1. Introduction
Ion channels are proteins found in cell membranes that create openings in the membrane to allow cells to communicate with each other and with the outside to transform signals and to conduct tasks together [
1,
2]. They have an aqueous pore that becomes accessible to ions after a change in the protein structure that makes ion channels open. Ion channels permit the selective passage of charged ions formed from dissolved salts, including sodium, potassium, calcium, and chloride ions that carry electrical current in and out of the cell.
To unravel how ion channels operate, one needs to understand the physical structure of ion channels, which is defined by the channel shape and the spatial distribution of permanent and polarization charge. The shape of a typical ion channel is often approximated as a cylindrical-like domain with a non-uniform cross-sectional area. Within a large class of ion channels, amino acid side chains are distributed mainly over a “short” and “narrow” portion of the channel, with acidic side chains contributing permanent negative charges and basic side chains contributing permanent positive charges, analogous to the doping of semiconductor devices, e.g., bipolar PNP and NPN transistors.
The spatial distribution of side chains in a specific channel defines the permanent charge of the channel. The spatial distribution of permanent charge forms (most of) the electrical structure of the channel protein. The spatial distribution of mass forms the structure studied so successfully by molecular and structural biologists. Ions that move through channels are often only an Angstrom or so away from the permanent charges residing on acid and base side chains. In addition, electrical forces are in general much stronger than entropic forces. Thus, in most cases, the electrical structure is more important in determining how ions go through a channel than the mass structure. Sometimes, the dielectric properties (“polarization”) of the protein contribute a charge that is significant. Then, the spatial distribution of dielectric properties becomes an important part of the electrical structure.
The most basic function of ion channels is to regulate the permeability of membranes for a given species of ions and to select the types of ions and to facilitate and modulate the diffusion of ions across cell membranes. At present, these permeation and selectivity properties of ion channels are usually determined from the current–voltage (I–V) relations measured experimentally [
2,
3]. Individual fluxes carry more information than the current, but it is expensive and challenging to measure them [
4,
5]. Indeed, the measurement of unidirectional fluxes by isotopic tracers allowed the early definition of channels and transporters and is a central subject in the history of membrane transport, as described in textbooks—for example, [
6,
7,
8,
9]. The precise definition and use of unidirectional fluxes are dealt with at length in the paper [
5]. The I–V relation defines the function of the channel structure, namely the ionic transport through the channel. That transport depends on driving forces expressed mathematically as boundary conditions. The multi-scale feature of the problem with multiple physical parameters allows the system to have great flexibility and to exhibit vibrant phenomena/behaviors—a great advantage of “natural devices” [
10]. On the other hand, the same multi-scale feature with multiple physical parameters presents an extremely challenging task for anyone to extract meaningful information from experimental data, also given the fact that the internal dynamics cannot be measured with present techniques. The general inverse problem is challenging, although specific inverse problems have been successfully solved with surprisingly little difficulty using standard methods and software packages [
11].
To understand the importance of the relation of current and permanent charges, that is, the I–Q relation, we point out that the role of permanent charges in ionic channels is similar to the role of doping profiles in semiconductor devices. Semiconductor devices are similar to ionic channels in the way that they both use atomic-scale structures to control macroscopic flows from one reservoir to another. Ions move a lot like quasi-particles move in semiconductors. In a crude sense, holes and electrons are the cations and anions of semiconductors. Semiconductor technology depends on the control of migration and diffusion of quasi-particles of charge in transistors and integrated circuits. Doping is the process of adding impurities into intrinsic semiconductors to modulate its electrical, optical, and structural properties [
12,
13]. In a crude sense, doping provides the charges that acid and base side chains provide in a protein channel.
Ion channels are almost always passive and do not require a source of chemical energy (e.g., ATP hydrolysis) in order to operate. Instead, they allow ions to flow passively driven by a combination of the transmembrane electrical potential and the ion concentration gradient across the membrane. For other fixed physical quantities, the total current depends on the transmembrane potential and the permanent charge . For fixed , a reversal potential is a transmembrane potential that produces zero current . Similarly, for fixed transmembrane potential , a reversal permanent charge is a permanent charge that produces zero current .
The
Goldman–Hodgkin–Katz (GHK) equation for reversal potentials involving multiple ion species [
14,
15] is used to determine the reversal potential across ion channels. The GHK equation is an extension of the Nernst equation—the latter is for one ion species. The classical derivations were based on the incorrect assumption that the electric potential
is linear in
X—the coordinate along the length of the channel. This assumption is particularly unfortunate because it is the change in the shape of the electrical potential
that is responsible for so many of the fascinating behaviors of transistors or ionic systems [
16,
17,
18,
19,
20,
21]. There was no substitute for GHK equations until authors of [
22,
23] recently offered equations derived from self-consistent Poisson–Nernst–Planck (PNP) systems, to the best of our knowledge.
In this work, focusing on basic understanding of possible effects of unequal diffusion coefficients and, as a starting point, we will use the classical PNP model with a piecewise constant permanent charge and a cylinder-like channel with variable cross-sectional area. The classical PNP model treats ions as point charges. Among many limitations, gating and selectivity cannot be captured by the simple classical PNP model. However, the basic finding on reversal potentials and their dependence on permanent charges and on ratios of diffusion constants seems important and some are non-intuitive and deserving of further investigation. In the future, more structural detail and more correlations between ions should be taken into considerations in PNP models such as those including various potentials for ion-to-ion interaction accounting for ion sizes effects and voids [
24,
25,
26,
27,
28,
29,
30,
31,
32].
There have been great achievements in analyzing the PNP models for ionic flows through ion channels [
5,
28,
33,
34,
35,
36], etc. Although mathematical analysis plays a powerful and unique role to explain mechanisms of observed biological phenomena and to discover new phenomena, numerical simulations are needed to fit actual experimental data and study cases where analytical solutions do not exist. Furthermore, numerical observations may give clues for more theoretical investigations. Indeed, numerical and analytical studies are linked; any progress in one catalyzes work in the other.
This paper is a mathematical study on some aspects of ionic flows via the PNP models. It uses established mathematical methods and analytical results [
23,
33] that are derived without further assumption from their underlying physical models. The numerical results, throughout the paper, are gained from the algebraic systems (
15), (
22), (
23) and (
27), obtained from reduced matching systems of analytical results in [
23,
33]. The nonlinear algebraic systems are then solved by the MATLAB
® (Version 9.5) function
fsolve that uses the trust–region dogleg algorithm. The trust–region algorithm is a subspace trust–region method and is based on the interior-reflective Newton method described in [
37]. Our numerical results indicate that current–voltage and current-permanent charge and even zero-current relations depend on a rich interplay of boundary conditions and the channel geometry arising from the mathematical properties analyzed in [
23,
33,
34,
38]. Although the work here is presented in the context of biological ion channels, it is clear that the results apply to the artificial channels that are now being studied for their engineering applications.
The highlights of our studies in this paper as well as in [
23,
33,
34,
38] applied to the setup of this paper include:
- (i)
a mathematically derived system for the zero-current condition (see System (
15)) that can be used to determine the reversal potential in terms of other parameters (see Display (
22));
- (ii)
an examination on how the reversal potential depends on permanent charge: its sign and its monotonicity in permanent charge (see
Section 2.2); and a comparison between this reversal potential and that from GHK in the special setting (see
Section 2.3);
- (iii)
a characterization of monotonic dependencies of the reversal potential on the ratio of diffusion coefficients in terms of different conditions on the boundary concentrations (see
Section 2.2), as well as effects of un-equal diffusion coefficients on signs of zero-current flux and its dependence on permanent charge (see
Section 2.1);
- (iv)
numerical spatial profiles under the zero current condition of the concentrations and electric potential, and hence the profiles of the electrochemical potentials for several choices of permanent charges that reveal special features of permanent charge effects (see
Section 2.4, particularly, Remark 3);
- (v)
numerical and analytical studies of I–V and I–Q relations, and zero-voltage current and its rich dependence on permanent charge (see
Section 3.3).
Furthermore, there are several qualitatively important but non-intuitive results discussed in this work. These qualitative results may be helpful in guiding experimentation and some might not be apparent in intuitive thinking about ion channel behavior. Here are some examples:
- a.
The zero-current flux
J has the same sign as that of
(see
Section 2.1).
- b.
The magnitude of the ratio between of the two diffusion coefficients affects the monotonicity of the zero-current flux
J in
Q (see
Section 2.1).
- c.
I–Q curves are not monotonic in general (see
Section 3.2).
- d.
Rich phenomena of interplay between boundary conditions and diffusion coefficients in terms of monotonicity of zero-voltage current on permanent charge (
Section 3.3).
To this end, we would like to emphasize that applying the geometric analysis allows us to identify and formulate quantities and properties that are crucial to biology, while also providing quantitative and qualitative understanding and predictions.
This paper is organized as follows. The classical PNP model for ionic flows is recalled in
Section 1.1 to prepare the stage for investigations in later sections. In
Section 2, we study zero current problems to investigate the corresponding fluxes and reversal potentials
. In particular, we compare a special case of the reversal potential with the GHK equation. Some other numerical observations are also provided to study profiles of relevant physical quantities in
Section 2.4. In
Section 3, we first recall the analytical results in [
33] when diffusion constants are also involved. Then, numerical observations are provided to examine behaviors of current, voltage, and permanent charge with respect to each other in some general cases. Some concluding remarks are provided in
Section 4.
1.1. Poisson–Nernst–Planck Models for Ionic Flows
The PNP system of equations has been analyzed mathematically to some extent, but the equations have been simulated and computed to a much larger extent [
39,
40,
41,
42,
43]. One can see from these simulations that macroscopic reservoirs must be included in the mathematical formulation to describe the actual behavior of channels [
24,
44]. For an ionic mixture of
n ion species, the PNP type model is, for
,
where
with
being a three-dimensional cylindrical-like domain representing the channel of length
,
is the permanent charge density of the channel (with unit
),
is the relative dielectric coefficient (with unit 1),
is the vacuum permittivity,
(coulomb) is the elementary charge,
is the Boltzmann constant,
T is the absolute temperature (
kelvin, for water);
is the electric potential (with the unit
), and, for the
k-th ion species,
is the concentration (with unit M),
is the valence (the number of charges per particle with unit 1), and
is the electrochemical potential (with unit
) depending on electrical potential
and concentrations
. The flux density
(with unit
) is the number of particles across each cross-section in per unit time,
is the diffusion coefficient (with unit
), and
n is the number of distinct types of ion species (with unit 1).
Ion channels have narrow cross-sections relative to their lengths. Therefore, three-dimensional PNP type models can be reduced to quasi-one-dimensional models. The authors of [
45] first offered a reduced form, and, for a particular case, the reduction is precisely verified by the mathematical analysis of [
46]. The quasi-one-dimensional steady-state PNP type is, for
where
X is the coordinate along the channel,
is the area of cross-section of the channel over location
X, and
(with unit
) is the total flux through the cross-section. Equipped with System (
2), we impose the following boundary conditions, for
,
One often uses the electroneutrality conditions on the boundary concentrations because the solutions are made from electroneutral solid salts,
The electrochemical potential
for the
k-th ion species consists of the ideal component
and the excess component
, i.e.,
. The excess electrochemical potential
accounts for the finite size effect of ions. It is needed whenever concentrations exceed, say 50 mM, as they almost always do in technological and biological situations and often reach concentrations
or more. The classical PNP model only deals with the ideal component
, which reflects the collision between ions and water molecules and ignores the size of ions; that is,
where
is a characteristic concentration of the problems, and one may consider
For given
,
,
’s and
’s, if
is a solution of the boundary value problem (BVP) (
2) and (
3), then the electric current
is
For an analysis of the BVP (
2) and (
3), we work on a dimensionless form. Set
In terms of the new variables, the BVP (
2) and (
3) becomes, for
,
with the boundary conditions
Remark 1. The actual dimensional forms of quantities have been used for all figures throughout the paper, that is,and we take , and and, for diffusion constants [31], 1.2. Setup of the Problem
We now designate the case we will study in this paper. We will investigate a simple setup, the classical PNP model (
9) with ideal electrochemical potential (
5), and the boundary conditions (
10). More precisely, we assume
- (A0)
The ionic mixture consists of two ion species with valences ;
- (A1)
for is a constant and ;
- (A2)
Electroneutrality boundary conditions (
4) hold;
- (A3)
The permanent charge
Q is piecewise constant with one nonzero region; that is, for a partition
of
,
where
is a constant.
We assume that
in System (
14) is small. The assumption is reasonable since, if
and
, then
[
47]. The assumption that
is small enables one to treat System (
14) of the dimensionless problem as a singularly perturbed problem that can be analyzed by the theory of geometrical singular perturbations (GSP). GSP uses the modern invariant manifold theory from nonlinear dynamical system theory to study the entire structure, i.e., the phase space portrait of the dynamical system, and is not to be confused with the classical singular perturbation theory that uses, for example, matched asymptotic expansions.
We rewrite the classical PNP system (
9) into a standard form of singularly perturbed systems and turn the boundary value problem to a connecting problem. We refer the readers to the papers [
33] and [
36] (with insignificantly altered notations) for details. Denote the derivative with respect to
x by overdot and introduce
. System (
9) becomes, for
System (
14) will be treated as a dynamical system with the phase space
and the independent variable
x is viewed as time for the dynamical system.
A GSP framework for analyzing BVP of the classical PNP systems was developed first in [
33,
35] for ionic mixtures with two types of ion species. The model of ion channel properties involves coupled nonlinear differential equations. Until accomplished, it was not apparent that any analytical results could be found, let alone the powerful ones provided by geometrical singular perturbation. This GSP framework was extended to an arbitrary number of types of ion species successfully only when
two special mathematical structures of the PNP system were revealed [
36]. One special structure is
a complete set of integrals (or conserved quantities) for the
limit fast (or inner) system that allows a detailed analysis of a singular layer component of the full problem. It should pointed out that most of the integrals are NOT conserved for the physical problem since, no matter how small it is,
is NOT zero. The GSP allows one to make conclusion about the BVP for
small from information of
limit systems. The other special structure is that
a state-dependent scaling of the independent variable turns the nonlinear limit slow (or outer) system to a linear system with constant coefficients. The coefficients do depend on unknown fluxes to be determined as a part of the whole problem, and this is the mathematical reason for the rich dynamics of the problem. As a consequence of the framework, the existence, multiplicity, and spatial profiles of
the singular orbits—zeroth order in
approximations of the BVP—are reduced to a system of nonlinear algebraic equations that involves all relevant quantities altogether. This system of nonlinear
algebraic equations defines the physical framework of the problem precisely. The system shows explicitly what has been guessed implicitly “everything interacts with everything else” and, in the cases analyzed in this paper, the system shows quantitatively how those interactions occur. This geometric framework with its extensions to include some of the effects of ion size [
28,
29,
32] has produced a number of results that are central to ion channel properties [
5,
23,
30,
34,
38,
48]; for example, it was shown in [
34] that
a positive permanent charge may enhance anion flux as well as cation flux; and,
in order to optimize effects of the permanent charge, the channel should have a short and narrow neck within which the permanent charge is confined; and, it was shown in [
38] that
large permanent charge is responsible for the declining phenomenon—decreasing flux with increasing transmembrane electrochemical potential. We refer the readers to the aforementioned papers for more details on geometric singular perturbation framework for PNP as well as concrete applications to ion channel problems.
In this paper, we will apply some results and follow the notations in [
23,
33] for analytical results where the quantities are all in their dimensionless forms. In addition, for simplicity, we use the letters
l,
r and
where
,
,
.
Remark 2. We remind the readers that the quantities , and I are dimensionless quantities corresponding to the dimensional quantities , and , respectively, obtained from Display (8). We switch from dimensional form to the dimensionless form and vice versa several times throughout the paper. Dimensionless variables are convenient for illustrating and analyzing mathematical and general physical relations. Dimensional variables are necessary for showing how evolution has exploited those general relations. 2. Zero Current Problems with General Diffusion Constants
In this section, we study how boundary concentrations, electric potential, permanent charges, and diffusion constants work together to produce current reversal. Throughout this section, in order to express the effects of diffusion constants on zero-current flux and reversal potential, we study and compare the results for different cases of diffusion constants where and where , to indicate and emphasize the differences.
Diffusion is the phenomenon through which the spatial distribution of solute particles varies as a result of their potential energy. It is a spontaneous process that acts to eliminate differences in concentration and eventually leads a given mixture to a state of uniform composition. Fick’s first law [
49] describes diffusion of uncharged particles by
, where
c is the concentration,
is the diffusion constant, and
t is time. Frequently, the determination of diffusion constants involves measuring sets of simultaneous values of
t,
c, and
x. These measured values are then applied to a solution of Fick’s law to get the diffusion constants. Many techniques are available for the determination of diffusion constants of ions (charge particles) in aqueous solutions [
31,
50,
51,
52,
53], etc. When diffusion constants are equal, classical electrochemistry tells that many electrical phenomena “disappear” altogether, e.g., the “liquid junction” is zero. If the diffusion constants of potassium and chloride are equal, classical electrochemistry says that KCl acts nearly as an uncharged species. Indeed, this is the basis for the saturated KCl salt bridge used in a broad range of electrochemical experiments for many years. Therefore, the equal diffusion constants case is quite degenerate. Experimental measurements are exclusively performed under isothermal conditions to avoid deviation of
values. Nevertheless, even diffusion constants of certain ionic species may differ from one method to another, even when all other parameters are held constant. Everything becomes much more complicated mathematically when the diffusion constants are not equal, however. This complexity is what makes many biological and technological devices interesting, useful, and valuable. Some kinds of selectivity depend on the non-equality of diffusion constants as well.
Applying GSP theory to the classical PNP system (
2) for two ion species with diffusion constants
, the authors of [
23] obtained an algebraic matching system with eleven equations and eleven unknowns for zero current problems and singular orbits on
. They further reduced the matching system for the case where two ion valences satisfy
. It follows that the reduced matching system for zero current
when
is
where
and,
A is the geometric mean of concentrations at
, that is,
and
Note that, if
is uniform, then
is the ratio of the length with the cross-section area of the potion of the channel over
. The original of this quantity
has its root in Ohm law for resistance of a uniform resistor. It turns out that the quantities
and
together with the value
are key characteristics for the shape and the permanent charge of the channel structure (see Section 4 in [
34] for more detailed and concrete results about the roles of
and
on the fluxes).
To this end, we recall three relevant results from [
23] on which most of our analytical and numerical studies are based.
For fixed
and
,
A can actually be solved from
, where
is defined in Display (
16) with the properties stated in the next theorem.
Theorem 1 (Theorem 3.4 in [
23]).
The solution of satisfies- (a)
and ,
- (b)
if , then ,
- (c)
if , then ,
- (d)
if , then has the same sign as that of ,
where .
For fixed and , the reversal potential can also be determined and enjoy properties stated in the next two theorems. Recall that we denote by J.
Theorem 2 (Theorem 4.2 in [
23]).
For the reversal potential , one has- (i)
if , then , and, hence, ;
- (ii)
if , then , and, hence, ;
- (iii)
and .
Theorem 3 (Theorem 4.4 in [
23]).
For any given , one has- (i)
if , then is increasing in for and decreasing in for ;
- (ii)
if , then, for , is increasing in for and decreasing in for ;
- (iii)
if , then, for , is increasing in for and decreasing in for .
In what follows, numerical simulations are conducted with the help of analysis on System (
15). The combination of numerics and analysis gives a better understanding of the zero-current problems and compliments some analytical results obtained in [
23]. For our numerical simulations, we choose
,
in Display (
13) and
for simplicity and for definiteness.
2.1. Zero-Current Flux .
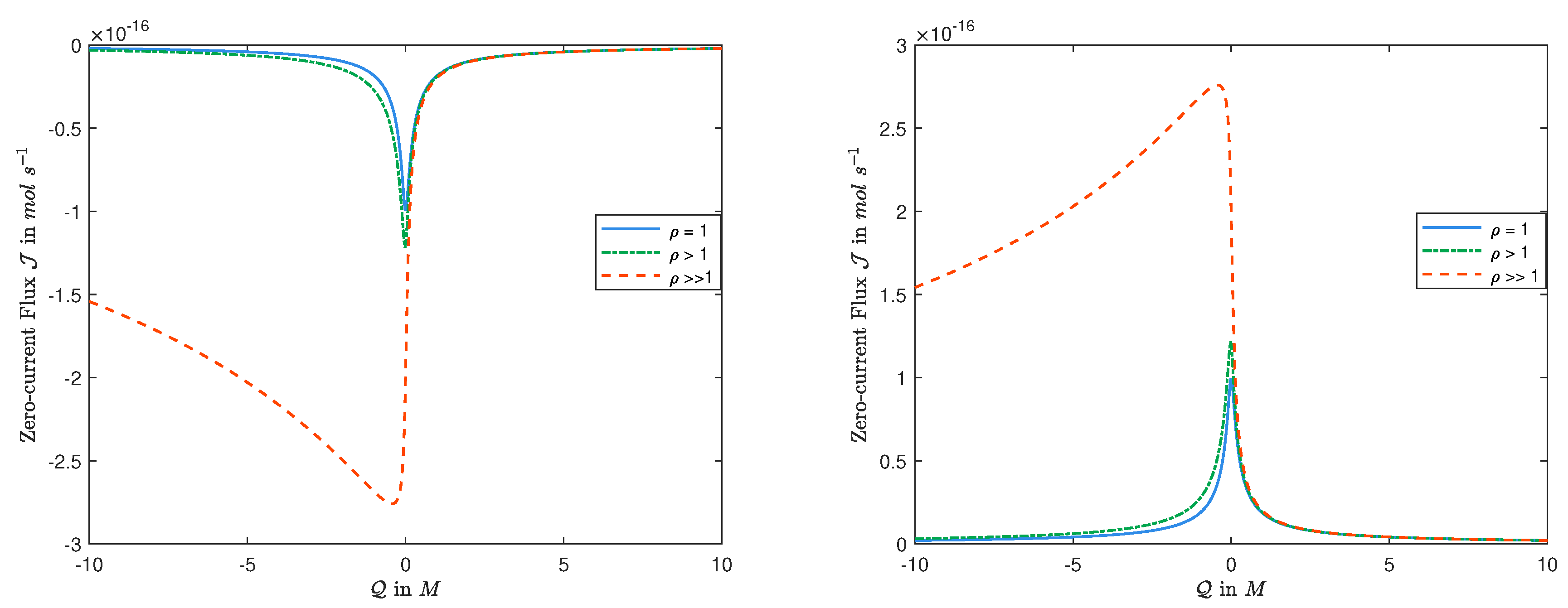
We aim to clarify the relationships of ion fluxes with permanent charge and diffusion constants when current is zero.
Recall that fluxes
and
are equal for this case and let
J denote it. For any permanent charge
, once a solution
of System (
15) is obtained, it follows from matching equations (see Appendix in [
23]) that
J is given by
2.1.1. Sign of Zero-Current Flux
It was observed in [
22] that the Nernst–Planck equation in Display (
9) (with dimensionless quantities) gives, for
,
Therefore, the sign of flux
depends only on the boundary conditions
l,
r and
V. Note that Equation (
21) holds for any condition, not just zero-current condition.
For zero-current problem,
depends on
l,
r,
,
, and
Q as well, in general. Thus, the sign of zero-current flux
J seems to depend on all quantities and to be difficult to figure out. It is not the case. A consequence of Display (
20) together with Theorem 1 is that:
The latter follows directly from Theorem 1 that, for zero-current,
has the same sign as that of
. This is consistent with observations in
Figure 1 where
is fixed, and
varies from the same value to
, and to a random large value.
2.1.2. Dependence of Zero-Current Flux J on and ’s
Concerning the dependence of the zero-current flux J on , we have the following:
- (i)
If , then the zero-current flux J is an even function in , and it is monotonic for .
In this case,
and, hence, it follows from Theorem 1 that
A is an even function in
and is monotonic in
for
, and thus is the zero-current flux
J from Display (
20).
- (ii)
If , then the zero-current flux J is not an even function in and the monotonicity of the zero-current flux J in seems to be more complicated.
In this case, it can be seen that
in Display (
16) is not an even function in
, and, hence, the zero-current flux
J is not. We would like to point out that, it follows from [
38], for fixed
, no matter how large, one always has
as
that is consistent with the observations in
Figure 1.
- (iii)
Another fascinating result is that the magnitude of affects the monotonicity of the zero-current flux J in .
In this case, if one fixes
, and let
increases from small values to
, (i.e.,
), then it follows from Display (
20) that there is a meaningful change in the monotonicity of the zero-current flux
J, for small values of
that is not intuitive.
Let us consider the case where
and
is small. Recall that
A is the geometric mean of concentrations at
. It follows from System (
15) and (
16) that, as
increases,
- (a)
A increases if (that is ), and consequently the zero-current flux J decreases;
- (b)
A decreases if (that is ), and, hence, the zero-current flux J increases.
Thus, depending on the size of
, the zero-current flux
J may increase or decrease in
, which is also consistent with the observations in
Figure 1. The analysis for the case with
is similar.
It seems likely that the engineering, like evolution, will use these mathematical properties to control the qualitative properties of channels, technological, and biological.
2.2. Reversal Potential .
Experimentalists have long identified reversal potential as an essential characteristic of ion channels [
54,
55]. Reversal potential is the potential at which the current reverses direction, i.e.,
that produces zero current
. Using dimensionless form of quantities (see Remark 2), it follows from System (
15) and (
16) (where there are two ion species with valences
) that for general permanent charge
with arbitrary diffusion constants [
23], the variable
A (the geometric mean of concentrations at
) can be solved uniquely from
in System (
15), and the reversal potential is then
where
B,
,
, and
are defined in Displays (
18) and (
19).
2.2.1. Range of Reversal potential
For fixed l, r, and for any given , it follows from Theorem 2 that there exists a unique reversal potential such that . As , then gets close to the boundary values, i.e., .
2.2.2. Zero Reversal Potential
One particular case is when the reversal potential is zero. To examine under what conditions one obtains , it follows Theorem 2 that,
- (i)
if , then for ,
- (ii)
if , then there is a , such that ,
- (iii)
if , then there is a , such that .
Considering the second case above, the observations in
Figure 2 show that, as
increases, magnitude of the corresponding
becomes larger. In fact, as
, then
.
2.2.3. Reversal Potential for
For , one has from Theorem 2. Therefore,
- (i)
if , then ,
- (ii)
if , then has the same sign as that of .
Let us consider the case where
. In that case,
has the same sign as that of
. This is reasonable, since, for
, we have
(since all but
are independent of
in Equation (
21)), and to help
more than
to get
for zero current conditions, one needs to increase
V when
(and decrease
V when
), and that is why
for
(and
for
). This is consistent with observations in
Figure 2 as well. The analysis for the other case with
is similar.
2.2.4. Monotonicity of with Respect to
It follows from Theorem 3 that
This analytical result does not allow conclusions about the case for
, however. The observations in
Figure 2 and
Figure 3 show that the result holds for any
and
Q. Thus, we have
We remark that, in
Figure 3, we take
,
, and
and
which are diffusion constants of, say, Na
and Cl
, respectively (see the solid line), and
and
, where
is the diffusion constants of Ca
(see the dashed line).
2.2.5. Dependence of on
Let us discuss the dependence of
on
for effects of
and
. It follows from Proposition 4.6 in [
23] that
This feature reveals a fantastic aspect that is not intuitive immediately. Recall Equation (
21). Given the boundary values and diffusion constants, the values one obtains for all terms in Equation (
21) except
are independent of
[
36]. The relation surely holds for the zero-current condition, i.e.,
with
. Now, let us fix
and increase
(so
is increasing). Then,
increases since all but
in Equation (
21) are independent of
. Consequently, to meet the zero-current condition, we need to increase
. Intuitively increasing
seems to lead to an increase in
. This intuition agrees with the result for
. However, for the case with
, the result is the exact opposite of the intuitive result. That is, for
, it says, as
increases,
decreases. This counterintuitive behavior could be explained by the fact that
depends on
, and reducing
could still increase
. In fact,
will result in reducing
, but
changes in a way that consequently increases
.
To illustrate the counterintuitive behavior, we provide a numerical result in
Figure 4. We choose
,
and
for Na
as mentioned in Remark 1. Now, suppose that
, and consider the boundary concentrations
,
and
. In this case,
and
. Now, if we increase
to
, which is Cl
diffusion constant, then
and
. These values make sense now, based on the above discussion. Note that we just pictured the middle part of the channel in
Figure 4 since the sides are almost identical. One should notice that it is hard to realize, from
Figure 4, how
will result in reducing
. The complicated behavior discussed above convinces us that detailed analytical studies, even for special cases, could be critical for the design and interpretation of numerical results.
2.3. A Comparison with Goldman–Hodgkin–Katz Equation for .
In this section, we first recall the GHK equation [
14,
15], which relates the reversal potential with the boundary concentrations and the permeabilities of the membrane to the ions. If the membrane is permeable to only one ion, then that ion’s Nernst potential is the reversal potential at which the electrical and chemical driving forces balance. The GHK equation is a generalization of the Nernst equation in which the membrane is permeable to more than just one ion. The derivation of GHK equation assumes that the electric field across the lipid membrane is constant (or, equivalently, the electric potential
is linear in
x in the PNP model). Under the assumption, the I–V (current–voltage) relation is given by
For the case where
and
, the GHK equation for the reversal potential is
The assumption that the electric potential
is linear is not correct when applied to channels in proteins. This is because proteins have specialized structure and spatial distributions of permanent charge (acid and base side chains) and polarization (polar and nonpolar side chains).
Experimental manipulations of the structure of channel proteins show that these properties control the biological function of the channel. The GHK equation does not contain variables to describe any of these properties and so cannot account for the biological functions they control. A linear
is widely believed to make sense without channel structure presumably, in particular, where
. However, this is not correct either. It follows from Formula (
22) for
that the zeroth order in
approximation of the reversal potential in this case is
Figure 5 compares
in Formula (
24) with
from the GHK-equation in Display (
23). It can be seen from the left panel that, when
l and
r are not far away from each other (for example
,
), then the two curves have almost the same behavior. However, when we reduce
L from
to
, then the right panel shows a significant difference between the two graphs.
In
Figure 6, we arrange a simple numerical result for the case where
to compare the graphs of
, obtained from Formula (
22), for various values of permanent charge
. We consider
,
, and
for some values of
, i.e.,
.
2.4. Profiles of Relevant Physical Quantities
It follows that, for any given
Q, once a solution
of Equations (
15) and (
16) is determined, all the other unknowns can be settled, and, hence, the approximation of the solution of boundary value problem can be obtained. We consider the dimensional form of quantities, and fix
to numerically investigate the behavior of
and
throughout the channel.
Figure 7 and
Figure 8 graph the cases with small permanent charge
when
,
,
and
. In this case, we obtain
and
.
Furthermore,
Figure 9 and
Figure 10 show graphs of concentrations, electrical potential, and electrochemical potentials versus
X, where
,
,
and diffusion constants are the same as the previous one. In this case, we obtain
and
.
Remark 3. We end this section with a few of the remarks on some important features captured in Figure 7, Figure 8, Figure 9 and Figure 10. It follows from the Nernst–Planck equation that has the same sign as that of or the opposite sign as that of ; in particular, is always monotonically increasing or decreasing. For the zero-current situation, the reversal potential depends on ALL other parameters; and so it seems that it would be hard to make general conclusions about , for example, about its monotonicity. This is not true. In fact, in Section 2.1, we have concluded that the sign of zero-current flux J is the same as that of , and, hence, has the opposite sign as that of . For the case considered in this part, , one has , independent of the value of Q. Therefore, for , and, hence, ’s are increasing for zero-current situation when , independent of Q, as shown in Figure 8 for and in Figure 10 for . On the other hand, as one changes the value of Q, the profiles of concentrations ’s and electrical potential may vary from monotone to non-monotone, as shown between Figure 7 for and Figure 9 for . 3. Current–Voltage and Current-Permanent Charge Behaviors
Ionic movements across membranes lead to the generation of electrical currents. The current carried by ions can be examined through current–voltage relation or I–V curve. In such a case, voltage refers to the voltage across a membrane potential, and current is the flow of ions through channels in the membrane. Another important piece of data are current-permanent charge (I–Q) relation. Dependence of current on membrane potentials and permanent charge is investigated in this section for arbitrary values of diffusion constants.
To derive the I–V and I–Q relations, we rely on [
33] where the authors showed that the set of nonlinear algebraic equations is equivalent to one nonlinear equation for
A, the geometric mean of concentrations at
defined in Equation (
17). All other quantities or variables such as fluxes, profiles of electric potential
and concentrations
can then be obtained in terms of
A. It is crucial to realize that this is a specific result, not available for general cases. One can only imagine that the resulting simplification produces controllable and robust behavior that proved useful as evolution designed and refined protein channels. The reduction allowed by this composite variable can be postulated to be a “design principle” of channel construction, in technological (engineering) language, or an evolutionary adaptation, in biological language. In particular, the current
I can be explicitly expressed in terms of boundary conditions, permanent charge, diffusion constants, and transmembrane potential in the special case that allows the determination of
A. In what follows, we derive flux and current equations—when diffusion constants are involved as well—in terms of boundary concentrations, membrane potential, and permanent charge. The I–V, I–Q, J–V, and J–Q relations are investigated afterward in
Section 3.2.
3.1. Reduced Flux and Current Equations
In this section, for simplicity, in addition to the assumptions at the beginning of the setup section (
Section 1.2), we will also assume that
,
and
, in particular,
and
(see Display (
19)). It was shown in [
33] that the BVP (
9) and (
10) can be reduced to the algebraic equation
where
,
,
and
N are defined in Display (
18), and
Once
A is solved from Equation (
25), we can obtain the flux densities and current equations as follows:
For any given , there exists a solution for the flux J and current I. The numerical results in the next section give us more information on “current–voltage” and “current-permanent charge” relations.
3.2. Current–Voltage and Current-Permanent Charge Relations
3.2.1. Dependence of Current on Diffusion Constants
Now, we reveal a feature of the theoretical results that is not intuitive. Suppose that
is given (
V is still free and is allowed to take any value!). It follows from Display (
18) for the definition of
N that there exists an
A so that
. It consequently follows from Equation (
26) that, if
, then
. Therefore, from Display (
27),
, which implies
3.2.2. I–V Curves and I–Q Curves
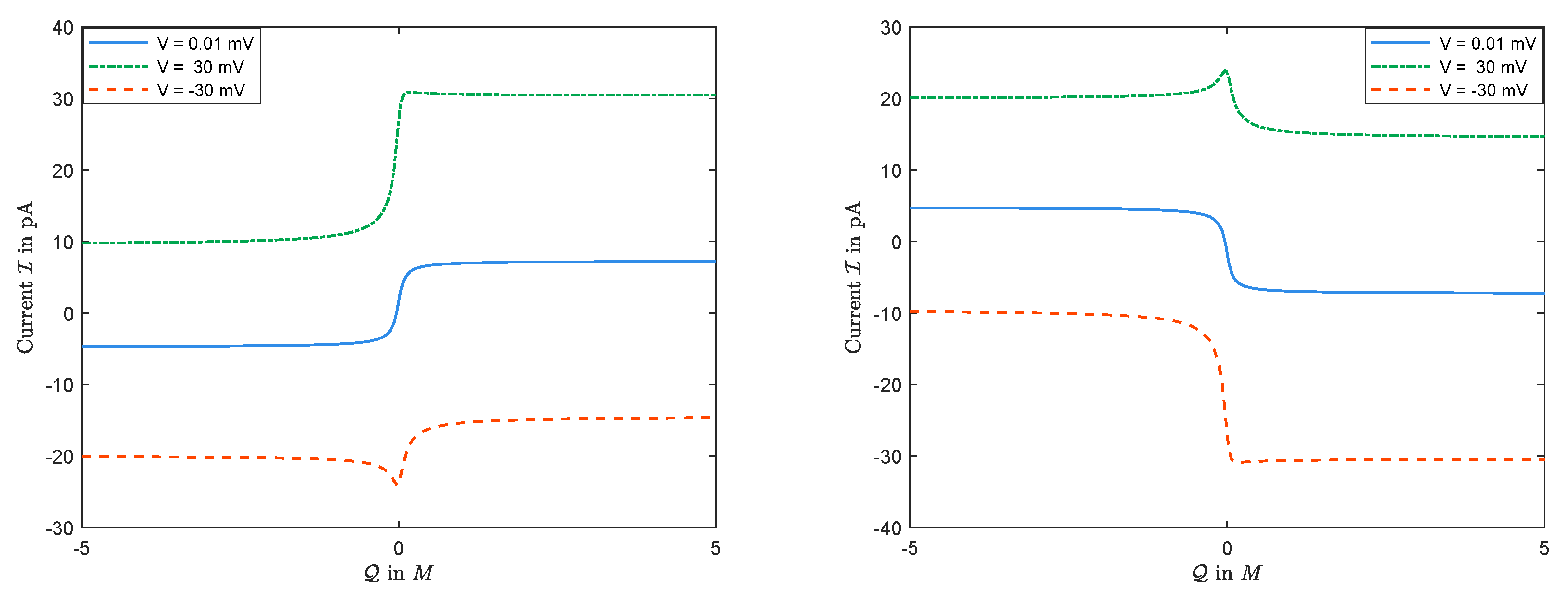
Figure 11 is a numerical simulation from Equations (
25) and (
26) of the I–V curves for several values of
with
and
. One may suspect, based on the numerical observations, that the value of current
, obtained from Display (
27), is unique for any
, and is monotonically increasing in
. However, this may not be correct, in general. This is important since the opening and closing properties of channels might be thought to arise from non-unique solutions [
16,
17].
Now, for I–Q relations, our numerical experiments shows that:
Recall that Equation (
21), in dimensional form, gives
The sign of
is determined by the boundary conditions, independent of the permanent charge. Nevertheless, as expected and seen in
Figure 12, the magnitudes of
’s, and, consequently, the sign and the size of the current
do depend on
in general. (Here,
would be the nonzero value of the permanent charge in dimensional form.) Treating
as a parameter, the current
is a function of
. The numerical observations in
Figure 12 indicate that,
- (i)
there exists some such that, for , has a unique maximum;
- (ii)
there exists some such that, for , has a unique minimum.
In particular, I–Q curves are not monotonic in general.
In addition, we claim based on numerical observations (not proven though) that there exists , such that
- (i)
for any given where , the corresponding current is non-monotonic in , but
- (ii)
for any where , the corresponding current is monotonic in .
In particular, it can be seen in
Section 3.3 that current is monotonic in
for
. In the end, we would like to mention that the diffusion constants affect the values
and
above.
3.3. Zero-Voltage Current
The different permeability of the membrane determines the zero membrane potential (voltage) to different types of ions, as well as the concentrations of the ions, the permanent charge, and the shape of the channel. Denote current
, and the fluxes
, for
, to include the dependence on
Q too. We call
the
zero-potential current and
the
zero-potential fluxes, respectively, when
. For any given value of membrane potential
V, approximation formulas for the current
, for small and large values of permanent charge
Q, are provided in [
34,
38], respectively.
It follows from [
34] that, for
small values of
Q, applying
, zero-potential current
, and zero-potential fluxes
(in dimensionless forms as mentioned in Remark 2) are
For
large positive values of
, with
(where
is small), it follows from [
38] that zero-potential current
and zero-potential fluxes
are
where
It can be readily seen from Equations (
28) that, for small values of
Q, the zero-potential current
is increasing in
Q when
and is decreasing in
Q if
.
However, for large values of the permanent charge Q, the zero-potential current depends on Q in a much richer way. To state the results, we need the following lemma.
Lemma 1. There are and with so that for and for .
It follows from Equations (
29) and Lemma 1 that, for large values of
Q (small values of
),
- (i)
if (so that ), then, for arbitrary and , the zero-potential current is decreasing in (increasing in Q) when , and is increasing in (decreasing in Q) when ;
- (ii)
if (so that ), then,
- (a)
for , the zero-potential current is decreasing in (increasing in Q) for , and is increasing in (decreasing in Q) for ;
- (b)
for , the zero-potential current is increasing in (decreasing in Q) for , and is decreasing in (increasing in Q) for .
Figure 13 illustrates some of the above conclusions. In addition, it suggests that the monotonicity of
holds for all values of permanent charge, not only for small or large values. We emphasize that the monotonicity of current
I with respect to permanent charge
Q is just true for zero membrane potential, i.e.,
. Indeed, one should recall from
Section 3.2 that, when
, then the current
I is not monotonic in
Q.