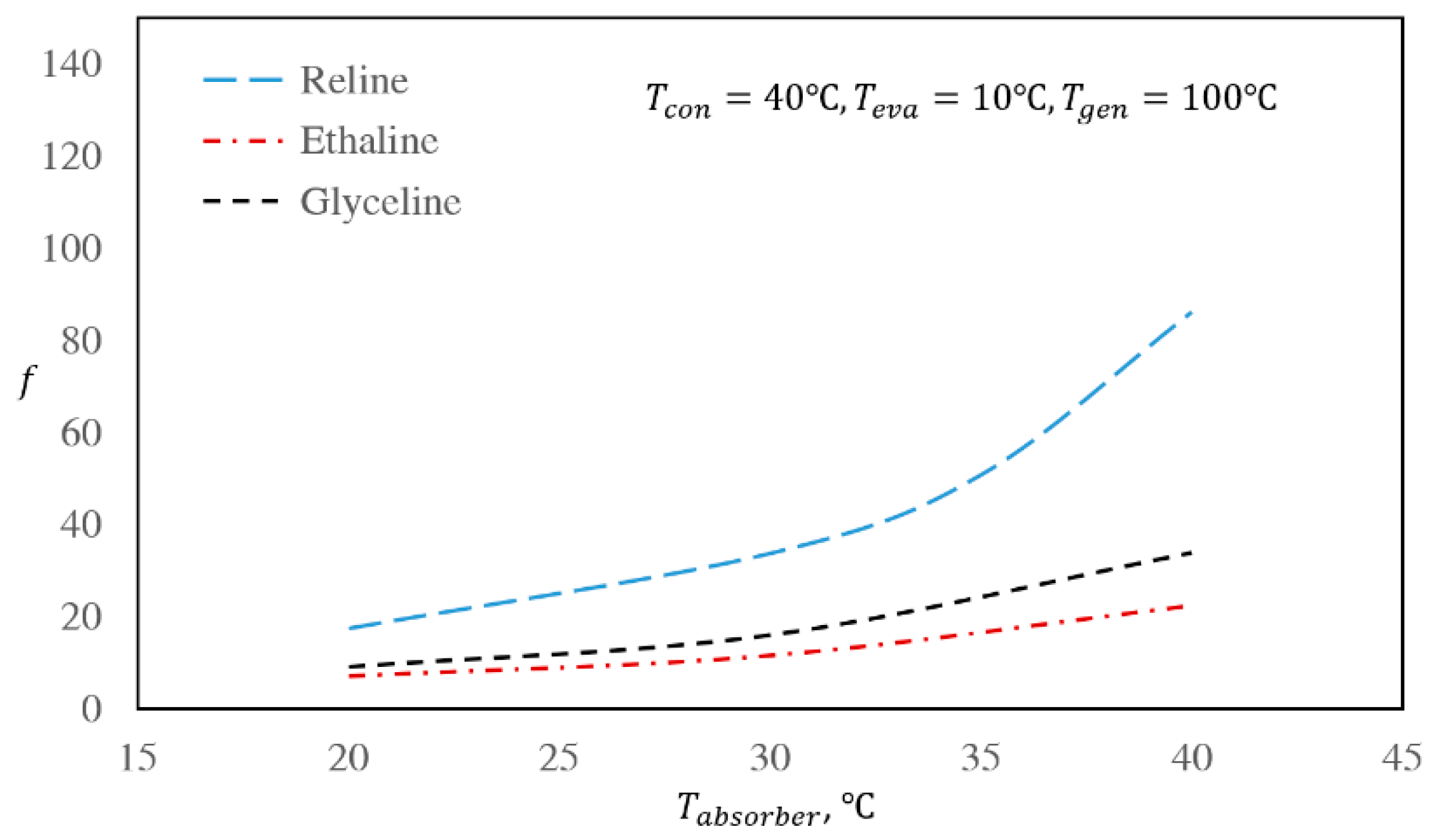
3.3. The Effect of Regenerator and Absorber Temperatures
3.4. Analysis by Experimental Design
3.5. Effect of Pump Work
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Substance | |||||
|---|---|---|---|---|---|
| Reline | 30.43067 | 0.275520 | −6.4000 | −4.400 | |
| Ethaline | 23.31867 | 0.320320 | −9.5000 | −4.300 | |
| Glyceline | 25.11867 | 0.410253 | −15.0000 | −4.100 | |
| Water | 32.24685 | 0.001928 | 1.0557 | −0.036 | |
Appendix B
| Working Pair | Temperature | Pressure (kPa) | x5 (Mass%) |
|---|---|---|---|
| Reline/water | 322.87 | 1.227 | 97.46 |
| Ethaline/water | 328.03 | 1.227 | 92.38 |
| Glyceline/water | 326.32 | 1.227 | 94.50 |
| Working Pair | Temperature (K) | Pressure (kPa) | x2 (Mass%) |
|---|---|---|---|
| Reline/water | 303.15 | 1.227 | 94.57 |
| Ethaline/water | 303.15 | 1.227 | 84.37 |
| Glyceline/water | 303.15 | 1.227 | 88.61 |
References
- Berlitz, T.; Plank, H.; Ziegler, F.; Kahn, R. An ammonia-water absorption refrigerator with a large temperature lift for combined heating and cooling. Int. J. Refrig. 1998, 21, 219–229. [Google Scholar] [CrossRef]
- Bianco, V.; Cascetta, F.; Marino, A.; Nardini, S. Understanding energy consumption and carbon emissions in Europe: A focus on inequality issues. Energy 2019, 170, 120–130. [Google Scholar] [CrossRef]
- Wu, W.; Wang, B.; Shi, W.; Li, X. An overview of ammonia-based absorption chillers and heat pumps. Renew. Sustain. Energy Rev. 2014, 31, 681–707. [Google Scholar] [CrossRef]
- Zheng, D.; Meng, X. Ultimate refrigerating conditions, behavior turning and a thermodynamic analysis for absorption–compression hybrid refrigeration cycle. Energy Convers. Manag. 2012, 56, 166–174. [Google Scholar] [CrossRef]
- Abedin, R.; Heidarian, S.; Flake, J.C.; Hung, F.R. Computational Evaluation of Mixtures of Hydrofluorocarbons and Deep Eutectic Solvents for Absorption Refrigeration Systems. Langmuir 2017, 33, 11611–11625. [Google Scholar] [CrossRef]
- Wang, M.; Becker, T.M.; Infante Ferreira, C.A. Assessment of vapor–liquid equilibrium models for ionic liquid based working pairs in absorption cycles. Int. J. Refrig. 2018, 87, 10–25. [Google Scholar] [CrossRef]
- Wu, W.; You, T.; Zhang, H.; Li, X. Comparisons of different ionic liquids combined with trans-1,3,3,3-tetrafluoropropene (R1234ze(E)) as absorption working fluids. Int. J. Refrig. 2018, 88, 45–57. [Google Scholar] [CrossRef]
- Smith, J.M.; Van Ness, H.C.; Abbott, M.M. Introduction to Chemical Engineering Thermodynamics, 7th ed.; McGraw-Hill: Boston, MA, USA, 2005. [Google Scholar]
- Yokozeki, A. Theoretical performances of various refrigerant–absorbent pairs in a vapor-absorption refrigeration cycle by the use of equations of state. Appl. Energy 2005, 80, 383–399. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, D. Performance simulation of the absorption chiller using water and ionic liquid 1-ethyl-3-methylimidazolium dimethylphosphate as the working pair. Appl. Therm. Eng. 2011, 31, 3316–3321. [Google Scholar] [CrossRef]
- Sujatha, I.; Venkatarathnam, G. Comparison of performance of a vapor absorption refrigeration system operating with some hydrofluorocarbons and hydrofluoroolefins as refrigerants along with ionic liquid [hmim][TF2N] as the absorbent. Int. J. Refrig. 2018, 88, 370–382. [Google Scholar] [CrossRef]
- Arshi Banu, P.S.; Sudharsan, N.M. Review of water based vapour absorption cooling systems using thermodynamic analysis. Renew. Sustain. Energy Rev. 2018, 82, 3750–3761. [Google Scholar] [CrossRef]
- Sujatha, I.; Venkatarathnam, G. Performance of a vapour absorption heat transformer operating with ionic liquids and ammonia. Energy 2017, 141, 924–936. [Google Scholar] [CrossRef]
- Yokozeki, A.; Shiflett, M.B. Water Solubility in Ionic Liquids and Application to Absorption Cycles. Ind. & Eng. Chem. Res. 2010, 49, 9496–9503. [Google Scholar] [CrossRef]
- Moreno, D.; Ferro, V.R.; de Riva, J.; Santiago, R.; Moya, C.; Larriba, M.; Palomar, J. Absorption refrigeration cycles based on ionic liquids: Refrigerant/absorbent selection by thermodynamic and process analysis. Appl. Energy 2018, 213, 179–194. [Google Scholar] [CrossRef]
- Zhao, B.-Y.; Xu, P.; Yang, F.-X.; Wu, H.; Zong, M.-H.; Lou, W.-Y. Biocompatible Deep Eutectic Solvents Based on Choline Chloride: Characterization and Application to the Extraction of Rutin from Sophora japonica. ACS Sustain. Chem. Eng. 2015, 3, 2746–2755. [Google Scholar] [CrossRef]
- Zhang, Q.; De Oliveira Vigier, K.; Royer, S.; Jérôme, F. Deep eutectic solvents: syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef]
- Kunz, W.; Häckl, K. The hype with ionic liquids as solvents. Chem. Phys. Lett. 2016, 661, 6–12. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep Eutectic Solvents Formed between Choline Chloride and Carboxylic Acids: Versatile Alternatives to Ionic Liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef]
- Phadtare, S.B.; Shankarling, G.S. Halogenation reactions in biodegradable solvent: Efficient bromination of substituted 1-aminoanthra-9,10-quinone in deep eutectic solvent (choline chloride : urea). Green Chem. 2010, 12, 458–462. [Google Scholar] [CrossRef]
- Ilgen, F.; Ott, D.; Kralisch, D.; Reil, C.; Palmberger, A.; König, B. Conversion of carbohydrates into 5-hydroxymethylfurfural in highly concentrated low melting mixtures. Green Chem. 2009, 11, 1948–1954. [Google Scholar] [CrossRef]
- Imperato, G.; Höger, S.; Lenoir, D.; König, B. Low melting sugar–urea–salt mixtures as solvents for organic reactions—estimation of polarity and use in catalysis. Green Chem. 2006, 8, 1051–1055. [Google Scholar] [CrossRef]
- Gorke, J.T.; Srienc, F.; Kazlauskas, R.J. Hydrolase-catalyzed biotransformations in deep eutectic solvents. Chem. Commun. 2008, 1235–1237. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Zhang, H.; Row, K.H. Application of deep eutectic solvents in the extraction and separation of target compounds from various samples. J. Sep. Sci. 2015, 38, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.-L.; Peng, X.; Huang, Y.-Y.; Li, L.; Wei, Z.-F.; Zu, Y.-G.; Fu, Y.-J. Green and efficient extraction of bioactive flavonoids from Equisetum palustre L. by deep eutectic solvents-based negative pressure cavitation method combined with macroporous resin enrichment. Ind. Crop. Prod. 2015, 70, 142–148. [Google Scholar] [CrossRef]
- Yao, X.-H.; Zhang, D.-Y.; Duan, M.-H.; Cui, Q.; Xu, W.-J.; Luo, M.; Li, C.-Y.; Zu, Y.-G.; Fu, Y.-J. Preparation and determination of phenolic compounds from Pyrola incarnata Fisch. with a green polyols based-deep eutectic solvent. Sep. Purif. Technol. 2015, 149, 116–123. [Google Scholar] [CrossRef]
- Nam, M.W.; Zhao, J.; Lee, M.S.; Jeong, J.H.; Lee, J. Enhanced extraction of bioactive natural products using tailor-made deep eutectic solvents: Application to flavonoid extraction from Flos sophorae. Green Chem. 2015, 17, 1718–1727. [Google Scholar] [CrossRef]
- Shahbaz, K.; Mjalli, F.S.; Hashim, M.A.; AlNashef, I.M. Prediction of deep eutectic solvents densities at different temperatures. Thermochim. Acta 2011, 515, 67–72. [Google Scholar] [CrossRef]
- Leron, R.B.; Li, M.-H. High-pressure density measurements for choline chloride: Urea deep eutectic solvent and its aqueous mixtures at T = (298.15 to 323.15)K and up to 50MPa. J. Chem. Thermodyn. 2012, 54, 293–301. [Google Scholar] [CrossRef]
- Leron, R.B.; Soriano, A.N.; Li, M.-H. Densities and refractive indices of the deep eutectic solvents (choline chloride+ethylene glycol or glycerol) and their aqueous mixtures at the temperature ranging from 298.15 to 333.15 K. J. Taiwan Inst. Chem. Eng. 2012, 43, 551–557. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 70–71. [Google Scholar] [CrossRef]
- Prausnitz, J.M.; Lichtenthaler, R.N.; Azevedo, E.G.d. Molecular Thermodynamics of Fluid-Phase Equilibria, 3rd ed.; Prentice Hall PTR: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
- Myint, P.C.; McClelland, M.A.; Nichols, A.L. Application of the Peng–Robinson Equation of State to Energetic Materials RDX and TNT: Pure Components, Liquid Mixtures, and Solid Mixtures. Ind. Eng. Chem. Res. 2016, 55, 2252–2266. [Google Scholar] [CrossRef]
- Joback, K.G.; Reid, R.C. Estimation of pure component properties from group contribution. Chem. Eng. Commun. 1987, 57, 233–243. [Google Scholar] [CrossRef]
- Borgnakke, C.; Sonntag, R.E. Fundamentals of Thermodynamics, 8th ed.; Wiley: Hoboken, NJ, USA, 2013. [Google Scholar]
- Haghbakhsh, R.; Parvaneh, K.; Raeissi, S.; Shariati, A. A general viscosity model for deep eutectic solvents: The free volume theory coupled with association equations of state. Fluid Phase Equilib. 2018, 470, 193–202. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Raeissi, S. Modeling the Phase Behavior of Carbon Dioxide Solubility in Deep Eutectic Solvents with the Cubic Plus Association Equation of State. J. Chem. Eng. Data 2018, 63, 897–906. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Raeissi, S.; Parvaneh, K.; Shariati, A. The friction theory for modeling the viscosities of deep eutectic solvents using the CPA and PC-SAFT equations of state. J. Mol. Liq. 2018, 249, 554–561. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Raeissi, S. Modeling vapor-liquid equilibria of mixtures of SO2 and deep eutectic solvents using the CPA-NRTL and CPA-UNIQUAC models. J. Mol. Liq. 2018, 250, 259–268. [Google Scholar] [CrossRef]
- Kontogeorgis, G.M.; Voutsas, E.C.; Yakoumis, I.V.; Tassios, D.P. An Equation of State for Associating Fluids. Ind. & Eng. Chem. Res. 1996, 35, 4310–4318. [Google Scholar] [CrossRef]
- Michelsen, M.L.; Hendriks, E.M. Physical properties from association models. Fluid Phase Equilib. 2001, 180, 165–174. [Google Scholar] [CrossRef]
- Elliott, J.R.; Suresh, S.J.; Donohue, M.D. A simple equation of state for non-spherical and associating molecules. Ind. Eng. Chem. Res. 1990, 29, 1476–1485. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Parvaneh, K.; Shariati, A. Viscosities of Pure Ionic Liquids Using Combinations of Free Volume Theory or Friction Theory with the Cubic, the Cubic Plus Association, and the Perturbed-Chain Statistical Associating Fluid Theory Equations of State at High Pressures. Ind. Eng. Chem. Res. 2017, 56, 2247–2258. [Google Scholar] [CrossRef]
- Keshtkari, S.; Haghbakhsh, R.; Raeissi, S.; Florusse, L.; Peters, C.J. Vapor–liquid equilibria of isopropyl alcohol+propylene at high pressures: Experimental measurement and modeling with the CPA EoS. J. Supercrit. Fluids 2013, 84, 182–189. [Google Scholar] [CrossRef]
- Parvaneh, K.; Haghbakhsh, R.; Rahimpour, M.R. High pressure viscosity modeling of pure alcohols based on classical and advanced equations of state. J. Taiwan Inst. Chem. Eng. 2016, 58, 57–70. [Google Scholar] [CrossRef]
- Huang, S.H.; Radosz, M. Equation of state for small, large, polydisperse, and associating molecules. Ind. Eng. Chem. Res. 1990, 29, 2284–2294. [Google Scholar] [CrossRef]
- Kontogeorgis, G.M.; Michelsen, M.L.; Folas, G.K.; Derawi, S.; von Solms, N.; Stenby, E.H. Ten Years with the CPA (Cubic-Plus-Association) Equation of State. Part 1. Pure Compounds and Self-Associating Systems. Ind. Eng. Chem. Res. 2006, 45, 4855–4868. [Google Scholar] [CrossRef]
- Guo, P.; Tu, H.; Wang, Z.; Wang, Q. Calculation of thermodynamic properties of water by the CPA equation of state. Nat. Gas Ind. B 2017, 4, 305–310. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Rojas, R.E. Critical Properties of Ionic Liquids. Revisited. Ind. Eng. Chem. Res. 2009, 48, 6890–6900. [Google Scholar] [CrossRef]
- Yadav, A.; Pandey, S. Densities and Viscosities of (Choline Chloride + Urea) Deep Eutectic Solvent and Its Aqueous Mixtures in the Temperature Range 293.15 K to 363.15 K. J. Chem. Eng. Data 2014, 59, 2221–2229. [Google Scholar] [CrossRef]
- Leron, R.B.; Li, M.-H. High-pressure volumetric properties of choline chloride–ethylene glycol based deep eutectic solvent and its mixtures with water. Thermochim. Acta 2012, 546, 54–60. [Google Scholar] [CrossRef]
- Yadav, A.; Kar, J.R.; Verma, M.; Naqvi, S.; Pandey, S. Densities of aqueous mixtures of (choline chloride+ethylene glycol) and (choline chloride+malonic acid) deep eutectic solvents in temperature range 283.15–363.15K. Thermochim. Acta 2015, 600, 95–101. [Google Scholar] [CrossRef]
- Yadav, A.; Trivedi, S.; Rai, R.; Pandey, S. Densities and dynamic viscosities of (choline chloride+glycerol) deep eutectic solvent and its aqueous mixtures in the temperature range (283.15–363.15)K. Fluid Phase Equilib. 2014, 367, 135–142. [Google Scholar] [CrossRef]
- Leron, R.B.; Wong, D.S.H.; Li, M.-H. Densities of a deep eutectic solvent based on choline chloride and glycerol and its aqueous mixtures at elevated pressures. Fluid Phase Equilib. 2012, 335, 32–38. [Google Scholar] [CrossRef]
- Kaushik, S.C.; Tomar, C.S.; Chandra, S. Coefficient of performance of an ideal absorption cycle. Appl. Energy 1983, 14, 115–121. [Google Scholar] [CrossRef]
- Mjalli, F.S.; Mousa, H. Viscosity of aqueous ionic liquids analogues as a function of water content and temperature. Chin. J. Chem. Eng. 2017, 25, 1877–1883. [Google Scholar] [CrossRef]
- Wimby, J.M.; Berntsson, T.S. Viscosity and density of aqueous solutions of lithium bromide, lithium chloride, zinc bromide, calcium chloride and lithium nitrate. 1. Single salt solutions. J. Chem. Eng. Data 1994, 39, 68–72. [Google Scholar] [CrossRef]
- Chen, W.; Liang, S.; Guo, Y.; Tang, D. Thermodynamic analysis of an absorption system using [bmim]Zn2Cl5/NH3 as the working pair. Energy Convers. Manag. 2014, 85, 13–19. [Google Scholar] [CrossRef]
- Khuri, A.I.; Mukhopadhyay, S. Response surface methodology. WIREs Comput. Stat. 2010, 2, 128–149. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Peyrovedin, H.; Raeissi, S.; Duarte, A.R.C.; Shariati, A. Investigating the performance of novel green solvents in absorption refrigeration cycles: Energy and exergy analyses. Int. J. Refrig. 2020. In Press. [Google Scholar] [CrossRef]

| DES Name | HBD | HBA | HBA/HBD Molar Ratio | Mw | Freezing Point (K) | |
|---|---|---|---|---|---|---|
| Reline | Urea | Choline chloride | 1:2 | 86.580 | 285.00 4 | 1.19790 2 |
| Ethaline | Ethylene glycol | Choline chloride | 1:2 | 87.920 | 207.14 1 | 1.11704 3 |
| Glyceline | Glycerol | Choline chloride | 1:2 | 107.933 | 271.82 1 | 1.19123 3 |
| DES | β | Tc (K) | Pc (MPa) | ω | ||||
|---|---|---|---|---|---|---|---|---|
| Reline 1 | 28.31 | 0.0657 | 0.116 | 0.5458 | 1001.70 | 644.44 | 5.0149 | 0.6167 |
| Ethaline 1 | 29.28 | 0.0733 | 0.835 | 0.5456 | 1001.70 | 602.00 | 4.1661 | 0.8747 |
| Glyceline 1 | 26.53 | 0.0829 | 0.810 | 0.5585 | 1018.30 | 680.67 | 3.4183 | 1.1780 |
| Water 2 | 1.22777 | 0.0145 | 1.180 | 0.0250 | 140.36 | 647.10 | 22.055 | 0.345 |
| Solution | Temperature Range (K) | Pressure Range (MPa) | DES Mole Fraction Range | Density Range | Ndp | 103k0 | 105k1 | Ref |
|---|---|---|---|---|---|---|---|---|
| Reline/water | 293.15–363.15 | 0.1–50 | 0–1 | 965.0–1205.8 | 682 | −1.66406 | −1.33 | [29,50] |
| Ethaline/water | 283.15–363.15 | 0.1–50 | 0–1 | 965.0–1130.6 | 781 | −2.40793 | −1.44 | [30,51,52] |
| Glyceline/water | 283.15–363.15 | 0.1–50 | 0–1 | 965.0–1202.4 | 808 | −2.61677 | -1.39 | [30,53,54] |
| Working Pair | COP | f | x5 Mass% | x2 Mass% | Qg (kW) |
|---|---|---|---|---|---|
| Reline/water | 0.705 | 33.73 | 97.46 | 94.57 | 3052.35 |
| Ethaline/water | 0.713 | 11.53 | 92.38 | 84.37 | 3017.36 |
| Glyceline/water | 0.716 | 16.06 | 94.50 | 88.61 | 3004.06 |
| LiBr/water * | 0.780 | 4.08 | 66.29 | 50.02 | 3012.95 |
| [bmim][BF4]/water * | 0.544 | 13.00 | 96.88 | 89.42 | 4320.04 |
| [emim][BF4]/water * | 0.525 | 18.20 | 98.56 | 93.14 | 4476.38 |
| [emim][C2H5SO4]/water * | 0.569 | 13.57 | 97.92 | 90.70 | 4130.23 |
| [mmim][(CH3)2PO4]/water * | 0.662 | 5.32 | 93.66 | 76.06 | 3550.00 |
| [bmim][I]/water * | 0.534 | 23.70 | 98.75 | 94.58 | 4400.94 |
| [choline][Gly]/water * | 0.446 | 4.79 | 93.22 | 73.76 | 5269.28 |
| [choline][CH3SO3]/water * | 0.636 | 7.32 | 94.78 | 81.83 | 3695.13 |
| [choline][Lac]/water * | 0.659 | 7.79 | 96.71 | 84.29 | 3566.16 |
| [bmim][(C4H9)2PO4]/water * | 0.532 | 11.17 | 91.82 | 83.6 | 4417.48 |
| [eeim][(C2H5)2PO4]/water * | 0.565 | 12.38 | 95.99 | 88.24 | 4159.47 |
| [emim][(C2H5)2PO4]/water * | 0.588 | 7.75 | 90.28 | 78.63 | 3996.77 |
| [emim][(CH3)2PO4]//water * | 0.691 | 8.66 | 98.13 | 86.79 | 3401.01 |
| Temperature | Min (°C) | Max (°C) |
|---|---|---|
| Tevaporator | 5 | 15 |
| Tregenerator | 60 | 150 |
| Tabsorber | 20 | 50 |
| Tcondenser | 20 | 50 |
| Working Pair | Standard Deviation | R-Squared Value | Mean Value |
|---|---|---|---|
| Reline/water | 0.0048 | 0.9978 | 0.64 |
| Ethaline/water | 0.0126 | 0.9454 | 0.68 |
| Glyceline/water | 0.0013 | 0.9996 | 0.67 |
| Working Pair | Optimum Value of COP | f | Teva (°C) | Tgen (°C) | Tabs (°C) | Tcon (°C) | Qe (kW) | Qg (kW) |
|---|---|---|---|---|---|---|---|---|
| Reline/water | 0.816 | 244.3 | 10.00 | 60.00 | 20.00 | 40.00 | 2150.9 | 2635.91 |
| Ethaline/water | 0.776 | 30.8 | 10.00 | 60.00 | 20.00 | 40.00 | 2150.9 | 2771.78 |
| Glyceline/water | 0.786 | 47.9 | 10.00 | 60.00 | 20.00 | 40.00 | 2150.9 | 2736.51 |
| Working Pair | WP (W) | Heat ratio | WP/Qg (%) |
|---|---|---|---|
| Reline/water | 175.82 | 0.70 | 0.006 |
| Ethaline/water | 64.87 | 0.71 | 0.002 |
| Glyceline/water | 85.17 | 0.72 | 0.003 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haghbakhsh, R.; Peyrovedin, H.; Raeissi, S.; Duarte, A.R.C.; Shariati, A. Energy Conservation in Absorption Refrigeration Cycles Using DES as a New Generation of Green Absorbents. Entropy 2020, 22, 409. https://doi.org/10.3390/e22040409
Haghbakhsh R, Peyrovedin H, Raeissi S, Duarte ARC, Shariati A. Energy Conservation in Absorption Refrigeration Cycles Using DES as a New Generation of Green Absorbents. Entropy. 2020; 22(4):409. https://doi.org/10.3390/e22040409
Chicago/Turabian StyleHaghbakhsh, Reza, Hamed Peyrovedin, Sona Raeissi, Ana Rita C. Duarte, and Alireza Shariati. 2020. "Energy Conservation in Absorption Refrigeration Cycles Using DES as a New Generation of Green Absorbents" Entropy 22, no. 4: 409. https://doi.org/10.3390/e22040409
APA StyleHaghbakhsh, R., Peyrovedin, H., Raeissi, S., Duarte, A. R. C., & Shariati, A. (2020). Energy Conservation in Absorption Refrigeration Cycles Using DES as a New Generation of Green Absorbents. Entropy, 22(4), 409. https://doi.org/10.3390/e22040409







