Entropy in Heart Rate Dynamics Reflects How HRV-Biofeedback Training Improves Neurovisceral Complexity during Stress-Cognition Interactions
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Protocol
2.3. Heart Rate Variability Biofeedback (HRVB) Training Procedure
2.4. Psychological Tests
2.5. Heart Rate Recordings and Analyses
2.6. Statistical Analyses
3. Results
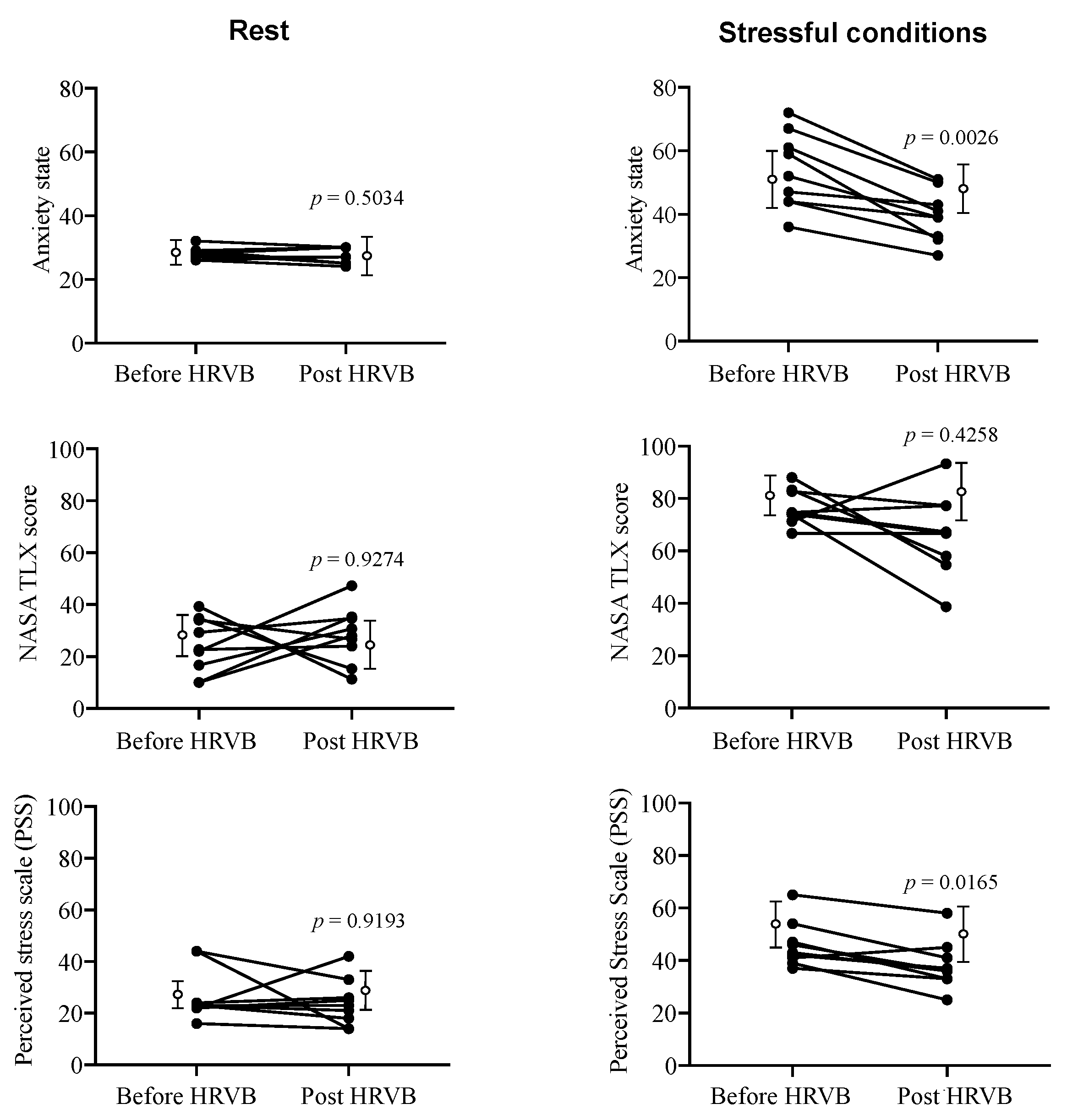
3.1. Psychological Markers
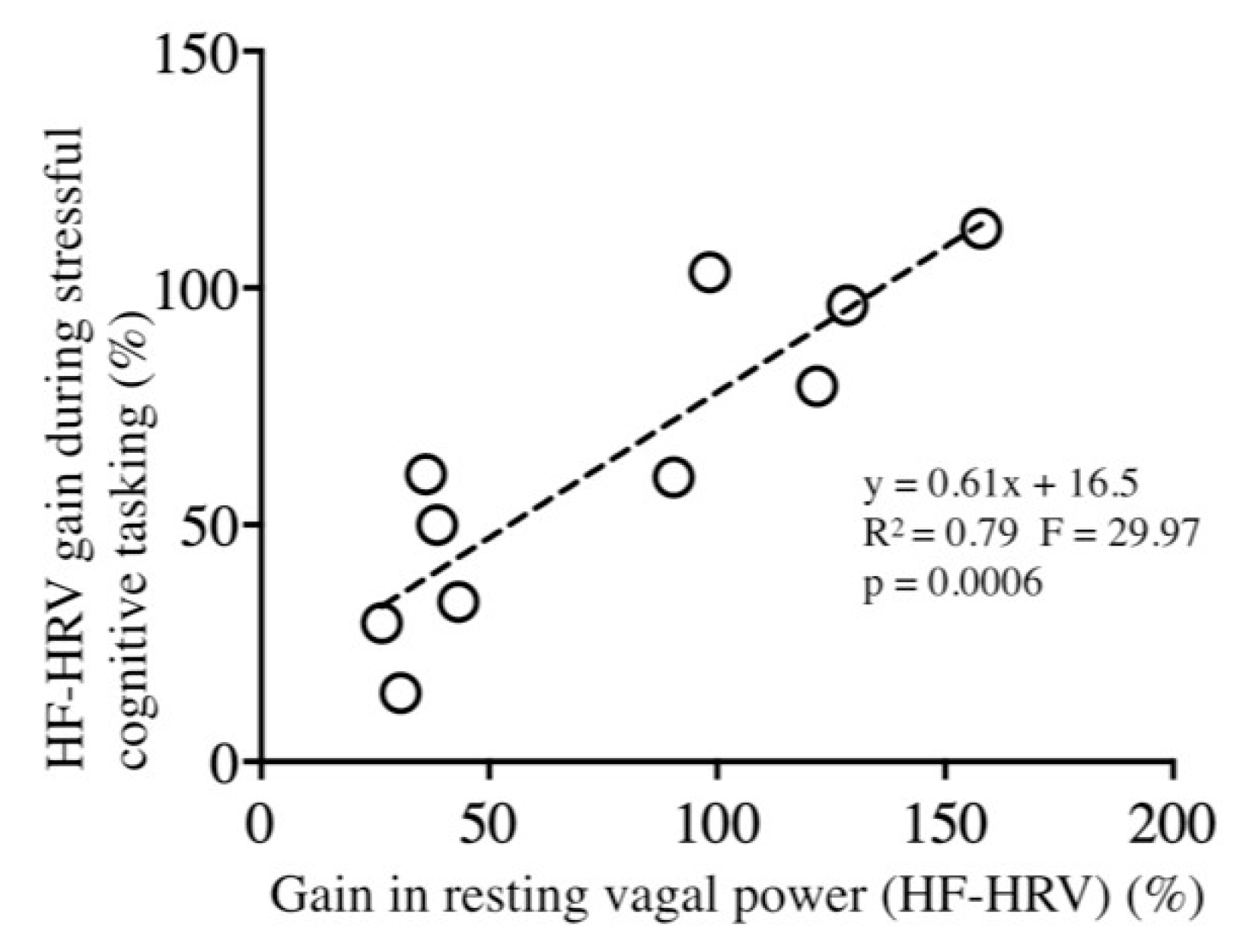
3.2. HRV-Based Autonomic Markers
4. Discussions
Author Contributions
Funding
Conflicts of Interest
References
- Wayne, P.M.; Manor, B.; Novak, V.; Costa, M.D.; Hausdorff, J.M.; Goldberger, A.L.; Ahn, A.C.; Yeh, G.Y.; Peng, C.-K.; Lough, M.; et al. A systems biology approach to studying Tai Chi, physiological complexity and healthy aging: Design and rationale of a pragmatic randomized controlled trial. Contemp. Clin. Trials 2013, 34, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, P.; Parati, G.; Faini, A. Can the Detrended Fluctuation Analysis Reveal Nonlinear Components of Heart Rate Variabilityf. In Proceedings of the 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; Volume 2019, pp. 6351–6354. [Google Scholar]
- Castiglioni, P.; Faini, A. A Fast DFA Algorithm for Multifractal Multiscale Analysis of Physiological Time Series. Front. Physiol. 2019, 10, 115. [Google Scholar] [CrossRef] [PubMed]
- Torre, K.; Vergotte, G.; Viel, É.; Perrey, S.; Dupeyron, A. Fractal properties in sensorimotor variability unveil internal adaptations of the organism before symptomatic functional decline. Sci. Rep. 2019, 9, 15736. [Google Scholar] [CrossRef]
- Park, G.; Thayer, J.F. From the heart to the mind: Cardiac vagal tone modulates top-down and bottom-up visual perception and attention to emotional stimuli. Front. Psychol. 2014, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Porges, S.W. The polyvagal theory: New insights into adaptive reactions of the autonomic nervous system. Clevel. Clin. J. Med. 2009, 76, S86–S90. [Google Scholar] [CrossRef] [PubMed]
- Rollin McCraty the Coherent Heart: Heart–Brain Interactions, Psychophysiological Coherence, and the Emergence of System-Wide Order. Available online: https://www.heartmath.org/research/research-library/basic/coherent-heart-heart-brain-interactions-psychophysiological-coherence-emergence-system-wide-order/ (accessed on 16 May 2018).
- Thayer, J.F.; Lane, R.D. A model of neurovisceral integration in emotion regulation and dysregulation. J. Affect. Disord. 2000, 61, 201–216. [Google Scholar] [CrossRef]
- Benarroch, E.E. The Central Autonomic Network: Functional Organization, Dysfunction, and Perspective. Mayo Clin. Proc. 1993, 68, 988–1001. [Google Scholar] [CrossRef]
- Benarroch, E. Central Autonomic Control. In Primer on the Autonomic Nervous System; David, R., Phillip, L., Ronald, P., Eds.; Academic Press: Cambridge, MA, USA, 2012; pp. 9–12. [Google Scholar]
- Ellis, R.J.; Thayer, J.F. Music and Autonomic Nervous System (Dys)function. Music Percept. 2010, 27, 317–326. [Google Scholar] [CrossRef]
- Beauchaine, T.P.; Thayer, J.F. Heart rate variability as a transdiagnostic biomarker of psychopathology. Int. J. Psychophysiol. 2015, 98, 338–350. [Google Scholar] [CrossRef]
- Thayer, J.F.; Lane, R.D. Claude Bernard and the heart-brain connection: Further elaboration of a model of neurovisceral integration. Neurosci. Biobehav. Rev. 2009, 33, 81–88. [Google Scholar] [CrossRef]
- Thayer, J.F.; Sternberg, E. Beyond heart rate variability: Vagal regulation of allostatic systems. Ann. N. Y. Acad. Sci. 2006, 1088, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Critchley, H.D.; Mathias, C.J.; Josephs, O.; O’Doherty, J.; Zanini, S.; Dewar, B.-K.; Cipolotti, L.; Shallice, T.; Dolan, R.J. Human cingulate cortex and autonomic control: Converging neuroimaging and clinical evidence. Brain 2003, 126, 2139–2152. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Metzger, C.D.; Glover, G.H.; Duyn, J.H.; Heinze, H.-J.; Walter, M. Association between heart rate variability and fluctuations in resting-state functional connectivity. Neuroimage 2013, 68, 93–104. [Google Scholar] [CrossRef]
- Gianaros, P.J.; Wager, T.D. Brain-Body Pathways Linking Psychological Stress and Physical Health. Curr. Dir. Psychol. Sci. 2015, 24, 313–321. [Google Scholar] [CrossRef]
- Sakaki, M.; Yoo, H.J.; Nga, L.; Lee, T.-H.; Thayer, J.F.; Mather, M. Heart rate variability is associated with amygdala functional connectivity with MPFC across younger and older adults. Neuroimage 2016, 139, 44–52. [Google Scholar] [CrossRef]
- Wei, L.; Chen, H.; Wu, G.-R. Structural Covariance of the Prefrontal-Amygdala Pathways Associated with Heart Rate Variability. Front. Hum. Neurosci. 2018, 12, 12. [Google Scholar] [CrossRef]
- Young, H.; Benton, D. We should be using nonlinear indices when relating heart-rate dynamics to cognition and mood. Sci. Rep. 2015, 5, 16619. [Google Scholar] [CrossRef]
- Young, H.A.; Benton, D. Heart-rate variability: A biomarker to study the influence of nutrition on physiological and psychological health? Behav. Pharmacol. 2018, 29, 140–151. [Google Scholar] [CrossRef]
- Dimitriev, D.A.; Saperova, E.V.; Dimitriev, A.D. State Anxiety and Nonlinear Dynamics of Heart Rate Variability in Students. PLoS ONE 2016, 11, e0146131. [Google Scholar] [CrossRef]
- Blons, E.; Arsac, L.M.; Gilfriche, P.; McLeod, H.; Lespinet-Najib, V.; Grivel, E.; Deschodt-Arsac, V. Alterations in heart-brain interactions under mild stress during a cognitive task are reflected in entropy of heart rate dynamics. Sci. Rep. 2019, 9, 18190. [Google Scholar] [CrossRef] [PubMed]
- Cacioppo, J.T.; Burleson, M.H.; Poehlmann, K.M.; Malarkey, W.B.; Kiecolt-Glaser, J.K.; Berntson, G.G.; Uchino, B.N.; Glaser, R. Autonomic and neuroendocrine responses to mild psychological stressors: Effects of chronic stress on older women. Ann. Behav. Med. 2000, 22, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, R.J.; Gatzke-Kopp, L.M.; Roos, L.E.; Skowron, E.A. Resting sympathetic arousal moderates the association between parasympathetic reactivity and working memory performance in adults reporting high levels of life stress. Psychophysiology 2017, 54, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Vaschillo, E.G.; Vaschillo, B.; Lehrer, P.M. Characteristics of Resonance in Heart Rate Variability Stimulated by Biofeedback. Appl. Psychophysiol. Biofeedback 2006, 31, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Lehrer, P.M.; Gevirtz, R. Heart rate variability biofeedback: How and why does it work? Front. Psychol. 2014, 5, 756. [Google Scholar] [CrossRef]
- McCraty, R.; Shaffer, F. Heart Rate Variability: New Perspectives on Physiological Mechanisms, Assessment of Self-regulatory Capacity, and Health risk. Glob. Adv. Health Med. 2015, 4, 46–61. [Google Scholar] [CrossRef]
- Chen, S.; Sun, P.; Wang, S.; Lin, G.; Wang, T. Effects of heart rate variability biofeedback on cardiovascular responses and autonomic sympathovagal modulation following stressor tasks in prehypertensives. J. Hum. Hypertens. 2016, 30, 105–111. [Google Scholar] [CrossRef]
- McCraty, R.; Tomasino, D. Emotional Stress, Positive Emotions, and Psychophysiological Coherence. In Stress in Health and Disease; Wiley-VCH: Weinheim, Germany, 2006; pp. 342–365. [Google Scholar]
- McCraty, R.; Zayas, M.A. Cardiac coherence, self-regulation, autonomic stability, and psychosocial well-being. Front. Psychol. 2014, 5, 5. [Google Scholar] [CrossRef]
- Deschodt-Arsac, V.; Lalanne, R.; Spiluttini, B.; Bertin, C.; Arsac, L.M. Effects of heart rate variability biofeedback training in athletes exposed to stress of university examinations. PLoS ONE 2018, 13, e0201388. [Google Scholar] [CrossRef]
- Schumann, A.; Köhler, S.; Brotte, L.; Bär, K.-J. Effect of an eight-week smartphone-guided HRV-biofeedback intervention on autonomic function and impulsivity in healthy controls. Physiol. Meas. 2019, 40, 064001. [Google Scholar] [CrossRef]
- Spielberger, C. Manual for the State-Trait Anxiety Inventory (STAI); Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Hart, S.G. Nasa-Task Load Index (NASA-TLX); 20 Years Later. Proc. Hum. Factors Ergon. Soc. Annu. Meet. 2006, 50, 904–908. [Google Scholar] [CrossRef]
- Cohen, M.A.; Taylor, J.A. Short-term cardiovascular oscillations in man: Measuring and modelling the physiologies. J. Physiol. 2002, 542, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Pasadyn, S.R.; Soudan, M.; Gillinov, M.; Houghtaling, P.; Phelan, D.; Gillinov, N.; Bittel, B.; Desai, M.Y. Accuracy of commercially available heart rate monitors in athletes: A prospective study. Cardiovasc. Diagn. Ther. 2019, 9, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Gilgen-Ammann, R.; Schweizer, T.; Wyss, T. RR interval signal quality of a heart rate monitor and an ECG Holter at rest and during exercise. Eur. J. Appl. Physiol. 2019, 119, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- TaskForce Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [CrossRef]
- Tarvainen, M.P.; Ranta-Aho, P.O.; Karjalainen, P.A. An advanced detrending method with application to HRV analysis. IEEE Trans. Biomed. Eng. 2002, 49, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [CrossRef] [PubMed]
- Richman, J.S.; Lake, D.E.; Moorman, J.R. Sample entropy. Meth. Enzymol. 2004, 384, 172–184. [Google Scholar]
- Costa, M.; Goldberger, A.L.; Peng, C.-K. Multiscale Entropy Analysis of Complex Physiologic Time Series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef]
- Wu, S.-D.; Wu, C.-W.; Lin, S.-G.; Lee, K.-Y.; Peng, C.-K. Analysis of complex time series using refined composite multiscale entropy. Phys. Lett. A 2014, 378, 1369–1374. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.-K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef]
- Humeau-Heurtier, A. The Multiscale Entropy Algorithm and Its Variants: A Review. Entropy 2015, 17, 3110–3123. [Google Scholar] [CrossRef]
- Huang, N.; Shen, Z.; Long, S.R.; Wu, M.L.C.; Shih, H.H.; Zheng, Q.; Yen, N.C.; Tung, C.-C.; Liu, H.H. The empirical mode decomposition and the Hilbert spectrum for nonlinear and non-stationary time series analysis. Proc. R. Soc. Lond. Ser. A Math. Phys. Eng. Sci. 1998, 454, 903–995. [Google Scholar] [CrossRef]
- Gow, B.J.; Peng, C.-K.; Wayne, P.M.; Ahn, A.C. Multiscale Entropy Analysis of Center-of-Pressure Dynamics in Human Postural Control: Methodological Considerations. Entropy 2015, 17, 7926–7947. [Google Scholar] [CrossRef]
- Beauchaine, T.P. Respiratory Sinus Arrhythmia: A Transdiagnostic Biomarker of Emotion Dysregulation and Psychopathology. Curr. Opin. Psychol. 2015, 3, 43–47. [Google Scholar] [CrossRef]
- Thayer, J.F.; Hansen, A.L.; Saus-Rose, E.; Johnsen, B.H. Heart rate variability, prefrontal neural function, and cognitive performance: The neurovisceral integration perspective on self-regulation, adaptation, and health. Ann. Behav. Med. 2009, 37, 141–153. [Google Scholar] [CrossRef]
- Pyne, J.M.; Constans, J.I.; Wiederhold, M.D.; Gibson, D.P.; Kimbrell, T.; Kramer, T.L.; Pitcock, J.A.; Han, X.; Williams, D.K.; Chartrand, D.; et al. Heart rate variability: Pre-deployment predictor of post-deployment PTSD symptoms. Biol. Psychol. 2016, 121, 91–98. [Google Scholar] [CrossRef]
- Kim, D.; Kang, S.W.; Lee, K.-M.; Kim, J.; Whang, M.-C. Dynamic correlations between heart and brain rhythm during Autogenic meditation. Front. Hum. Neurosci. 2013, 7, 7. [Google Scholar] [CrossRef]
- Mulcahy, J.S.; Larsson, D.E.O.; Garfinkel, S.N.; Critchley, H.D. Heart rate variability as a biomarker in health and affective disorders: A perspective on neuroimaging studies. Neuroimage 2019, 202, 116072. [Google Scholar] [CrossRef]
- Vigo, D.E.; Guinjoan, S.M.; Scaramal, M.; Siri, L.N.; Cardinali, D.P. Wavelet transform shows age-related changes of heart rate variability within independent frequency components. Auton. Neurosci. 2005, 123, 94–100. [Google Scholar] [CrossRef]
- Iyengar, N.; Peng, C.K.; Morin, R.; Goldberger, A.L.; Lipsitz, L.A. Age-related alterations in the fractal scaling of cardiac interbeat interval dynamics. Am. J. Physiology-Regul. Integr. Comp. Physiol. 1996, 271, R1078–R1084. [Google Scholar] [CrossRef] [PubMed]
- Huikuri, H.V.; Mäkikallio, T.H.; Peng, C.K.; Goldberger, A.L.; Hintze, U.; Møller, M. Fractal correlation properties of R-R interval dynamics and mortality in patients with depressed left ventricular function after an acute myocardial infarction. Circulation 2000, 101, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, P.C.; Amaral, L.A.N.; Goldberger, A.L.; Havlin, S.; Rosenblum, M.G.; Struzik, Z.R.; Stanley, H.E. Multifractality in human heartbeat dynamics. Nature 1999, 399, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, P.; Lazzeroni, D.; Coruzzi, P.; Faini, A. Multifractal-Multiscale Analysis of Cardiovascular Signals: A DFA-Based Characterization of Blood Pressure and Heart-Rate Complexity by Gender. Available online: https://www.hindawi.com/journals/complexity/2018/4801924/ (accessed on 31 January 2020).



| Markers | Before HRVB | Post HRVB | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | CV (%) | Mean | SD | CV (%) | Effect size | p Value | |
| RMSSD (ms) | ||||||||
| rest | 27.4 | 16.9 | 61.6 | 38.0 | 22.0 | 57.8 | −0.541 small | 0.007 |
| stress | 34.5 | 15.4 | 44.4 | 45.4 | 17.4 | 38.4 | −0.662 med. | 0.002 |
| LF-HRV (ms2) | ||||||||
| rest | 824 | 653 | 79.3 | 1161 | 647 | 55.8 | −0.418 small | 0.230 |
| stress | 1268 | 957 | 75.5 | 1070 | 732 | 68.4 | 0.232 small | 0.925 |
| HF-HRV (ms2) | ||||||||
| rest | 352 | 465 | 132.2 | 697 | 736 | 105.6 | −0.560 small | 0.008 |
| stress | 472 | 394 | 83.3 | 925 | 709 | 76.7 | −0.790 med. | 0.020 |
| LF/HF | ||||||||
| rest | 3.23 | 1.39 | 43.0 | 2.51 | 1.29 | 51.4 | 0.084 small | 0.050 |
| stress | 3.08 | 2.24 | 72.8 | 1.60 | 1.21 | 75.6 | 0.732 med. | 0.021 |
| Entropy index | ||||||||
| rest | 6.86 | 0.29 | 4.23 | 7.00 | 0.32 | 4.57 | −0.478 small | 0.889 |
| stress | 7.33 | 0.94 | 12.90 | 8.43 | 0.89 | 10.53 | −1.198 large | 0.003 |
| Variables | Sensitivity | Specificity | Youden Index | AUC | p Value |
|---|---|---|---|---|---|
| RMSSD (ms) | |||||
| rest | 0.589 | 0.567 | 0.156 | 0.648 | 0.255 |
| Stress-task | 0.617 | 0.588 | 0.204 | 0.694 | 0.135 |
| LF-HRV (ms2) | |||||
| rest | 0.594 | 0.571 | 0.165 | 0.657 | 0.227 |
| stress-task | 0.528 | 0.521 | 0.049 | 0.546 | 0.722 |
| HF-HRV (ms2) | |||||
| rest | 0.713 | 0.674 | 0.318 | 0.722 | 0.088 |
| stress-task | 0.708 | 0.684 | 0.392 | 0.731 | 0.075 |
| LF/HF | |||||
| rest | 0.611 | 0.583 | 0.194 | 0.685 | 0.155 |
| stress-task | 0.774 | 0.785 | 0.560 | 0.824 | 0.013 |
| Entropy index | |||||
| rest | 0.704 | 0.644 | 0.349 | 0.793 | 0.097 |
| stress-task | 0.813 | 0.799 | 0.612 | 0.818 | 0.010 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deschodt-Arsac, V.; Blons, E.; Gilfriche, P.; Spiluttini, B.; Arsac, L.M. Entropy in Heart Rate Dynamics Reflects How HRV-Biofeedback Training Improves Neurovisceral Complexity during Stress-Cognition Interactions. Entropy 2020, 22, 317. https://doi.org/10.3390/e22030317
Deschodt-Arsac V, Blons E, Gilfriche P, Spiluttini B, Arsac LM. Entropy in Heart Rate Dynamics Reflects How HRV-Biofeedback Training Improves Neurovisceral Complexity during Stress-Cognition Interactions. Entropy. 2020; 22(3):317. https://doi.org/10.3390/e22030317
Chicago/Turabian StyleDeschodt-Arsac, Veronique, Estelle Blons, Pierre Gilfriche, Beatrice Spiluttini, and Laurent M. Arsac. 2020. "Entropy in Heart Rate Dynamics Reflects How HRV-Biofeedback Training Improves Neurovisceral Complexity during Stress-Cognition Interactions" Entropy 22, no. 3: 317. https://doi.org/10.3390/e22030317
APA StyleDeschodt-Arsac, V., Blons, E., Gilfriche, P., Spiluttini, B., & Arsac, L. M. (2020). Entropy in Heart Rate Dynamics Reflects How HRV-Biofeedback Training Improves Neurovisceral Complexity during Stress-Cognition Interactions. Entropy, 22(3), 317. https://doi.org/10.3390/e22030317





