Topographic Reconfiguration of Local and Shared Information in Anesthetic-Induced Unconsciousness
Abstract
1. Introduction
2. Materials and Methods
2.1. Anesthetic Protocol
2.2. fMRI Data Acquisition and Preprocessing
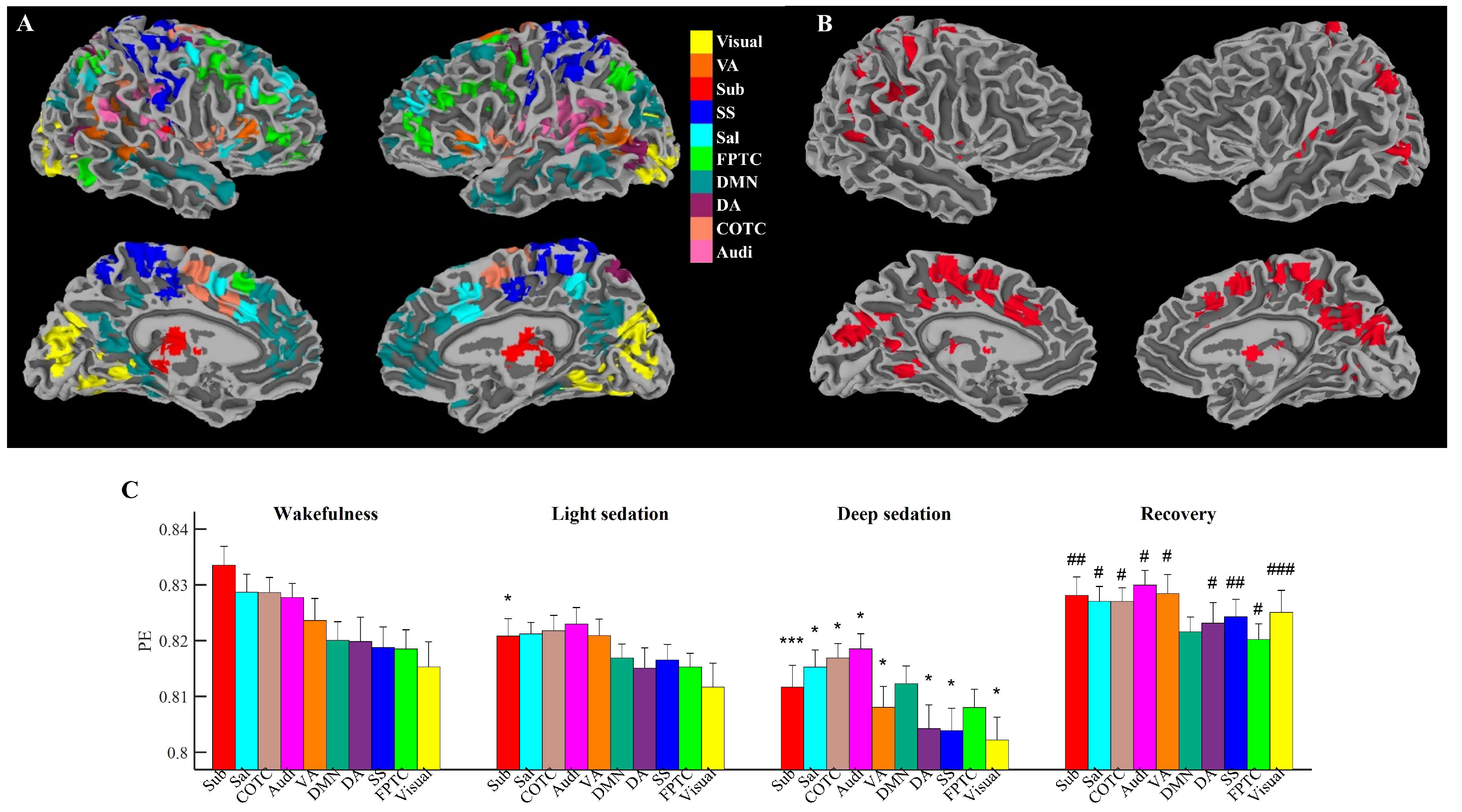
2.3. Definition of Functional Networks
2.4. Ordinal Pattern Analysis
2.5. Topographic Similarity and Relationship between PE and SMI
2.6. Statistical Analysis
3. Results
3.1. Propofol Differentially Suppresses Local Information in Functional Areas
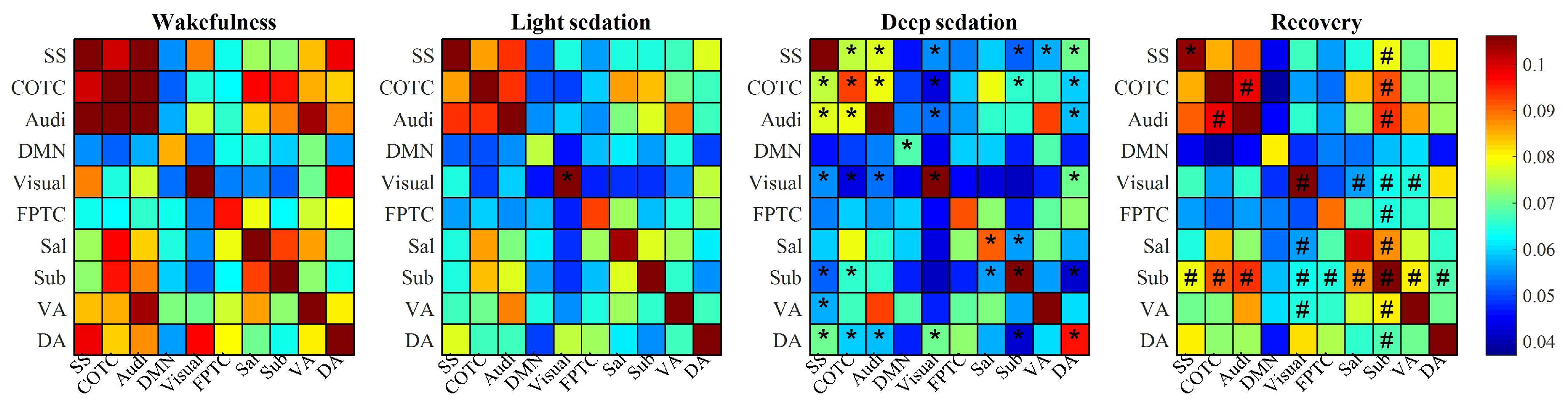
3.2. Functional Connections Are Differentially Affected by Propofol
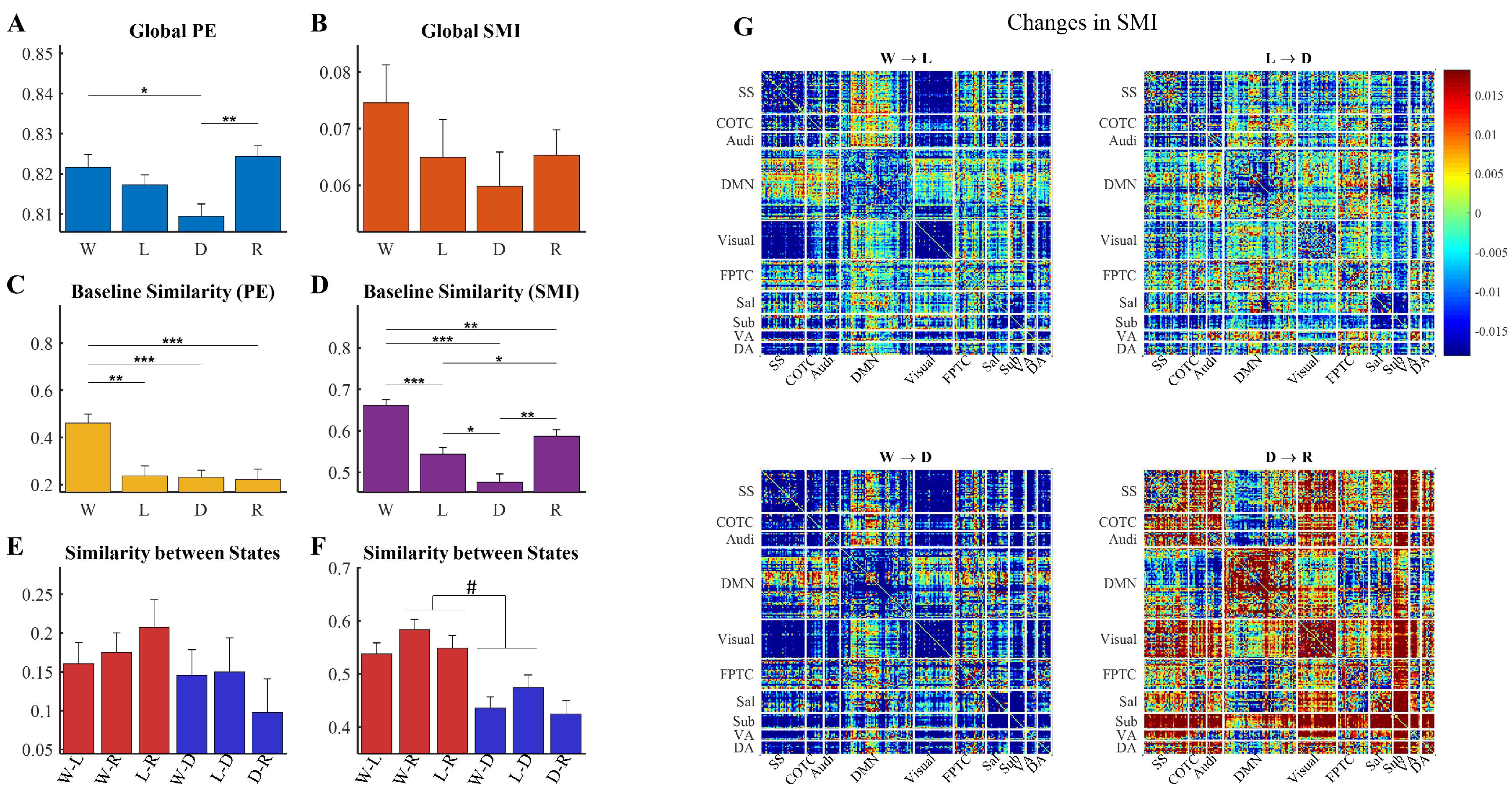
3.3. Topographic Structure of Shared Information Is Associated with States of Consciousness
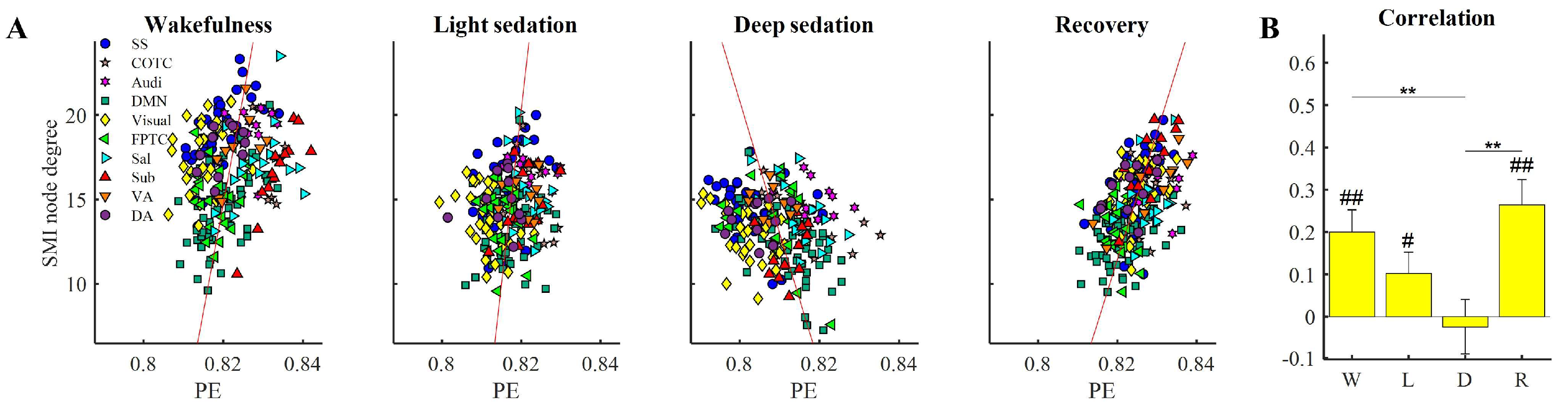
3.4. Altered Relationship between Local and Shared Information
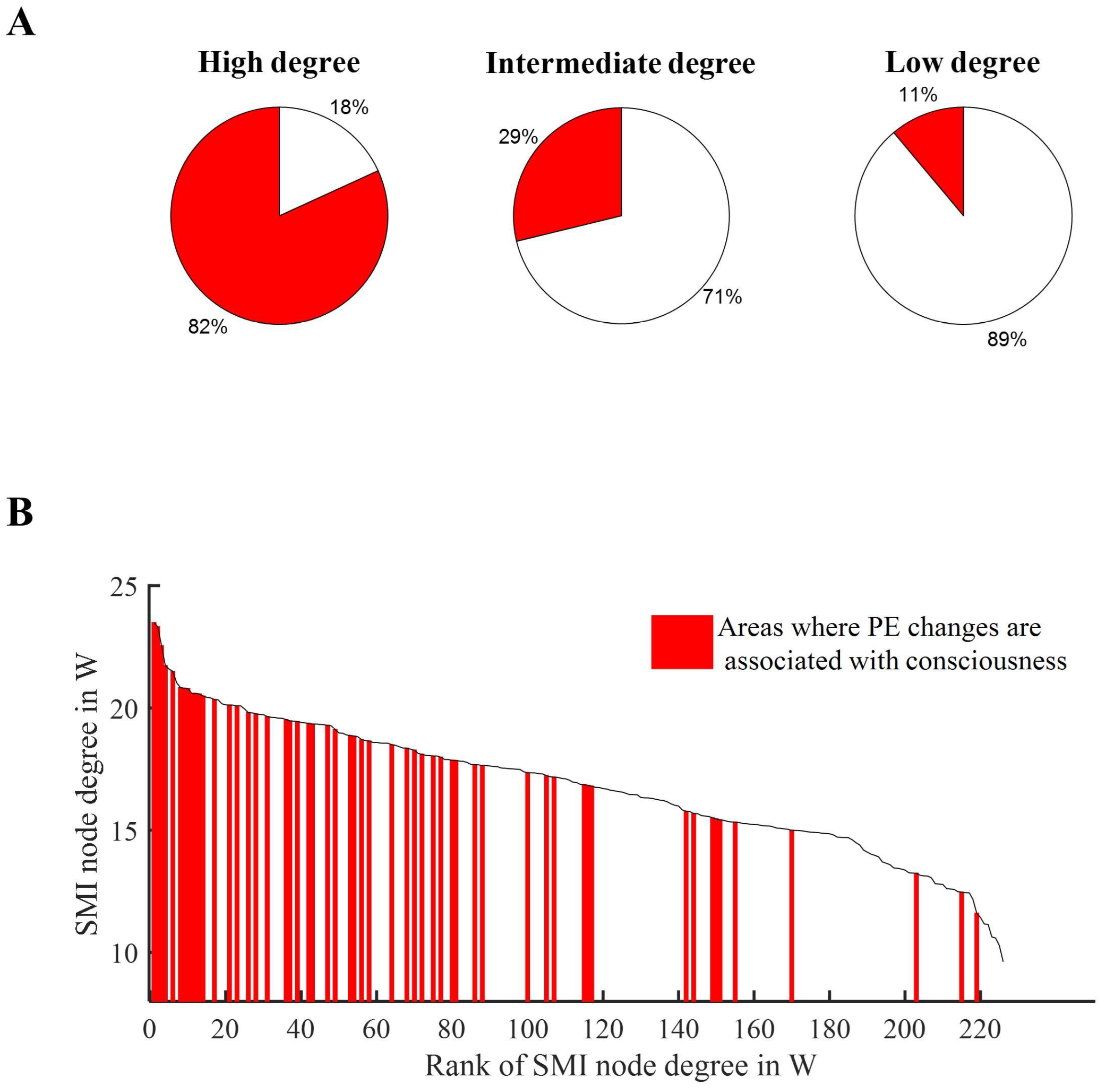
3.5. PE Changes in High Degree Nodes Are Associated with States of Consciousness
4. Discussion
4.1. Differential Effect of Propofol Sedation on Brain Regions
4.2. Topographic Structure of Functional Connectivity Characterizes States of Consciousness
4.3. Propofol Sedation Disrupts the Relationship between Local and Shared Information
4.4. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liang, Z.; Wang, Y.; Sun, X.; Li, D.; Voss, L.J.; Sleigh, J.W.; Hagihira, S.; Li, X. EEG Entropy Measures in Anesthesia. Front. Comput. Neurosci. 2015, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Noh, G.J.; Choi, B.M.; Ku, S.W.; Joo, P.; Jung, W.S.; Kim, S.; Lee, H. Suppressed Neural Complexity during Ketamine- and Propofol-Induced Unconsciousness. Neurosci. Lett. 2017, 653, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Bandt, C.; Pompe, B. Permutation Entropy: A Natural Complexity Measure for Time Series. Phys. Rev. Lett. 2002, 88, 174102. [Google Scholar] [CrossRef] [PubMed]
- Olofsen, E.; Sleigh, J.W.; Dahan, A. Permutation Entropy of the Electroencephalogram: A Measure of Anaesthetic Drug Effect. Br. J. Anaesth. 2008, 101, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Noh, G.J.; Joo, P.; Choi, B.M.; Silverstein, B.H.; Kim, M.; Wang, J.; Jung, W.S.; Kim, S. Diversity of Functional Connectivity Patterns Is Reduced in Propofol-Induced Unconsciousness. Hum. Brain Mapp. 2017, 38, 4980–4995. [Google Scholar] [CrossRef] [PubMed]
- Shalbaf, R.; Behnam, H.; Sleigh, J.W.; Steyn-Ross, A.; Voss, L.J. Monitoring the Depth of Anesthesia Using Entropy Features and an Artificial Neural Network. J. Neurosci. Methods 2013, 218, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Li, X.; Liang, Z.; Voss, L.J.; Sleigh, J.W. Multiscale Permutation Entropy Analysis of EEG Recordings during Sevoflurane Anesthesia. J. Neural Eng. 2010, 7, 046010. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Niu, Y.; Miao, L.; Cao, R.; Yan, P.; Guo, H.; Li, D.; Guo, Y.; Yan, T.; Wu, J.; et al. Decreased Complexity in Alzheimer’s Disease: Resting-State FMRI Evidence of Brain Entropy Mapping. Front. Aging Neurosci. 2017, 9, 378. [Google Scholar] [CrossRef] [PubMed]
- King, J.R.; Sitt, J.D.; Faugeras, F.; Rohaut, B.; El Karoui, I.; Cohen, L.; Naccache, L.; Dehaene, S. Information Sharing in the Brain Indexes Consciousness in Noncommunicative Patients. Curr. Biol. 2013, 23, 1914–1919. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lauer, K.K.; Ward, B.D.; Roberts, C.J.; Gollapudy, S.; Rohloff, R.; Gross, W.; Xu, Z.; Chen, G.; Binder, J.R. Fine-Grained Parcellation of Brain Connectivity Improves Differentiation of States of Consciousness during graded propofol sedation. Brain Connect. 2017, 7, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lauer, K.K.; Douglas Ward, B.; Roberts, C.; Liu, S.; Gollapudy, S.; Rohloff, R.; Gross, W.; Chen, G.; Xu, Z.; et al. Propofol attenuates low-frequency fluctuations of resting-state fMRI BOLD signal in the anterior frontal cortex upon loss of consciousness. Neuroimage 2017, 147, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Liu, X.; Mashour, G.A.; Hudetz, A.G. Timescales of Intrinsic BOLD Signal Dynamics and Functional Connectivity in Pharmacologic and Neuropathologic States of Unconsciousness. J. Neurosci. 2018, 38, 2304–2317. [Google Scholar] [CrossRef] [PubMed]
- Marsh, B.; White, M.; Morton, N.; Kenny, G.N.C. Pharmacokinetic Model Driven Infusion of Propofol in Children. Br. J. Anaesth. 1991, 67, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Glover, G.H.; Li, T.Q.; Ress, D. Image-Based Method for Retrospective Correction of Physiological Motion Effects in FMRI: RETROICOR. Magn. Reson. Med. 2000, 44, 162–167. [Google Scholar] [CrossRef]
- Fox, M.D.; Snyder, A.Z.; Vincent, J.L.; Corbetta, M.; Van Essen, D.C.; Raichle, M.E. From The Cover: The Human Brain Is Intrinsically Organized into Dynamic, Anticorrelated Functional Networks. Proc. Natl. Acad. Sci. USA 2005, 102, 9673–9678. [Google Scholar] [CrossRef] [PubMed]
- Fox, M.D.; Raichle, M.E. Spontaneous Fluctuations in Brain Activity Observed with Functional Magnetic Resonance Imaging. Nat. Rev. Neurosci. 2007, 8, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Chai, X.J.; Castañán, A.N.; Öngür, D.; Whitfield-Gabrieli, S. Anticorrelations in Resting State Networks without Global Signal Regression. Neuroimage 2012, 59, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- He, B.J. Scale-Free Properties of the Functional Magnetic Resonance Imaging Signal during Rest and Task. J. Neurosci. 2011, 31, 13786–13795. [Google Scholar] [CrossRef] [PubMed]
- Stephens, G.J.; Honey, C.J.; Hasson, U. A Place for Time: The Spatiotemporal Structure of Neural Dynamics during Natural Audition. J. Neurophysiol. 2013, 110, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Power, J.D.; Cohen, A.L.; Nelson, S.M.; Wig, G.S.; Barnes, K.A.; Church, J.A.; Vogel, A.C.; Laumann, T.O.; Miezin, F.M.; Schlaggar, B.L.; et al. Functional Network Organization of the Human Brain. Neuron 2011, 72, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.W.; Bassett, D.S.; Power, J.D.; Braver, T.S.; Petersen, S.E. Intrinsic and Task-Evoked Network Architectures of the Human Brain. Neuron 2014, 83, 238–251. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zhang, J.; Longtin, A.; Dumont, G.; Duncan, N.W.; Pokorny, J.; Qin, P.; Dai, R.; Ferri, F.; Weng, X.; et al. Is There a Nonadditive Interaction Between Spontaneous and Evoked Activity? Phase-Dependence and Its Relation to the Temporal Structure of Scale-Free Brain Activity. Cereb. Cortex 2017, 27, 1037–1059. [Google Scholar] [CrossRef] [PubMed]
- Hudetz, A.G.; Liu, X.; Pillay, S.; Boly, M.; Tononi, G. Propofol Anesthesia Reduces Lempel-Ziv Complexity of Spontaneous Brain Activity in Rats. Neurosci. Lett. 2016, 628, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Hudetz, A.G.; Liu, X.; Pillay, S. Dynamic Repertoire of Intrinsic Brain States Is Reduced in Propofol-Induced Unconsciousness. Brain Connect. 2015, 5, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Bruhn, J.; Röpcke, H.; Hoeft, A. Approximate Entropy as an Electroencephalographic Measure of Anesthetic Drug Effect during Desflurane Anesthesia. Anesthesiology 2000, 92, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Schartner, M.; Seth, A.; Noirhomme, Q.; Boly, M.; Bruno, M.A.; Laureys, S.; Barrett, A. Complexity of Multi-Dimensional Spontaneous EEG Decreases during Propofol Induced General Anaesthesia. PLoS ONE 2015, 10, e0133532. [Google Scholar] [CrossRef] [PubMed]
- Flores, F.J.; Hartnack, K.E.; Fath, A.B.; Kim, S.-E.; Wilson, M.A.; Brown, E.N.; Purdon, P.L. Thalamocortical Synchronization during Induction and Emergence from Propofol-Induced Unconsciousness. Proc. Natl. Acad. Sci. USA 2017, 114, E6660–E6668. [Google Scholar] [CrossRef] [PubMed]
- Alkire, M.T.; Haier, R.J.; Fallon, J.H. Toward a Unified Theory of Narcosis : Brain Imaging Evidence for a Thalamocortical Switch as the Neurophysiologic Basis of Anesthetic-Induced Unconsciousness. Conscious. Cognit. 2000, 386, 370–386. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.N.; Lydic, R.; Schiff, N.D. General Anesthesia, Sleep, and Coma. N. Engl. J. Med. 2010, 27, 2638–2650. [Google Scholar] [CrossRef] [PubMed]
- Dueck, M.H.; Petzke, F.; Gerbershagen, H.J.; Paul, M.; Heßelmann, V.; Girnus, R.; Krug, B.; Sorger, B.; Goebel, R.; Lehrke, R.; et al. Propofol Attenuates Responses of the Auditory Cortex to Acoustic Stimulation in a Dose-Dependent Manner: A FMRI Study. Acta Anaesthesiol. Scand. 2005, 49, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Fiset, P.; Daloze, T.; Plourde, G.; Meuret, P.; Bonhomme, V.; Hajj-ali, N.; Backman, S.B.; Evans, A.C. Brain Mechanisms of Propofol-Induced Loss of Consciousness in Humans: A Positron Emission Tomographic Study. J. Neurosci. 1999, 19, 5506–5513. [Google Scholar] [CrossRef] [PubMed]
- Tagliazucchi, E.; Chialvo, D.R.; Siniatchkin, M.; Amico, E.; Brichant, J.; Bonhomme, V.; Noirhomme, Q.; Laufs, H.; Laureys, S. Large-Scale Signatures of Unconsciousness Are Consistent with a Departure from Critical Dynamics. J. R. Soc. Interface 2016, 13, 20151027. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Hudetz, A.G.; Lee, J.; Mashour, G.A.; Lee, U.; The ReCCognition Study Group. Estimating the Integrated Information Measure Phi from High-Density Electroencephalography during States of Consciousness in Humans. Front. Hum. Neurosci. 2018, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Deco, G.; Jirsa, V.K. Ongoing Cortical Activity at Rest: Criticality, Multistability, and Ghost Attractors. J. Neurosci. 2012, 32, 3366–3375. [Google Scholar] [CrossRef] [PubMed]
- Stam, C.J.; van Straaten, E.C.W.; Van Dellen, E.; Tewarie, P.; Gong, G.; Hillebrand, A.; Meier, J.; Van Mieghem, P. The Relation between Structural and Functional Connectivity Patterns in Complex Brain Networks. Int. J. Psychophysiol. 2016, 103, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Hudetz, A.G.; Humphries, C.J.; Binder, J.R. Spin-Glass Model Predicts Metastable Brain States that Diminish in Anesthesia. Front. Syst. Neurosci. 2014, 8, 234. [Google Scholar] [CrossRef] [PubMed]
- Stam, C.J. Modern Network Science of Neurological Disorders. Nat. Rev. Neurosci. 2014, 15, 683–695. [Google Scholar] [CrossRef] [PubMed]
- Wollstadt, P.; Sellers, K.K.; Rudelt, L.; Priesemann, V.; Hutt, A.; Fröhlich, F.; Wibral, M. Breakdown of Local Information Processing May Underlie Isoflurane Anesthesia Effects. PLoS Comput. Biol. 2017, 13, e1005511. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, U.; Antkowiak, B. Molecular and Neuronal Substrates for General Anaesthetics. Nat. Rev. Neurosci. 2004, 5, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, H.C.; Akabas, M.H.; Goldstein, P.A.; Trudell, J.R.; Orser, B.A.; Harrison, N.L. Emerging Molecular Mechanisms of General Anesthetic Action. Trends Pharmacol. Sci. 2005, 26, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Sanders, R.D.; Tononi, G.; Laureys, S.; Sleigh, J.W. Unresponsiveness ≠ Unconsciousness. Anesthesiology 2012, 116, 946–959. [Google Scholar] [CrossRef] [PubMed]
- Mashour, G.A.; LaRock, E. Inverse Zombies, Anesthesia Awareness, and the Hard Problem of Unconsciousness. Conscious. Cognit. 2008, 17, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Noreika, V.; Jylhänkangas, L.; Móró, L.; Valli, K.; Kaskinoro, K.; Aantaa, R.; Scheinin, H.; Revonsuo, A. Consciousness Lost and Found: Subjective Experiences in an Unresponsive State. Brain Cognit. 2011, 77, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Kaisti, K.K.; Metsähonkala, L.; Teräs, M.; Oikonen, V.; Aalto, S.; Jääskeläinen, S.; Hinkka, S.; Scheinin, H. Effects of Surgical Levels of Propofol and Sevoflurane Anesthesia on Cerebral Blood Flow in Healthy Subjects Studied with Positron Emission Tomography. Anesthesiology 2002, 96, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Kondo, Y.; Hirose, N.; Maeda, T.; Suzuki, T.; Yoshino, A.; Katayama, Y. Changes in Cerebral Blood Flow and Oxygenation during Induction of General Anesthesia with Sevoflurane versus Propofol. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2016; Volume 876, pp. 479–484. [Google Scholar]
- Supp, G.G.; Siegel, M.; Hipp, J.F.; Engel, A.K. Cortical Hypersynchrony Predicts Breakdown of Sensory Processing during Loss of Consciousness. Curr. Biol. 2011, 21, 1988–1993. [Google Scholar] [CrossRef] [PubMed]
- Purdon, P.L.; Pierce, E.T.; Mukamel, E.A.; Prerau, M.J.; Walsh, J.L.; Wong, K.F.K.; Salazar-Gomez, A.F.; Harrell, P.G.; Sampson, A.L.; Cimenser, A.; et al. Electroencephalogram Signatures of Loss and Recovery of Consciousness from Propofol. Proc. Natl. Acad. Sci. USA 2013, 110, E1142–E1151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, Z.; Chen, Y.; Zhang, J.; Ghinda, D.; Nikolova, Y.; Wu, J.; Xu, J.; Bai, W.; Mao, Y.; et al. Breakdown in the Temporal and Spatial Organization of Spontaneous Brain Activity during General Anesthesia. Hum. Brain Mapp. 2018, 39, 2035–2046. [Google Scholar] [CrossRef] [PubMed]
- Kuizenga, K.; Kalkman, C.J.; Hennis, P.J. Quantitative Electroencephalographic Analysis of the Biphasic Concentration-Effect Relationship of Propofol in Surgical Patients during Extradural Analgesia. Br. J. Anaesth. 1998, 80, 725–732. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Huang, Z.; Liu, X.; Lee, U.; Hudetz, A.G. Topographic Reconfiguration of Local and Shared Information in Anesthetic-Induced Unconsciousness. Entropy 2018, 20, 518. https://doi.org/10.3390/e20070518
Lee H, Huang Z, Liu X, Lee U, Hudetz AG. Topographic Reconfiguration of Local and Shared Information in Anesthetic-Induced Unconsciousness. Entropy. 2018; 20(7):518. https://doi.org/10.3390/e20070518
Chicago/Turabian StyleLee, Heonsoo, Zirui Huang, Xiaolin Liu, UnCheol Lee, and Anthony G. Hudetz. 2018. "Topographic Reconfiguration of Local and Shared Information in Anesthetic-Induced Unconsciousness" Entropy 20, no. 7: 518. https://doi.org/10.3390/e20070518
APA StyleLee, H., Huang, Z., Liu, X., Lee, U., & Hudetz, A. G. (2018). Topographic Reconfiguration of Local and Shared Information in Anesthetic-Induced Unconsciousness. Entropy, 20(7), 518. https://doi.org/10.3390/e20070518





